Abstract
As commissioned by the Federation of European Laboratory Animal Science Associations, these working group recommendations define the requirements to achieve the humane killing of fish, compare methods of killing, recommend methods of killing depending on context, and detail protocols leading to good practice. With a review of current practices in a fish laboratory and available literature to guide the recommendations, the concept of ideal euthanasia is discussed, and the dilemma of a prompt but stressful death versus a slow but stress-free experience is introduced. Noticeably, the context of fish killing varies widely whether to satisfy European Directive requirements, efficacy for species and/or developmental stages, scientific needs, health and safety, or animal welfare. Examples in the recommendations are based on the most common laboratory fish species, such as zebrafish Danio rerio, and the most commonly used methods of killing, such as overdose of anaesthesia, hypothermic shock, electrical stunning, and concussion – percussive blow to the head. Practical applications of completion of death, refinements, and protocols for good practice are proposed for all developmental stages and depending on the potential fate of the carcass as a scientific sample.
Introduction
Fish have become a major animal model in biomedical research. The termination of life (i.e. killing) of the laboratory animals on welfare grounds or for the purpose of sampling is unavoidable. The process should follow the ethical principles suggested by the Greek etymology of the word euthanasia: ‘good death’ or humane killing, i.e. death must be induced and completed without causing distress, suffering, or pain. The European Directive 2010/63/EU 1 lists some methods of humane killing for laboratory fish. Member states, research establishments, and scientific projects may allow or require other techniques, based on scientific needs, health and safety recommendations, or animal welfare experimental data. For example, some fish species display aversion towards the most used chemical anaesthetics.2–4 The alternatives are physical methods (such as concussion, electrical stunning, or hypothermic shock), which perceived speeds of action (i.e. the time between procedure and assumed loss of consciousness) seem fast enough to prevent any prolonged discomfort. However, these methods could induce a short discomfort. Moreover, humane killing should be a painless experience, but not necessarily a short one. 5 Could humane killing be slow if it remains a stress-free, peaceful, and relaxing experience? Humane means that the killing should be performed with compassion and benevolence. Therefore, in any case, which methods of fish euthanasia would be deemed good practice?
Aims of the working group
To address these questions, the Federation of European Laboratory Animal Science Associations (FELASA) constituted a working group to recommend choices of techniques depending on the species, the stage of development, staff and environment safety, the fate of the carcass, the reliability and efficacy of the protocol, and animal welfare cost. The aim of the working group was to propose good practice guidance on all available chemical and physical methods of humane killing of laboratory fish, focusing on practices in the laboratory. The working group did not consider fishing practices for the purpose of meat harvesting or angling, or issues related to the non-availability of anaesthetics in some countries. The initial step of the working group was to collect information on current practices, in order to identify the list of fish species used in the laboratory and the most relevant issues regarding laboratory fish humane killing. In this document, first, the main results of a survey on euthanasia practices in European and North American laboratories are described. 4 Second, the completion of death is addressed, before humane killing is discussed with the concept of ideal fish euthanasia. Finally, recommendations for good practice of physical (hypothermic shock, electrical stunning, concussion/percussive blow to the head) and chemical (e.g. anaesthesia) methods and their completion are detailed.
Survey on laboratory fish euthanasia
An electronic survey regarding species used for research, methods of euthanasia, health monitoring, and biosecurity in fish laboratories was conducted during winter 2018. 4 A total of 145 laboratories from 23 countries participated, including 111 from Europe and 24 from North America. A wide range of fish species was declared used for biomedical research, and the most common species was the zebrafish (Danio rerio), mentioned by 86% of the responding laboratories. Euthanasia of zebrafish will therefore be specifically discussed in this manuscript.
Regarding methods of euthanasia, the survey results revealed that the majority of respondents killed fish using an anaesthetic overdose, with tricaine (also known as MS222) being the most common anaesthetic agent. About one-third of laboratories killed fish with a hypothermic shock, all of which held zebrafish or medaka (Oryzias latipes). Concussion/percussive blow to the head was performed in 22% of the responding facilities, including mainly laboratories using larger fish such as Atlantic cod (Gadus morhua). Only 3% of the respondents electrically stunned fish.
As defined by the Directive 2010/63/EU, 1 death shall be confirmed using an adjunctive method. According to the survey participants, completion methods were not performed by 42% for adult fish and by 70% for fry capable of independent feeding. This seems to be in breach of compliance, although the survey did not allow collection of further details or analysis. Still, it is difficult to analyse the causes for such a high level of worrying answers from the participants. One obvious reason is the lack of publications on successful protocols for fish euthanasia. In consequence, laboratories who optimize euthanasia protocols, either for untested species or with new information for already well documented species, are encouraged to publish these, as it will save time, resources, and animal life (Refine, Reduce) for other laboratories, reduce non-protocol variation, and improve reproducibility and compliance.
Completion of death
Defining the sequence between completion of death and sampling is essential to preserve fish welfare and sample quality, even if the criteria indicating death is reached can be difficult to determine. For example, fish exhibiting ceased opercular movement may still elicit flight reflexes, indicating that some neural activity remains.6,7 An animal appearing unconscious might simply be unable to move, e.g. due to chemicals preventing neuromuscular action or physical restraint in chilled water. Safety margins must therefore be applied, and fish must not be able to recover from euthanasic procedures. Consequently, once all reflexes (including the startle reflex) 7 have permanently disappeared, the process of euthanasia must be completed by destruction of the brain or by confirming the permanent cessation of blood circulation. The latter can be achieved by ablation of the gills (a more practical approach for bigger fish) or by cutting through the fish to severe the dorsal aorta (e.g. decapitation or severing the caudal peduncle; see Figure 1).

