Abstract
The present study reports the clinical data of a patient with renal venous malformation misdiagnosed as carcinoma. CT revealed hematocele in the left renal pelvis and ureter. CTU: the left renal pelvis and calyces showed a slightly high density shadow, a size of about 2.6 cm*1.5 cm, and mild-to-moderate enhancement was found at the edge of the lesion. Enhanced MR showed that irregular mass abnormal signal was observed in the lower calyx of the left kidney and the lesions were cast, with short T1 and slightly long T2 signals. The secondary bleeding or mucus of low-grade malignant tumor became suspicious. The patient underwent cystoscopy and left ureteroscopy under general anesthesia on December 3, 2021. Bloody urine can be seen on the left side, and multiple blood clots in the left renal pelvis can be detected. After washing, dark red bloody necrotic substances can be seen. Pathology suggests that renal venous malformation, accompanied by bleeding and thrombosis, is located in the renal medulla, involving the renal calyx, rupture and bleeding of the renal calyx, and obvious local bleeding of surrounding renal tissue. Follow-up for more than 1 year showed that the patient's condition was stable. When patients have renal colic with hematuria, enhanced CT suggests that renal mass is mild-to-moderate continuous enhancement, enhanced MRI suggests short T1 and long T2, considering that the mass may be accompanied by bleeding, and ureteroscopy suggests that dark red bloody necrotic substances should be considered in the diagnosis of renal venous malformation.
Introduction
Venous malformations (VMs) are congenital vascular malformations that predominantly or exclusively affect the venous system. VMs have an estimated incidence of 1/5000–1/10,000 births and an estimated prevalence of 1%. 1 VMs are composed of structurally aberrant vessels that frequently lack muscular layer. The most widespread VM classification divides vascular anomalies in vascular tumors (hemangiomas and others) and VMs. 2
VMs have nothing to do with angiomas or hemangiomas, which are vascular tumors. 3 VMs are very often misnamed and incorrectly managed. Although lots of literature underlines the unique features that distinguish VMs from hemangiomas, however, VMs are often ignored or vaguely assimilated to the improper term angioma. 4 Lots of authors considered as a tumor the diagnosis of cavernous angioma. We now know that cavernous angiomas should be named VMs and that they are indeed malformations and not tumors.
VMs are most typically superficially located in the skin, soft tissue, or the subcutis. They can rarely affect the mucosae or deeper structures. 5 However, renal venous malformation is extremely rare, and the related literature only belongs to a case report. 6 It is usually divided into capillary venous malformation and cavernous venous malformation. 7 Renal venous malformation has usually single lesion, with a size of about 1–2 cm. It can occur in any part of the kidney, which is more common in the renal pelvis mucosa, subepithelial layer, renal calyx, or renal cortex. 7 There is no difference in incidence between gender and age. 8 Clinical manifestations may include hematuria, low back pain, or no symptom. 5 , 9 Its manifestations in imaging examination are often similar to those of renal pelvis malignant tumors. 8 It is very difficult to carry out clinical diagnosis for renal venous malformation, which is easy to cause misdiagnosis. Therefore, most patients eventually undergo nephrectomy on the affected side, which has a certain impact on their lives. 10
This paper reported the clinical data of a patient with renal venous malformation, which initially presented as renal pelvis carcinoma but pathologically confirmed as renal venous malformation after operation.
