Abstract
Objectives
We evaluated the impact of a 2014 New York City health code change requiring laboratories to indicate if a patient is pregnant or probably pregnant in the electronic laboratory report (ELR) when reporting syphilis and hepatitis B virus (HBV) cases to the New York City Department of Health and Mental Hygiene (DOHMH).
Methods
We calculated the number of pregnant persons with syphilis or HBV infection reported to DOHMH from January 1, 2013, through June 30, 2018. We compared the proportion in which the first report to DOHMH was an ELR with pregnancy indicated before and after the policy change. We calculated time between first ELR with pregnancy indicated and subsequent reporting by a method other than ELR and the proportion of cases in which ELR with pregnancy indicated was the only report source.
Results
A total of 552 new syphilis and 8414 HBV-infected cases were reported to DOHMH. From January 2013–June 2014 (pre-change) to January 2017–June 2018 (post-change), the proportion of cases in which ELR with pregnancy indicated was the first report to DOHMH increased significantly (14.7% [23/156] to 46.2% [80/173] for syphilis; 8.0% [200/2498] to 45.3% [851/1879] for HBV infection [P < .001]). Median time between first ELR with pregnancy indicated and subsequent reporting by a method other than ELR was 9.0 days for syphilis and 51.0 days for HBV infection. ELR with pregnancy indicated was the only report for 43.1% (238/552) of syphilis cases and 23.4% (1452/6200) of HBV cases during the study period.
Conclusion
Including pregnancy status with ELR can increase the ability of public health departments to conduct timely interventions to prevent mother-to-child transmission.
Mother-to-child transmission of syphilis and hepatitis B virus (HBV) infection can result in severe morbidity and mortality in infants. Untreated maternal syphilis during pregnancy can lead to neonatal death, stillbirth or fetal loss, and miscarriage. 1 Among infants with congenital syphilis, complications include preterm birth, low birth weight, and abnormalities such as osteochondritis, hemolytic anemia, hydrocephalus, and severe mental disability. 2 -5 Among infants infected with HBV at birth, up to 90% will develop chronic infection, 25% of whom will die prematurely from HBV-related complications, including liver disease and liver cancer. 6
Prenatal identification of syphilis and HBV infection is critically important for timely implementation of prevention efforts, including screening and treatment of infected pregnant women and HBV postexposure prophylaxis (PEP) of newborns, to minimize the risk of congenital infections and associated complications. Routine prenatal screening for syphilis and HBV infection is recommended nationally and is required in New York State. Screening for syphilis is required at first prenatal care examination, and screening for HBV is required at the time of blood draw during prenatal care. Syphilis screening is repeated at delivery, but HBV screening is repeated only if the prenatal HBV result is not available. 7 -10 Early treatment of pregnant women infected with syphilis by using benzathine penicillin G prevents most cases of congenital syphilis. 11 Administration of HBV PEP to the infant, consisting of HBV immune globulin and the first dose of HBV vaccine within 12 hours of birth, is 94% effective at preventing perinatal HBV transmission. 12 Identification of maternal HBV infection during pregnancy is also necessary for assessing the need for prenatal antiviral treatment of women with high HBV viral load, to further reduce the likelihood of mother-to-child transmission. 12
Dual laboratory and health care provider reporting of pregnant women and girls with (1) syphilis or (2) HBV infection is mandated in New York State. 9,13 Once reports are received, the New York City Department of Health and Mental Hygiene (DOHMH) conducts patient-level case management for pregnant women and girls with these infections. For syphilis, case management involves ensuring necessary treatment and follow-up testing during pregnancy; for HBV infection, case management involves providing PEP at birth as well as medical referrals, as needed. Case management for both conditions must begin during the prenatal period to implement the interventions needed to prevent mother-to-child transmission.
