Abstract
Continuous monitoring of physiological parameters in non-human primates (NHPs) necessitates a precise, non-invasive, and convenient method. This study aimed to validate the use of smartwatches with integrated pulse oximetry and heart rate (HR) monitoring capabilities for use in NHPs. Currently, the clinical standard for non-invasive continuous monitoring of peripheral oxygen saturation (SpO2) in NHPs has been the use of a transmittance pulse oximeter (TPO) affixed to a location of highly vascularized tissue. In a clinical setting, HR is monitored through electrocardiogram (ECG) or associated with SpO2 measurement from a TPO probe utilizing photoplethysmography technology. Challenges in obtaining precise readings with TPOs stem from technological limitations and probe placement restrictions. To address these limitations, simultaneous HR and SpO2 measurements were obtained from 15 cynomolgus macaques (Macaca fascicularis) using the Apple Watch 7 (AW 7), Apple Watch 9 (AW 9), and a clinical-grade TPO probe with integrated optical HR measurement technology (iM70, ELAN). Arterial blood gas (ABG) analysis was used as a reference method for SpO2. We found that a TPO device significantly underestimated SpO2 compared to the AW 7 and AW 9 when referenced against ABG values. Smartwatch-derived HR and SpO2 measurements demonstrated good agreement and minimal bias compared to the gold standard method. Overall, the AW 7 and AW 9 exhibited good agreement with clinical reference standards for HR and good agreement with the gold standard for SaO2 in sedated cynomolgus macaques.
Introduction
Physiological monitoring of non-human primates (NHPs) is integral to translational research, ensuring both scientific rigor and the welfare of the animals involved. A key component of this monitoring involves accurately measuring blood oxygen levels. In healthy NHPs, arterial hemoglobin should maintain an oxygen saturation level exceeding 95% oxygen. 1 When oxygen saturation falls below 90%, the animal is considered hypoxic, and intervention is required to improve oxygenation. 2 In the absence of real-time physiological monitoring, blood oxygen levels may drop below this threshold before cyanosis becomes visually appreciable by clinicians. Pulse oximetry is valuable for tracking relative changes in blood oxygen, offering an objective measure that reduces clinicians’ dependence on visual signs of hypoxia, which may not be obvious or present. Therefore, accurate monitoring of oxygen saturation in NHPs is crucial for early detection of hypoxemia and intervention.
One such monitoring technique is through arterial blood gas (ABG) analysis, which is performed using a blood gas analyzer, and is currently the gold standard for assessing blood oxygen levels in humans 3 and NHPs. 4 Using a sample of arterial blood, blood gas analyzers calculate the arterial oxygen saturation of hemoglobin (SaO2), providing a complete analysis of the blood oxygen level composed of both hemoglobin-bound and plasma-dissolved oxygen. While this approach to measuring oxygen saturation is accurate, there are clinical limitations. Firstly, acquiring a sample of arterial blood requires the puncture or cannulation of a peripheral artery. Performing this type of sampling is technically challenging due to the deep anatomical location of arteries. 5 Arterial puncture is also considered invasive and presents some risk of arterial injury, hematoma, thrombosis, and other adverse events for the patient.6–8 Furthermore, ABG offers data only at the specific moment of blood sampling. Continuous monitoring of a patient’s condition would necessitate repetitive punctures or cannulation of a peripheral artery and the analysis of fresh arterial blood samples at regular intervals. This approach becomes expensive and impractical in a clinical setting.
Due to these limitations, measurement of blood oxygen levels is performed using photoplethysmography (PPG) technology integrated into a transmittance pulse oximeter (TPO) device. This approach is a non-invasive, inexpensive alternative to ABG for continuous monitoring of arterial oxygen saturation. In human patients, pulse oximetry is considered a fifth vital sign. 9 Implementation of this technology for monitoring oxygen saturation has also allowed for earlier detection of deterioration of oxygen saturation and overall improved patient outcomes in humans. 10
Pulse oximetry relies on the pulsatile characteristics of arterial blood and the distinct absorption properties of oxyhemoglobin and deoxyhemoglobin across red and infrared wavelengths to estimate peripheral oxygen saturation of hemoglobin (SpO2) as a percentage 11 rather than the direct measurements involved in ABG. 4 The current clinical standard for non-invasive continuous blood oxygen saturation monitoring in NHPs involves using a TPO on highly vascularized tissue, such as the tongue, outer ear, cheek, or fingertip. 4
Unfortunately, TPO devices are impractical for monitoring NHPs as their attachment to a finger or similar area and connection to a multiparameter monitor restrict ambulatory movement and are unsuitable for use in awake animals. Given these limitations, alternative monitoring methods must be investigated.
