Abstract
Objectives:
Electronic case reporting (eCR), a cornerstone of the Centers for Disease Control and Prevention’s (CDC’s) Data Modernization Initiative, automates bidirectional information sharing between electronic health records and public health agencies for reportable conditions. eCR grew rapidly in response to the COVID-19 pandemic. CDC and the Florida Department of Health (FDOH) collaborated to compare the completeness and timeliness of eCR with that of traditional reporting methods for COVID-19–related patient encounters at 4 health care organizations in Florida in December 2020.
Methods:
Electronic initial case reports (eICRs) were matched to corresponding (ie, for same patient encounter) electronic laboratory reports (ELRs) or manually transmitted documents. We extracted and compared selected data from each report type across matched reports for completeness and timeliness.
Results:
Most (>98%) eICRs provided earlier notification of COVID-19 to the local public health department than corresponding ELRs or manually transmitted documents. Additionally, eICRs provided more data on race and ethnicity (>90%) than ELRs (71%) or manually transmitted documents (<5%).
Conclusions:
Advancing implementation of eCR nationwide may provide more complete and timely case data than ELR or manually transmitted documents to guide public health action.
Keywords
The Centers for Disease Control and Prevention (CDC) launched the Data Modernization Initiative in 2019 to create an integrated and real-time surveillance infrastructure to monitor and protect against health threats. Rapid and robust surveillance and reporting of cases of diseases and conditions is a cornerstone of data modernization efforts. In case reporting, information pertaining to cases of reportable diseases and conditions is reported by health care providers, laboratories, hospitals, and other partners to local and state health departments for investigation and action. The list of reportable diseases and conditions is determined by local laws; among these, the Council of State and Territorial Epidemiologists and CDC have identified a subset of reportable diseases and conditions that are considered nationally notifiable. 1 Electronic case reporting (eCR) automates bidirectional information sharing between electronic health records (EHRs) and public health agencies for reportable conditions and is a cornerstone of the Data Modernization Initiative. 2 Nationwide response to the COVID-19 pandemic emphasized the need for modernized public health data infrastructure and processes, including rapid and robust case reporting, and resulted in substantial resource allocation to support the implementation of eCR in state and local public health jurisdictions. In addition, the burden of reporting on health care providers was immense and the desire to automate the reporting resulted in the rapid implementation of eCR in response to the COVID-19 pandemic. 3
Until implementation of eCR, case reports for reportable conditions from clinical providers were typically manually transmitted via telephone, fax, or mail. This process interrupts clinical workflows, 4 and reports can be inaccurate, incomplete, delayed, or not transmitted. 5 Furthermore, public health staff must manually enter information from the transmitted documents into surveillance systems. A limited number of jurisdictions used data from EHRs to assist with case reporting prior to the COVID-19 pandemic for selected conditions,6,7 but no large-scale efforts have been made to automate case reporting from EHRs until eCR. Although public health agencies receive laboratory results (in electronic laboratory reporting [ELR]) automatically, these reports are not full case reports and do not include necessary information about the patient and condition, which has challenged the timeliness and completeness of surveillance data nationwide and presented a major hurdle in response to situations such as the COVID-19 pandemic. Thus, eCR developed a vital component, a shared services infrastructure, for interoperability between health care providers and public health agencies, that supports rapid case reporting and notification. Prior to the COVID-19 pandemic, fewer than 200 health care facilities nationwide had begun implementation of eCR for 5 or 6 reportable conditions. 8 In this study, CDC and the Florida Department of Health (FDOH) collaborated to compare the completeness and timeliness of eCR with that of traditional reporting methods for COVID-19–related patient encounters at 4 health care organizations (HCOs) in Florida in December 2020.
Methods
CDC and FDOH identified information from December 2020 electronic initial case reports (eICRs), ELRs, and manually transmitted documents to assess differences in completeness of data on race and ethnicity and timeliness in COVID-19 reporting from 4 participating HCOs. Part of eCR, eICRs are electronically transmitted documents that contain a standard set of data elements, vocabularies, and value sets related to information commonly found in EHRs and necessary for public health reporting. The 4 HCOs’ EHR-generated eICRs that were routed to FDOH and predetermined data elements in reports were automatically processed into the state surveillance system. Similarly, laboratories transmitted ELRs to FDOH in batches on an hourly or daily basis; these ELRs were automatically processed into the state surveillance system. ELRs included in this study were transmitted to public health hourly, 3 times per day, or once per day (depending on rules established by the transmitting HCO). Manually transmitted documents (eg, medical record excerpts and health care provider notes, demographic summary sheets) sent to county health departments via email or fax were manually uploaded into the same surveillance system. FDOH provided all eICRs, ELRs, and manually transmitted documents it received in December 2020; the sample period was chosen to ensure adequate sample size during a period of parallel production for participating HCOs (ie, both eICR and manually transmitted documents were being generated and transmitted). All reports were transferred to CDC using a secure file transfer protocol.