Zebrafish killed by overdose of anaesthesia completed by severing of the caudal peduncle.
The choice of completion method often depends on whether the time frame to death completion must be shortened because fresh samples are requested, for experimental or health monitoring purposes. For example, only fresh samples can be fixed for histopathology whereas polymerase chain reaction (PCR) can often be performed on samples of lesser quality.8–9 Taking the example of fixing a zebrafish for histopathology, the steps could consist of: (a) The killing – induction of euthanasia and confirmation of cessation of all reflexes; (b) The completion – after a further 5–10 min in euthanasic condition (e.g. anaesthetic bath), complete the killing by severing the caudal peduncle caudally to the anal fin (this also removes the tail to facilitate formalin diffusion; see Figure 1); (c) The sampling – nick the coelomic cavity wall to facilitate formalin diffusion and set the dead fish in a formalin pot.
Whether fresh samples are required or not, fish must remain in their euthanasic environment (e.g. anaesthetic bath) until death is completed, i.e. rigor mortis onset is confirmed or the permanent cessation of blood circulation and of all reflexes is confirmed and non-reversible. 1 In practice, this can be achieved by following a few steps: (a) The killing – after induction of euthanasia, confirm fish lost opercula movement; (b) Keep fish in the euthanasic condition (e.g. anaesthetic bath) long enough to reach a lethal overdose and warrant non-recovery (e.g. this period can be prolonged overnight as long as anaesthetic potency is preserved); (c) The completion – perform destruction of the brain or confirm the permanent cessation of blood circulation or keep fish in the euthanasic condition during destruction of the fish carcass (e.g. maceration, freezing) without interruption of the euthanasic condition in order to prevent any risk of recovery.
More generally, the euthanasia protocol of choice may depend on whether the fish tissue will be used for specific assays. In all cases, the method of euthanasia must be reliable enough to ensure death is achieved promptly and/or free of distress and pain. One application is concussion or percussive blow to the head promptly followed by destruction of the brain by pithing (‘Ikejime’). Here, as a final step, spine pithing (‘Tanaguchi’) is also recommended to ensure no central nervous system remains active. 10 Another completion challenge is raised with embryonic and larval forms in which the heartbeat may stop but recover after removal of the euthanasic condition. 11 In such forms, completion of death may be achieved only by destruction of the carcass or the central nervous system. Note that heart beating in adults is not always indicative of life either, because myocardial contractions may continue for up to 4 hours in decapitated goldfish. 12
Requirements for ideal fish euthanasia
Completion is only one of several challenges associated with the current available legislation for fish euthanasic procedures. With over 30,000 fish species documented to date, the differences in size, physiology, and living conditions exclude the possibility of a single euthanasia protocol suitable for all species. Furthermore, within species, one protocol may not be suitable for all life stages. While the number of species subjected to laboratory experiments is only a fraction of the total number, there are still considerable interspecies differences that need to be taken into consideration. The optimal euthanasic procedure for the individual species and life stage in question should therefore be assessed on a small group of animals before upscaling. The requirements of an ideal protocol are discussed below and summarized in Table 1. Each method is then assessed against these requirements in corresponding tables.
Requirements for an ideal euthanasic procedure for laboratory fish.

Choice of euthanasic method could affect experimental outcome
Fish are used in a wide variety of research studying behaviour, genetics, development, neuroscience, reproduction, carcinogenesis, toxicology, nutrition, and more. In some studies, all data are gathered while the animal is still alive, whereas in others, the animal or its tissues are utilized post mortem. In the latter case, physiological characteristics of the cells, gene expression levels, hormone levels or else may need to be conserved through the euthanasic procedure. Depending on the endpoints of a given experiment, it can therefore be of crucial importance that the method does not affect experimental outcome, e.g. by stressing the animal, mechanically damaging cells/tissue, or inducing cytotoxicity. As an example, if the experimental endpoint is brain histology, then concussion could be a detrimental choice of euthanasic method.
Applying the 3Rs (Replace, Reduce, Refine; first described by Russel and Burch (1959)) 13 is a goal for all stages of animal care and use. Optimizing the euthanasia protocols to individual species and life stages will minimize the adverse impact on the animals (Refinement). Table 2 gives further tips to refine the process. 14 Furthermore, standardizing and carefully choosing a method of euthanasia that does not affect the experimental outcome will make results more easily reproducible across laboratories and Reduce the number of animals used for research. Similarly, the ideal euthanasic procedure should be easily reproducible so that it reduces the variation of experience and outcome between individuals. The reproducibility of the technique is characterized by its reliability. The procedure is deemed reliable when death is consistently achieved for all individuals within a short time frame, in an irreversible manner. A reliable procedure should also be easy to perform with a low risk of error, in all circumstances. The reliability of methods first selected by literature review should be tested and validated in-house before large scale experimental use to adjust for possible species or strain differences or other conditions.
Tips to refine the euthanasia protocol.