Reflectance pulse oximetry (RPO) as an alternative method for measuring SpO2 is becoming increasingly accepted within and beyond clinical settings. The wide range of probe placement options and integration into wearable devices like smartwatches drive the popularity of RPO technology. Unlike TPOs, RPOs have their LED and photodiode components positioned adjacent to one another rather than in a reciprocal position. Because the LED and sensor diodes are situated on one face without attachment or clipping to the skin or tissue, RPO can be incorporated more seamlessly into wearable devices and may facilitate longitudinal monitoring. Smartwatch utilization for SpO2 monitoring has been validated in adult human patients, including healthy individuals and those with medical conditions.12–14 Smartwatches have also demonstrated accurate detection of hypoxemia compared to standard fingertip TPOs in humans. 15 Beyond human applications, smartwatches with integrated RPO have recently exhibited high accuracy in assessing cat oxygen saturation levels. 5 While the use of wearable RPOs for monitoring SpO2 is promising, this technology has yet to be validated for use in NHPs.
Another potential use for wearable monitoring devices is non-invasive measurement of heart rate (HR). HR can also be assessed using a similar PPG technology employed in TPO and RPO. Electrocardiogram (ECG) technology has been used as the clinical standard for evaluating cardiovascular parameters like HR. This technology relies on the heart’s electrical activity and involves placing multiple bioelectrodes at particular anatomical locations on the patient. Although effective, this technology lacks the portability and convenience of a single PPG probe or wearable device. While ECG is considered the gold standard for HR measurement, PPG for HR measurement has been demonstrated in human patients to be reliable in healthy patients 16 and patients with atrial fibrillation. 17 Wrist-worn reflectance PPG for HR measurement has also been validated in healthy human volunteers.18–20 Wearable reflectance PPG technology, like those integrated into smartwatches, has yet to be validated for HR accuracy in NHPs.
Here we aimed to evaluate the accuracy of smartwatches for physiological monitoring in NHPs. This study had two distinct aims. The first aim was to assess the accuracy of smartwatches in measuring SpO2 in anesthetized NHPs compared to the clinical non-invasive standard (TPO) and the gold standard (ABG analysis). We hypothesized that the SpO2 measurements obtained from smartwatches would not significantly differ from those obtained via ABG analysis. The second aim was to determine the accuracy of smartwatches equipped with HR monitoring capabilities compared to a clinical PPG HR sensor. It was hypothesized that there would be no significant difference in HR measurements between the two monitoring methods.
Methods
Study population
This study was approved by the Queen’s University Animal Care Committee (Protocol no. 2023-2456) prior to initiation, and all procedures were in compliance with the Canadian Council on Animal Care Policies and Guidelines. The study was carried out in parallel with the animals’ semi-annual physical exams and tuberculosis testing. A total of 15 cynomolgus macaques (Macaca fascicularis), comprising 8 females and 7 males, from the Queen’s University NHP colony were studied. The mean age of the animals was 3.2 years (1.9–10.5 years), with a mean weight of 3.3 kg (2.2–4.8 kg). All animals were either group or pair housed (continuous full contact) on a 12:12-h light cycle with access to daily enrichment through foraging, puzzle toys, mirrors, swings, platforms, and climbing structures. Animals were fed a standard diet of high-protein monkey chow (LabDiet 5045, PMI, St. Louis, MO) supplemented with fresh fruits, vegetables, nuts, and seeds, with municipal city water available ad libitum.
Data collection
On the collection date, the animals underwent a minimum 4-h fasting period to reduce the risk of anesthesia-induced aspiration. Physical exams preceded the initiation of this study protocol, and all animals were assessed as healthy and in good body condition. All data collection occurred between January 8 and January 19 2024, between 8 a.m. and 2 p.m.
Subjects were sedated with a combination of ketamine (KO, Ketaset, 100 mg/ml; Zoetis Canada, Kirkland, QC) 1.5 mg/kg and dexmedetomidine 0.03 mg/kg (n = 15) (Dexvetidine, 0.5 mg/ml; Modern Veterinary Therapeutics, Miami, FL, USA)(Killoran et al., 2021). At the conclusion of data collection, animals were administered a reversal agent, atipamezole (Revertor, 5 mg/ml; Modern Veterinary Therapeutics, Miami, FL, USA), at a dosage of 0.3 mg/kg, before being returned to their home cage. Drug dosages were calculated based on previously documented weights for each animal and delivered intramuscularly into the biceps femoris muscle.
NHPs were injected within their cages using standard squeeze-back caging. Once in lateral recumbency, the animals were moved to the designated data collection area. The animals then underwent a series of assessments, including weighing, temperature measurement using a rectal thermometer, manual auscultation of heart and respiration rates, and a physical examination.
