Abstract
Objective:
The objective of this study was to describe the capture-recapture method used by the Philadelphia Department of Public Health to enhance surveillance of perinatal hepatitis B virus (HBV), report on results and limitations of the process, and determine why some HBV-positive mother-infant pairs were not initially identified by Philadelphia’s Perinatal Hepatitis B Prevention Program (PHBPP).
Methods:
We performed capture-recapture retrospectively for births in 2008 and 2009 in Philadelphia and prospectively for births from 2010 to 2014 by independently matching annual birth certificate data to PHBPP and HBV surveillance data. We compared the number of HBV-positive mother-infant pairs identified each year to the point estimates and lower-limit estimates calculated by the Centers for Disease Control and Prevention for the Philadelphia PHBPP.
Results:
Of 156 605 pregnancy outcomes identified between 2008 and 2014, we found 1549 HBV-positive mother-infant pairs. Of 705 pairs that were initially determined, 358 (50.7%) were confirmed to be previously unidentified HBV-positive pairs. Reasons for failing to identify these mother-infant pairs prior to capture-recapture included internal administrative issues (n = 191, 53.4%), HBV testing and reporting issues (n = 92, 25.7%), and being lost to follow-up (n = 75, 20.9%). Each year that capture-recapture was used, the number of pairs identified by the Philadelphia PHBPP exceeded the Centers for Disease Control and Prevention’s lower-limit estimates for HBV-positive mother-infant pairs.
Conclusions:
Capture-recapture was useful for identifying HBV-positive pregnant women for Philadelphia’s PHBPP and for highlighting inadequacies in health department protocols and HBV testing during pregnancy. Other health departments with access to similar data sources and staff members with the necessary technical skills can adapt this method.
Keywords
In the absence of postpartum prophylaxis, >40% of infants born to women with hepatitis B virus (HBV) become infected with the virus. 1,2 Most perinatal HBV infections result in chronic disease, which is associated with progressive liver disease, liver cancer, and a 25% increased risk of premature death in children and adults. 3,4 To reduce perinatal HBV transmission in the United States, the Centers for Disease Control and Prevention (CDC) has provided funding for all state health departments and several city health departments since 1990 to maintain Perinatal Hepatitis B Prevention Programs (PHBPPs) in their jurisdictions. 5 A goal of PHBPPs is to identify HBV-positive pregnant women and ensure that their exposed infants receive timely postpartum prophylaxis with hepatitis B immunoglobulin, the HBV vaccine series, and postvaccination serologic testing. 5 PHBPPs are cost-effective, contribute to a substantial reduction in perinatal HBV transmission, 5,6 and play an important role in reducing the overall rate of new HBV infections in the United States.
Annually, CDC estimates the expected burden of HBV-positive mother-infant pairs for each PHBPP jurisdiction, based on the racial/ethnic distribution of childbearing women and the known hepatitis B surface antigen (HBsAg) seroprevalence of each racial/ethnic group in that jurisdiction. 5 CDC provides these burden values to each jurisdiction in the form of point estimates (target number of pairs to be identified) and lower-limit estimates. Many jurisdictions are hindered in their ability to identify and manage all cases of HBV-positive pregnant women by issues such as underreporting of HBV test results and inadequate prenatal testing for HBV. Also, although prenatal testing for HBsAg is recommended for all pregnant women, 7 fewer than half of the positive HBV test results for pregnant women in the United States are promptly reported to health departments and entered into their PHBPP. 5,8
In Philadelphia, Pennsylvania, women with HBV infection are primarily identified by the Philadelphia Department of Public Health (PDPH) from routine laboratory-based reporting, but these reports rarely include pregnancy status. If data on pregnancy status are missing, PDPH staff members contact health care providers and/or patients to ascertain the pregnancy status of any woman aged 14-44 for whom a new positive HBV laboratory test has been reported. It is important to ascertain pregnancy status because women of childbearing age constitute approximately 25% of all positive HBV laboratory reports to PDPH each year. PDPH also identifies some HBV-infected women via their prenatal health care providers, who occasionally alert PDPH about HBV-positive pregnant women. These identification methods rely on universal routine prenatal testing for HBsAg, which is not always achieved.