Animal welfare during the euthanasic procedure
Fish are vertebrates protected by legislation and, as sentient animals, must be treated with the same consideration and care as mammals. 1 To optimize animal welfare throughout the euthanasic procedure, including during the events leading to slaughter, stress and handling should be kept to a justifiable minimum. While some handling might be unavoidable, such as netting fish from a tank, or specifically demanding (e.g. concussion), careful planning and training will ensure a swifter and more gentle process. Further, it is crucial that the euthanasia protocol is optimized for the individuals at hand. For instance, there is evidence that the commonly used anaesthetic tricaine induces aversive behaviour in several fish species.3,15–18 Using an unsuited protocol may therefore lead to unnecessary and unacceptable distress for the animal. For euthanasia to be humane, the loss of consciousness should precede the loss of motor activity (muscle movements).
In addition to species, the age of the animal needs to be taken into consideration as methods may work differentially at different life stages. For instance, while hypothermic shock kills adult zebrafish in a matter of seconds, zebrafish larvae can still recover after 60 min.18,19 The ideal euthanasia protocol should induce unconsciousness or death instantly, without signs of distress, panic, or pain. In absence of an ideal solution, sometimes the ethical dilemma resides between choosing a fast aversive protocol or a slow peaceful one. The option deemed to induce the least distress should be chosen. As fish are not able to communicate their feelings verbally, one must rely on behavioural and physiological clues to assess their state of wellbeing. 20 Typical aversive behaviour is rapid opercular movement, piping, twitching, erratic swimming or gasping at the surface.6,7,15–17 However, here too, are species-specific differences. 3
It is therefore of great importance that the operators are well educated on the biology and behaviour of the species at hand. Furthermore, knowledge about animal welfare, compassion for fish, as well as rigorous training in the techniques and skills required to execute the procedures, should be a pre-requisite for anyone performing euthanasia. This is especially important when there are large numbers of animals being euthanized simultaneously or within a short time frame. On such occasions (e.g. laboratory generation change, termination of large-scale experiments, eradication of pathogens in the facility, or facility closure), cautions should be taken so that practical considerations are not prioritized over the welfare of the individual animal.
Management, preparation, and consequences of the euthanasic procedure
In addition to animal welfare and experimental outcome, an ideal euthanasic procedure should also protect the operator and the environment. Procedures using high doses of anaesthetics could potentially be toxic to the personnel and wildlife. As well as protective gear for the operator, strict protocols for handling and disposal of the chemicals must be in place. Sometimes, animals themselves can be a threat to the operator. For instance, some fish are of such a size that handling demands physical strength. To ensure a calm and effective process, protecting both the animal and the operators, careful planning and increased personnel are essential. If compatible with the experimental design, sedation prior to euthanasia may be advisable. Unexpected events (e.g. power failure, emergency situations, bad weather, malfunctioning equipment, fish escaping net, anaesthesia process taking longer than usual) may occur and must be prepared for. To avoid complications while performing euthanasia, the procedure should be easily and reliably performed and utilize simple equipment not requiring advanced maintenance. This will aid getting back on track in case of unexpected adverse events during the procedure.
Some methods of euthanasia, such as concussion, can be brutal to watch and perform, even though they may be humane. In addition to demanding high technical skill, the use of force may elicit emotional aversion for the operator. When performing these methods, it is therefore crucial that the operator is well trained and comfortable with the procedure. As a rule, the more secure and calm the operator feels during the procedure, the more efficiently and precisely it will be performed, which benefits both the animal, the experiment, and the workers. Finally, as animal captivity and the use of animals in research is a topic receiving much public attention and outrage, it is beneficial if the procedure can be performed in such a manner that the animal welfare is understandable by the public.
Hypothermic shock
Studies on euthanasia of adult zebrafish by hypothermic shock have shown a fast loss of consciousness and no evidence of dermal or muscular cryo-injury when the fish is kept away from ice contact.3,11,19,21 Moreover, the potential acquisition of samples not polluted by chemicals is very relevant for some scientific perspectives, assuming the water used to make the ice is controlled and unpolluted. This makes the technique a valuable, inexpensive, reproducible, operator and environment friendly, uncomplicated to perform, and easy to set up option for adult zebrafish, as discussed in Table 3. The experiments demonstrating the efficacy of the method mostly focused on Danio rerio, and it is not advisable to use this method on larger fish (>5 cm body length, including D. rerio). 22 For other small, tropical species, the literature is scarce, 23 and recommendations at this point cannot be made due to lack of published data confirming the results for other small tropical fish species. It is therefore advisable that laboratories that work with such species test, document, and publish the potential suitability of this method. Hypothermic shock is only an option for tropical species: it should not be used for cold water species.
Ideal euthanasia versus hypothermic shock in zebrafish.

Danio rerio are ectotherms, their internal physiological sources of heat are relatively small, which means that their body temperature is dependent on the outside environment. Rapid and severe changes in the temperature of their environment will subject the fish to a hypothermic shock, which will result in loss of consciousness and cessation of heartbeat. For a guidance protocol, see Table 4 and ensure the zebrafish is held at >26°C up until immersion in ice chilled water at <2°C. In the experience of the working group, these temperatures are easily reached in practice and allow compliance with the amended Directive 2010/63/EU. 22 If the fish holding tank is not warm enough, there is a risk the difference of water temperatures may not suffice to induce a shock and a prompt death. Studies have shown that adult zebrafish kept at >26°C lose reflexes and/or die in 30 seconds following sudden immersion into ice chilled water.19,21 However, a proportion of fish (39%; Wilson et al., 2009) 15 display adverse behaviour for a few seconds. 6 This suggests this method of euthanasia is not ideal. Indeed, there is no knowledge on the experience of the adult zebrafish or potential distress, suffering, or pain until loss of consciousness. Following immersion in ice chilled water, zebrafish movements and the lack of movements thereafter may be triggered by hypothermic shock, independently of consciousness.