Health care provider reporting in New York City to DOHMH is required by law for reportable health conditions but is not fully enforced; as such, health care providers have been able to get away with not reporting. Health care providers do not always report pregnant women and girls with syphilis and HBV infection as required, and when cases are reported, reports may not indicate if the person is pregnant. As such, an amendment to Article 13 of the New York City Health Code in 2008 required laboratories to indicate pregnancy status “if known” on all positive test results for “clinically relevant” diseases. 14 Despite the 2008 amendment, many laboratories reported pregnancy status only if it was explicitly indicated by the ordering health care provider on the laboratory requisition. To increase reporting of pregnancy when it was not included on the requisition form, Article 13 of the New York City Health Code was further amended in July 2014 to also require reporting of pregnancy when pregnancy is “probable.” 14 In August 2014, the DOHMH sent a letter to laboratory directors that included a technical guide on how to identify and indicate probable pregnancy when reporting test results (J. Varma, personal communication, August 1, 2014). The guide included examples of probable pregnancy, pregnancy-related diagnostic codes on a test requisition, tests ordered as part of a prenatal panel, or specimens submitted from labor and delivery units or freestanding birthing centers. More than 2000 pregnancy-related codes are in the International Statistical Classification of Diseases and Related Health Problems, 10th Revision (ICD-10), some of which are general (eg, pregnant state, incidental [Z33.1]) and some of which are disease-specific (eg, syphilis complicating pregnancy, first trimester [O98.111]). 15,16 If pregnancy is known or probable pregnancy indicators are present, laboratories are instructed to indicate pregnancy by inserting the text “pregnant, prenatal, or probable pregnant” into the electronic laboratory report (ELR)–relevant clinical information field of the electronic data reporting message (specifically, the Observation Request segment 13 in the Health Level-7 standard).
We evaluated the effect of the New York City Health Code change on timely identification of syphilis and HBV infections among pregnant women and girls by measuring (1) the proportion of reports for syphilis and HBV infection among pregnant women and girls for which ELR with pregnancy indicated was the first source of report, (2) the time between the first ELR with pregnancy indicated and any subsequent report(s) by methods other than ELR (“non-ELRs” [eg, paper laboratory reports, paper provider reports, electronic provider reports, telephone, medical record review, jail import record]), (3) the proportion of reports for syphilis and HBV infection among pregnant women and girls for which ELR with pregnancy indicated was the only source of report, (4) the accuracy of pregnancy as reported by laboratories, and (5) compliance by laboratories reporting probable pregnancy. To our knowledge, this study is the first to evaluate the public health effect of a policy change requiring laboratories to indicate if a patient is pregnant or probably pregnant when electronically reporting results for syphilis and HBV infections. Although other reportable diseases can cause congenital infection, we selected syphilis and HBV for this analysis because these are the only diseases used by the DOHMH ELR team for testing laboratory compliance of pregnancy reporting since the health code amendment was passed.
Methods
Routine DOHMH Surveillance for Syphilis and HBV Infection During Pregnancy
As part of routine surveillance to prevent perinatal transmission of syphilis and HBV infection, all persons with syphilis and pregnant women and girls with HBV infection are reported to DOHMH by health care providers, laboratories, and other external sources. DOHMH investigates all reports of suspected new cases of syphilis and, for women and girls of childbearing age, determines whether the person is pregnant if this information is missing from the report or confirms that the person is pregnant. When reports of HBV infection with pregnancy indicated are received, DOHMH investigates to confirm that the person is pregnant. For both conditions, “true” pregnancy is confirmed through outreach to the medical provider, medical record review, and/or patient interview. Women and girls with syphilis and HBV infection in pregnancy reported during the prenatal or postnatal period and residing in New York City are classified using the Council of State and Territorial Epidemiologists case definitions for syphilis and HBV infection. 17 -19
Our analysis included new cases of syphilis or HBV infection among pregnant women and girls that were reported from January 1, 2013, through June 30, 2018. Report dates were defined as diagnosis date for syphilis and as DOHMH classification date for HBV infection. For syphilis only, study data also included women and girls reported to DOHMH by laboratories or health care providers without pregnancy information, who were subsequently identified as pregnant through DOHMH investigation or other methods. Because this analysis was considered a routine surveillance activity, institutional review board approval was not sought.
Measures of ELR With Pregnancy Indicated
We calculated the proportion of cases in which ELR with either syphilis or HBV infection, as well as pregnancy indicated, was the first of 1 or more reports to DOHMH by comparing the number of cases for which ELR with pregnancy indicated was the first report (numerator) with the total number of cases reported by any method (denominator). We calculated the proportions on a semiannual basis for reports from January 2013 through June 2018; we also calculated the proportions for the 18-month period before the July 2014 New York City Health Code amendment (January 2013 through June 2014) and compared those proportions with the 18-month period at the end of the evaluation period (January 2017 through June 2018), using Pearson χ2 analysis, with P < .05 considered significant.
