Abstract
Objectives:
In the United States, universal screening for latent tuberculosis (TB) infection among people with HIV is recommended, but the percentage receiving screening is unknown. This study assessed screening for latent TB infection among people with HIV enrolled in Medicaid during 2006-2010.
Methods:
We used nationwide fee-for-service Medicaid records to identify people with HIV, measure screening for latent TB infection, and examine associated demographic, social, and clinical factors. We used logistic regression analysis to calculate odds ratios (ORs) and 95% confidence intervals (CIs). We created 2 multivariate models to prevent collinearity between variables for length of HIV infection.
Results:
Of 152 831 people with HIV, 26 239 (17.2%) were screened for latent TB infection. The factor most strongly associated with screening was TB exposure or suspected TB (OR = 3.78; 95% CI, 3.27-4.37). Significant demographic characteristics associated with screening included being African American (OR = 1.28; 95% CI, 1.24-1.32) or ≤20 years of age (OR = 1.35; 95% CI, 1.28-1.42). Significant clinical and social factors associated with screening included poor housing conditions, low body mass index, chemotherapy treatment, and use of certain substances (ORs ranged from 1.24 [95% CI, 1.20-1.27] to 1.47 [95% CI, 1.22-1.76]). The screening rate for latent TB infection was higher among people with newly diagnosed HIV infection than among those with established infection (OR = 1.37; 95% CI, 1.32-1.41) and among people with a longer established HIV infection than among those with shorter HIV infection (OR = 1.24; 95% CI, 1.23-1.26 for each additional year).
Conclusion:
Screening for latent TB infection among fee-for-service Medicaid beneficiaries with HIV was suboptimal, despite the presence of demographic, social, or clinical characteristics that should have increased the likelihood of screening. The lack of certain data in Medicaid may have resulted in an underestimation of screening.
Keywords
During the 1980s and 1990s, infection with HIV contributed to increases in the number of active cases of tuberculosis (TB) in the United States. For people living with HIV, the dangers of TB coinfection are well known, including faster HIV disease progression, greater severity of infection, and increased mortality. 1 –3 Moreover, for people with TB, HIV infection can cause latent TB infection to progress to active TB much faster than among HIV-negative people. 4 –7 More recently, data from 2015 have shown that the historic decrease in active TB cases in the United States has stalled. This has caused public health experts to give renewed attention to all populations at increased risk of progression from latent TB infection to active TB, including people infected with HIV. 8
Most cases of active TB in the United States are among people born outside of the country and among them, active TB is caused primarily by reactivation of latent TB infection. 8 Modeling studies show that TB will not be eliminated in the United States unless treatment increases among people with latent TB infection. 9 US Preventive Services Task Force guidelines recommend screening for latent TB infection among asymptomatic adults at increased risk for infection. 10
Screening for both active and latent TB infections in people with HIV prevents morbidity and is cost effective. 11,12 Because of the association between HIV and TB infection, the Centers for Disease Control and Prevention (CDC) and the Infectious Diseases Society of America have recommended since 1998 that all people with newly diagnosed HIV infection should be screened for TB and that those determined to have active or latent TB infection should be treated. 13 –16
The percentage of people living with HIV in the United States who are screened for latent TB infection is unknown, except for clinics participating in the Ryan White HIV/AIDS Program, a federal grant system that funds HIV care to uninsured or underinsured people with HIV. 1 Clinics participating in the Ryan White HIV/AIDS Program use program performance measures to assess the quality of care among grantees, and one possible performance measure is screening for latent TB infection. Outside of grantees in the Ryan White HIV/AIDS Program, the amount of screening and factors that might influence practitioners to screen people with HIV for latent TB infection are not well understood. We examined the frequency of screening for latent TB infection as well as factors associated with screening among people with HIV enrolled in fee-for-service Medicaid programs. Medicaid is among the largest providers of health care for people with HIV in the United States, covering 44% of people with HIV and an estimated 22% of people at the time of their HIV diagnosis. 18,19
Methods
We analyzed data from all fee-for-service Medicaid claims of beneficiaries who met inclusion criteria during a 5-year period, from January 1, 2006, through December 31, 2010, for all 50 states and the District of Columbia (n = 25 304 108). To be included in our study, a beneficiary had to meet the following criteria: (1) enrolled in Medicaid fee-for-service insurance; (2) enrolled in the same state for ≥24 consecutive months (or until death) between January 1, 2006, and December 31, 2010; (3) had 1 or more diagnosis codes for HIV; and (4) had both a date of HIV diagnosis and a date on which he or she either (a) was no longer enrolled in Medicaid, (b) died, or (c) reached the end of the study period. The data used in this study were de-identified by the Centers for Medicare & Medicaid Services and were subject to a data-use agreement between CDC and the Centers for Medicare & Medicaid Services. As such, CDC deemed the study exempt from institutional review board review.
