Abstract
This paper reviews the methods and approaches used to humanely anesthetize (render unconscious) and or euthanize (kill) laboratory fish (in research settings), with a specific focus on the fathead minnow. We surveyed the literature (333 scientific studies published 2004–2021) to examine euthanasia methods used for various life stages. Our findings showed that many published scientific papers do not provide an adequate description of anesthesia or euthanasia methods, particularly for larval fathead minnows. Over the two decades there was a 20% increase in the number of papers that described their euthanasia method(s). In addition, the review shows evidence that younger minnows require higher concentrations of anesthetic (compared with adults) for effective euthanasia. Recommendations from the review include the use of a two-step euthanasia method (immersion in anesthetic followed by spinal severance and/or exsanguination). As well, it is recommended that details of anesthesia and euthanasia are more fully captured in published scientific manuscripts to allow for comparison among studies and for progress in animal welfare methods. Specific research questions remain on whether rapid cooling is a humane first-step euthanasia method, better investigations into understanding when anesthesia has occurred in fish, and research into methods of euthanasia in larval and juvenile fish.
Introduction
Researchers are obligated to ensure that euthanasia of test animals is humane, minimizes distress, and meets current guidelines and research. Periodic review of the literature is an important part of ensuring that best practices for euthanasia are being used in the lab.1 –3 Refinement of euthanasia methods to minimize pain and distress can also benefit researchers by decreasing variability and improving the quality of their scientific results. 4
The use of fish in research is common and continues to increase in aquaculture, biomedical/biological research, and ecotoxicology.3,5 –8 Decreasing the reliance on mammalian species is part of the reason for increased fish use in research.3,7 Fish also act as sentinel species in aquatic ecotoxicology regulatory testing.3,9 However, there is still less known about euthanasia methods and considerations for fish compared with mammals and other vertebrates.2,5,7 In fish, euthanasia methods are wide ranging, and include chemical methods, physical methods, or a combination of the two. Euthanasia methods result in death by causing hypoxia (direct or indirect), and/or by disrupting/depressing the brain and central nervous system. 10
Recent research and discussion have highlighted the need to update and standardize husbandry and euthanasia methods for zebrafish, a member of the Cyprinidae family and common model species in aquatic toxicology and biomedical research.11 –15 Certain anesthetics, such as MS-222 (tricaine-methane-sulfonate), were found to be less effective and aversive to zebrafish.7,12,16 In addition, the immersion anesthetics MS-222 and eugenol were found to be ineffective on larval zebrafish when using adult dosages.11,17
The review includes the current methods and knowledge of euthanasia in fish, as well as how this has been reported in the literature and to what extent. The first objective of this research paper is to review current knowledge of euthanasia methods used for fish in research with a specific focus on fathead minnow. Fathead minnow (Pimephales promelas) are small freshwater fish also belonging to the Cyprinidae family and are one of the most commonly used fish models in aquatic toxicology. 9 The second objective is to survey the euthanasia methods used on fathead minnows in scientific studies from 2004 to 2021 to ascertain whether any consensus exists between methods used for various life stages.
Current euthanasia research: considerations and recommendations
Physical euthanasia
Physical methods of euthanasia result in direct physical destruction of the brain and include a properly articulated blow to the head resulting in unconsciousness, decapitation/cervical dislocation and pithing. 10 To ensure brain death more than one physical technique is required in a two-step format; for instance, a blow to the head followed by pithing or cervical dislocation/decapitation.10,18 Decapitation or cervical dislocation alone are not acceptable forms of euthanasia.10,18 A number of methods involve first anesthetizing the fish using immersion anesthetic followed by a physical method such as pithing, decapitation/cervical dislocation, freezing or exsanguination.10,18 These are not exhaustive descriptions and acceptable two-step methods are outlined by the Canadian Council on Animal Care (CCAC) and the American Veterinary Medical Association (AVMA) panel on euthanasia.
While two-step physical methods of euthanasia result in certain brain death, the effectiveness of rapid cooling is still under debate. Rapid cooling, or hypothermal shock, involves sudden placement of fish in an ice water slurry (no direct contact with ice), resulting in a rapid transition from an acclimatized temperature to 2–4°C. 10 Recent changes to guidelines now support the use of hypothermic shock as an acceptable one-step or two-step method of euthanasia for small-bodied tropical and subtropical fish, such as zebrafish, which cannot survive temperatures below 4°C.10,15,19 Acceptance of the one-step method is dependent upon life stage and duration in ice water. 10 Currently, this method is not acceptable for small-bodied temperate, cool, or cold-water fish that can survive below 4°C, or for medium and large bodied fish. 10 Rapid cooling is also not an acceptable method of fish euthanasia according to European guidelines. 20
Fathead minnows are a temperate species and therefore are not a candidate for hypothermal shock as an acceptable euthanasia method. However, Hirakwa and Slinas 21 compared temperature tolerance between wild and laboratory-bred fathead minnows and found the upper thermal limit of the wild fish was significantly higher than that of the laboratory stock. While the lower thermal limit of the fish was not investigated, it may be of interest to consider that laboratory-bred lines of fathead minnow may be less tolerant to sudden large drops in temperature (from 25°C to 2°C) owing to the stability of laboratory conditions. 21 More studies are needed to investigate whether laboratory stocks held at constant temperatures lose tolerance to large drops in temperatures and if this would make lab acclimatized fathead minnows a candidate for rapid cooling as an acceptable form of euthanasia.