Protocol for hypothermic shock of adult zebrafish.

Hypothermia is only a fast method of euthanasia in adult zebrafish. Early developmental stages require prolonged exposure to ice chilled water, or they may recover (e.g. more than one hour for zebrafish at least up to 7 dpf).18,19 Considering that aversive behaviour is detected in zebrafish immersed in ice chilled water at 4 dpf, 18 we do not recommend using this technique for the euthanasia of larval zebrafish. Wallace et al. (2018) 19 showed that hypothermic shock efficacy varies significantly along the third and fourth weeks post fertilization, with 14 dpf zebrafish requiring at least one hour of exposure when 30 seconds might be enough for 28 dpf zebrafish. It is to be noted that acquisition of metamorphosis may explain the difference of required exposure length. Metamorphosis time scale depends on strain, feeding and temperature. Therefore, when the technique is to be used during the third or fourth week post fertilization (from 17 dpf), 22 acquisition of metamorphosis should be assessed, and the efficacy of the euthanasic technique and its averseness trialled prior routine use.
Electrical stunning
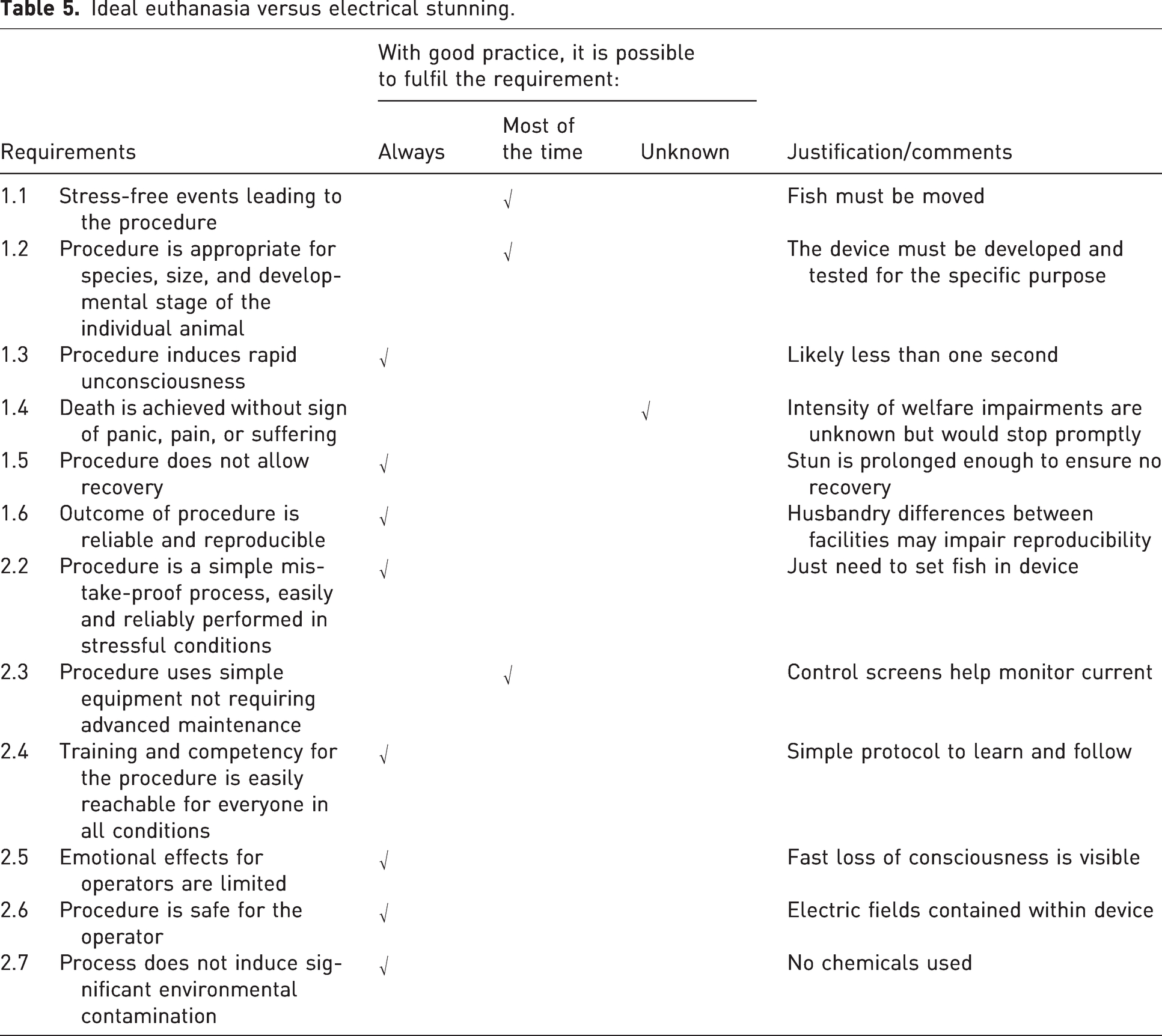
Building an electrical stunning device requires sound competency in electronic engineering and being well conversed and experienced with the effects of electric waveforms and electric fields on fish welfare. Failing this, the device might constitute a hazard for the fish and the operator. The method is currently limited in its practical application in laboratories as only a few species are documented for electrical treatment (mainly farmed fish). Further, there is no international database collating success and failure data that would help optimizing good practice protocols, and the effects of electricity on the physiology of the fish remain largely unknown and may potentially compromise sample quality for some scientific purposes. However, electrical stunning is a chemical-free method and presents advantages compared with other methods. It is recommended for the slaughter of farmed fish destined for humane consumption and is also a useful method in laboratories.24,25 For example, zebrafish eggs and larvae (<5 dpf) can be killed in one minute with the appropriate device. This is particularly relevant considering the lack of efficacy of other methods for early developmental stages. 18 Similar efficacy has been demonstrated in other fish species embryos (e.g. Oncorhynchus mykiss). 26 Table 5 lists how electrical stunning meets requirements for an ideal euthanasia. Nonetheless, the experience of the fish during the procedure remains unknown. Therefore, it is required that the stun induces a loss of perception and consciousness (electro-narcosis) in less than one second. The brief latency to loss of consciousness is the ethical argument balancing the potential discomfort due to the stun. If this loss of consciousness is not achieved in less than one second, then that electrical stunning device may not be refined enough to be recommended.24,25 The stun may induce an initial body reaction, possibly due to electricity-induced muscle contractions. The current administration is then prolonged until animals die and are unable to recover. The voltages used for electrical stunning should not result in animal injuries such as broken spine and haemorrhages.