To prepare watch placement sites, an 8- × 8-cm patch at the medial brachial region of each arm was shaved with 10-blade electric clippers. A smartwatch with integrated RPO was affixed over the shaved patches of each arm. The watches utilized were the 7th generation 41 mm Apple Watch (Apple Inc., Cupertino, CA, USA) and the 9th generation 45 mm Apple Watch (Apple Inc., Cupertino, CA, USA). Both smartwatches were affixed simultaneously and randomly assigned to either the left or right arm of each animal, with assignment generated by Microsoft Excel. To increase the pressure of the watch probes against the underlying skin and increase arm diameter, a clean roll of 2-inch vet wrap (Vetrap, 3M Canada, London, ON, CAN) was positioned underneath each arm, and the watch strap (Apple Watch Nylon Sport Loop, Apple Inc., Cupertino, CA, USA) was secured around both the arm and the vet wrap. A TPO (EDAN SpO2, EDAN USA, San Diego, CA, USA) clip with integrated HR measurement capabilities connected to a multiparameter monitor (iM70 VET Veterinary Monitor, EDAN USA, San Diego, CA, USA) was affixed to the cheek, with the LED positioned inside the mouth. The TPO device was randomly assigned to either the left or right cheek. A non-invasive blood pressure cuff (SurgiVet Advisor Vital Signs Monitor, Smiths Medical PM, Waukesha, WI, USA) was affixed to the base of the tail and set to an automatic 5-min inflation cycle.
Data collection commenced after confirming the accurate positioning of both watches and the TPO probe by obtaining readings from all devices. SpO2 was measured from the watches using the Blood Oxygen App (Health App, Apple Inc., Cupertino, CA, USA) with manual initiation of readings that took 15 s to produce a value. Upon completing an SpO2 reading, the watches simultaneously recorded HR. TPO-measured SpO2 and PPG-measured HR were manually recorded each time the watches obtained a blood oxygen value. HR from the multiparameter monitor, TPO-measured SpO2, and the time was recorded every time the watches completed a blood oxygen reading attempt, approximately every 30 s, giving a total of 19 data points per animal. At any point during data collection, if the animals displayed signs of decreased level of sedation, data collection was halted, and the most recent data point obtained was considered the final data point for that animal. If the watches could not produce a reading or the measurement was unsuccessful, it was recorded as a failed attempt. After a failed attempt, adjustments were made to the watches by moving the watch face 3–5 cm either proximally or distally or by altering the tension of the watch band until a successful reading was obtained. If the TPO probe failed to produce readings, the clip was either adjusted on the same cheek or relocated to the opposite side of the face.
Immediately following the successful acquisition of readings from all oximetry devices, blood was obtained from a femoral artery for ABG analysis. This procedure involved palpation of the femoral pulse and aseptic preparation of the collection site with isopropyl alcohol. Approximately 1 ml of blood from the artery was drawn into a red-top tube (BD Vacutainer® Serum Tube, Becton, Dickinson and Company, Mississauga, ON, CAN). Confirmation of arterial blood was done through visual inspection of blood brightness. Immediately following collection, blood was transferred to a 0.5 m lithium-heparin collection tube (BD Microtainer® Lithium Heparin, Becton, Dickinson and Company, Mississauga, ON, CAN) and drawn into a 1 ml syringe. After discarding the first drop, blood from the syringe was transferred to a test card and inserted into the blood gas analyzer (Heska Element POC, Heska Corporation, Barrie, ON, CAN) for analysis. Arterial blood samples were analyzed within 1 min of collection.