Electrical stunning is a physical method of euthanasia that is often used in the capture of wild fish. 22 This electrofishing method stuns the fish with a controlled electrical current. Both electrical stunning and anesthetic overdose are approved for use in fish as the first step in European euthanasia guidelines. 20 This technique has been successfully used in some farmed fish species, but more research is needed on the use of electrical stunning for euthanasia of laboratory fish. 23 There might be a place for electrical stunning as a euthanasia method in a research setting, but more work/research needs to be done to ensure operator safety and to establish appropriate current/voltage/timing for various species. There is also a need to ensure that the fish is truly being rendered unconscious and that appropriate follow-up methods (two-step euthanasia) are being employed to ensure death. 23
Chemical euthanasia
Chemical euthanasia uses an overdose of anesthetic usually 5–10× the dose required for anesthesia.10,24 With an overdose of chemical anesthetic, deep anesthesia is achieved in several seconds, compared to minutes with an anesthetic dose. A variety of anesthetics are commonly used for anesthetizing and euthanizing fish, the most common being MS-222.24,25 Other commonly used anesthetics include benzocaine (parent compound of MS-222), lidocaine, clove oil and its derivatives (eugenol, isoeugenol), 2 phenoxyethanol, metomidate, etomidate, and Quinaldine sulfate. Immersion anesthetic involves submerging the fish in water containing a known dose of anesthetic for a specified amount of time, and this is the most common method of fish euthanasia. Injecting fish with anesthetic is also becoming more common, but this method is mainly useful for larger species.10,24 Depending on the dose of the anesthetic, life stage, and species, immersion should be for 10–30 min after the cessation of opercular movement.10,18 The AVMA 2020 guidelines and the following review papers should be referred to for details on various anesthetics, dosages, and mechanisms of action.24,26 –29
Anesthetics with longer recovery times and quicker induction may be better for euthanizing fish, since it is less likely for the fish to regain consciousness. 16 For instance, clove oil and its main active component, eugenol, have been shown to have a shorter induction time and longer recovery times compared with MS-222.30 –32 Sladky et al. 31 attribute the deeper anesthesia achieved with clove oil to the oil’s ability to adhere to the gills. It should be mentioned, however, that even though the food and drug administration qualify clove oil and its derivatives as ‘generally regarded as safe’, studies done by the National Toxicology Program have found the clove oil derivatives methyleugenol and eugenol to be carcinogenic.33 –36
Behavioral responses to anesthesia
Distress behaviors exhibited by fish exposed to certain euthanasia methods are being investigated as a potential indication of harm and discomfort. Exposure to anesthetics has been found to cause aversive or avoidant behaviors, which can vary by species.7,16,37,38 Previously, anesthetics were chosen based on researcher-focused criteria which include: a short induction and recovery time, non-toxic and biodegradable ingredients, minimal impact on experimental endpoints, cost effectiveness, and availability.16,18,39 Sometimes anesthetics are chosen out of habit or researchers are bound by regulatory statutes.5,14,40
Recently, focus has shifted back to the test animal and how it responds to the anesthetic. Researchers use specialized chambers to measure fish aversion to different anesthetics.7,37 The chambers have a gradient stream on each side, one having control water, the other an anesthetic, a positive control (HCl), or negative control (water). Zebrafish (Danio rerio) were found to display aversive behavior to MS-222 and benzocaine but not to etomidate. 7 Using the same experimental set-up, carp (Cyprinus carpio), a species closely related to zebrafish, displayed avoidance behavior toward etomidate, but not MS-222 or benzocaine. 37 Fathead minnows (P. promelas) did not display aversion to any of the compounds and, in fact, spent significantly more time in the MS-222 than in the control water. However, fathead minnows were also unreactive to the positive control (HCl) treatment contrary to the other species tested. The authors found the results inconclusive for fathead minnows, but the AVMA concluded that fathead minnows show no aversion to MS-222, which is unlike other species.10,37
‘Conditioned place avoidance’ experiments have also been used to investigate whether fish feel aversion to certain anesthetics.16,38 Zebrafish placed in testing chambers with light and dark sides have a natural preference for the light side, which can be reinforced by feeding only on that side. 16 When zebrafish were anesthetized on the preferred light side with MS-222 the fish subsequently spent significantly less time in the light area. However, when clove oil or metomidate hydrochloride was used fish displayed less evidence of conditioned place aversion. 16
Studies have also compared anesthetics with rapid cooling methods. Fish have been observed to display more distress behaviors to anesthetics than to the rapid cooling. Bony bream (Nematolosa erebi) treated with benzocaine (chemically similar to MS-222) showed more distress behaviors and longer duration of stress behaviors when compared with rapid chilling. 41 Likewise, zebrafish exposed to MS-222 exhibited significantly more distress behaviors, including twitching and abnormal swimming, compared with the rapid cooling method. 12
The results discussed above all found that zebrafish displayed aversion to MS-222, but unfortunately MS-222 is the only anesthetic legally permitted for use in fish research in many regions. 14 In a survey of zebrafish labs between February and March 2016, 93% of these labs used MS-222 as an anesthetic agent. 14 MS-222 was also reported to be the most common anesthetic used for euthanizing larvae (58%) and broodstock (76%). Possible aversion to MS-222 in some species and its effect on fish anesthesia and euthanasia is an area requiring further study. Rapid cooling is now an accepted method (one- or two-step) for euthanasia in the United States and Canada and, of the labs surveyed, 47% and 40%, respectively, euthanized larvae and adults respectively using this method.10,14,15 Rapid cooling is not accepted in any step of euthanasia methods for fish in European guidelines. 20
Ensuring loss of consciousness
In many cases it is assumed that because the fish is immobile it has been rendered insensible and that the administered agents are also acting as analgesics, but this may not be the case. 28 Depression of the brain and central nervous system by anesthetics results in unconsciousness, but the level of pain relief and muscle relaxation can vary. 29
Metomidate induces rapid anesthesia but it does not provide adequate analgesia or surgical level anesthesia; therefore it is not recommended as an anesthetic for painful procedures.28,42,43 Adult fathead minnows exposed to concentrations of metomidate ranging from 4 mg/l to 32 mg/l achieved deep anesthesia (defined in the study as ‘total loss of equilibrium and cessation of locomotion’) but they reportedly maintained rapid reflex movements if disturbed. 42 At the highest concentration of metomidate (32 mg/l) only 30% survived after 20 min of immersion in the anesthetic, which suggests it may be used as an anesthetic for euthanasia alone, but that any painful procedures should not be performed until medullary collapse is evident. 42
Eugenol, one of the active ingredients in clove oil, causes immobilization of fish but not a complete loss of consciousness, as they respond to physical sensations during deep anesthesia.31,44 Sladky et al. found that adult Red pacu (Piaractus brachypomus) deeply anesthetized with eugenol reacted more strongly to needle punctures than those anesthetized by other agents. Barbas et al. measured brain activity in juvenile Tambaqui (Colossoma macropomum) and found that although they were immobilized, there was a lack of central nervous system depression and their neuronal activity was comparable to a seizure invoking drug. 44 This calls into question the anesthetic properties and use of eugenol and other clove oil derivatives, as AVMA guidelines state that euthanasia agents that cause convulsions before unconsciousness are unacceptable. 10
MS-222 has been found to block action potentials in neurons of rainbow trout (Oncorhynchus mykiss) while not paralyzing the fish at low doses (156 mg/l); however, with increasing doses their skeletal muscle action potentials were impacted, with full paralysis at 830 mg/l. 45 In a different study rainbow trout treated with 150 mg/l of MS-222 experienced a depression in brain activity, exhibiting very linear progression to unconsciousness and deep anesthesia within 5 min of immersion in the anesthetic. 46 Sladky et al. found Red pacu anesthetized with MS-222 did not show reactions to needle punctures, unlike clove oil treated counterparts. 31 Together this suggests MS-222 causes unconsciousness as well as fish immobilization.