Case
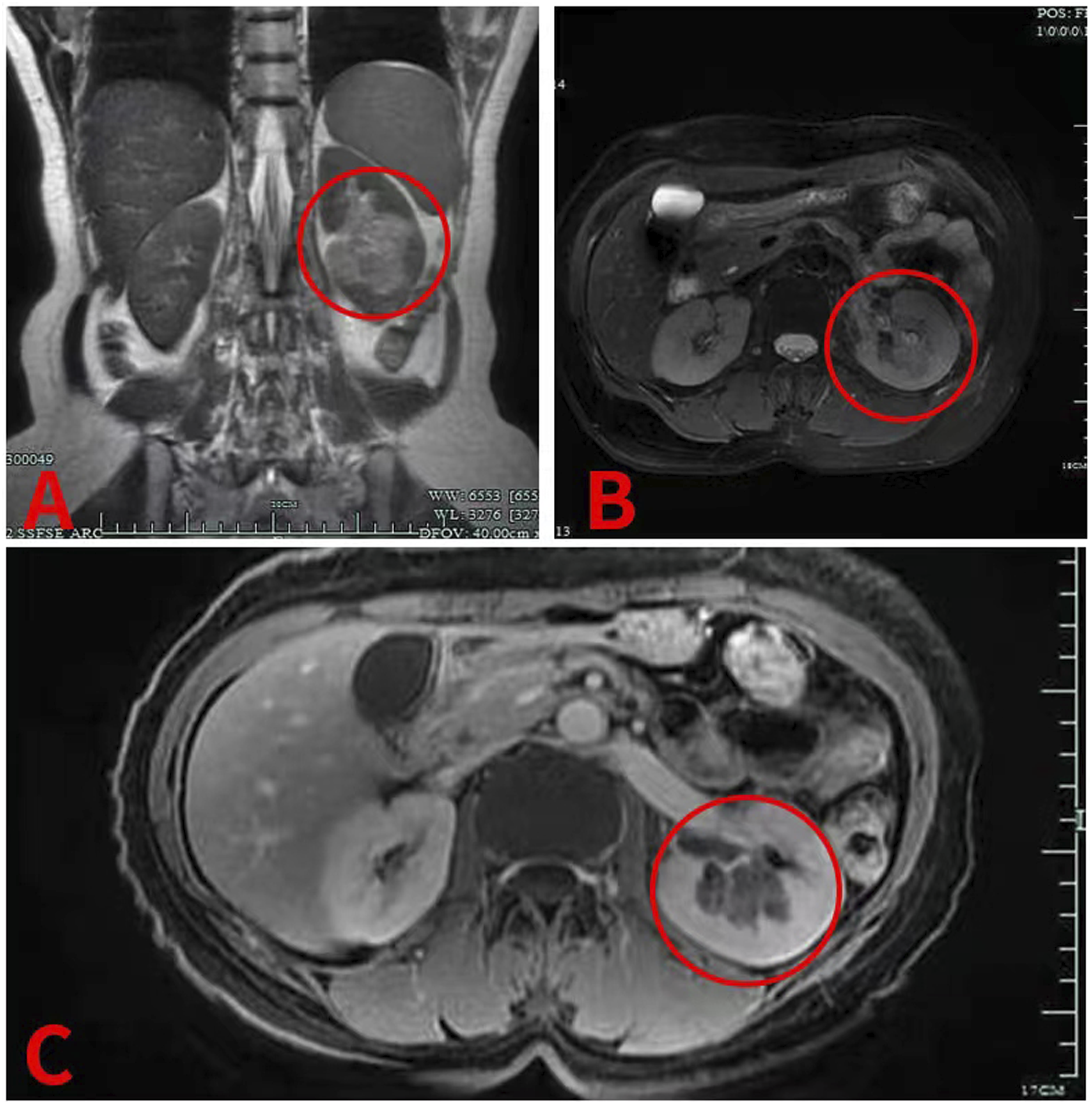
The patient, a 52-year-old female, was admitted in Hangzhou Hospital of Traditional Chinese Medicine on December 29, 2021 with “left renal colic with hematuria for more than 1 week” as the chief complaint. More than 1 week ago, the patient had left waist pain without obvious inducement, which was manifested as paroxysmal colic, and the degree was severe and unbearable. After rest, there was no improvement, no obvious radiation pain, gross hematuria, small strip blood clots, no frequent urination, urgent urination and pain, and no chills and fever. The patient sought medical treatment in a local hospital, and CT examination of the urinary system showed hematocele in the left renal pelvis and ureter and renal pelvis tumor with bleeding was considered. After spasmolytic and analgesic treatment, the symptoms of patient were improved slightly. For further treatment, the patient went to the department in Hangzhou Hospital of Traditional Chinese Medicine for treatment, and the outpatient department planned to accept patients for “renal colic and hematuria to be checked.” The patient had been in good health in the past and denied the history of hypertension and diabetes, as well as the history of surgery, trauma, and smoking. The patient had long-term exposure to dyes, leather, and had occupational exposure to aromatic amines. Admission examination results are as follows: T: 37°C P: 78 R: 20 BP: 135/75 Abdominal soft, no tenderness, liver and spleen under the rib, positive knocking pain in left kidney area, positive tenderness in left ureter path, no difference in right side. Laboratory examination after admission: Blood routine: erythrocyte 3.44*1012/L; hemoglobin 99 g/L. Urine routine: microscopic examination of red blood cells + + + +/HP. No obvious abnormalities were found in liver and kidney function ions, coagulation function, electrocardiogram, and chest X-ray. CTU (see Figure 1): Left kidney occupied with hemorrhage and adjacent calyx involved. Hemorrhage of the renal pelvis and calyceal in the lower pole of left kidney with decreased perfusion of peripheral renal cortex is likely to cause tumor. Enhanced MR (see Figure 2) showed abnormal signal in the lower calyx area of the left kidney, and