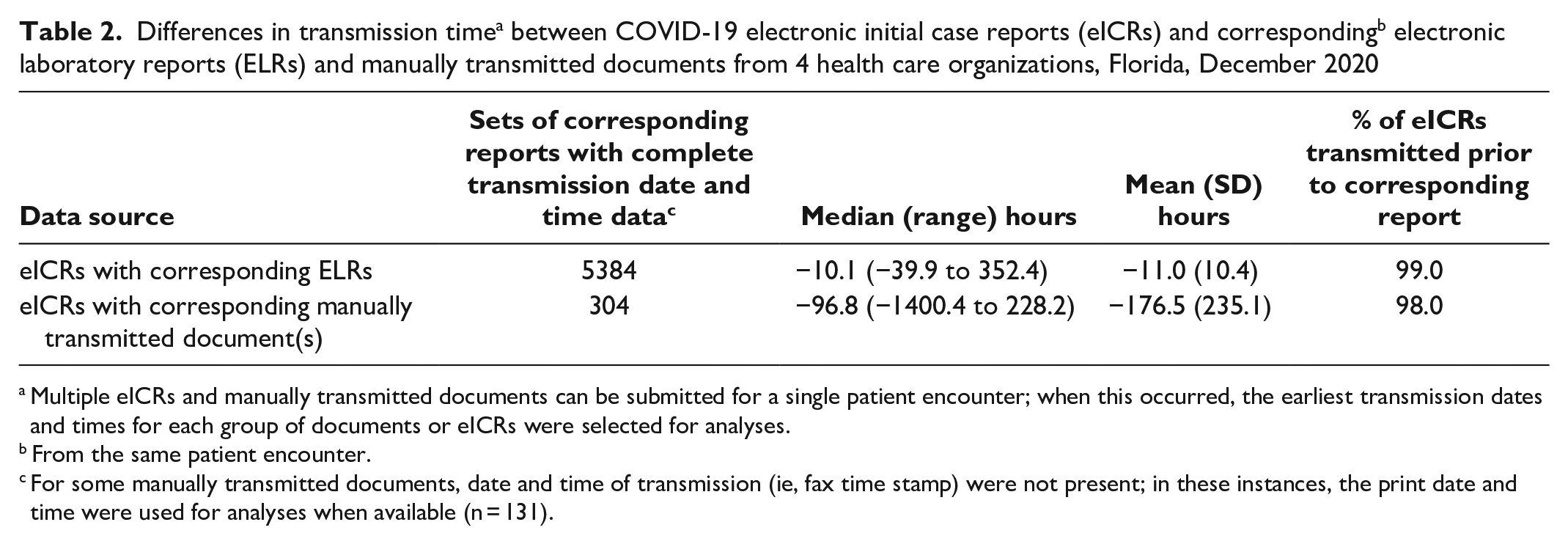
CDC received 10 814 eICRs, 49 991 ELRs, and 3367 manually transmitted documents and filtered them to select appropriate documents and reports for inclusion in this study (Figure). We stored files in secure, restricted-access folders to protect personally identifiable information. The study included files transmitted by 4 HCOs that implemented eCR for COVID-19, and 2 HCOs continued to provide manually transmitted documents in parallel with eICRs. Among the eICRs, ELRs, and documents received from FDOH, we selected only clinical documents, ELRs, and eICRs pertaining to patient encounters for COVID-19 that occurred in December 2020. We extracted data on patient and visit identifiers, race and ethnicity, and report transmission date and time from eICRs by XPath queries using Python 3.8.7 (Python). FDOH exported ELR data to comma-separated values files from the public health surveillance system. Three authors (L.C., G.M., and B.C.) and a group of trained data extractors extracted the same data twice by hand from 589 manually transmitted documents and then compared them using a multistep validation process to ensure data quality. 9 We cleaned and standardized data for consistency within and across sources.