These studies suggest that visual/behavioral indicators alone are not a good measure of loss of consciousness and highlight the importance of conducting studies that measure neurophysiological changes when determining the best and most humane approaches for anesthesia and euthanasia.46 –48 Analgesia needs to be investigated by neurophysiological means, such as through measurement of brain activity or other parameters that measure action on the peripheral and central nervous system. 28 Continued research in this area to ensure that visual indicators of deep anesthesia translate to appropriate loss of consciousness are imperative to ensuring that the most humane approaches to anesthesia and euthanasia are being implemented.
Impact of euthanasia methods on physiological parameters
Studies have identified that different modes of euthanasia can have varying impacts on histopathological and physiological parameters.49 –52 Differences in both dosage and length of time in the anesthetic have an influence on physiological blood chemistry parameters and histological changes in gill and nerve tissue.49,50,52 –54 As stated in the CCAC guidelines under the principles of the Three Rs, ‘any factor that disturbs the normal physiological balance of an animal has an effect on the studies being conducted and therefore should be avoided or minimized for scientific and ethical reasons …’. 4 It seems reasonable that this would extend to the method of euthanasia used, so great care should be taken to minimize changes in physiological endpoints.
When an overdose of anesthetic is used exclusively for euthanasia, it is often recommended that fish are kept in the anesthetic for 10–30 min after cessation of opercular movement. 10 Anesthesia doses of various anesthetics can impact cortisol levels and other stress markers over time and could be an important consideration for researchers when sampling blood and tissues. Holloway et al. investigated the impact that eugenol anesthesia had on rainbow trout blood chemistry over time. 50 Fish euthanized 10 min after induction of deep level anesthesia had significantly lower plasma cortisol than those euthanized directly after anesthesia; however, thyroid hormones were significantly higher in the 10-min anesthetic group compared with control fish. 50 Channel catfish (Ictalurus punctatus) exposed to optimal doses of anesthetics for the purpose of anesthesia, but not euthanasia, showed significant differences in circulating plasma cortisol at 10-, 20-, and 30-min exposure time points. MS-222 and quinaldine exposed catfish had significantly higher cortisol levels than catfish treated with clove oil or metomidate (which did not affect cortisol levels). 53 Fathead minnows exposed to anesthetic doses and simultaneously exposed to handling and crowding stress displayed differences in plasma cortisol and an increase in stress markers between the anesthetics tested. 42 Treatment with MS-222 while experiencing stress increased plasma cortisol and decreased degranulation of neutrophil primary granules; treatment with eugenol also significantly increased plasma cortisol but did not impact degranulation of neutrophil primary granules. Metomidate treatment, on the other hand, prevented both increases in plasma cortisol and decreases in degranulation. 42 Considering the above findings, care should be taken to keep consistent time in anesthetic, or a two-step euthanasia method (avoiding prolonged anesthetic exposure) should be used when collecting tissues.
Anesthetic doses more typical of euthanasia (versus anesthesia), where fish are sampled directly after loss of equilibrium and/or response to tactile stimuli, show no differences in blood chemistry parameters between anesthetics.42,50,51,53 Fathead minnows exposed to MS-222, metomidate, or eugenol at concentrations causing euthanasia showed no significant difference in blood cortisol levels. 42 However, Davis et al. reported a significant increase in plasma cortisol measured in zebrafish euthanized with MS-222 compared with those euthanized with an equipotent dose of clove oil. 54 It should be noted that MS-222 was unbuffered in this study (justified by previous work by Wilson et al. 12 finding that MS-222 is aversive to zebrafish whether buffered or not), which may have contributed to the observed cortisol increase.
Studies have also found that when comparing physical methods of euthanasia with immersion anesthetics there were significant differences in plasma glucose, cortisol,50,52 hematocrit 51 plasma ions, metabolites, lactic acid, enzymes, 52 histopathology of gill and nerve tissue, 49 and gill tissue adenosine triphosphate: adenosine diphosphate levels. 51 Parrott et al. found that fathead minnow larvae euthanized via spinal severance had significantly more diversity in their gut microbiome compared with larvae euthanized with MS-222. 55 Ayala-Soldado et al. reported that eugenol and MS-222 used at doses appropriate for euthanizing zebrafish resulted in significant alterations and lesions in the histopathology of gill and nerve tissue compared with control fish that were first anesthetized with MS-222 and then euthanized by decapitation. 49 Taken together, these studies show that caution must be undertaken when comparing results from fish studies that use different euthanasia methods prior to sampling. It is important to recognize the effects that different euthanasia methods may have on endpoint measurements in fish and there is a need to develop detailed standardized sampling practices to increase consistency and reproducibility between studies.