Data analysis
The sample size was calculated (Sample Size Calculator, GIGAcalculator, 2017) to identify differences of 4% or 5 beats per minute (bpm) between measurement types or against the clinical standards for SpO2 and HR. For these calculations, the type I error rate was 5% (0.05), with a power of 80% (type II error rate). All data was processed by GraphPad Prism 10 (Version 10.1.1, GraphPad Software LLC, 2024), Excel (Microsoft Excel for Mac, Version 16.40, Microsoft Corporation, 2020), or R Studio (RStudio, Version 2023.12.1, Posit Software, PBC, 2023). Shapiro–Wilk normality tests were performed on SpO2 and HR data across all tested devices. Non-normally distributed data was subjected to nonparametric Mann–Whitney U tests. The clinical relevance of accuracy of the SpO2 measurement devices were assessed following the procedures outlined in the International Organization for Standardization (ISO) 80601-2-61:2017 (CSA, 2017). According to this standard, accurate pulse oximetry equipment should exhibit a root mean square error (RMSE) of less than or equal to 4% across a range of 70%–100% SaO2. Lower RMSE values indicate better accuracy and precision. Calculation of RMSE values allowed for the comprehensive assessment of tested pulse oximeter devices against the reference standard (ABG). Bootstrapping was employed with 50,000 replicates to estimate the confidence intervals (CIs) for the RMSE values obtained in this study. Clinical relevance of HR measurement accuracy of the smartwatches was assessed using standards outlined by the Association for the Advancement of Medical Instrumentation standards for HR meters and cardiac monitors. 21 Here, achieving a mean absolute error (MAE) of less than 5 bpm or a mean absolute percentage error (MAPE) of less than 10% is necessary to meet the standard of accuracy. MAE and MAPE values are indicators of the average magnitude of measurement errors, with smaller values indicating more accurate performance of tested devices against a reference method. PPG-measured HR was used as the reference standard for MAE and MAPE calculations. Paired SpO2 measurements, SpO2–SaO2 measurements, and paired (HR) measurements obtained from different