From 2005 to 2007, the number of HBV-positive pregnant women identified in Philadelphia decreased from 138 to 116, whereas the 2007 CDC point estimates and lower-limit estimates for Philadelphia were 218 and 142, respectively. To reduce the discrepancies between the expected and observed numbers of HBV-positive pregnant women identified in Philadelphia, PDPH devised a novel method—capture-recapture—to identify pregnant women with HBV infection who had not been reported to PDPH or whose data had not been entered into its PHBPP. Some epidemiologists have advocated the use of capture-recapture methods to assess the completeness of population size estimates, noting that they offer the potential to reduce the costs of evaluating disease burden and the bias in estimates of disease incidence. 9 In this case, PDPH performed capture-recapture by matching data from birth certificates, the HBV Surveillance Registry, and PHBPP records.
The objective of this study was to describe the capture-recapture method used by PDPH, report on the results and limitations of the process, and determine why some HBV-positive mother-infant pairs were not initially identified by Philadelphia’s PHBPP. We hypothesized that the capture-recapture method would increase the identification of HBV-positive women who were pregnant or who recently gave birth and were not previously identified by PHBPP.
Methods
We performed capture-recapture retrospectively for births in 2008 and 2009 and prospectively, on an annual or biannual basis, for births from 2010 to 2015. We compared the number of mother-infant pairs identified each year with the point estimates and lower-limit estimates calculated by CDC for the Philadelphia PHBPP. The PDPH Institutional Review Board reviewed and approved this analysis.
Data Sources
We obtained data for this study from 3 sources: the PHBPP database, the HBV Surveillance Registry database, and electronic birth certificates. We used the PHBPP database for information about mother-infant pairs residing in Philadelphia and tracked by PHBPP between 2008 and 2014. The PHBPP database contained HBV laboratory testing results, infant vaccination information, and records related to the completion of PHBPP case management. Mother-infant pairs who had not been identified by PDPH were not included in the database.
The HBV Surveillance Registry database includes laboratory testing and surveillance-related information on Philadelphia residents. We used data reported from 2001 to 2015 on women who had tested positive for HBsAg, hepatitis B envelope antigen, anti-HBe antibody, anti-HBs antibody IgM, HBV DNA, or HBV genotype. When available, we also used negative test results for our analysis.
Electronic birth certificates provided information about births to Philadelphia residents from 2008 to 2014 (unpublished data, PDPH, 2008-2014). For our analysis, we counted nonsingleton births only once.
Data Matching
We independently matched annual birth certificate data to HBV surveillance data and then to PHBPP data. We performed the matches using maternal date of birth, name, address, and maiden name. We used the SAS 10 SPEDIS (spelling distance) function to select records, and we manually confirmed that the records were appropriately matched. We also made some additional matches through manual review. When we matched these records to PHBPP data, we were able to identify some mother-infant pairs that were already known to PHBPP. Birth certificates that matched to HBV surveillance data but not to PHBPP data were considered to represent mother-infant pairs that were newly identified by the capture-recapture method.
Capture-Recapture Confirmation
We confirmed a mother-infant pair that was newly identified through capture-recapture as an additional PHBPP case if the mother had ≥2 positive HBV tests indicative of current infection (ie, HBsAg, hepatitis B envelope antigen, HBV DNA, HBV genotype) prior to delivery with ≥6 months apart or 1 positive HBV test result during pregnancy with no subsequent negative HBsAg results. For HBV-positive women who had not had laboratory test results reported to PDPH during their pregnancy, we obtained those test results through medical chart abstraction from prenatal, delivery, and infant medical records, and we included those results in the confirmation assessment. We excluded from the final PHBPP count women with negative or nondefinitive HBV test results during pregnancy.
Missed Identification
We determined the reasons why mother-infant pairs had not been identified by PHBPP prior to capture-recapture. We identified the reasons retrospectively after completing the matching process. We performed database matching on a single occasion for the 2008 and 2009 births and subsequently annually. We organized the reasons for missed identification into internal PDPH issues and external health care provider-based issues.
On a sporadic and ongoing basis, PDPH used this information to develop strategies and adapt existing protocols to improve PHBPP identification methods. PDPH initiated meetings to improve interdepartmental communications in 2010, began altering protocols in 2011, and optimized the PHBPP database beginning in 2015. We reassessed the reasons for missed identification once per year to determine whether the PDPH responses resulted in changes in these reasons.
Statistical Methods
We used Fisher exact χ2 testing to assess the changes in internal reasons for missed identification after PDPH implemented improvement strategies. We used the Welch t test to assess the impact of capture-recapture on the PHBPP population. For all analyses, we considered P < .05 to be statistically significant. We performed data extraction, manipulation, and analyses using SAS. 10
Results
During 2008 to 2014, a total of 156 605 pregnancy outcomes among Philadelphia residents were identified in birth certificates. Of these, 1552 births occurred to women who were among the 7429 with a positive HBV laboratory test captured by HBV surveillance during 2001 to 2015 (Figure 1). The PHBPP program had already identified 844 of the 1552 births (54.4%) using traditional methods, leaving 708 previously unidentified mother-infant pairs. We attributed their identification to the capture-recapture method.