Life stage consideration and indicators of death
There is evidence to suggest that larval stages of fish have a higher tolerance to anesthetic than juvenile or adult fish and may need higher doses and increased time in anesthetic.10,11,17,56 These studies have mainly been done on zebrafish, although Rombough 17 also references unpublished data on rainbow trout. Recommended doses of MS-222 for euthanasia are 5–10 times the dose used for anesthesia in a given species. 10 The typical dose of MS-222 used for euthanasia for adult zebrafish is 200–300 mg/l.11,12,57 Rombough found that the average median lethal concentration of MS-222 for three-day post fertilization zerbrafish larvae was 1633 mg/l and this fell to 730 mg/l at nine days post fertilization. 17 Unpublished preliminary experiments found that newly hatched rainbow trout have an LC50 (LC50 = the Lethal Concentration that kills 50 % of the fish) in the range of 500–1000 mg/l of MS-222, whereas a typical lethal concentration used to euthanize salmonids is 400–500 mg/l.17,26 It is speculated that the higher LC50 in larvae is due to different mechanisms of death between adult and larval fish. Fish gill ventilation is stopped by MS-222, which is effective for euthanasia of adults; however, many larval fish are not solely dependent on their gills and can acquire oxygen via cutaneous gas exchange.17,58 It is also speculated that changes in the biotransformation pathways over development may explain differences in sensitivities between different developmental stages, as biotransformation enzyme activity varies with life stage. 17
When exploring different modes of euthanasia, one must also be aware of indicators diagnosing death. AVMA guidelines outline general criteria for determining fish death and concede that due to the vast number of fish species and their physiological and anatomical variance, not all criteria are applicable to all species. 10 The indicators they describe can be applied to many common species of fish and include: immobility and no reaction to stimuli, flaccid body (unless rigor mortis has set in), inconsistent or complete loss of opercula movement, and absence of eye roll reflex. 10 However, these criteria can also be true of fish that are simply deeply anesthetized, as outlined in various reviews of described stages of anesthesia.25,26,40,59,60 Assessing larval fish is more complicated, mainly because it is difficult to observe whether the opercula are still beating and if there is a loss of eye roll reflex. The absence of a startle reflex has been used to indicate death and is easy to observe in larval fish; however, it could also represent a deep state of anesthesia.25,61 The presence or absence of a heartbeat in fish is not necessarily a clear indication of life either as, as acknowledged in the AVMA guidelines, the heart can continue beating after brain death, and even after it has been removed from the body. 10 Thus, fish displaying a heartbeat are not necessarily alive – but a prolonged state of cardiac arrest provides strong evidence that the fish is dead. 10 However, a few studies have also noted that after exposing fish to anesthetics to the point of cessation of heartbeat, the heartbeat returned upon placement back into control water.11,17,56 These observations highlight the importance of having in place a two-step euthanasia procedure with the first step resulting in deep unconsciousness followed by an adjunctive step (such as flash freezing) that ensures brain death, particularly for larval fish.
Survey of published studies using fathead minnows
A survey of the literature was conducted by examining studies that used fathead minnows for lethal endpoints to determine the euthanasia procedure used. Studies from five scientific journals with focus on fish biology or ecotoxicology were scanned covering the period of January 2004 up to June 2021. The journals were chosen as they are typically where toxicology and biology research on fathead minnows is published, and thus euthanasia and anesthesia methods are used and reported in the papers. Additional details of the search criteria and methodology used are detailed in the Supplementary material provided online.
The survey of the literature found that, overall, for those studies that reported euthanasia methods or end of life sampling, 88% of the time immersion anesthetic was used in all or one of the steps (Figure 1(a)), with MS-222 being the most commonly used immersion anesthetic (91%) (Figure 1(b)). This finding is in agreement with Neiffer and Stamper’s review of the literature specific to euthanasia in fish in general, not specific to fathead minnow. 24
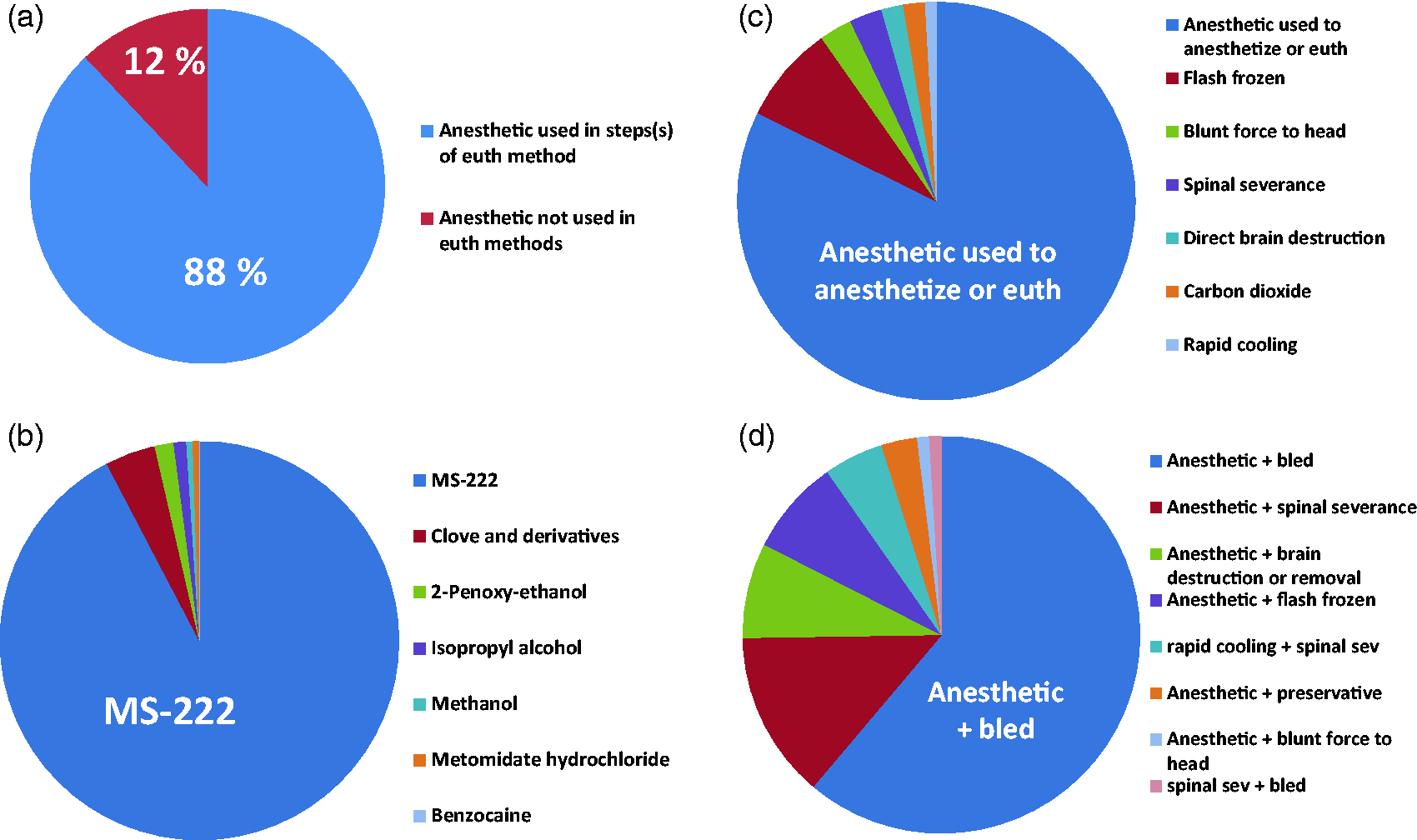