the secondary bleeding or mucus of low-grade malignant tumor became suspicious. Renal artery CTA: The distal blood vessels in the posterior segment of the left renal artery were poorly displayed. Combined with CTU and enhanced MRI, the possibility of renal pelvis malignant tumor was considered. Moreover, the patient's persistent hematuria did not improve significantly, and the blood routine showed that hemoglobin decreased. In view of the above, the patient had indications for cystoscopy + ureteroscopy + left nephrectomy. After perfect preoperative preparation, the patient underwent cystoscopy and left ureteroscopy under general anesthesia on December 3, 2021. Bloody urine in the bladder and strip blood clot deposition were found. Spray urine on the right ureter, bloody urine can be seen on the left side, and multiple irregular blood clots in the left renal pelvis can be detected. After washing, dark red bloody necrotic substances can be seen. Considering the possibility of renal pelvis malignant tumor in combination with preoperative enhanced CT, enhanced MR, and had occupational exposure to aromatic amines, the patient's family members were informed of the relevant conditions during the operation and agreed to undergo resection of the left kidney, ureter, and partial bladder. The operation went smoothly. Pathology (see Figure 3) suggests that renal venous malformation (size 2.2 cm*2.5 cm*1.1 cm), with bleeding and thrombosis. The lesion is located in renal medulla, involving the renal calyx, rupture and bleeding of renal calyx, and obvious local bleeding of surrounding renal tissue. Ureteral mucosa showed chronic inflammatory changes. No significant lesions were found at the incision margin of bladder. Follow-up for more than 1 year showed that the patient's condition was stable. CTU: A plain scan showed that the left renal pelvis and calyces showed a slightly high-density shadow, with a CT value of about 62HU, a size of about 2.6 cm*1.5 cm, and blurred edges, mainly in the middle and lower poles of the renal pelvis and calyces B and C. Enhanced scan showed that mild-to-moderate enhancement was found at the edge of the lesion; after enhancement, the left renal cortex in the lower pole was diffusely low enhanced. Excretion phase showed a