Selection and matching of COVID-19 electronic initial case reports (eICRs), electronic laboratory reports (ELRs), and manually transmitted documents from 4 health care organizations, Florida, December 2020.
CDC reviewed this activity and determined that it was consistent with applicable federal law and CDC policy (45 CFR part 46, 21 CFR part 56; 42 USC §241[d]; 5 USC §552a; 44 USC §3501 et seq). Institutional review board determination was not required because the study did not involve research among human subjects and was conducted as part of public health surveillance.
We applied a multitiered algorithm that included patient and visit identifiers to match eICRs to corresponding ELRs or manually transmitted documents for the same patient encounter (Box; Figure). To determine completeness of data on race and ethnicity, we calculated the proportions of eICRs and corresponding ELRs and manually transmitted documents that contained valid (ie, values not missing or listed as unknown) information on race, ethnicity, and both race and ethnicity. Health Level Seven International (HL7) eCR and ELR implementation guides identify possible values for race and ethnicity.10,11 We calculated the proportions of records with “unknown” documented for race, ethnicity, and both race and ethnicity. In cases where multiple files were manually transmitted for a patient encounter, we extracted available race and ethnicity data from all files for analyses.
Algorithms used to match COVID-19 electronic initial case reports (eICRs) to electronic laboratory reports (ELRs) or manually transmitted documents.

We used date and time of transmission extracted from these files to determine the mean and median differences in transmission times between the eICRs and corresponding ELRs and between the eICRs and corresponding manually transmitted documents. In cases where multiple eICRs or multiple manually transmitted documents were transmitted for a patient encounter, the earliest transmission time for the eICRs and the earliest transmission time for the manually transmitted documents were used for analyses.
Results
For the 8558 eICRs received for COVID-19 in December 2020, data on race and ethnicity were present in 93.0% (n = 7955) and 95.0% (n = 8132) of eICRs, respectively (Table 1). Fewer than 1% of these eICRs contained a value of “unknown” for race or ethnicity. Of these eICRs, 5395 (63.0%) had corresponding ELRs and 339 (4.0%) had corresponding manually transmitted documents. For those matched to an eICR, 71.4% (n = 3860) of ELRs and 4.7% (n = 16) of manually transmitted documents contained data on race and ethnicity. In addition to values listed in HL7 implementation guides, combinations of listed values (eg, African American, Other) were present