Euthanasia methods described in studies with fathead minnows from five different biology/toxicology journals from 2004 to 2021. (a) Percentage of studies that used an immersion anesthetic in their euthanasia (euth) process, includes one-, two- and three-step euthanasia processes (n = 216). (b) Percentage of specific anesthetic used in studies that reported the use of anesthetics (n = 199). (c) For studies using a one-step euthanasia method only, the percentage using each method is displayed in the pie chart and (d) For studies using two- and three-step euthanasia methods, the percentage that used specific methods for the first two steps is shown. In cases where studies used more than one life stage or more than one euthanasia method, each method was counted as a separate method. MS-222: tricaine-methane-sulfonate.
In all of the studies reviewed, 46% of the time a one-step method was used and 54% of the time a two- or three-step euthanasia method was used (Figure 2). When a one-step euthanasia method was used, most (82%) of the time it was an immersion anesthetic (Figure 1(c)). Similarly, when a two-step euthanasia process was employed, 61% of the studies used an immersion anesthetic followed by exsanguination (Figure 1(d)). It should be noted that in many cases, the exsanguination step was not always framed as part of the euthanasia process but more commonly part of the ‘sampling’ process (fish were bled, brains removed). However, exsanguination ensures the death of the animal, so it was considered part of the euthanasia process in the data analysis. As outlined in the CCAC guidelines, a two-step euthanasia process with anesthesia as the first step is preferred whenever possible. Considering this, it is surprising that just over half of the studies that reported euthanasia methods achieved this. As highlighted above, when working with small fish such as fathead minnow, especially larval stages, a two-step method more adequately ensures that the fish have been euthanized. The AVMA (2020) does allow for immersion anesthetics to be used as a one-step method with the caveats that the fish species are not hypoxia tolerant, and that they are in the anesthetic a minimum of 30 min after cessation of opercula movement. 10 In cases where fish are being sampled, it is unlikely that they would be held this long in anesthetic.

Euthanasia methods described in studies with fathead minnows from five different biology/toxicology journals from 2004 to 2021. Percentage of studies that reported one-, two- and three-step euthanasia methods (N = 234). Studies that did not report details of euthanasia were not included. In cases where studies used more than one life stage or more than one euthanasia method, each method was counted as a separate method.
In addition, out of all the described euthanasia methods (in the reviewed studies where methods were described), only three out of 238 made reference to how long fish were held in the immersion anesthetic bath. As highlighted above, in the ‘Impact of euthanasia methods on physiological parameters’ section, physiological impacts are known to occur based on the dosage, anesthetic of choice, and amount of time held in the anesthetic. Providing as many details as possible of the euthanasia process, such as duration of time in anesthetic, would allow better comparisons between studies and reduce inconsistencies. ARRIVE (Animal Research: reporting of in vivo experiments) guidelines established by the National Center for the replacement, refinement and reduction of animal research (NC3Rs) have a series of recommendations that encompass experimental design, data analysis, and description of details to report for experimental procedures. 62 The experimental procedure section of the ARRIVE guidelines covers euthanasia and describes in detail what should be outlined in the manuscript, including: euthanasia method and which standards the methods comply with, anesthetic used with dose, measures taken to reduce pain and distress, timing of euthanasia and timing of tissue collection after euthanasia. 62 The goals of the ARRIVE guidelines are ‘maximizing the quality and reliability of published research, and enabling others to better scrutinize, evaluate and reproduce it’. 62
Five studies in the survey of the literature reported using rapid cooling as the first step in a two-step euthanasia process on fathead minnow. This is noteworthy because both the CCAC 2005 and AVMA 2020 guidelines do not support the use of hypothermic shock in a temperate species of fish. Perhaps the use of this euthanasia method was justified because lab fathead minnow strains that have been held in the lab for many generations and acclimatized to 25°C are thought to be adequately shocked by a large change in temperature.