decrease in parenchymal density and a filling defect adjacent to the calyx. MRI: A and B plain scan MRI showed that irregular mass abnormal signal was observed in the lower calyx of the left kidney, with the largest slice size of about 3.8 mm × 3.1 mm. The lesions were cast, with short T1 and slightly long T2 signals, slightly high signal on DWI, and low signal on ADC. C Enhanced MR showed that mild enhancement of heterogeneity and decreased perfusion of adjacent renal parenchyma were observed after enhancement. Pathological hints: (staining, H & E; magnification, ×100): The arrow is renal venous malformation with bleeding and thrombosis. The lesion is located in the renal medulla, involving the renal calyx, with rupture and bleeding of the renal calyx, and local bleeding of surrounding renal tissue is obvious.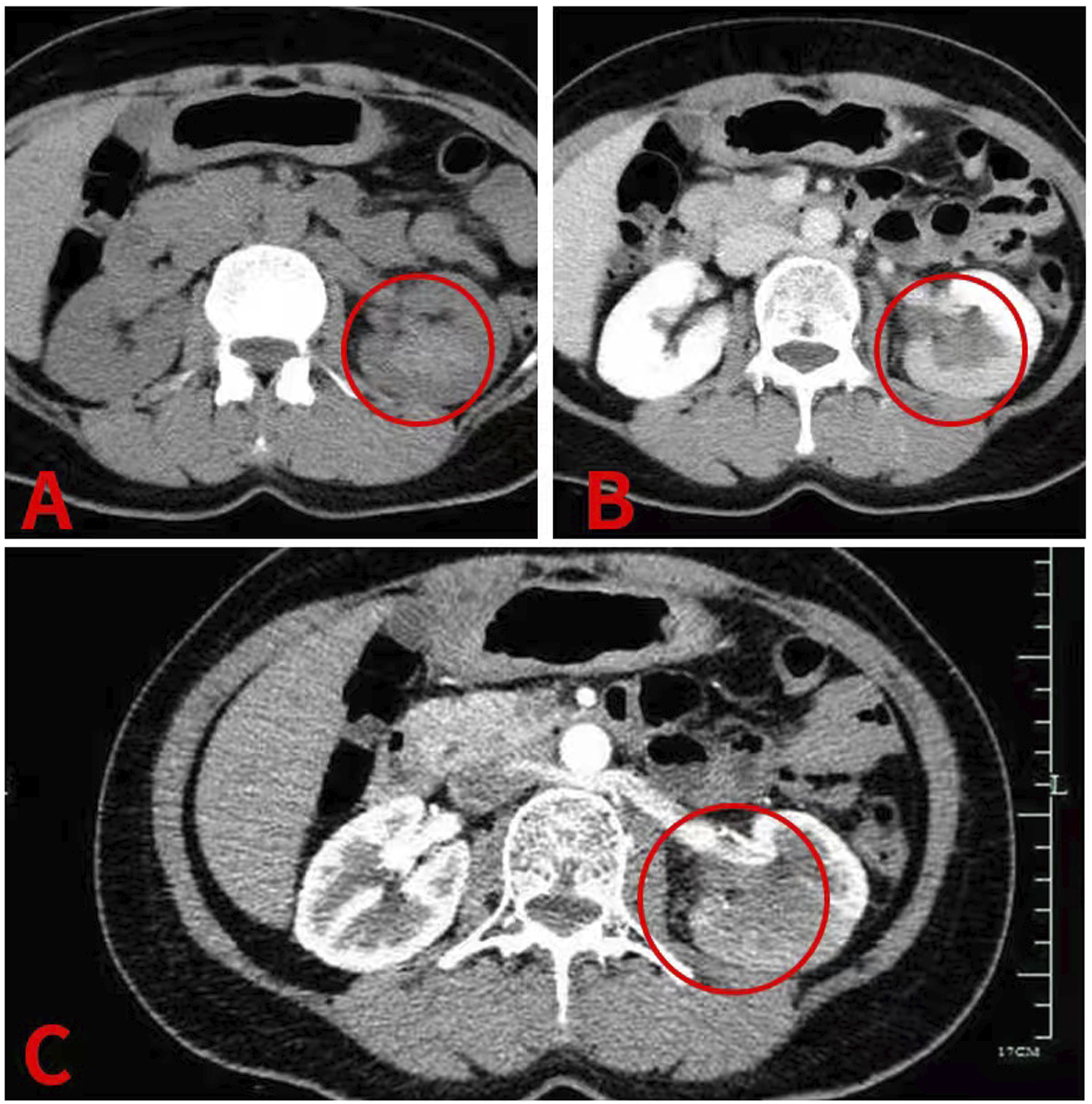

Discussion
Renal venous malformation is a rare renal vascular malformation. Renal venous malformation originated from embryonic residual tissue of monoenergetic angioblasts that did not develop into normal blood vessels. 8 Histologically, renal cavernous venous malformation is composed of cavernous vascular lumen which is composed of epithelial cells and connective tissue separation. There are blood and small thrombi in the lumen, which grow in the way of sprouting and pushing the surrounding tissues, pushing the surrounding tissues to form a pseudocapsule, which does not communicate with the surrounding blood vessels. 8 The pathogenesis of renal venous malformation remains unclear. Zhao M et al. believed that patients with end-stage renal disease are prone to renal epithelial and interstitial lesions, resulting in renal venous malformation. 11 It was common in the renal pelvis (48.7%), followed by renal medulla (42.1%) and renal cortex (9.2%). 12 There is no difference in the incidence between men and women, which is usually unilateral and single, with only 12% of bilateral tumors. The diameter of lesion can range from several millimeters to 10 cm. 5 , 7 This patient is a 52-year-old woman with unilateral onset. The lesion is located in the renal pelvis and calyces, with a diameter of about 3 cm. There is no chronic disease in this patient, and no potential pathogenic factors have been found.