measurement methods were also analyzed using the methods described by Bland et al. 22 Bland–Altman plots serve to visually illustrate agreement between two quantitative measurement methods, depicting the average difference (bias) between a method and a reference method plotted against the mean of the two measurements. This analysis identified discrepancies in means (systematic error or bias), established 95% limits of agreement, and calculated standard deviations (SDs). Positive bias indicates the overestimation of one method against the reference method. Negative bias indicates an underestimation of one method against the reference method. Means were regressed against the percent difference of the means to quantify the assessment of agreement obtained from Bland–Altman analyses. The Bland–Altman analysis did not include the time points with an associated failed reading (dropout).
Results
Animals
The animals recovered from sedation without complications. Physical exams revealed no abnormal findings and ABG analyses were within normal limits. Hematocrit levels (mean = 39.85%, SD +/− 3.924%) indicated no anemia. 1
Dropout rate
The dropout rate for each device, indicating failed SpO2 measurements or unsuccessful blood oxygen measurements, was calculated as the number of failed measurements divided by the total attempted measurements for each device. The clinical standard, TPO, had a dropout rate of 0.79% (2 out of 252), while AW 7 and AW 9 had higher dropout rates of 11.81% (30 out of 224) and 8.23% (21 out of 233), respectively.
Smartwatch and TPO SpO2 against the gold standard (SaO2)
For each animal, an SaO2 value was collected simultaneously with an SpO2 measurement from the TPO, AW 7, and AW 9 devices, resulting in a total of 15 paired measurements (Table 1, Figure 1). Across all animals, the average SaO2 obtained from ABG analysis was 98%. Only SpO2 data points collected in conjunction with an SaO2 measurement (at arterial blood collection time) were used for analysis against ABG. An unpaired, nonparametric Mann–Whitney U test indicated no statistically significant differences in the SpO2 data obtained from the AW 7 and the AW 9 at the time of SaO2 collection (p = 0.8802). Therefore, only the SpO2 data from the AW 9 device was used for further analysis against SaO2.