Among cases in which ELR with pregnancy indicated was the first source of report from January 2013 through June 2018, we calculated the median and mean number of days from the initial ELR to the subsequent non-ELR; we did not include cases for which ELR with pregnancy indicated was the only reporting source in this measure.
We calculated the proportion of cases reported only by ELR with pregnancy indicated from January 2013 through June 2018 by comparing the number of cases for which ELR with pregnancy indicated was the only source of report (numerator) with the total number of cases reported by any method (denominator). For HBV infection, our analysis excluded cases that were only reported postnatally, because multiple other mechanisms are in place for DOHMH to identify pregnant persons with HBV infection after delivery aside from ELR or health care provider reporting (eg, universal reporting of maternal HBV infection status on the newborn metabolic screening form and mandatory reporting of HBV PEP administered to infants born to HBV-positive mothers).
We calculated the positive predictive value of pregnancy as reported through ELR from July 2014 through June 2018 by comparing the number of HBV ELRs with pregnancy indicated that were verified as true pregnancies (numerator) with the total number of HBV ELRs received with pregnancy indicated (denominator). Because true pregnancy is not assessed for syphilis test results if the ELR does not correspond to a new case of syphilis, we did not calculate the positive predictive value of pregnancy for ELRs of syphilis.
After the health code amendment was passed in July 2014, DOHMH’s ELR team informed all commercial and hospital-based local, regional, and national laboratories located in New York City or that routinely report results for New York City residents of the amendment; the team also sent notifications about the new pregnancy reporting requirement to laboratories during a 1-year period before amendment enactment via multiple email notifications and a DOHMH ELR newsletter. The ELR team conducted targeted outreach through email and telephone calls to those laboratories that had reported syphilis or HBV infection to DOHMH before the 2014 health