Abstract

For centuries, scientists have been intrigued by the interactions between macro- and microorganisms. The holobiont concept sees humans and animals as holobionts, entities composed of the host and its associated microbial communities. 1 The gut microbiome, which includes communities and genetic material of bacteria, viruses, fungi, archaea, and protozoa, forms a significant part of this. These microorganisms aid digestion, immune regulation, and protect against pathogen invasion, but their imbalances are associated with various disorders like carcinogenesis and with inflammatory, metabolic, and infectious diseases. Recently, research has expanded beyond the gut, exploring the influence of the intestinal microbiota on other organs. 2 By promising insights into health and as a potential target for novel therapeutics, microbiome research is currently capturing both scientific and public interest. 3
In the 19th century, scientists were already debating whether life without bacteria was possible. German researchers Nuttall and Thierfelder (1895) demonstrated that guinea pigs could live without bacteria. 4 This pioneering achievement launched the new field of study named gnotobiology. Here, we focus on its application in commonly used laboratory rodents, mice and rats. Gnotobiotic conditions involve known microbial compositions and include germ-free animals devoid of all living microorganisms or animals colonized with defined ones. Early studies focused on microbial roles in nutrient metabolism, immune development, and overall impact on host physiology. Today, gnotobiotic animals provide controlled environments for studying the microbiome’s impact on host health and disease, enabling precise manipulation of microbial communities for experimental investigation.