Ideal euthanasia versus electrical stunning.
Concussion/percussive blow to the head
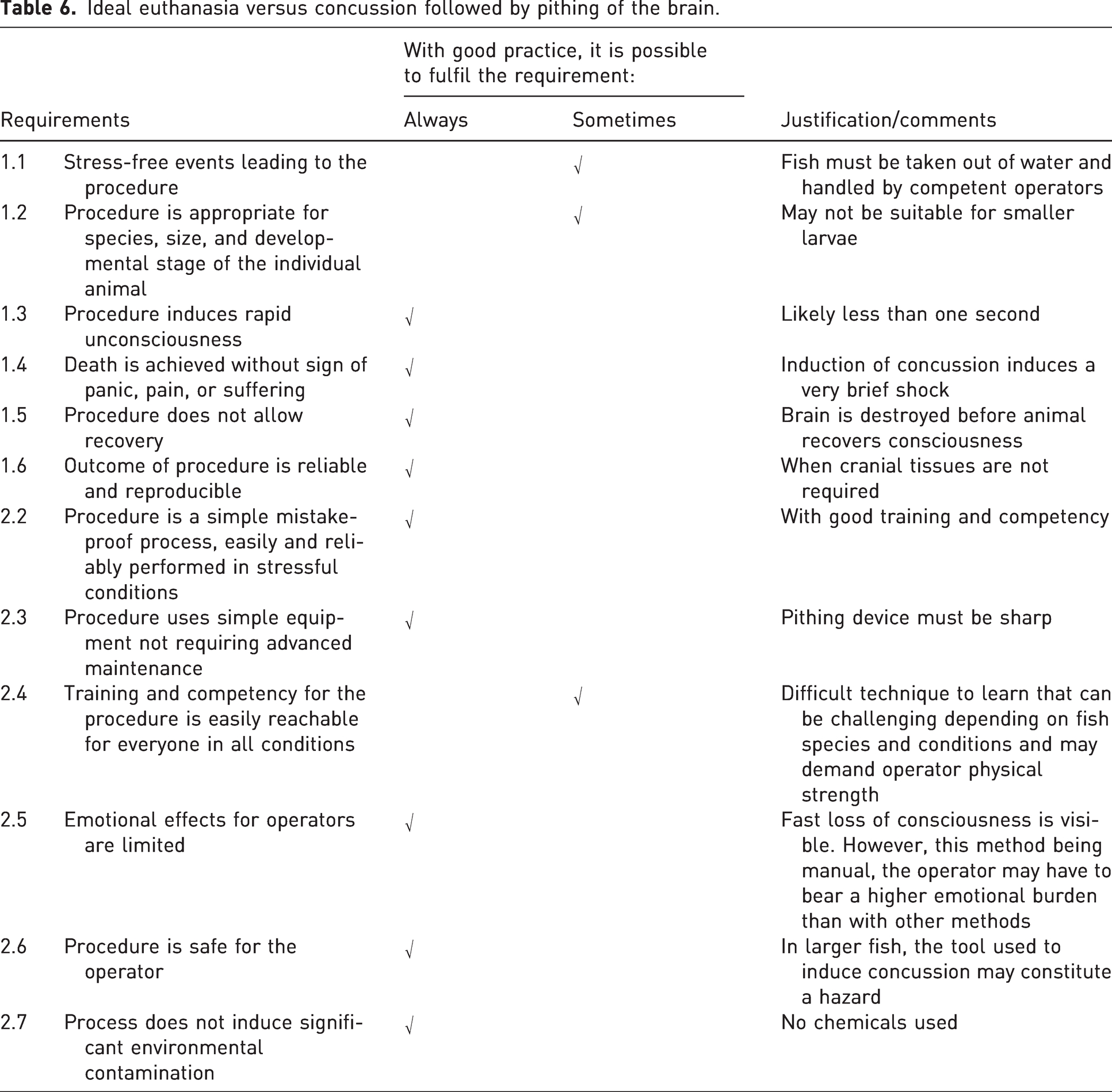
Concussion or percussive blow to the head should only be carried out by experienced personnel capable of doing this in a way that the fish immediately loses consciousness. 20 Prior to using the technique on a new species, recognition of anatomical structures for correct placement of the blow should be performed by dissection of a cadaver’s cranium and with the help of available graphics such as https://www.ikijime.com/all-fish/ and Figure 2. 27 This method is suitable for calm, slow, and easily handled species. To aid this in some species (e.g. cod and trout), a wet cloth can be used to cover the eyes of the fish. For other species, sedation, or other means of restraint, may be used to set the animal steady before percussion. Immediately after the blow, death must be completed rapidly by a secondary method before the animal recovers consciousness, e.g. by decapitation or pithing of the central nervous system (i.e. inserting a sharp instrument into the cranium to destroy the brain by rotated and back and forth movements). When performed competently, this method is fast and valuable, as discussed in Table 6 and detailed in Table 7, and can be used in all circumstances not requiring intact central nervous system samples.28,29 The method is rarely used on smaller fish such as zebrafish but can be performed as described in Table 8.