We based HIV diagnosis on the presence of a diagnosis code from the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) or a Medicare Severity Diagnosis Related Group code for HIV or AIDS 20,21 (Table 1). We considered the following to indicate an HIV diagnosis: (1) the presence of 1 claim with a diagnosis code from the inpatient files or long-term care files or (2) the presence of 2 claims on different days from the other therapy file (a file that contains claims for various services, including clinic visits and hospital outpatient claims). This same method has been used by other researchers examining administrative data for people with HIV. 24,25 In this study, we referred to beneficiaries whose files contained these codes as people with HIV.
Codes and coding systems used to determine diagnosis with HIV, screening for HIV, and screening for latent TB infection among people living with HIV who were screened or not screened for latent TB infection in Medicaid, 2006-2010a

Abbreviations: CPT-4, Current Procedural Terminology, Fourth Edition; DRG, Diagnosis Related Group; HCPCS, Healthcare Common Procedure Coding System; ICD-9-CM, International Classification of Diseases, Ninth Revision, Clinical Modification; TB, tuberculosis.
We defined screening for latent TB infection after HIV diagnosis by the presence of a procedure code for latent TB infection screening 1 or more days after the earliest available HIV diagnosis code. The presence of a code from the Current Procedural Terminology, Fourth Edition, 22 or an ICD-9-CM procedure code for either an interferon-gamma release assay or a tuberculin skin test indicated screening for latent TB infection (Table 1).
To understand factors that might influence the screening of people with HIV for latent TB infection, we examined the following beneficiary characteristics: race/ethnicity, sex, age, residence in the 5 states with the largest number of people with HIV (California, Florida, Illinois, New York, and North Carolina), 26 as well as the presence of an ICD-9-CM code indicating the following: living in poor housing conditions or in a noncorrectional residential institution; having a low body mass index, suspected TB or TB exposure, a personal history of TB, cell-mediated immunity disorder, certain autoimune diseases, renal/kidney disease, diabetes, pneumoconiosis, or sarcoidosis; using certain substances (including alcohol, drugs, and current/past tobacco); and receiving chemotherapy treatment (Table 2). We chose these factors because they increase the likelihood of acquiring TB (eg, homelessness, communal housing, exposure to TB), increase the likelihood of transitioning from latent to active TB infection (eg, reduced cell-mediated immunity, immunosupressive medication, personal history of TB), or are associated with more severe TB (eg, low body mass index, tobacco use, drug use). 27,28
ICD-9-CM codes used to identify people with clinical and social conditions that are associated with TB infection among people living with HIV who were screened or not screened for latent TB infection in Medicaid, 2006-2010a

Abbreviations: ICD-9-CM, International Classification of Diseases, Ninth Revision, Clinical Modification; TB, tuberculosis.
a Data source: National Center for Health Statistics. 20
To assess whether the duration of HIV infection was associated with screening for latent TB infection, we distinguished people with newly diagnosed HIV infection from people with established HIV infection. We deemed people to have newly diagnosed HIV infection if they had an HIV screening code up to 90 days before their first recorded HIV diagnosis code during the study period. HIV screening codes included codes from Current Procedural Terminology, Fourth Edition, and codes from the Healthcare Common Procedure Coding System for either HIV antigen or antibody tests (Table 1). 23 We chose a 90-day interval to adhere to the recommendations for entry into care for people with newly diagnosed HIV infection, as well as the linkage-to-care performance measure used by the HIV/AIDS Bureau. 17,29,30 We also examined whether the duration of time as a Medicaid beneficiary with documented HIV infection influenced screening for latent TB infection. We calculated the documented length of HIV infection as the amount of time between a beneficiary’s first HIV diagnosis code and the date on which he or she (1) was no longer enrolled in Medicaid, (2) died, or (3) reached the end of the study period. To account for possibly nonlinear time trends, we included in the models both the length of documented HIV infection in years and the length of documented HIV infection in years squared.