The majority of the 333 studies examined reported the details of the euthanasia process (71%) (Figure 3(a)). However, 29% of the studies reviewed did not describe the details of the euthanasia process. For all studies, if more than one life stage of fathead minnow was used, or if more than one method of euthanasia was described, these were counted as separate methods. Reporting of euthanasia methods in the studies varied year-to-year, but it did show slow trends of general improvements over time (Figure 3(c)). With changes in guidelines and increasing focus on animal welfare, it was expected that reporting of euthanasia methods would also increase. And indeed Figure 3(c) shows that there has been some progress over the past two decades, with trends of increased reporting of euthanasia methods fully in published peer-reviewed manuscripts. The percentage of studies that reported their euthanasia method has increased from 62% in 2004 to 81% in 2021 (see yellow trendline in Figure 3(c), p = 0.074, r2 = 0.1857; full data are in Supplementary material Figure S1). However, our literature survey showed that there were still big differences in reporting euthanasia methods across the various fish life stages. Euthanasia methods were most often reported when the fish being used were adults (Figure 3(b)). For experiments that used larval fathead minnow, euthanasia methods were described only half of the time (Figure 3(b)).
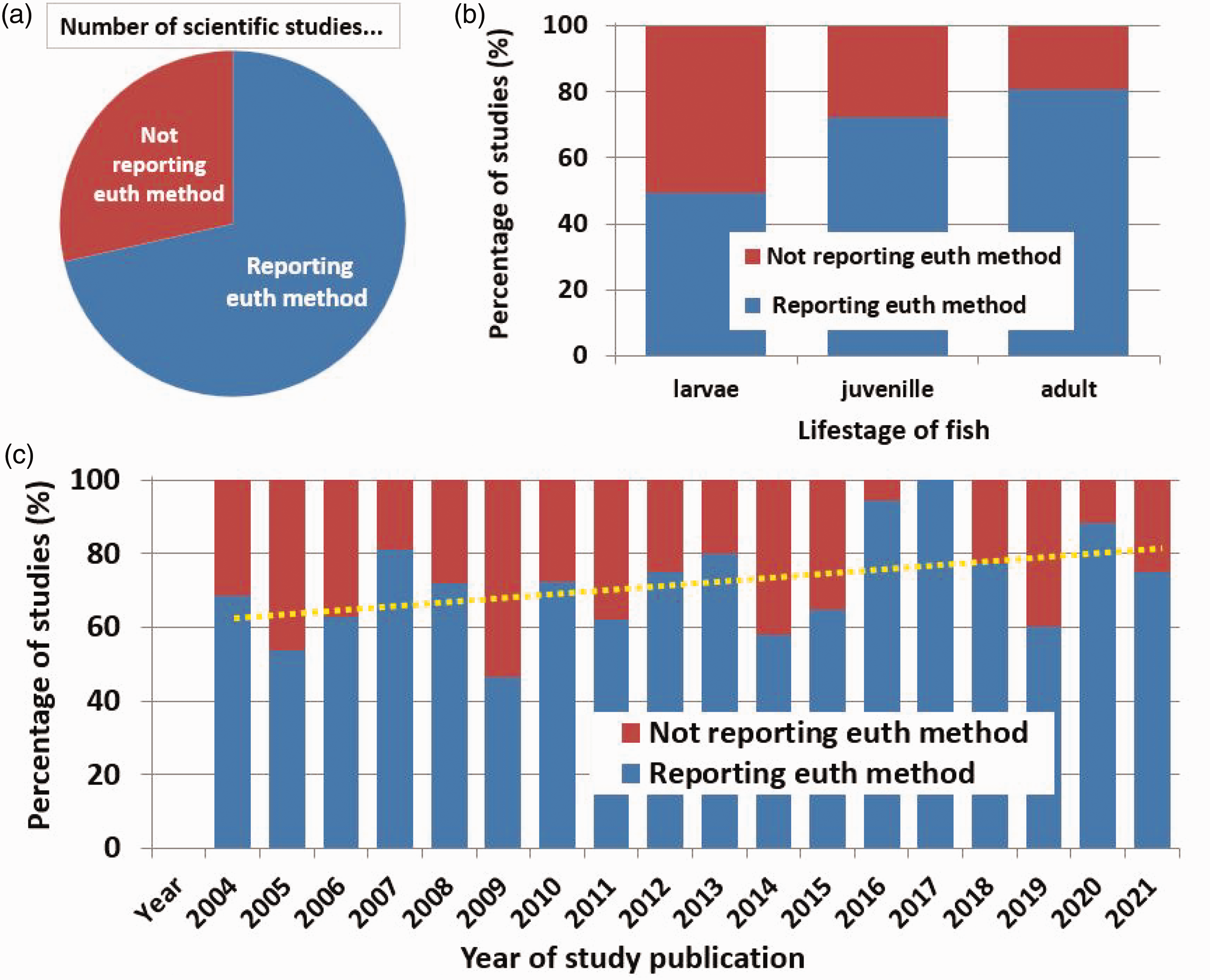
Euthanasia methods described in studies with fathead minnows from five different biology/toxicology journals from 2004 to 2021. (a) Percentage of studies that reported euthanasia methods and percentage that did not report methods. (b) Percentage of studies that reported euthanasia (euth) methods for each life stage: larvae (n = 83), juvenile (n = 58) and adult (n = 192). In cases where studies used more than one life stage or more than one euthanasia method, each method was counted as a separate method and (c) Percentage of studies that reported euthanasia methods for each year examined: 2004 (n = 19), 2005 (n = 13), 2006 (n = 27), 2007 (n = 21), 2008 (n = 25), 2009 (n = 15), 2010 (n = 18), 2011 (n = 37), 2012 (n = 20), 2013 (n = 15), 2014 (n = 19), 2015 (n = 17), 2016 (n = 18), 2017 (n = 12), 2018 (n = 22), 2019 (n = 10), 2020 (n = 17) and 2021 (n = 8). The dotted yellow line shows the trend (line of best fit) for the percentage of studies that report their method of euthanasia in each year.