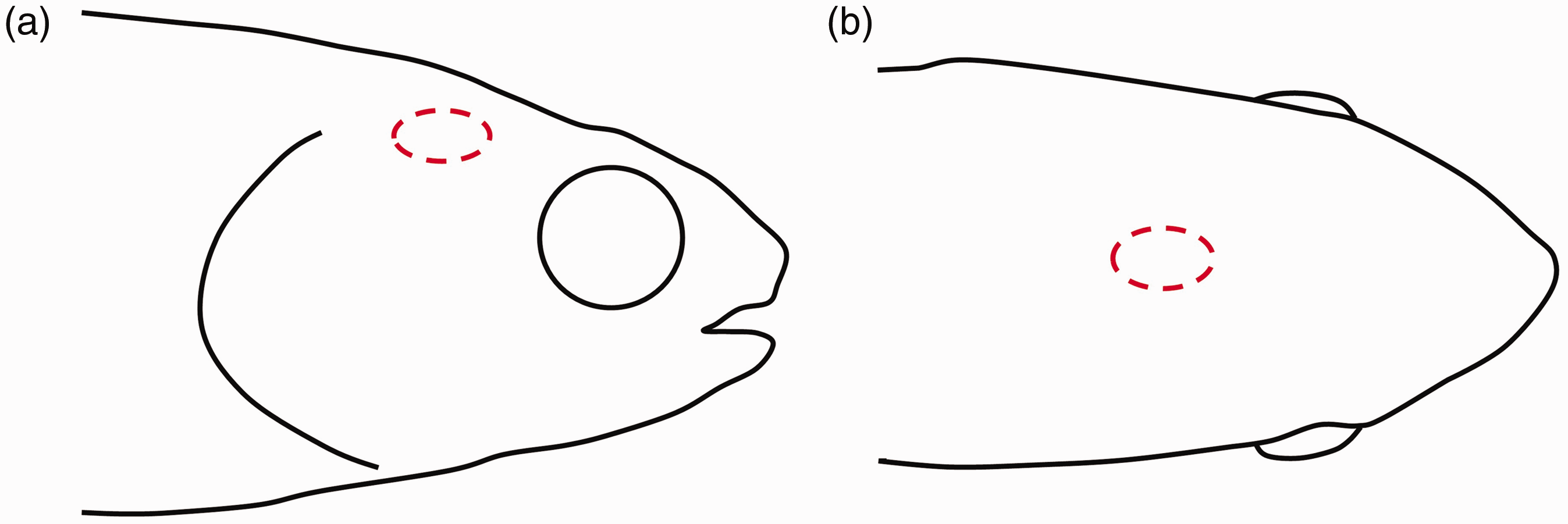
Outline of a trout head with the brain location circled in dotted red: (a) Lateral view and (b) Dorsal view.
Ideal euthanasia versus concussion followed by pithing of the brain.
Protocol for concussion followed by pithing of the brain of adult trout.

Protocol for concussion followed by pithing of the brain of adult zebrafish.

Decapitation
Used alone (i.e. without prior loss of consciousness), decapitation is generally not an acceptable method of euthanasia unless the procedure is ethically justified with a harm-benefit analysis of the programme of work considering the aims of the experiment and practicalities due to challenging conditions. For example, decapitation may be justifiable when the aim of the killing process is to collect fresh brain samples, uncontaminated by chemicals, in species unsuitable for hypothermic shock and docile enough not to require chemical sedation. The main caveat is that the fish brain may remain conscious for a longer period compared with concussion. Decapitation consists of the severing of the central nervous system, as close as possible to the brain, and of major blood vessels, inducing rapid loss of consciousness and exsanguination. Decapitation is mainly used as a technique to confirm death following the killing of the fish. Cervical dislocation, spinal transection, and spinal ablation are neurologically speaking no different to decapitation, but they induce a slower death due to the slower bleeding; these are therefore not recommended methods.