Statistical Analysis
We used logistic regression analysis to examine differences between people with HIV who were screened for latent TB infection and those who were not screened. We used bivariate and multivariate models with accompanying odds ratios (ORs) and 95% confidence intervals (CIs) to obtain the associations between screening for latent TB infection and factors of interest. Given the large sample size of this study, we retained all variables examined in bivariate models in multivariate models to prevent overreliance on P values, and we considered P < .05 to be significant. We ran 2 separate multivariate models to prevent collinearity between the 2 variables for length of HIV infection. We conducted all statistical analyses by using SAS version 9.4. 31
Results
For the years 2006-2010, a total of 152 831 eligible Medicaid beneficiaries had 1 or more diagnosis codes indicating HIV infection, of whom 26 239 (17.2%) were screened 1 or more times for latent TB infection, and 126 592 (82.8%) had no record of screening for latent TB infection. All screenings for latent TB infection occurred in outpatient settings. Each beneficiary contributed a mean of 3.5 (standard deviation [SD] ±1.4) years to the study.
People with HIV who were screened for latent TB infection had higher odds of being ≤20 years of age (OR = 1.35; 95% CI, 1.28-1.42) and lower odds of being 45-64 years of age (OR = 0.79; 95% CI, 0.77-0.81) or ≥65 (OR = 0.16; 95% CI, 0.14-0.19) compared with those who were not screened. People with HIV who were screened had higher odds of being non-Hispanic African American (OR = 1.28; 95% CI, 1.24-1.32) or non-Hispanic American Indian/Alaska Native (OR = 1.30; 95% CI, 1.10-1.53) and lower odds of being Hispanic (OR = 0.80; 95% CI, 0.76-0.84) compared with those who were