Once larval fish are free swimming and no longer dependent on their yolk for nutrition, they are considered to be ‘fish’ and are covered under CCAC guidelines. 18 Larval fathead minnow for the purpose of this study were classed as 2 dph (days post-hatch) to 20 dph, as healthy fathead minnow usually begin foraging for food by 2 dph 63 and are no longer subsisting on their yolk alone. 63 Though larval fish are protected under CCAC guidelines once they are free swimming, some argue that larvae should be protected even before this stage. 8 There may be an assumption that younger larval stages are easier to euthanize and do not require as much anesthetic as adult stages, and therefore details do not have to be described. However, larval fish often require higher doses of anesthetics to achieve anesthesia or death compared with their adult counterparts, as described in detail above in the ‘Life stage consideration and indicators of death’ section.10,17 Some euthanasia methods used on adult fish do not work on larval fish.10–11 This highlights why sharing details of euthanasia involving larvae and juvenile fish is just as pertinent as sharing the details of adult fish euthanasia, and that adhering to the ARRIVE guidelines on ethics and scientific integrity is important for all life stages of fish.
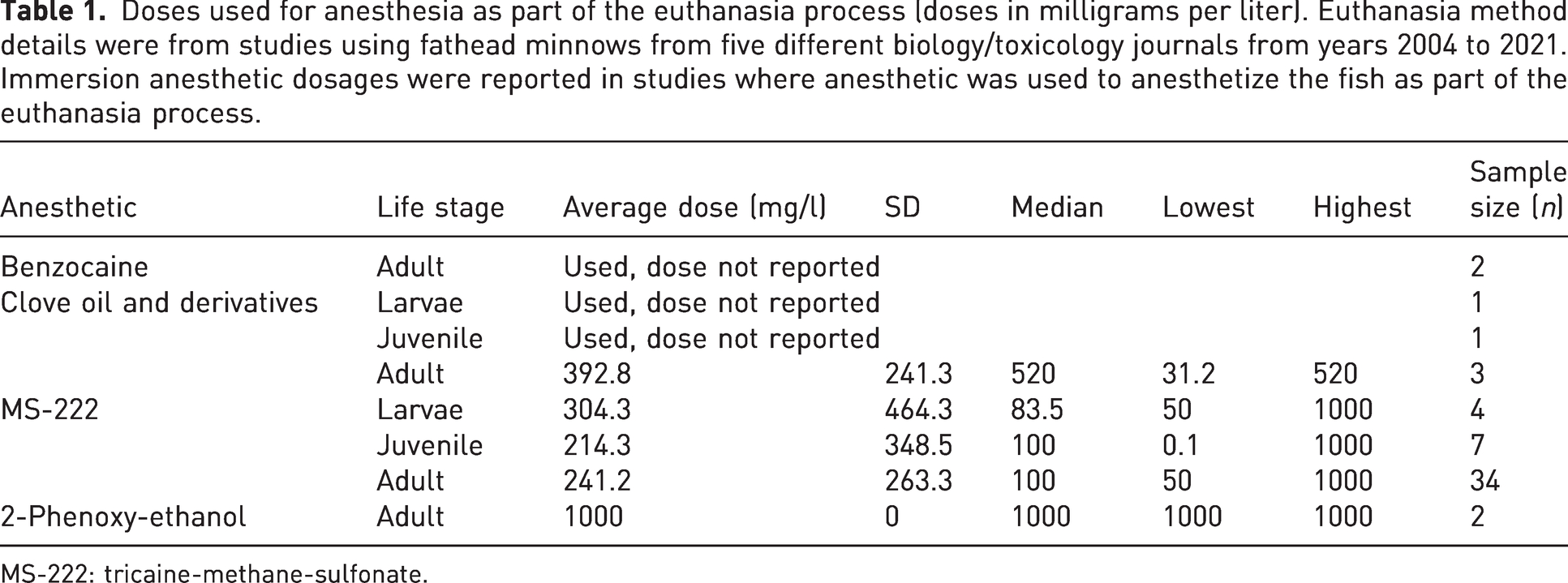
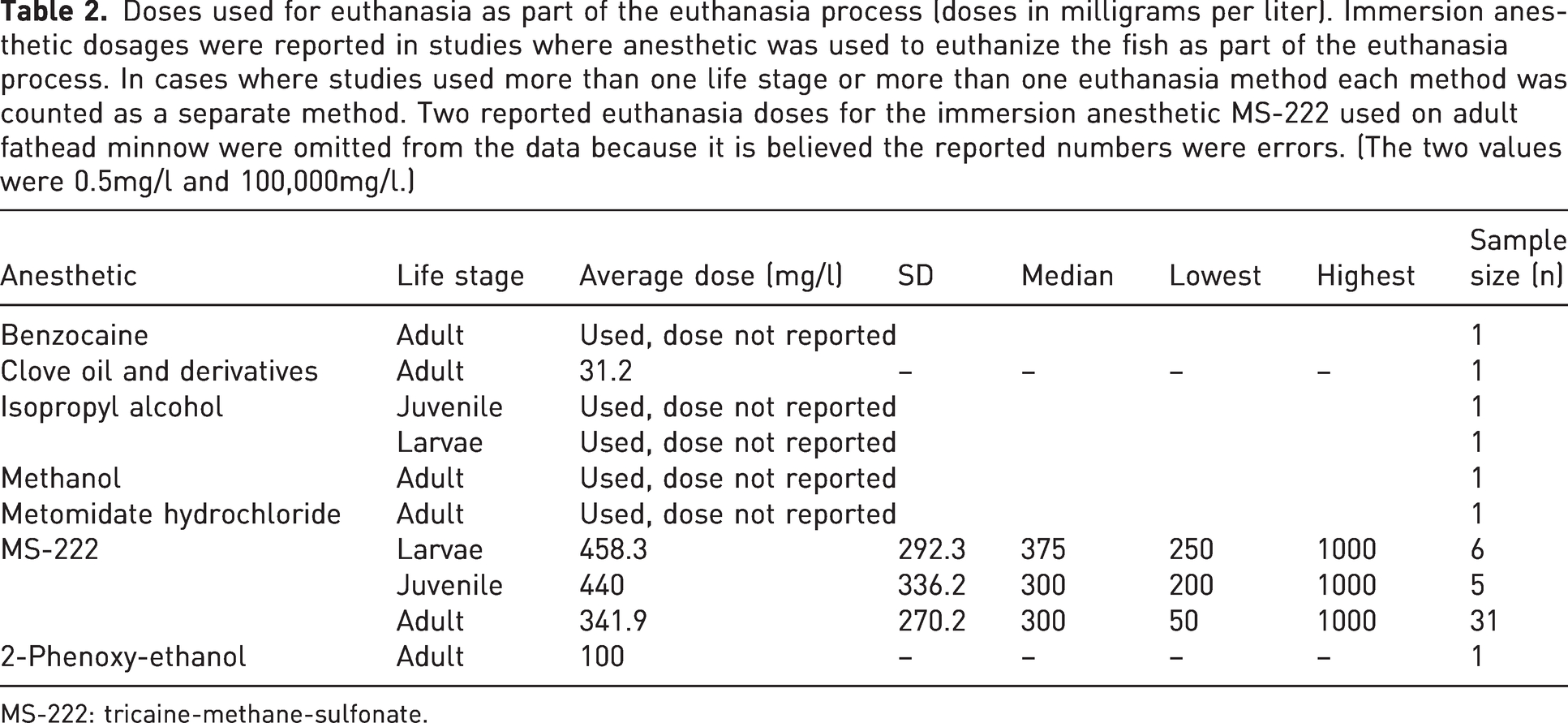
Dosages of anesthetics used in the euthanasia processes (of the studies examined) are provided in Table 1 and Table 2. MS-222, as previously mentioned, was by far the most commonly chosen and most of the data refer to its use. There was a wide variation in MS-222 doses found between studies, with the lowest dose being 50 mg/l and the highest 1000 mg/l. There does seem to be a trend of higher doses used on larval fish, although the sample size is much smaller. However, there is a considerable amount of variability. The dose of anesthetic used for euthanasia does seem to be higher overall when compared with the dose used to anesthetize the fish as a first step in the euthanasia process. For most species the AVMA guidelines for MS-222 suggest 250–500 mg/l, or 5–10 times the anesthetic dose, for a euthanasia dose. 10 Palic et al. reported the optimal anesthetic dose for adult fathead minnow when using MS-222 is 75 mg/l, making the euthanasia dose between 375 mg/l and 750 mg/l. 42 As discussed above, it could be assumed that an appropriate anesthetic dose for larval fathead minnow may be higher if secondary measures were not used to ensure death. Overall, both the range of doses for anesthesia and euthanasia in the studies that report them do fall within this dosing guideline. It would be beneficial, though, to better establish the doses for anesthesia and euthanasia in this species in order to reduce variation among researchers.
Doses used for anesthesia as part of the euthanasia process (doses in milligrams per liter). Euthanasia method details were from studies using fathead minnows from five different biology/toxicology journals from years 2004 to 2021. Immersion anesthetic dosages were reported in studies where anesthetic was used to anesthetize the fish as part of the euthanasia process.
MS-222: tricaine-methane-sulfonate.
Doses used for euthanasia as part of the euthanasia process (doses in milligrams per liter). Immersion anesthetic dosages were reported in studies where anesthetic was used to euthanize the fish as part of the euthanasia process. In cases where studies used more than one life stage or more than one euthanasia method each method was counted as a separate method. Two reported euthanasia doses for the immersion anesthetic MS-222 used on adult fathead minnow were omitted from the data because it is believed the reported numbers were errors. (The two values were 0.5mg/l and 100,000mg/l.)