Maintaining gnotobiotic animals requires strict isolation in specialized chambers called isolators, which provide a physical barrier and prevent the introduction of microorganisms. Sterilized equipment, food, and water maintain environmental sterility. Early challenges, such as understanding nutrition and the impact of sterilization on nutrients, hindered germ-free animal rearing. However, when these obstacles were overcome, successful breeding of germ-free rodents was established. 4
In Germany, a discussion has been initiated as to whether the germ-free status represents a burden for the animals, given the changes due to their sterile upbringing. Macroscopically, germ-free animals resemble those colonized with complex microbial communities. However, on a finer scale, germ-free animals have adapted to life in the absence of microbial stimuli (Figure 1). In the following, we explore this adaptation, drawing from our decades-long experience in the rearing of gnotobiotic animals. Importantly, experimental procedures on gnotobiotic animals that might involve pain, suffering, distress, or lasting harm and require project authorization, such as intentional colonization with harmful microorganisms, are not within the scope of this manuscript.
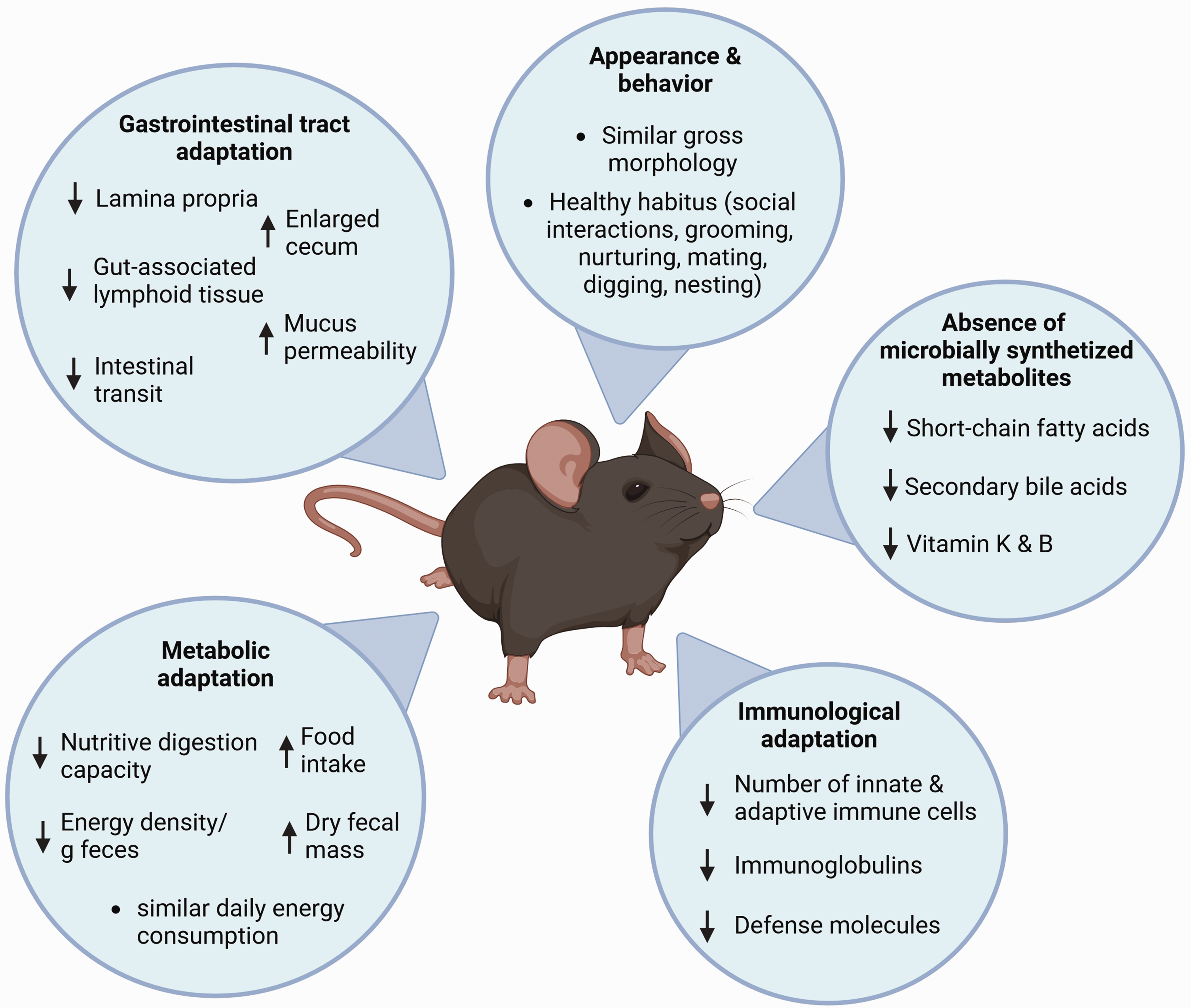
Adaptation of germ-free rodents to the germ-free environment. Illustration depicting general characteristics and prominent adaptations of gnotobiotic animals to the germ-free environment and the absence of microbial stimuli.
Appearance and behavior
Starting with the physiognomic aspects, the morphological appearance of germ-free rodents is comparable to their colonized conspecifics (Figure 2(a)). Numerous studies have documented differences in specific social activities and behaviors between germ-free and colonized rodents, highlighting the influence of gut microorganisms on brain