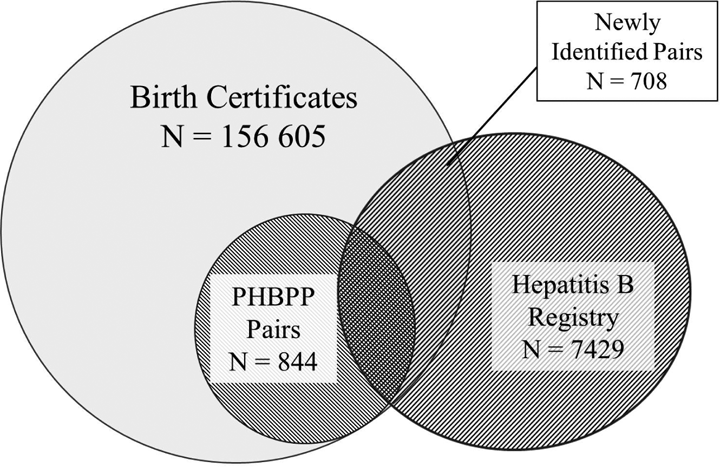
Previously unknown hepatitis B virus–positive mother-infant pairs newly identified through capture-recapture methods that matched birth certificates, PHBPP data, and Hepatitis B Surveillance Registry data, Philadelphia, Pennsylvania, 2008-2014. The scale in the figure is not proportional to the data. The Hepatitis B Surveillance Registry contained data for 2001-2015. Abbreviation: PHBPP, Perinatal Hepatitis B Prevention Program.
Using our confirmation definition, we found that 358 of the 708 mother-infant pairs (50.6%) identified through capture-recapture were newly identified PHBPP cases. The remaining 350 (49.4%) mother-infant pairs were not confirmed as PHBPP cases, because a subsequent negative HBsAg test was discovered, HBV testing was inadequate (eg, anti-HBe antibody only), no HBV testing was performed during pregnancy and previous testing had not demonstrated that the mother was chronically infected, or HBV testing was performed >6 months after birth.
We identified several reasons why the confirmed maternal HBV-positive capture-recapture mother-infant pairs were not initially identified by PHBPP. The most common reason was internal administrative error, which accounted for 191 of the 358 missed opportunities (53.4%) for identification. These errors included failures in protocol execution or in clerical procedures (eg, delayed referral of pregnant mothers to PHBPP from other PDPH departments). Other explanations for missed identification included mothers who had no or inadequate HBV testing during pregnancy (n = 81, 22.6%), mother-infant pairs lost to follow-up (n = 75, 20.9%), and mothers whose pregnancies and/or HBV laboratory results were not reported (n = 11, 3.1%).
After we determined the internal issues, PDPH made improvements to its HBV identification strategies, including altering existing protocols for mother-infant pair identification, improving interdepartmental communications and referrals of identified mother-infant pairs, and enhancing database use (by incorporating PHBPP into a more robust data system 11 that enabled a more streamlined and automated consolidation of data). These changes led to a proportional decline in these administrative errors, from 77.6% (52 of 67 missed identifications) in 2008 to 43.1% (25 of 58 missed identifications) in 2014 (P < .001).
Implementation of the capture-recapture method in 2008 resulted in an increase in the total annual mean number of maternal HBV-positive mother-infant pairs identified, from 123 per year during 2004-2007 to 160 per year during 2008-2014 (P = .002), a mean increase of 37 pairs identified per year (Figure 2). Because of this increase, PDPH exceeded its year-specific CDC lower-limit estimates for the expected burden of maternal HBV-positive mother-infant pairs by a mean of 35.3 (range, 11.0 to 51.0) during each year that capture-recapture was used (Figure 2).