The clinical manifestations of renal venous malformation have no characteristic manifestations. At present, most patients with renal venous malformation have no obvious symptoms, and they are only found by chance during physical examination. 13 A few patients may have hematuria (microscopic hematuria or gross hematuria), mostly intermittent hematuria, which is caused by the rupture of vascular epithelium of renal pelvis and calyceal mucosa, and blood clots may appear in patients with a large amount of hematuria, even endangering life. 7 Some patients also have low back pain (slight pain or renal colic), which is caused by blood clot blocking the ureter. 14 The clinical manifestations of renal venous malformation are similar to those of renal pelvis carcinoma, but it is difficult to distinguish it from renal pelvis carcinoma. Renal pelvis carcinoma may present as intermittent painless gross hematuria. However, renal pelvis carcinoma frequently has a history of exposure to aromatic amines. In addition, renal venous malformation should also be differentiated from renal angiomyolipoma, which usually has no obvious clinical symptoms. When a larger mass suddenly ruptures, patients may experience abdominal pain and hematuria. This patient has left renal colic without obvious inducement, which is severe and unbearable, accompanied by gross hematuria and small strip blood clots, which is similar to the symptoms reported by Lo C and Zhao X et al. This patient’s clinical manifestations are similar to those of renal pelvis carcinoma, combined with the patient’s occupation and long-term exposure to dyes, leather, and other aromatic amine substances, and it is difficult to differentiate from renal pelvis carcinoma, so that it is misdiagnosed as renal pelvis carcinoma.
The preoperative diagnosis of renal venous malformation is very difficult, and its imaging findings lack characteristics, and few patients can be diagnosed clearly before operation. 7 Ultrasound sonography (US) is usually the first routinely adopted imaging technique for soft tissue VMs. 15 In this case, urinary system enhanced CT and MRI were performed after admission without US because CT showed hematocele in the left renal pelvis and ureter, and renal pelvis tumor with bleeding was considered in the local hospital before admission.
CT has little role in diagnosis of soft tissue renal venous malformation. Zheng LP et al. believed that CT plain scan of renal venous malformation often shows equal density or low-density mass. If contrast is added, then a VM will typically appear as diffusely hyperdense just like hepatic angiomas (which most often are VMs) do. 16 This patient showed continuous enhancement on enhanced CT and did not show the characteristics of fast forward and slow out reported by previous scholars. However, the plain CT scan of renal pelvis carcinoma mostly showed isodensity or low-density mass, high-density bleeding was observed in the renal pelvis calyces, and also, the enhanced scan showed mild-to-moderate persistent enhancement. 17 Therefore, it was misdiagnosed as renal pelvis carcinoma. In addition, CT scan of renal angiomyolipoma showed that the mass was mainly of mixed density with fat density. However, in this case, CT showed that the mass had no fat density, so renal angiomyolipoma was ruled out.