code amendment and provided guidance on how to implement reporting of probable pregnancy. The ELR team requested that these laboratories demonstrate capture and successful transmission of pregnancy indicators via parallel testing of real and test messages through ELRs to DOHMH. The DOHMH ELR team and disease surveillance program staff members reviewed incoming reports to confirm successful transmission of the pregnancy indicators. Laboratory compliance with the health code changes is tracked by the ELR team, and monthly compliance reports are sent to the laboratories. Laboratory compliance with the health code change is calculated as the proportion of all laboratories reporting syphilis or HBV infection that successfully transmitted ELRs to DOHMH with probable pregnancy indicated.
Results
A total of 552 new cases of syphilis and 8414 cases of HBV infection among pregnant women and girls were reported to DOHMH via any method from January 2013 through June 2018. The proportion of new syphilis and HBV cases in which ELR with pregnancy indicated was the first source of report increased significantly from the 18 months before the amendment (January 2013–June 2014) to the last 18 months of the observation period (January 2017–June 2018); the proportion of new cases of syphilis increased significantly from 14.7% (23/156) to 46.2% (80/173) (P < .001), and the proportion of new cases of HBV infection increased significantly from 8.0% (200/2498) to 45.3% (851/1879) (P < .001) (Figures 1 and 2).
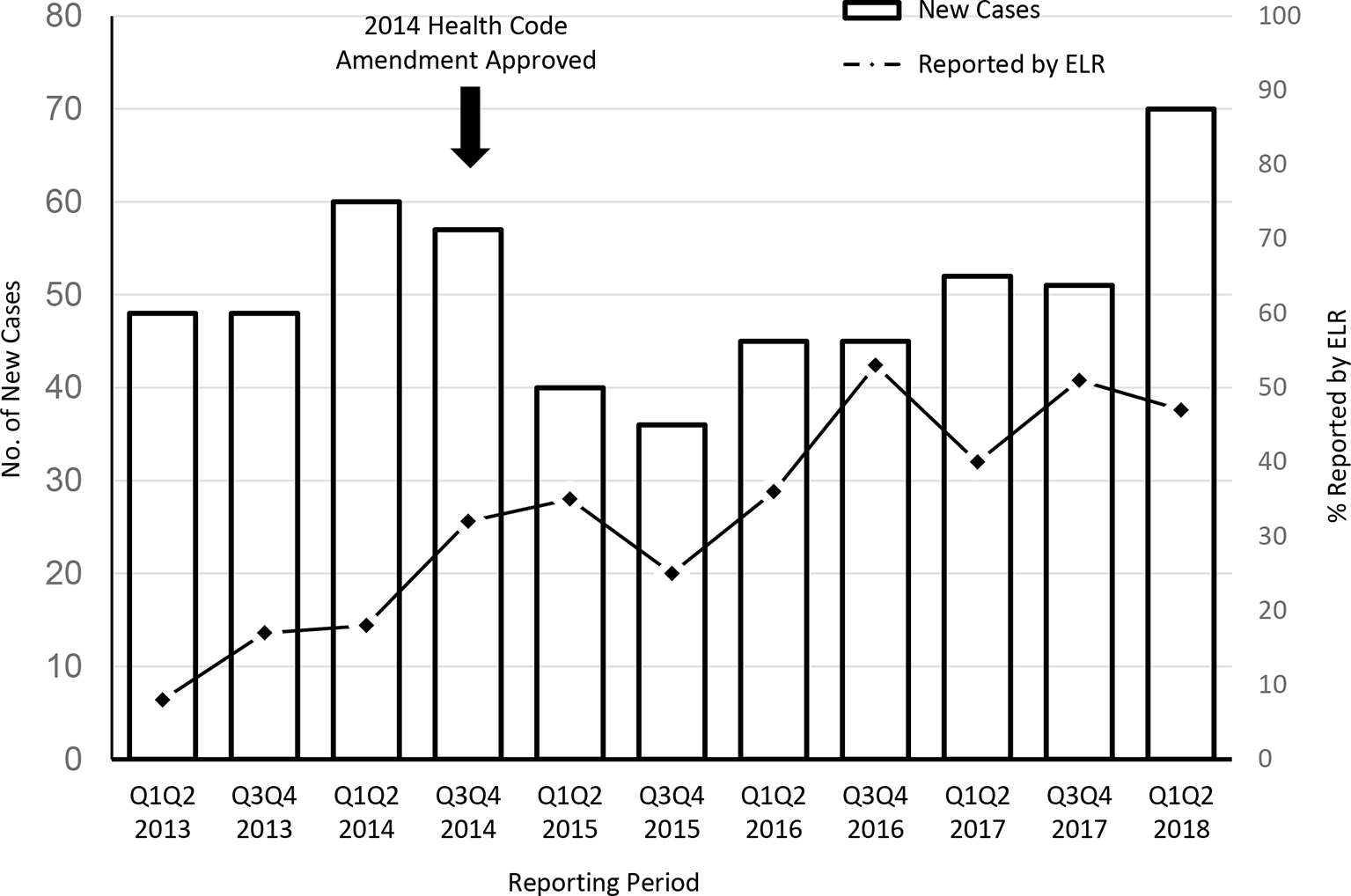
Number of new syphilis-infected pregnant women and girls and proportion initially reported as pregnant by electronic laboratory report (ELR) per half-year interval, New York City, January 1, 2013, through June 30, 2018. Data source: New York City Department of Health and Mental Hygiene. 20
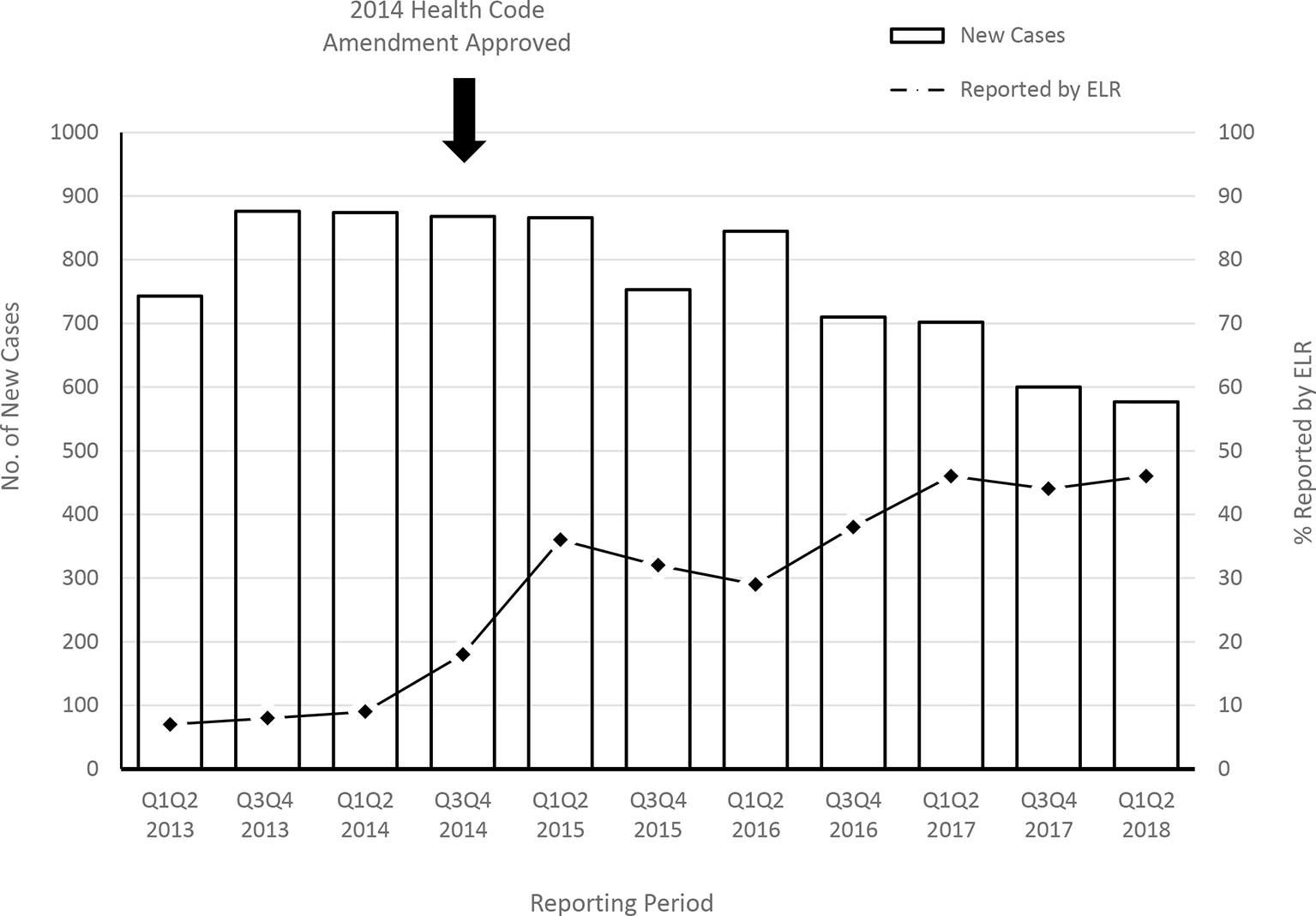
Number of pregnant women and girls infected with hepatitis B and proportion initially reported as pregnant by electronic laboratory report (ELR) per half-year interval, New York City, January 1, 2013, through June 30, 2018. Data source: New York City Department of Health and Mental Hygiene. 20