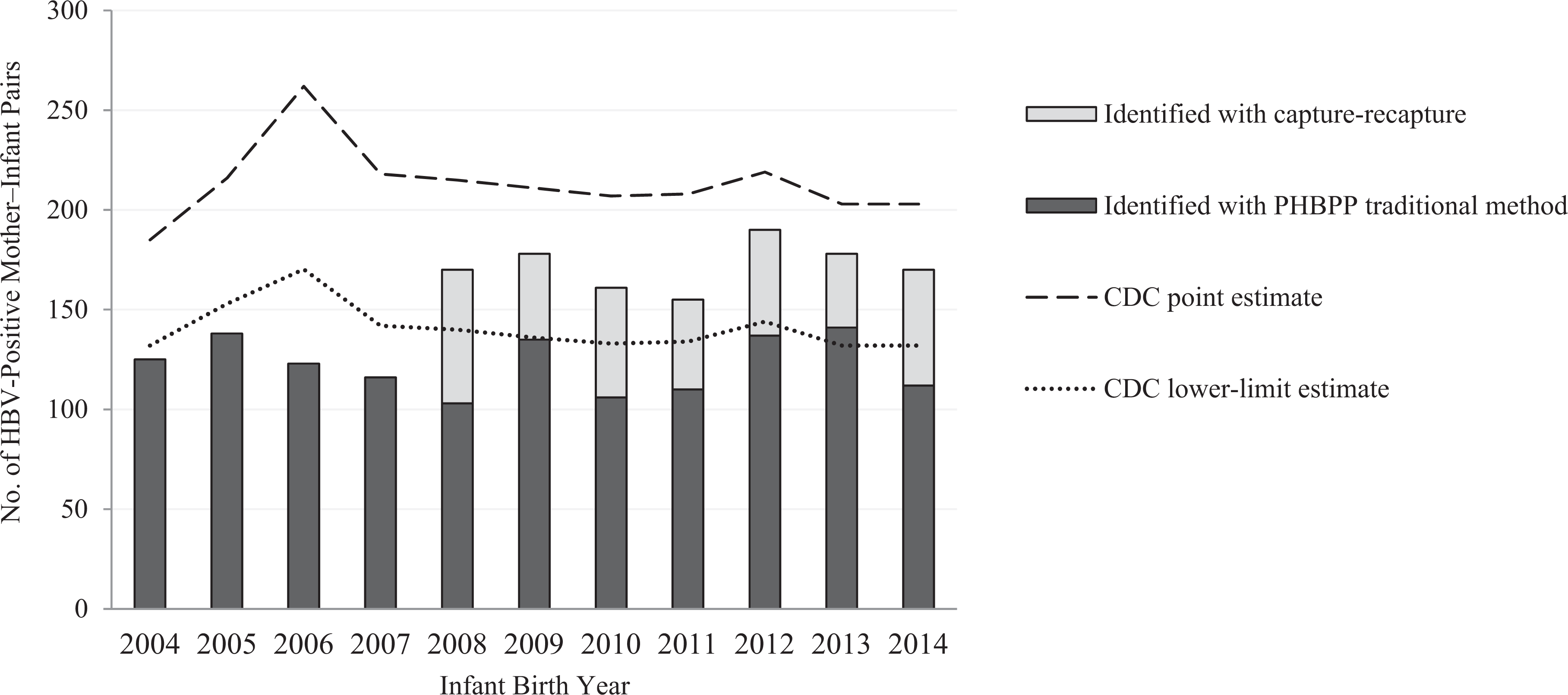
Annual incidence of HBV-positive mother-infant pairs, identified by PHBPP traditional and capture-recapture methods and compared with CDC point estimates and lower-limit estimates, Philadelphia, Pennsylvania, 2004-2014. Point (target) and lower-limit estimates of the expected number of HBV-positive pregnancies, based on racial/ethnic population distribution in the jurisdiction, were calculated annually by CDC for each PHBPP site. CDC estimates for 2013 were used for 2014 because 2014 estimates were not available. Data were obtained from birth certificates, the Philadelphia Hepatitis B Surveillance Registry, and unpublished PHBPP case management data. Abbreviations: CDC, Centers for Disease Control and Prevention; HBV, hepatitis B virus; PHBPP, Perinatal Hepatitis B Prevention Program.
Discussion
The capture-recapture method described in this study is a novel and effective way to identify HBV-positive women and their infants who were previously unrecognized by health departments. During the 7 years that this method was used, PDPH successfully identified and managed the cases of hundreds of additional mother-infant pairs that would otherwise have been undiscovered. In addition, use of the capture-recapture method helped PDPH detect and characterize internal and external issues that prevented the earlier identification of HBV-positive pregnant mothers, including delayed referral of HBV-positive pregnant mothers to PHBPP by other PDPH programs, poor or delayed laboratory results reporting, and inadequate HBV testing of pregnant mothers. Identification of these issues allowed PDPH to make improvements in its processes and activities related to HBV-positive mother-infant pair identification.