MRI represents the gold standard for the diagnosis and the quantification of the extension of the malformation as well as the degree of involvement of nearby structures. 16 MRI has demonstrated a 98.9% sensitivity with 98.9 positive predictive value in diagnosing VMs. 16 NAM S J et al. held that MRI manifestations of renal venous malformation are T1W1 low signal or iso-intensity and T2W1 high signal. If contrast is added, the VM is brightly enhanced, with clear boundary, which is similar to the lighting sign of hepatic hemangioma. 18 The two most typical features on MRI are venous lakes and phleboliths. MRI manifestations of renal pelvis carcinoma are T1W1 high signal or iso-intensity and T2W1 low or high signal. 17 In this case, enhanced MRI showed short T1 and long T2 (T1 high signal and T2 mixed signal). Considering that the mass may be accompanied by hemorrhage, mixed signal appeared. This patient did not have T1W1 low-signal intensity and T2W1 high-signal intensity that were reported by previous scholars. On the contrary, the MRI findings were similar to those of renal pelvis malignant tumor. Therefore, this patient is confused with renal pelvis malignant tumor of left lower calyx secondary bleeding on MRI.
Arteriography has no role with VMs. 19 On the other hand, phlebography with direct puncture of the venous malformation is very important. However, phlebographic studies are reserved for the management of VMs and not for the initial diagnosis. 19 In this patient, renal artery CTA showed that the distal blood vessels in the posterior segment of the left renal artery were poorly displayed.
Because the efficacy of renal venous malformation is exact and the prognosis is good, the focus of clinical treatment for renal venous malformation is preoperative diagnosis. 16 Zhao X et al. have reported that ureteroscopy is helpful for the diagnosis of renal pelvis venous malformation, and ureteroscopy shows dark red mass or lobulated sessile mass. 13 Chandran N et al. maintained that ureteroscopy is an invasive examination, biopsy is easy to cause bleeding, and most patients are misdiagnosed as renal pelvis carcinoma before operation, so ureteroscopy is not recommended considering tumor dissemination and metastasis. 6 Ureteroscopy in this patient is different from previous reports by scholars. There is no obvious mass in the renal pelvis under ureteroscopy in this patient, but there are multiple blood clots in the left renal pelvis. After washing, dark red bloody necrotic substances are seen, so it is possible to consider malignant tumor of renal pelvis.
Most VMs will not need any medication and typical indications to treat VMs are a significant cosmetic deformity, pain, the presence of functional disturbance or hemorrhage. However, other authors suggest early treatment of minor VMs to prevent their growth and consequent complications. 20 The treatment plan of renal venous malformation should be selected according to the disease progression. Patients with clear diagnosis, asymptomatic or mild hematuria, and small lesion can be treated conservatively and followed up regularly. Interventional therapy and partial nephrectomy are feasible for patients with renal function, repeated severe hematuria, and renal colic, so as to preserve nephron as much as possible and avoid unnecessary nephrectomy. 17 VMs are treated with drugs, laser, sclerosants or surgery. 20 Today, surgery plays a role in VM treatment. This patient had been misdiagnosed as having renal pelvis carcinoma, and resection of the left kidney + ureter + partial bladder was performed. Some scholars believe that single, localized VMs that have been treated successfully are not likely to recur. On the contrary, major infiltrating VMs will rarely be cured, irrespective of the technique that has been used. Smaller VMs are always easier to treat than major VMs. It might be advisable in major VMs to prescribe MRI once a year. Follow-up for more than 1 year showed that the patient's condition was stable.
Conclusion
In conclusion, clinicians should fully understand renal venous malformation, a rare kidney disease. Most patients were misdiagnosed because of the lack of specificity in clinical manifestations and imaging examination. Accord to that characteristics of this example, When patients have renal colic with hematuria, enhanced CT suggests that renal mass is mild-to-moderate continuous enhancement, enhanced MRI suggests short T1 and long T2, and ureteroscopy suggests that dark red bloody necrotic substances should be considered in the diagnosis of renal venous malformation. The focus of clinical diagnosis of renal venous malformation is preoperative diagnosis, and comprehensive diagnosis can minimize misdiagnosis.
Footnotes
Author contributions
Xinchun Zhang and Ning Wang: conceptualization, methodology, formal analysis, investigation, and writing—original draft. These authors contributed equally to this work. Bingkui Yang: validation, investigation, and writing—review and editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Zhejiang Provincial Health and Health Commission Medical and Health Science and Technology Plan Project Contract, 2020RC093, NO. 2021KY230; Hangzhou Medical and Health Science and Technology Project Contract, A20200066, B20210391.