MS-222: tricaine-methane-sulfonate.
Conclusions
Euthanasia methods that limit distress and pain experienced by fish are the best practice and using non-aversive anesthetics and euthanasia procedures should be prioritized. Increasing knowledge around species-specific reactions would increase scientific integrity, improve refinement practices, and advance fish-handling ethics. Furthermore, choosing methods that limit distress and impact on physiological parameters will increase the integrity, comparability, and reproducibility of the study. A two-step euthanasia process is superior in all cases and should use anesthetics with the quickest induction times and longest recovery.
Based on the research reviewed, the best euthanasia method for fathead minnow at all life stages is a two-step method – immersion in buffered MS-222 (as fathead minnow did not display any aversion) followed by flash freezing, pithing, spinal severance, or exsanguination. Neuro-physical tests should be implemented to investigate whether fathead minnow that have been held at 25°C for multiple lab generations may be potential candidates for euthanasia via rapid cooling as the first step in a two-step euthanasia method, especially at younger life stages.
We suggest that when writing manuscripts for publication, researchers should include as many details of the euthanasia method as possible (a suggestion that should be encouraged by journals as well). Details such as the anesthetic used, dose administered, buffering agent added, average time in anesthetic, and timing of sample collection should all be reported, not only for adult fish, but also for larval stages. Caution should be taken when comparing results between studies when these details are not given, as the method of euthanasia could impact measured parameters.
Continuing research into euthanasia methods will advance the field and close the gap between fish and other vertebrates. Future studies should focus on the neurophysical responses of fish to euthanasia methods using more conclusive indicators, such as electroencephalogram readings, rather than visual/behavioral indicators, whenever possible. In addition, confirmation that individual anesthetics are achieving surgical level anesthesia and analgesia is needed. Periodic reviews of the literature by researchers and review of updated guidelines established by governing bodies (like the AVMA and CCAC) will result in continual improvement in fish welfare and scientific integrity, in turn increasing the quality and reproducibility of studies utilizing fish.
Supplemental Material
sj-pdf-1-lan-10.1177_00236772241288146 - Supplemental material for Current research and guidelines for euthanasia in laboratory fish with a focus on fathead minnows
Supplemental material, sj-pdf-1-lan-10.1177_00236772241288146 for Current research and guidelines for euthanasia in laboratory fish with a focus on fathead minnows by Christine Lavalle, Stacey Clarence, Hufsa Khan, Kallie Shires and Joanne Parrott in Laboratory Animals
Footnotes
Data availability statement
The original summary data used in the review are available by contacting the first or last authors by email at
Declaration of conflicting interests
The author(s) declares no conflict of interest with respect to the research, authorship and/or publication of this article.
Ethics statement
Our study did not require an ethical board approval because it did not contain human or animal trials.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