With this information in mind, PDPH initiated several new strategies (outlined in the Methods section) to help identify HBV-positive pregnant mothers prior to delivery, which enabled more complete and timely postexposure prophylaxis for their infants. For example, PDPH migrated its PHBPP database into its HBV Surveillance Registry, which reduced delays in referrals of HBV-positive pregnant mothers to PHBPP from other PDPH departments. PDPH also worked with a CDC Division of Viral Hepatitis workgroup to encourage large reference laboratories to include pregnancy flags on electronic laboratory reports whenever positive maternal prenatal HBsAg test results were found. As more laboratories reported pregnancy status, women were identified earlier for case management, and PDPH relied less on PDPH staff members to find this information. In addition, PDPH’s CDC-funded enhanced viral hepatitis surveillance program, which conducts detailed investigations of approximately half of all newly reported HBV cases, identified additional HBV-positive pregnant women. Because many of these strategies are helping to identify more women and are identifying them earlier in their pregnancies than they are through capture-recapture and traditional methods, we expect that capture-recapture may identify a smaller proportion of HBV-positive pregnant women over time.
During the study period, PDPH found external issues that prevented the earlier identification of HBV-positive pregnant mothers. For example, a finding that was especially true for women known to be chronically HBV infected was that not all health care providers ordered an HBsAg test for every pregnant woman. PDPH used these findings to reach out to health care providers in its jurisdiction, to urge that HBV laboratory testing be ordered for every pregnancy, and to encourage the reporting of each pregnancy to PDPH.
Several resources are needed to perform the capture-recapture method, including electronic access to birth records and HBV Surveillance Registry data, staff members qualified to use the software, and staff members who can perform chart reviews. Although the initial matching to identify mother-infant pairs is not time intensive when the appropriate technical support is available, confirming that the mother is HBV positive requires manual chart review, which can be a prolonged process. The staff time required to initially review information about each mother-infant pair was usually several minutes. Furthermore, when necessary, medical chart review often took 15 to 30 minutes per mother-child pair. Because of delays in obtaining birth charts and laboratory results, however, medical chart review could take several months to complete.
Nevertheless, use of the capture-recapture method is not costly and is feasible for any local or state health department with access to the appropriate resources.
Given that the yield for case finding in this study was so high, with >50% of identified mother-infant pairs confirmed to be maternal HBV positive, capture-recapture has the potential to substantially improve the ability of health departments and PHBPPs to identify HBV-positive mothers and their infants. Alternatively, the capture-recapture method could be an effective and low-resource backup method for HBV-positive mother-infant pairs not previously identified through other methods. Also, because the capture-recapture method tends to identify reasons why mother-infant pairs have been missed, the more frequent application of the method may improve the timeliness of identifying these pairs, the appropriate treatment of mothers, and the necessary prophylaxis for their infants.
Limitations
The method used in this study had several limitations. Although reporting of positive HBV laboratory results is mandatory, some positive test results in pregnant women were not reported to PDPH; as such, these women would not have been identified through capture-recapture. However, because an increasing majority of laboratory reports are now delivered to PDPH electronically, we believe that this scenario was likely uncommon. In addition, some of our data suggest that inadequate HBV testing by health care providers did occur and that inadequate testing might have occurred at greater rates than found by the capture-recapture process. If so, some women may have had undiagnosed HBV infection or had HBV infection but incomplete testing, leaving them disqualified from PHBPP case management. Finally, the case definition that we used for inclusion of mother-infant pairs may have excluded some mothers who were HBV infected. However, the intensive chart reviews and investigations of laboratory results conducted for excluded cases would likely have identified most pregnant women with HBV infection; as such, the impact of the case definition on identifying HBV-infected mothers was likely minimal.
Conclusion
Capture-recapture was a useful tool for identifying HBV-positive pregnant women for a PHBPP, as well as for highlighting inadequacies in health department protocols and HBV testing during pregnancy. The capture-recapture method can be adapted by other health departments with access to similar data sources and to staff members with the necessary technical skills. For state and local health departments with funding restrictions, use of capture-recapture could improve identification of infants born to HBV-positive women, ensuring that more mother-infant pairs receive case management and appropriate HBV-related care.
Footnotes
Acknowledgments
We acknowledge the work of the PDPH Acute Communicable Disease and Immunization Programs in the handling and funding of PHBPP, specifically John Faherty, James Lutz, Dr Ami Patel, Fan Zhang, and Valerie Riis.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Centers for Disease Control and Prevention (CDC) cooperative agreement #1H23IP000735. The content of this article is the sole responsibility of the authors and does not necessarily represent the official views of CDC.