SpO2 measurements against SaO2.
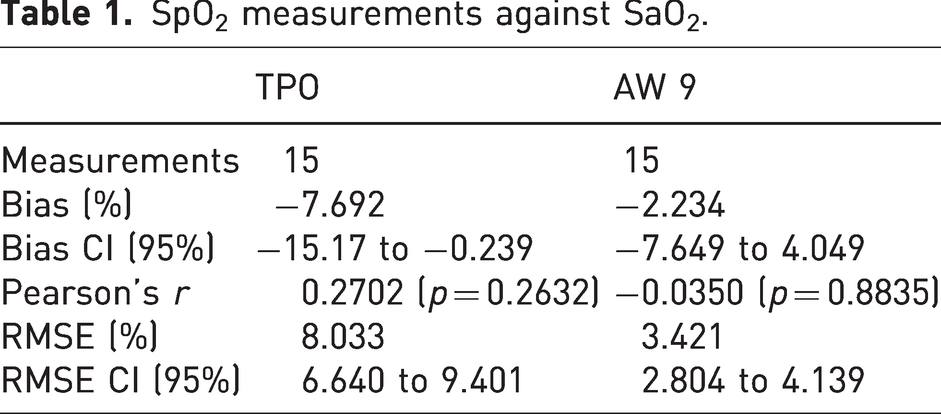
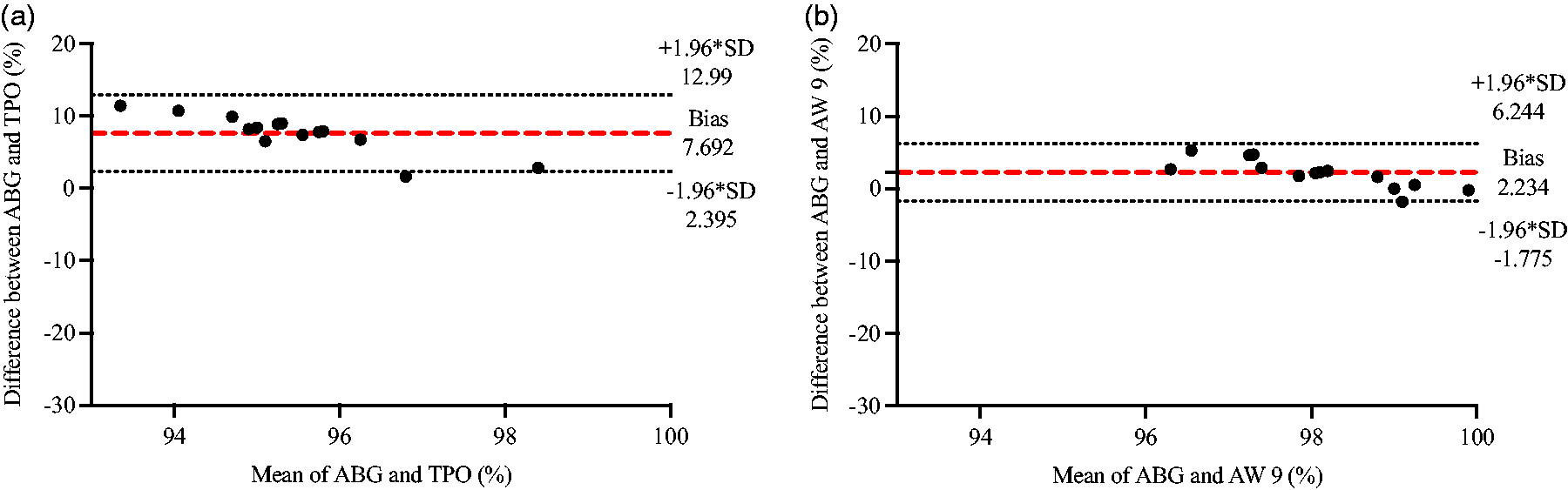
Bland–Altman plots comparing blood oxygen values obtained from ABG (SaO2) versus SpO2 measured by (a) the TPO and (b) the AW 9 (n = 15 for both). The dashed red line represents the mean bias of readings. Dashed gray lines represent the upper and lower 95% limits of agreement calculated as +/−1.96 times the SD.
Smartwatch SpO2 against the clinical standard (TPO)
For each animal, an SpO2 value was measured simultaneously from the AW 7, AW 9, and TPO, resulting in 224 and 233 paired measurements, respectively (Table 2, Figure 2). Between TPO and AW 7 there was a bias of 4.856% and a bias of 4.775% between TPO and AW 9.
AW 7- and AW 9-measured SpO2 against TPO-measured SpO2.
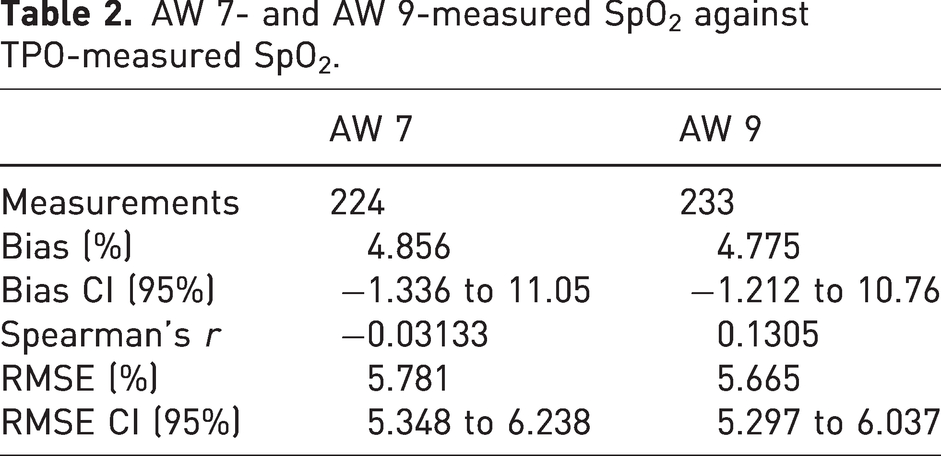
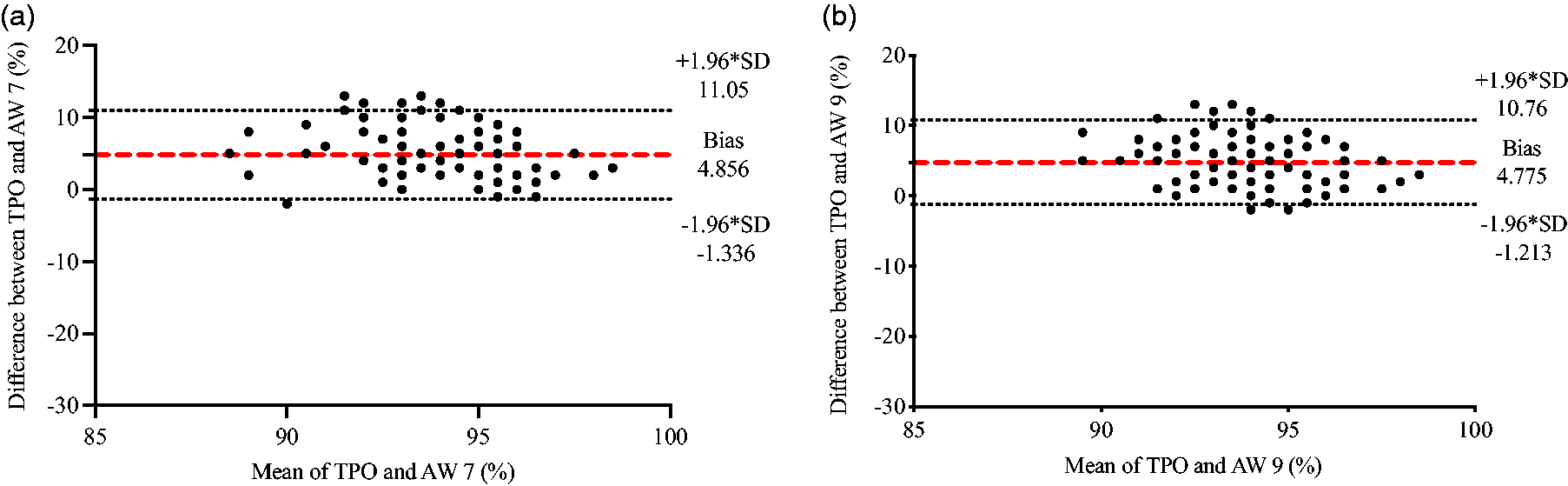
Bland–Altman plots comparing SpO2 values obtained from the TPO sensor versus measurements obtained from (a) the AW 7 (n = 224) and (b) the AW 9 (n = 233). The dashed red line represents the mean bias of readings. Dashed gray lines represent the upper and lower 95% limits of agreement calculated as +/−1.96 times the SD.
Smartwatch HR against the clinical standard (PPG)
For each device, HR was collected simultaneously to look for agreement between the TPO measurement and those of the AW 7 and AW 9. Between PPG and AW 7 there was a bias of 0.6429 and a bias of 0.4569 between PPG and AW 9 (Table 3, Figure 3). The average HR across all measurements was 88 bpm.
AW 7 and AW 9 against PPG HR measurements.
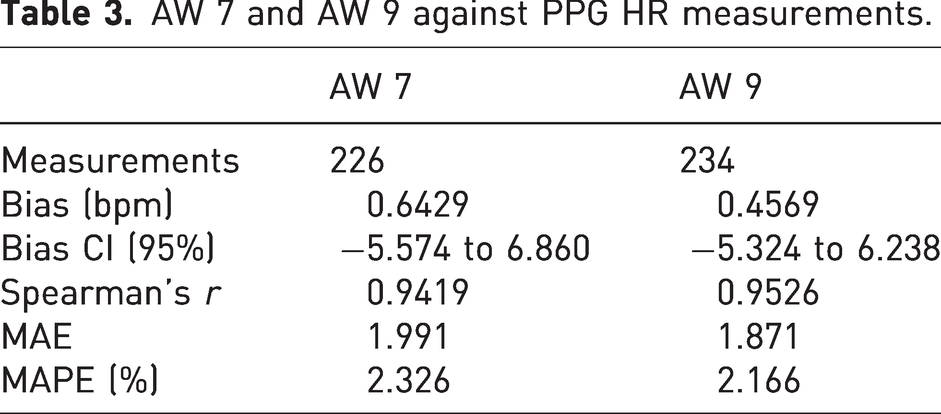
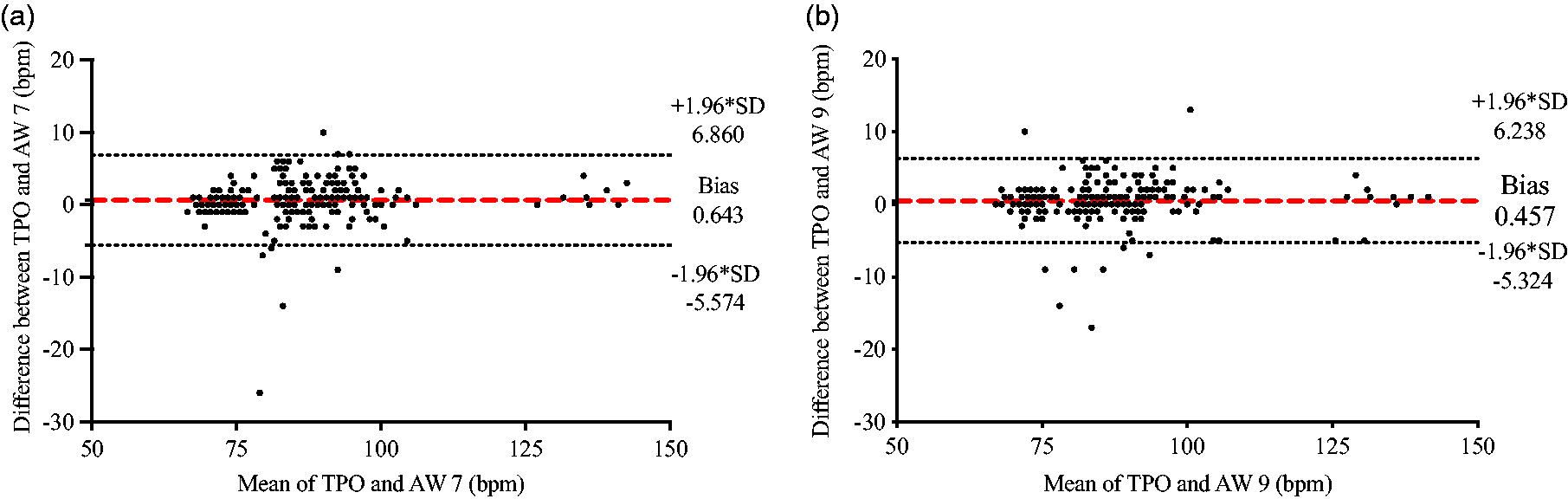
Bland–Altman plots comparing HR values obtained from the PPG sensor versus measurements obtained from (a) the AW 7 (n = 226) and (b) the AW 9 (n = 234). The red dashed line represents the mean bias of readings. Dashed gray lines represent the upper and lower 95% limits of agreement calculated as +/−1.96 times the SD.
Discussion