not screened. People with HIV who were screened also had lower odds of being male (OR = 0.74; 95% CI, 0.71-0.76) and lower odds of death during the study period (OR = 0.25; 95% CI, 0.23-0.27) compared with those who were not screened. People with HIV who were screened had lower odds of living in New York State (OR = 0.27; 95% CI, 0.26-0.28) and higher odds of living in California (OR = 1.07; 95% CI, 1.03-1.12), Florida (OR = 1.22; 95% CI, 1.17-1.27), or Illinois (OR = 1.32; 95% CI, 1.25-1.40) compared with those who were not screened (Table 3).
Demographic characteristics of people living with HIV who were screened or not screened for latent TB infection in Medicaid, 2006-2010a
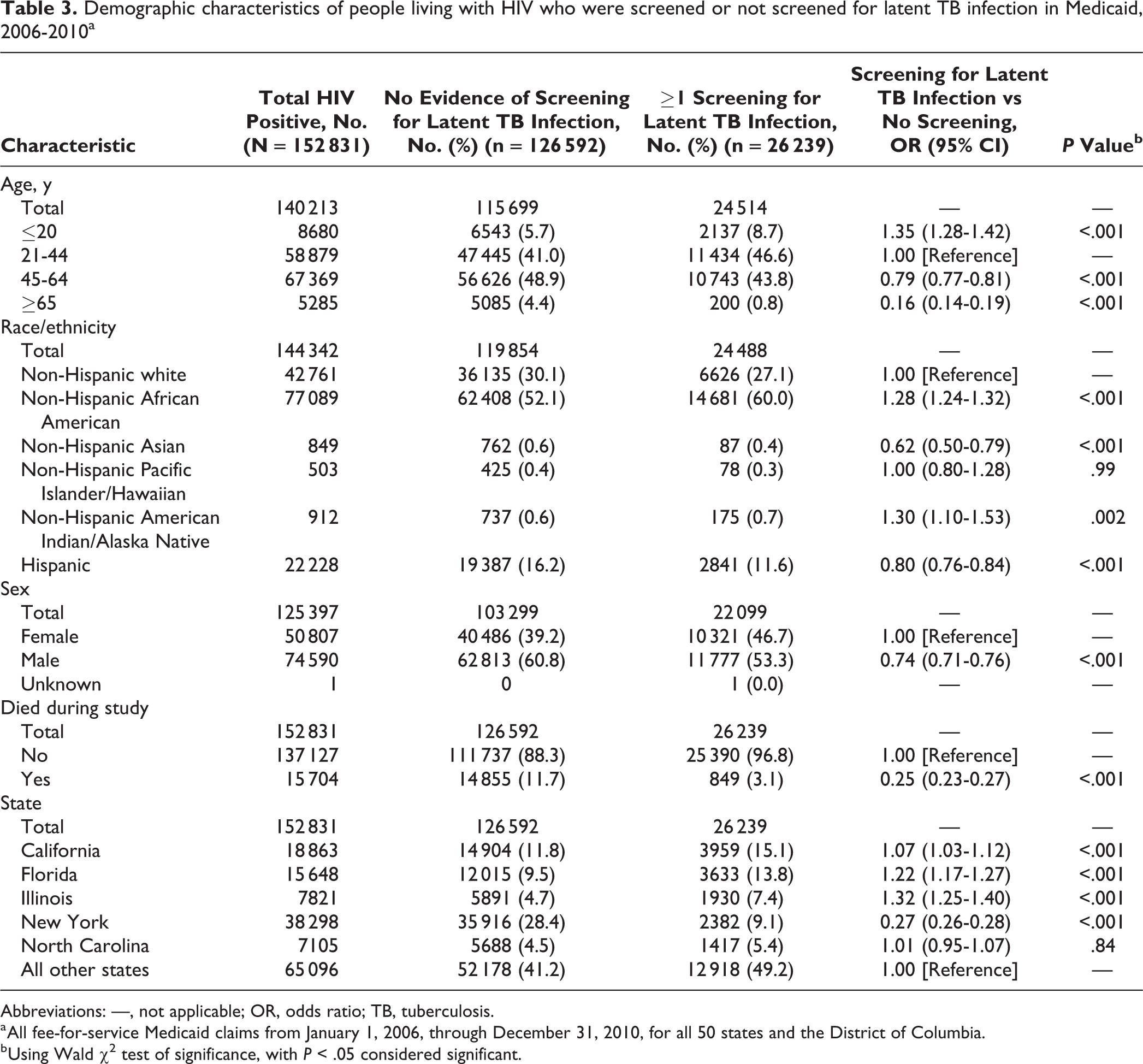
Abbreviations: —, not applicable; OR, odds ratio; TB, tuberculosis.
a All fee-for-service Medicaid claims from January 1, 2006, through December 31, 2010, for all 50 states and the District of Columbia.
b Using Wald χ2 test of significance, with P < .05 considered significant.
The factor that had the largest association with screening for latent TB infection was exposure to TB or suspected TB. People with HIV who had exposure to TB or suspected TB had nearly 4 times the likelihood of screening than those without this risk factor (OR = 3.78; 95% CI, 3.27-4.37). Other risk factors that increased the odds of screening for latent TB infection among people with HIV included poor housing conditions (OR = 1.47; 95% CI, 1.22-1.76), low body mass index (OR = 1.46; 95% CI, 1.13-1.90), chemotherapy treatment (OR = 1.38; 95% CI, 1.23-1.54), and sarcoidosis (OR = 1.35; 95% CI, 1.12-1.62). Other significant factors associated with increased odds of screening included certain autoimmune diseases, renal disease/kidney disease, and the use of certain substances (Table 4).