Overdose of anaesthesia
Considerations for all species
Despite the lack of publications demonstrating species-specific dose efficacy, euthanasia of laboratory fish can be performed by immersion in an overdose of anaesthetic. The operator must choose the suitable compounds, concentration, and exposure length, which all vary between fish species and are further confounded by a variety of physiological parameters (e.g. developmental stage, weight, body condition, species-specific behaviour) and environmental conditions (e.g. water temperature, hardness, pH, dissolved oxygen, salinity).5,20,30,31 The choice of compound depends as well on reported or detectable averseness of the compound for the species. 3 For a description of the most commonly used anaesthetics, readers are referred to the literature.30,31 When immersion is impractical or dangerous to operators (e.g. large fish), anaesthetic solutions can be applied directly to gills, with care to prevent unnecessary suffering due to restraining or high local concentrations. 30 The major disadvantages of overdose anaesthesia in large species are the slow speed of the process and the residue left in the fish (potentially compromising sample and data) and the environment. Apart for inducing an overdose, anaesthetics can be used to sedate fish prior to euthanasia by another technique, such as a physical method (e.g. concussion and decapitation) followed by a completion method.
Specificities for adult zebrafish
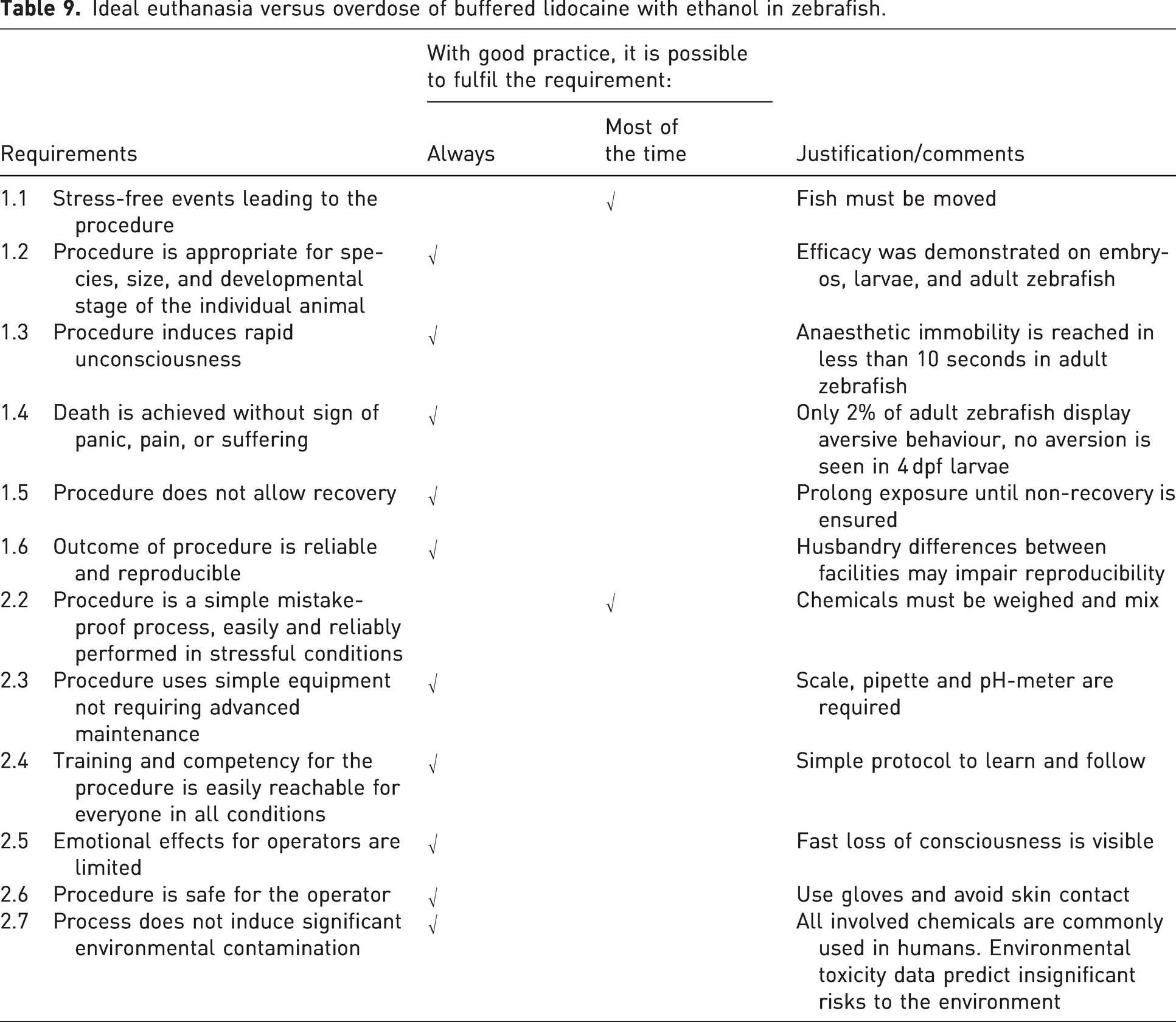
As mentioned above, the most common method for fish euthanasia is an anaesthetic overdose, with tricaine being the most utilized compound by far.2,4 For zebrafish, this is not ideal because tricaine can trigger aversive behaviour such as increased activity, piping, twitching, and erratic swimming when inducing anaesthesia and therefore euthanasia.15,17 Alternatives to tricaine have now been identified, allowing a more reliable, less aversive, and faster induction of anaesthetic overdose. 7 Indeed, adult zebrafish immersed in lidocaine hydrochloride (1 g/L) buffered with sodium bicarbonate (2 g/L) (with or without 50 mL/L ethanol 96%) lose opercular beat, righting and startle reflexes in less than 2 minutes. However, this protocol is only efficient when the pH of the anaesthetic bath is above 7.7, ensuring non-ionized lidocaine is available in high enough concentrations. The same study also revealed the relevance of monitoring the startle reflex and not to rely solely on more visibly obvious reflexes, because some drugs (e.g. tricaine, etomidate) only inhibit the startle reflex several minutes after cessation of other reflexes. Finally, signs of aversion with the buffered lidocaine were detected in less than 10% of adult zebrafish in these experiments and in only 2% of fish when ethanol was added to the anaesthetic solution. See Table 9 for details on how overdose of buffered lidocaine with ethanol meets requirements for ideal euthanasia of zebrafish. A protocol for adult zebrafish is proposed in Table 10.