This study evaluated the accuracy of wearable devices in monitoring HR and SpO2 in NHPs. Good agreement was observed between smartwatch measurements and reference standards in sedated NHPs. Our data demonstrated that smartwatches could serve as a reliable tool for tracking relative changes in blood oxygen levels, reinforcing their potential to provide an objective measure and reduce clinicians’ reliance on visual signs of hypoxia, which may not always be apparent.
The dropout rates across tested devices indicated varying success rates for capturing SpO2 measurements, with the AW 7 and AW 9 exhibiting significantly higher dropout rates in contrast to the clinical standard (Table 1). These dropout rates in the tested smartwatches reflected similar outcomes observed in humans23–25 and cats, 5 where high dropout rates have been repeatedly documented as a limitation to the applicability of wearable technology for SpO2 measurement in clinical settings.
We referenced ISO 80601-2-61:2019 to evaluate the clinical relevance of errors displayed by the TPO and smartwatches against the gold standard (ABG), which states that an RMSE of less than or equal to 4% against SaO2 is necessary for an acceptable accuracy of a pulse oximeter device. 26 We first compared SpO2 values obtained from both the AW 7 and AW 9 watches against the clinical standard (TPO) to evaluate the smartwatches against the clinical non-invasive standard. Significant biases were observed in the smartwatches (Table 2). SpO2 collected from the watches consistently overestimated SpO2 compared to the TPO (bias of AW 7 = 4.856%, AW 9 = 4.775%). When analyzed against SaO2, our results demonstrated that the AW 9 exhibited superior agreement with ABG measurements compared to the TPO device, with a bias of −1.800% versus −7.702%, respectively (Figure 1). The error observed from the AW 9 (3.421%) fell within the acceptable RMSE range for SpO2, whereas the error observed from the TPO device (8.033%) exceeded this threshold. Given that the SpO2 data obtained from the AW 7 was not significantly different from that of the AW 9, it is reasonable to assume that the AW 7 would yield the same results. These results indicated that the AW 7 and AW 9 measured SpO2 with greater accuracy when compared to the clinical standard device, TPO.