Association between social and clinical factors among people living with HIV (n = 152 831) and screening for latent TB infection, Medicaid, 2006-2010a
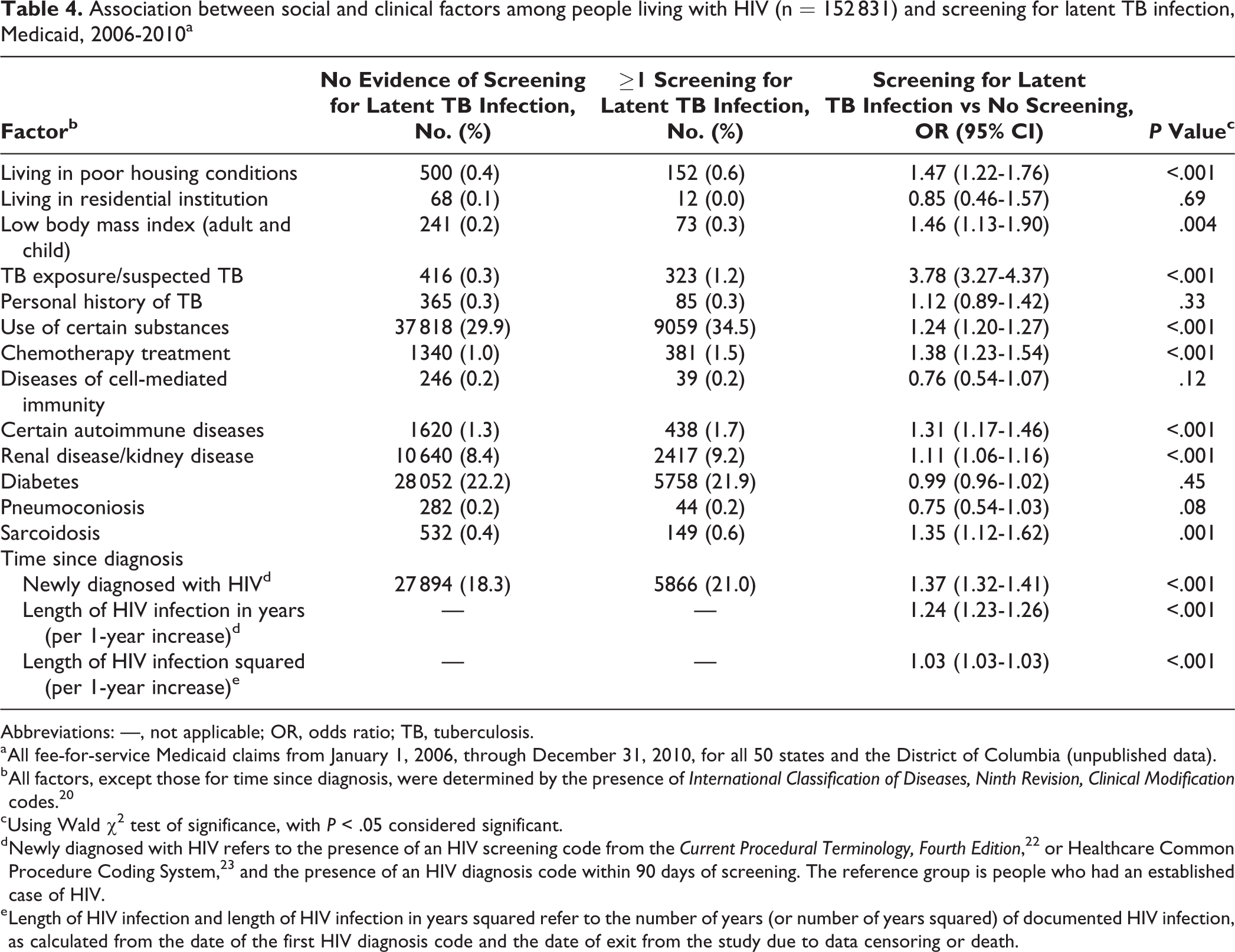
Abbreviations: —, not applicable; OR, odds ratio; TB, tuberculosis.
a All fee-for-service Medicaid claims from January 1, 2006, through December 31, 2010, for all 50 states and the District of Columbia (unpublished data).
b All factors, except those for time since diagnosis, were determined by the presence of International Classification of Diseases, Ninth Revision, Clinical Modification codes. 20
c Using Wald χ2 test of significance, with P < .05 considered significant.
d Newly diagnosed with HIV refers to the presence of an HIV screening code from the Current Procedural Terminology, Fourth Edition, 22 or Healthcare Common Procedure Coding System, 23 and the presence of an HIV diagnosis code within 90 days of screening. The reference group is people who had an established case of HIV.
e Length of HIV infection and length of HIV infection in years squared refer to the number of years (or number of years squared) of documented HIV infection, as calculated from the date of the first HIV diagnosis code and the date of exit from the study due to data censoring or death.
We identified 27 894 people with newly diagnosed HIV (18.3% of all people with HIV), of whom 5866 (21.0%) were screened for latent TB infection. Newly diagnosed people with HIV had higher odds of being screened than people with established HIV (OR = 1.37; 95% CI, 1.32-1.41) (Table 4). People with HIV who were screened for latent TB infection had a longer documented length of HIV infection, with a mean (SD) of 4.03 (±1.2) years, as compared with those who were not screened for latent TB infection, with a mean (SD) of 3.3 (±1.5) years of documented HIV infection (Satterthwaite P < .001). This longer length of documented HIV infection resulted in 1.2 times the odds of being screened for latent TB infection for each additional year of documented HIV infection (OR = 1.24; 95% CI, 1.23-1.26) (Table 4).