The number of days between the first case report received by DOHMH via ELR with pregnancy indicated and a subsequent non-ELR report for syphilis was 9.0 days (median) and 26.3 days (mean), and for HBV infection was 51.0 days (median) and 99.2 days (mean). The proportion of pregnant women and girls for whom ELR with pregnancy indicated was the only source of report from January 2013 through June 2018 was 43.1% (238 of 552) for syphilis and 23.4% (1452 of 6200) for HBV infection. Of 3459 HBV cases reported by ELR with pregnancy indicated from July 2014 through June 2018, 3239 reported true pregnancies, for a 93.6% positive predictive value of pregnancy.
We identified 43 laboratories that conduct testing for syphilis and HBV infection among pregnant women and girls and report to DOHMH. Of these laboratories, 39 (90.7%) successfully transmitted a probable pregnancy indication to DOHMH via ELR and were considered in compliance with the health code amendment as of June 30, 2018. The other 4 (9.3%) laboratories were not in compliance; 3 laboratories were awaiting system modification, and 1 was in a testing phase.
Discussion
Implementation of the 2014 New York City Health Code amendment to require laboratories to indicate probable pregnancy on ELRs with positive syphilis and HBV test results led to earlier identification of syphilis and HBV infection among pregnant women and girls than before the amendment. The median time from the first ELR with pregnancy indicated and subsequent non-ELRs was 9.0 days for syphilis and 51.0 days for HBV infection. Because of unreliable health care provider reporting, ELRs were often the only source of case reports to DOHMH during pregnancy (43.1% [238 of 552] for syphilis and 23.4% [1452 of 6200] for HBV infection). Health care providers do not consistently report pregnant women and girls infected with syphilis and HBV, and electronic data transmission from laboratories is fast; the inclusion of pregnancy indicators on laboratory reports can improve the timeliness of identification of these pregnant women and girls by bringing cases to the attention of health departments more quickly, improving the ability to carry out timely interventions to prevent mother-to-child transmission of syphilis and HBV infection.