Ideal euthanasia versus overdose of buffered lidocaine with ethanol in zebrafish.
Protocol for overdose of adult zebrafish with buffered lidocaine and ethanol.

Applications to zebrafish embryos and larvae
In the first days post fertilization, zebrafish gills are not fully functional and chemicals are mainly absorbed through the skin. 32 This limits the absorption of many compounds, compared with adults. Also, some compounds may not easily reach unhatched embryos. Therefore, anaesthetics used routinely in adult zebrafish may show different efficacy in early developmental stages. While few studies assess these differences, a recent study examined different methods of euthanasia for zebrafish embryos (<12 h and 24 h post fertilization (hpf)) and larvae (4 dpf), utilizing multiple laboratories across Europe to ensure reproducibility. 18 This confirmed that one hour immersion in lidocaine hydrochloride (1 g/L) buffered with sodium bicarbonate (2 g/L) and with 50 mL/L ethanol 96% added is so far the most reliable and least aversive protocol for overdose of anaesthesia in zebrafish <5 dpf. This suggests that the lidocaine protocol with added ethanol could be used in zebrafish of all developmental stages. However, immersion length would have to be adapted to the developmental stage.
Comparison of euthanasic methods
For early developmental stages, considering that hypothermic shock is inefficient, and concussion is not an option, only electrical stunning seems suitable when samples uncontaminated by anaesthetics are required. There is thus a need to understand better how electrical stunning may interfere with the quality of such samples. Taking the example of zebrafish, the main caveat with overdose of anaesthesia for embryos and larvae is the latency: one hour immersion does not allow fresh sampling. There may be room for improvement and shortening of protocols. Moreover, when fresh samples are not required, one hour immersion is acceptable only if fish do not show signs of discomfort. In practice, eggs and larvae could be left in the anaesthetic bath for hours before completion. An alternative is to use chemical destruction (e.g. bleach, ethanol, hydrogen peroxide). That may be suitable for very early developmental stages (e.g. zebrafish eggs) but not for larvae for which signs of aversion can be detected.
For adult fish, the choice of method (e.g. concussion, electrical stunning, and overdose of anaesthesia) should mainly depend on species characteristics and whether samples are required and for which purpose. Of course, the disadvantage of any anaesthetic protocol lays on fish and personnel exposure to chemicals. When experimental endpoints require anaesthetic-free samples, a physical method of euthanasia should be considered, such as concussion or electrical stunning, depending on species, local experience, and available equipment. In the case of adult zebrafish, hypothermic shock may also be considered.
For that particular species, the performance of the lidocaine protocol opens the debate of comparison between methods. Hypothermic shock may constitute a refinement to tricaine overdose, but lidocaine prepared as described above presents similar speeds to hypothermic shock (immobility or loss of equilibrium, respectively, is reached in less than 10 seconds).7,21 An added welfare benefit is that the fish experience is known and controlled with lidocaine: it is an induction of anaesthesia (i.e. analgesia, muscle relaxation, and loss of consciousness) and not a state of shock. As to whether hypothermic shock is at least as humane as an overdose of anaesthesia, the answer depends on the anaesthetic protocol at hand.
Regarding environmental impacts, the full life cycles of devices (e.g. ice maker and electrical stunner) and chemicals (i.e. raw material extraction, manufacturing, consumable distribution, use, disposal, end of life, and transport in between each step) remain under-investigated. For lidocaine, the environmental impact has been studied for its common use in humans, when the Predicted Environmental Concentration must remain inferior to the Predicted No Effect Concentration, below which no adverse effects on the environment are expected.33,34 There is also evidence that lidocaine is degraded in surface water, that it does not infiltrate into groundwater, and that its n-octanol/water partition coefficient indicates a low potential to bio-accumulate and to transfer from the aquatic environment into organisms in the wild.35–37 Finally, it is possible to carbon filter waste medicated water in the laboratory to control release of anaesthetics to the environment.38,39
Conclusion: how to apply these recommendations
The key to scientific progress is reproducibility of results. As euthanasia protocols may bias experimental data, standardization of methods across the international arena is crucial. One method of fish euthanasia does not suit all species, developmental stages, and experimental needs. Strain and water condition differences may also occur. All protocols must therefore be validated in-house before upscaling and set as standard. These protocols must consider all requirements for ideal euthanasia and completion. The overall aim is to apply the most humane methods in a standardized way.
Footnotes
Acknowledgement
The author(s) would like to thank Lynne Sneddon for providing advice regarding the concussion protocol for zebrafish.
Data availability
Original data are available on request.
Declaration of conflicting interests
R.M. is a director of Fish Management Systems Ltd., a company specializing in electric systems for fish management. The other authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical considerations
Ethical approval was not required for this publication.
Funding
FELASA supported the working group financially by covering meeting expenses and publication fees.