Adjusted odds ratios (aORs) for demographic, social, and clinical factors in the multivariate models were largely consistent in direction and significance with the results of the bivariate models. One exception was the association between Hispanic ethnicity and screening for latent TB infection. Compared with non-Hispanic white people with HIV, Hispanic people with HIV were 0.80 times as likely to screen for latent TB infection in the bivariate model, but 1.2 times as likely to screen in both multivariate models. The largest association in multivariate models was for exposure to TB or suspected TB; 5 times as many people with this risk factor were screened for latent TB infection compared with those without this risk factor (Model 1: aOR = 5.03; 95% CI, 4.22-6.01) (Model 2: aOR = 5.02; 95% CI, 4.20-6.00). Multivariate models also demonstrated that both newly diagnosed HIV (Model 1: aOR = 1.17; 95% CI, 1.12-1.22) and a longer length of HIV infection (Model 2: aOR = 2.18; 95% CI, 2.04-2.32) were significant predictors of screening for latent TB infection. In adjusted models, people who were newly diagnosed with HIV infection had lower odds of being screened for latent TB infection than were seen in the bivariate models (1.17 vs 1.37). The odds of length of HIV infection increasing the likelihood of screening for latent TB infection were higher in adjusted models than in unadjusted models (2.18 vs 1.24) (Table 5).
Adjusted odds of screening for latent TB infection among people living with HIV (n = 118 385), by demographic, social, and clinical factors and length of HIV diagnosis, Medicaid, 2006-2010a
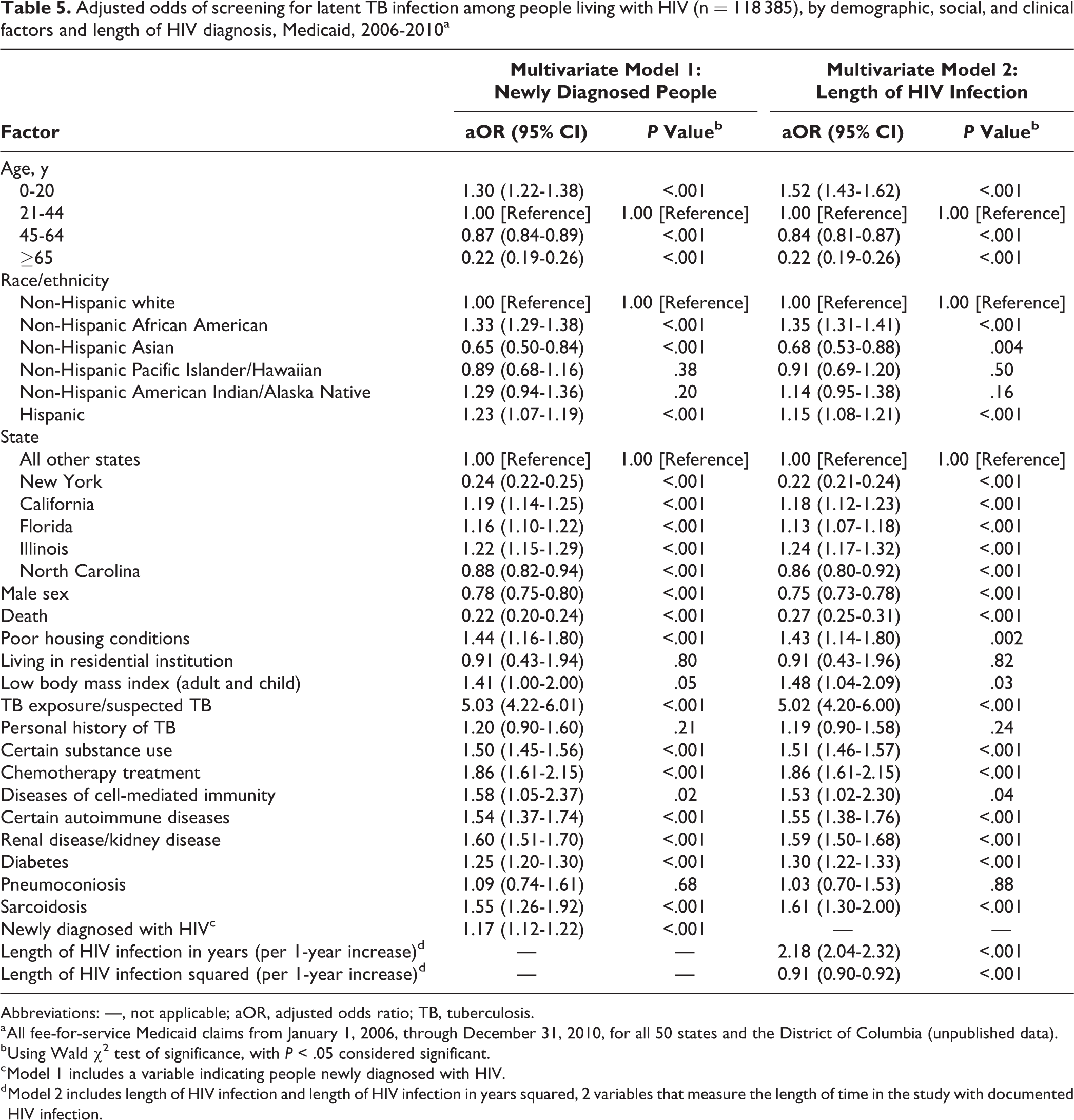
Abbreviations: —, not applicable; aOR, adjusted odds ratio; TB, tuberculosis.
a All fee-for-service Medicaid claims from January 1, 2006, through December 31, 2010, for all 50 states and the District of Columbia (unpublished data).
b Using Wald χ2 test of significance, with P < .05 considered significant.
c Model 1 includes a variable indicating people newly diagnosed with HIV.