in eICRs. Manually transmitted documents contained the following values for race: Asian, Black, Black Hispanic, Black or African American, Caucasian, Other, White Hispanic, White, or Caucasian. Values present for ethnicity in eICRs and ELRs included Hispanic, Hispanic or Latino, and not Hispanic or Latino. Manually transmitted documents contained the following values for ethnicity: American, Ashkenazi Jewish, Caucasian, Hispanic, and non-Hispanic.
Completeness of race and ethnicity data in COVID-19 electronic initial case reports (eICRs), electronic laboratory reports (ELRs), and manually transmitted documents from 4 health care organizations, Florida, December 2020
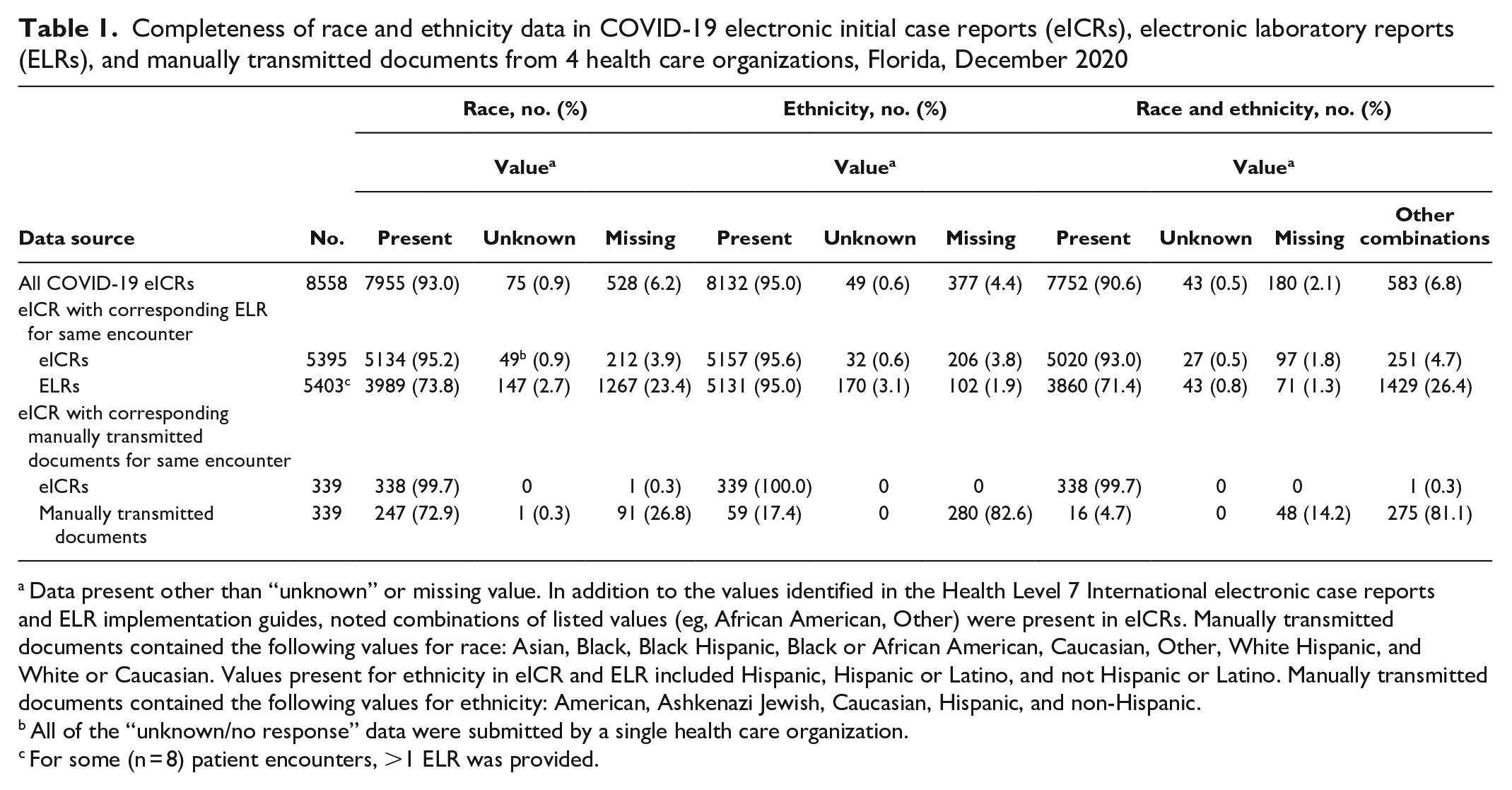
Data present other than “unknown” or missing value. In addition to the values identified in the Health Level 7 International electronic case reports and ELR implementation guides, noted combinations of listed values (eg, African American, Other) were present in eICRs. Manually transmitted documents contained the following values for race: Asian, Black, Black Hispanic, Black or African American, Caucasian, Other, White Hispanic, and White or Caucasian. Values present for ethnicity in eICR and ELR included Hispanic, Hispanic or Latino, and not Hispanic or Latino. Manually transmitted documents contained the following values for ethnicity: American, Ashkenazi Jewish, Caucasian, Hispanic, and non-Hispanic.
All of the “unknown/no response” data were submitted by a single health care organization.
For some (n = 8) patient encounters, >1 ELR was provided.
Most eICRs provided earlier notification to FDOH than corresponding ELRs or manually transmitted documents (Table 2). Overall, 99.0% of eICRs were transmitted before corresponding ELRs, with a mean difference in transmission time of 11.0 hours and a median difference in transmission time of 10.1 hours. The timeliness of ELR transmission differed by HCO, with the range of difference in transmission time between eICR and corresponding ELR of 35.9 hours (1.5 d) to 352.4 hours (14.7 d). Compared with manually transmitted documents, 98.0% of corresponding eICRs were transmitted earlier, with a mean difference in transmission time of 176.5 hours (7.4 d) and a median difference in transmission time of 96.8 hours (4.0 d).
Multiple eICRs and manually transmitted documents can be submitted for a single patient encounter; when this occurred, the earliest transmission dates and times for each group of documents or eICRs were selected for analyses.
From the same patient encounter.
For some manually transmitted documents, date and time of transmission (ie, fax time stamp) were not present; in these instances, the print date and time were used for analyses when available (n = 131).