In June 2018, 91% (39 of 43) of the laboratories were certified by DOHMH as reporting probable pregnancy in accordance with the 2014 health code amendment, demonstrating that this approach was feasible. With implementation of similar health code amendments and similar ELR systems, other public health jurisdictions could also leverage ELRs that include pregnancy status to improve disease surveillance and prevention activities. However, implementing the changes was resource intensive for both DOHMH and the laboratories. DOHMH has a dedicated ELR team that works directly with the laboratories to assist with implementation of new mandates; such resources may not be available at other state or local health departments. Although the DOHMH ELR team serves as a liaison to the laboratories, disease surveillance program staff member time is also required to assist with testing and to evaluate the quality of laboratory data received. Implementation of these changes also requires laboratory staff member time and financial resources to plan, test, and implement updates to laboratory requisition forms and laboratory information systems, and to make changes to web applications used by smaller laboratories for submitting ELRs.
Including pregnancy status presents several challenges for laboratories. Pregnancy status is often not submitted by health care providers on laboratory requisition forms, especially if pregnancy is not a required field. If laboratories cannot determine or do not receive or process probable pregnancy indicators in their information systems, they cannot include pregnancy status when sending ELR messages to public health agencies. Laboratories may not offer prenatal panels, or their laboratory information system may not be set up to receive pregnancy indicators such as ICD-10 codes. Even when laboratories do have the ability to receive this information, health care providers must order the tests as part of prenatal panels or include pregnancy-related diagnostic codes. Despite these barriers, laboratories are successfully submitting ELRs indicating pregnancy to DOHMH, and national commercial laboratories have the ability to send data on pregnancy status to health departments in other states and jurisdictions as well.
The proportion of pregnant women and girls infected with syphilis and HBV initially reported as pregnant by ELR in New York City began increasing in the year before the 2014 New York City Health Code amendment. This increase may be attributed to national efforts from a workgroup convened by the Centers for Disease Control and Prevention, Division of Viral Hepatitis, in 2012 to promote ELR of pregnancy indicators with reports of positive HBV test results. The workgroup included partners from health departments, commercial laboratories, and professional organizations. This collaboration led to the implementation of pregnancy reporting by 4 large nationwide laboratories; some laboratories that implemented pregnancy reporting for HBV infection as a result of these national efforts concurrently began indicating pregnancy on laboratory reports of syphilis infection.
In addition, the workgroup issued an official memorandum that supported pregnancy reporting and provided action steps for all laboratories, which was shared with New York City laboratories to facilitate their compliance with the 2014 health code amendment. 16,21
Limitations
This analysis had 2 limitations. Our evaluation did not include an analysis of the effect of the health code change on the number of syphilis and HBV infections in newborns, nor did it examine what proportion of results were received too late for intervention. Future studies might assess if timely laboratory reporting of syphilis and HBV infections with pregnancy indicated is associated with a decreased rate of perinatal infections.
Conclusions
This study demonstrates how public health policy can be used to assist public health prevention efforts and how electronic laboratory reporting could be used to improve the timeliness of disease investigations and implementation of known interventions. It also highlights the importance of strong collaborations among laboratories, public health surveillance, and information technology programs to implement changes related to electronic laboratory reporting for public health purposes.
The 2014 New York City Health Code amendment resulted in improvements in the timeliness of DOHMH’s identification of pregnant women and girls with new syphilis and HBV infections and the completeness of identification of pregnant women and girls with HBV infection. ELRs that include pregnancy status can improve the timely implementation of interventions, particularly medical treatment and health education, during pregnancy to help prevent mother-to-child transmission of syphilis and HBV infection. Although this analysis was limited to pregnancy reporting by ELR for syphilis and HBV infection, the 2014 New York City Health Code amendment can also assist with the surveillance and potential prevention of other infections, such as HIV, hepatitis C virus, and Zika virus.
Footnotes
Acknowledgements
The authors thank Preeti Patel, DrPH, for her insight on the statistical analysis.
Authors’ Note
Tim S. Liao (Bureau of Sexually Transmitted Infections) and Ariba Hashmi (Bureau of Immunization) contributed equally to this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