d Model 2 includes length of HIV infection and length of HIV infection in years squared, 2 variables that measure the length of time in the study with documented HIV infection.
Discussion
Our study found higher screening rates for latent TB infection among fee-for-service Medicaid beneficiaries living with HIV who also had clinical and social factors that increased the likelihood of acquiring TB infection, increased the likelihood of transitioning from latent to active TB disease, or were associated with more severe TB disease, as compared with people with HIV who did not have these clinical and social factors. Overall, screening for latent TB infection among people with HIV was suboptimal given that at least 1 lifetime screening for latent TB infection in this population is considered standard of care. 10 Additionally, the percentage of people with newly diagnosed HIV infection who were screened for TB in our study (21.0%) does not reflect adherence to the universal screening recommendation issued by CDC in 1999. 32 Although TB is less prevalent in the United States now than it was at the height of the HIV/TB syndemic in 1993, data from 2016 show that transmission and cases among people born in the United States still occurred. 33 Appropriate early treatment of HIV/TB coinfection reduces morbidity and mortality and advances the goal of TB elimination (<1 case of active TB per 1 million people) in the United States. 34
Published estimates of screening for latent TB infection rarely include categorization according to HIV status, making it difficult to compare the percentage of screening found in our study with other estimates of screening for latent TB infection in the United States. Most estimates of screening for latent TB infection have measured the lifetime screening percentage of people in the general population. One such estimate from the 2011-2012 National Health and Nutrition Examination Survey found that 64.2% of the US population had ever been administered a tuberculin skin test. 35 It is not surprising that lifetime TB screening percentages would exceed those seen during the mean length of time that individuals were in our study (3.5 [SD, 1.4] years).