Discussion
Multiple approaches to assessing completeness of public health data in EHRs and case reporting have been established.12,13 Comparisons of COVID-19–related eICRs to ELRs and manually transmitted documents for the same patient encounters in December 2020 at 4 HCOs in Florida provide evidence that eCR results in more complete information for race and ethnicity and earlier notification to public health agencies than ELRs or manually transmitted documents. The unique feature of this study was matching eICRs to corresponding reports and documents for direct comparison for the same patient encounter.
Extensive prior work identified data on race and ethnicity as important to the development and implementation of public health initiatives.14-17 The current study focused on completeness of race and ethnicity data in eCR. Although challenges to defining and operationalizing race and ethnicity in biomedical data have been identified and explored extensively,18,19 these constructs are still heavily used to explore trends and disparities in disease and health. Furthermore, missing data on race and ethnicity present a barrier to effective surveillance, resource allocation, and public health emergency response. Recent large-scale studies of COVID-19 case reports in 50 states demonstrated that data on race and ethnicity were missing from roughly one-third to almost one-half of case reports. 20 The growing body of evidence demonstrating racial and ethnic disparities in vaccination rates, cases of and severity of COVID-19 infections, and COVID-19 hospitalizations underscores the importance of availability of race and ethnicity information in case reports. 21 Modernizing case surveillance using eCR should lead to substantial improvement in data needed for case investigation and surveillance including race and ethnicity. 22
Timely availability of data is critical to effective public health surveillance and response activities. For example, receipt of patient information in the case report allowed public health officials to immediately engage in response activities such as contact tracing without having to first contact the health care provider for this information. Additionally, receipt of automated case reports allowed public health agencies to provide near–real-time accurate case counts, which were critical for decision makers when responding to the pandemic. Automating the submission of case reports reduced the time and effort of health care providers and allowed them to allocate resources that would otherwise have been dedicated to manual submission of case reports to other critical activities, such as direct patient care. In this study, almost all (99.0%) eICRs were transmitted earlier than their corresponding ELRs; the mean difference between corresponding reports was approximately 11 hours. Similarly, 98.0% of eICRs were transmitted prior to their corresponding manually transmitted documents, with eICRs providing notification an average of 176.5 hours (7.4 d) earlier.
The findings also indicated that timeliness of ELRs differed by HCO. These differences may be partially explained by laboratory facilities holding ELRs and transmitting them in batches at various time intervals (eg, every 6 or 12 h). Using information in the EHR captured as part of clinical workflow allows for reports of public health interest to be triggered instantaneously (eg, from predetermined triggers that could include problem lists, diagnoses, laboratory test orders, laboratory test results, medications, and vaccinations) and sent automatically at the time the data are saved into the patient’s EHR, providing information in near-real time to support surveillance and case investigation activities.
During the COVID-19 pandemic, rapid scale-up of eCR adoption resulted in the growth of eCR use from fewer than 200 health care facilities to more than 47 400 health care facilities as of November 13, 2024. 23 eCR now supports the reporting of 226 reportable conditions, including mpox, and public health agencies are working with HCOs to expand their eCR to include these conditions. 24 Recognizing the importance of achieving a nationwide capacity for reporting electronically to public health agencies, the Centers for Medicare & Medicaid Services included eCR as a required measure in its Promoting Interoperability Program regulations for hospitals, critical access hospitals, and Merit-based Incentive Payment System providers starting in January 2022. 25 This noted progress, coupled with findings presented in the present study, support what Mac Kenzie et al 26 described as the promise of nationwide eCR expansion for improving case surveillance in the United States. This progress includes (1) increasing the availability of quality and timely case information for public health action, (2) diminishing reporting burden for both health care and public health professionals, (3) offsetting costs and resources associated with manual reporting, and (4) providing an interoperable and scalable infrastructure that supports rapid reporting to improve outbreak detection and response, as well as supporting surveillance for other conditions such as opioid overdose and Parkinson’s disease.25,26 Moreover, eCR links efforts to support public health reporting requirements (eg, Reportable Conditions Knowledge Management System), 27 leverage a shared services platform used for public health surveillance (eg, Association of Public Health Laboratories’ Informatics Messaging Services platform), 28 and create a connected system that includes jurisdictional surveillance systems and vendor-provided EHR systems to support participation and enhance data available in the National Notifiable Diseases Surveillance System. 1
Limitations
Our findings were subject to at least 2 limitations. First, reports included in this study were from 4 HCOs in Florida and were limited to COVID-19 notifications; currently, more than 20 HCOs in Florida have implemented eCR. Therefore, these findings may not be representative of data in eICRs or timeliness of reporting for other HCOs or other jurisdictions, nor representative of findings for other reportable conditions. In particular, HCOs that use eCR may be even less likely than other HCOs to prioritize the transmission of timely or accurate data by other means (ie, ELR or manually transmitted documents). Second, data extracted and used for analyses were limited to a subset of those data elements that were automatically processed into the FDOH surveillance system: race, ethnicity, and transmission date and time. Analyses of other data variables could provide a more robust understanding of other aspects of data quality in eCR, including how well eCR supports data needed for complex diseases or conditions being reported.