Several factors may have contributed to variations in the TPO measurements that were not observed to the same extent in the smartwatches. Firstly, the degree of perfusion at the probe placement site could potentially have interfered with TPO accuracy. Due to its anatomical location (i.e., closer to the heart) and functional demands, the upper arm (location of smartwatch) will have a richer vascular supply with stronger and consistent fluctuations in blood flow to the tissue with each cardiac cycle. Tissue at the cheek (location of TPO probe) has a less extensive vascular network and less blood perfusion overall. A negative correlation has been observed between a reduction in peripheral perfusion and the degree of TPO accuracy.27,28 The underestimation of SpO2 by the TPO compared to the smartwatches may therefore have resulted from poorer perfusion at the anatomical location of the TPO probe placement site compared to the placement of the smartwatches. The nature of TPO probes requiring light transmission through tissue restricted where the probe could be placed while still allowing for signal detection: placement could not be standardized by placing all probes on the wrist.
HR measurements from both smartwatches demonstrated minimal bias (<0.7 bpm) and clinically insignificant error rates when measured against HR collected from the PPG sensor (Figure 3). The industry standard for the accuracy of cardiac monitors is set by the Association for the Advancement of Medical Instrumentation, which specifies that an absolute error <5 bpm or a relative error <10% indicates accuracy of tested devices. 21 Both smartwatches exhibited error levels that remained below these thresholds, demonstrating a MAE of less than 2 bpm and a MAPE of under 2.4%, indicating agreement with the reference standard (data not shown). Nonparametric Spearman correlation analysis also indicated strong positive correlations between smartwatches and the PPG sensor. These findings suggest that both tested smartwatches provided reliable and accurate HR measurements in NHPs sedated with a combination of ketamine/dexmedetomidine.
Several limitations should be acknowledged in this study. Firstly, to evaluate the accuracy of SpO2 measurement devices, adherence to ISO standards requires validation across a range of SaO2, from 70% to 100%. 26 We relied on the inherent variation in SaO2 levels in healthy animals breathing room air. SaO2 ranged from 93.0% to 99.9% in this study. Induction of hypoxia by manipulating SaO2 levels below 90% was beyond the scope of this study. Although the referenced guidelines are intended for application in human medicine, validation of smartwatches across a range of SaO2 levels in NHPs would be logical, as pulse oximeters (including those integrated into smartwatches) often exhibit reduced accuracy at lower oxygen saturation ranges.29,30 The accuracy of smartwatches at SaO2 levels that warrant medical intervention (<90%) in NHPs requires further investigation. ISO guidelines also specify a minimum of 200 SpO2–SaO2 measurement pairs to determine statistically significant accuracy. While our paired TPO-smartwatch measurements were sufficiently powered, we did not obtain enough arterial blood samples for a sufficiently powered comparison of these devices with ABG. This decision was based on the limited number of animals available for study, the invasive nature of arterial blood sampling, and the time constraints of short-acting sedation. Researchers may consider using a larger sample to enhance statistical power in future studies.
Furthermore, while efforts were made to standardize the fitting of the smartwatches to each arm, there may have been variation in the pressure applied to the watch during readjustment following a dropout. Because the watches were manually affixed and adjusted to the patient, standardization of the pressure placed on the watch face was not possible. Contact pressure between an RPO sensor and skin has been shown to affect the accuracy of HR and SpO2 measurements. 31 Inadequate pressure on the skin could result in increased errors, contributing to inconsistencies in the data collected from the watches. Future studies should establish standardized placement methods for the watches, ensuring more consistent pressure across subjects.
Given the accuracy of smartwatches for measuring HR and SpO2 in sedated NHPs, they could also be a valuable tool for the longitudinal monitoring of awake, ambulatory animals. However, several considerations need to be taken into account before deploying this technology in awake animals: for example, discrepancies in measurements have been found in HR and SpO2 between subjects at rest and during activity, 32 and the manual initiation for point-in-time measurements and a period of stillness ranging from 15 s (Apple Watch) up to approximately 30 s (Garmin, Withings) necessary to measure SpO2 could pose challenges.
Ensuring the accurate monitoring of HR and SpO2 in NHPs is crucial for detecting physiological changes that warrant clinical intervention. This study assessed the accuracy of wearable devices for HR and SpO2 monitoring in NHPs. We found good agreement and clinically insignificant errors between smartwatch SpO2 measurements and the gold standard. However, there was poor agreement and significant error between ABG measurements and TPO-derived SpO2, suggesting substandard performance of the clinical standard device in this study. Both tested smartwatches accurately measured HR when referenced against the gold standard. Significant dropout rates were observed in the tested smartwatches, suggesting technological limitations in commercially available smartwatches for widespread use in NHPs. Nonetheless, this research shows that, similar to advancements in wearable health monitoring technology for humans, there are promising applications for NHPs and other animals. These developments hold potential for enhancing research and clinical practices by reducing animal stress levels, improving veterinary care, and advancing the agricultural industry through real-time health monitoring.
Footnotes
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