The low percentage of screening in our study (17.1%) contrasts with the high level of TB screening reported by the Ryan White HIV/AIDS program. Grantees in the Ryan White HIV/AIDS Program reported median percentages of screening for latent TB infection of 73% in 2007 and 75% in 2011 among people with HIV who did not have previous documented TB screening or infection. 36,37 We believe that the difference in reported screening between our study of Medicaid beneficiaries and that reported by clinics participating in the Ryan White HIV/AIDS Program was caused by differences in data availability and quality assurance measures. The Ryan White HIV/AIDS Program calculates screening for latent TB infection by using clinical medical records, whereas we were able to do this only through administrative claims. Our study could not exclude beneficiaries whose clinical records noted previous testing for, or infection with, latent TB. In other words, our denominator was larger than that of Ryan White programs, which resulted in a lower screening percentage in our data. For example, our study found low screening percentages among adults aged >65, a population that is at high risk of latent TB infection. This finding could demonstrate an opportunity for increased screening among older Medicaid beneficiaries or could indicate that adults aged >65 have a record of screening for latent TB infection that is not available in the Medicaid data used in our analysis. In order to receive funding from the Health Resources and Services Administration, clinics participating in the Ryan White HIV/AIDS Program must create quality management programs with reportable performance measures. These performance measures can include screening for latent TB infection among grantees. 38 Although some Medicaid Managed care programs also include quality assessment and performance improvement requirements, Medicaid fee-for-service programs are not required to establish any quality improvement programs or initiatives, nor are they required to measure and report on performance measures. This would likely result in a lower emphasis on screening for latent TB infection among people enrolled in Medicaid fee-for-service programs as compared with those enrolled in a clinic receiving Ryan White HIV/AIDS Program funds.
In our study, most fee-for-service Medicaid beneficiaries were not screened for latent TB infection during 2006-2010, a troubling result given that Medicaid is the largest single source of health care for people with HIV. Under the Patient Protection and Affordable Care Act, states that expanded Medicaid offered coverage to previously ineligible low-income people, including people with HIV. 18,19 Historically, Ryan White programs served as the payer of last resort for people with HIV. Under Medicaid expansion, people with HIV who previously received services via Ryan White HIV/AIDS Program funds may now receive services under Medicaid instead, even if their medical home is the same. This transfer will shift patients from a high screening environment to a low screening environment. The Ryan White HIV/AIDS Program performance measures demonstrate that higher screening for latent TB infection is possible when Medicaid implements and reports HIV care indicators consistent with CDC’s TB screening recommendations.
Limitations
Our study had several limitations. Medicaid claims data are reimbursement based and contain information only for billed health services. As such, information on some demographic characteristics of interest (eg, country of birth, education level, income) and results of clinical tests (eg, viral load, CD4 T-cell, interferon-gamma release assay, tuberculin skin tests) were not available. Clinical information would have clarified the number of people with newly diagnosed HIV and whether health care providers were offering screening for latent TB infection based on the severity of HIV. Demographic information such as country of birth would have allowed us to examine whether those most at risk for latent TB infection (ie, people born outside of the United States) were prioritized for screening among people with HIV. Although administrative data based on claims are a useful source of diagnosis information, they often omit information on screening and other preventive health services. 39,40 The low percentage of screening for latent TB infection among people with HIV in our study may have resulted from underreporting of TB screening for reimbursement or incorrect coding for screenings. Additionally, data on services that enrollees received but that were not billed to Medicaid (eg, free screenings offered by state or city health department) were not captured in our data and could have resulted in an underestimation of screening rates. Lastly, approximately 4000 beneficiaries had their first HIV diagnosis code with 6 or fewer months left in the study period; thus, they may not have had enough time to be screened for latent TB infection.
No standard method exists for using administrative data to determine the numbers of people with newly diagnosed HIV. We created a novel method, based on a 90-day interval between screening and HIV diagnosis, to do so; however, other studies used different algorithms. 41,42 Our method resulted in a percentage of Medicaid beneficiaries with newly diagnosed HIV that was similar to the percentage reported by the Kaiser Family Foundation in 2004. 17 We did not examine the barriers to screening for latent TB infection among people with HIV, which would have been informative, especially in states with high levels of active TB cases (eg, Alaska, Georgia, and Texas), where people with HIV would be at greatest risk for acquiring TB. This topic should be explored in future research.
Conclusion
Our study demonstrated that gaps in screening for latent TB infection persist for people with HIV who are insured through Medicaid, despite the well-known association between HIV and TB infections and the presence of clinical and social factors that might predispose them to TB infection. Although progress has been made to reduce HIV/TB coinfections, practitioners must be vigilant to ensure continued reduction of cases of TB among people with HIV.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Cooperative Agreement Number U36/CCU300430 from the Centers for Disease Control and Prevention (CDC) and the Association of Schools and Programs of Public Health as well as by an appointment to the Research Participation Program at the Office of Infectious Diseases, CDC, administered by the Oak Ridge Institute for Science and Education through an interagency agreement between the US Department of Energy and CDC. The findings and conclusions of this publication do not necessarily represent the official views of CDC.