Conclusion
eCR provides data identified by public health agencies as needed across all reportable conditions. 3 These elements include data related to patient identity, demographic characteristics, and contact information; health care provider identity, facility, encounter type, and date; and clinical data such as symptoms, diagnoses, risk factors, laboratory tests, and medications. Furthermore, data transmitted to public health agencies through eCR are limited to data that are present in the EHR at the time of transmission. Of note, eCR is not intended to capture all condition-specific data necessary for investigation and surveillance activities for reportable conditions. Future evaluation efforts should seek to understand the usefulness of data captured through eCR for public health agencies.
The findings of our study suggest that eCR can result in more complete data on race and ethnicity and earlier transmission of reports to support public health surveillance and response. In addition, automating the electronic submission of case reports promises to reduce the burden on health care providers to report to public health agencies. We are still in the early stages of adoption and use of eCR for disease reporting to public health agencies. However, through increased availability of race and ethnicity data and faster reporting, data modernization through eCR contributes to a more comprehensive understanding of the incidence and prevalence of reportable conditions, supports the identification and addressing of salient health disparities in a more intentional and timely manner, and informs timely, data-driven decision-making.
Footnotes
Acknowledgements
The authors acknowledge the following people for their contributions to this research: Sachin Agnihotri, MS (Centers for Disease Control and Prevention [CDC]); Guillermo Avila (Chenega Professional and Technical Services LLC); Katherine Bruss, PsyD (CDC); Gregory Danyluk, MPH (Florida Department of Health [FDOH]); Leslie Dauphin, PhD (CDC); Joyce Dieterly, MPH (Strategic Innovation Solutions); Cory Ferdon, PhD (CDC); Amalie Frank, MS (FDOH); Sarah Gaunt (Lantana Consulting Group); Sudevi Ghosh (CDC); Kashaine Gray, MPH (CDC); Adiseshlu Gundlapalli, MD, PhD (CDC); Charles Heilig, PhD (CDC); Reynald Jean, MD, MPH (FDOH); Bernhard Kloppenburg, MPH (FDOH); Kimberly Knicely, PhD (CDC); Scott Lee, PhD (CDC); Adrean Mabry (CDC); Grace Mandel, MPH (CDC); Valerie Marshall, MA (The Evaluation Center at Western Michigan University); David Nitschke (CDC); Jill Raudabaugh, MPH (CDC); Preeti Ravindhran, MPH (CDC); Stephanie Shaw, MBA (CDC); Edhelene Rico, MPH (FDOH); Sarah Sobonya, PhD (CDC); Hassan Swann (Chenega Professional and Technical Services LLC); Taraesa Toney (Booz Allen Hamilton); Robin Tracy (Chenega Professional and Technical Services LLC); Thomas Troelstrup, MPH (FDOH); Abbigail Tumpey, MPH (CDC); Bradley Watts, PhD (The Evaluation Center at Western Michigan University); Alexander Whipp, MS (Booz Allen Hamilton); Lori Wingate, PhD (The Evaluation Center at Western Michigan University); and colleagues from the CDC Foundation.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by CDC to the Association of Public Health Laboratories (cooperative agreement no. OE20-2001) and to the Florida Department of Health (cooperative agreement no. CDC-RFA-CK19-1904). Contractual funding was provided by CDC for communications, technical writing, and business analytical support to Chenega Professional & Technical Services LLC (contract no. 75D30120C07723, HCPN1-2020-44296, and 200-2017-94367). Health information systems support was provided by Booz Allen Hamilton as part of a contract with CDC’s National Center for Emerging and Zoonotic Infectious Diseases (contract no. 75D301-20-F-07161).