function.5 –7 However, germ-free animals, across various mouse strains, consistently demonstrate a healthy habitus typical of mice, like interacting socially with cage-mates, 5 grooming and nurturing, 8 mating, digging, and nesting (Figure 2(b)). From our experience, gnotobiotic rodents show no elevated risk compared to colonized rodents in terms of nutritional status, response to handling, posture, alteration of skin or coat, cannibalism, or other criteria used for the severity assessment of rodents. We can even show that the percentage of total animal loss is notably lower in germ-free status rodents (Figure 2(c)). Furthermore, the reproduction rate of different germ-free rodents is comparable to that of colonized ones regarding the average litter size 9 (Figure 2(d)). Due to limited space in isolators, information on senescence under germ-free conditions is limited. However, a study by Gordon et al. showed that germ-free rodents have a longer life span than conventionally colonized control animals. 10
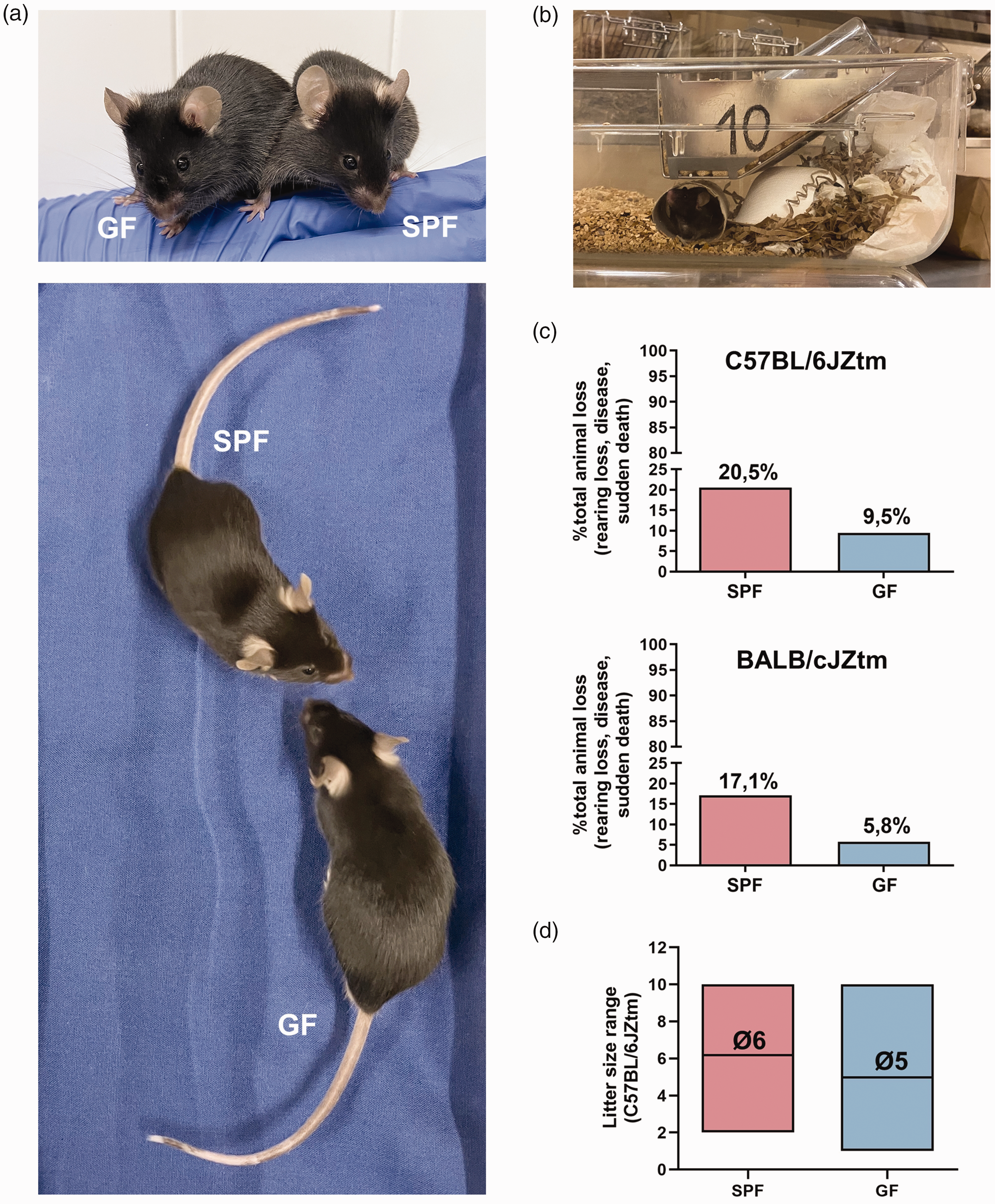
Comparison of biological features between germ-free (GF) and colonized (SPF) rodents: (a) representative photograph of morphological appearances of age- and sex-matched adult GF and SPF C57BL/6JZtm mice. (b) Representative photograph of a nest built by a GF C57BL/6JZtm mouse in an isolator. (c) Percentage of total animal loss including rearing loss, disease, and sudden death between SPF and GF mice in a 2-year period for two genetic backgrounds (C57BL/6J: SPF = 10,809, GF = 864, and BALB/cJ: SPF = 1308, GF = 378). Animals are inspected daily, and if pathological changes or illness are observed, they are euthanized upon reaching established humane endpoints and (d) Litter size range between SPF and GF C57BL/6JZtm mice. A total of 40 litters were analyzed for SPF and 152 for GF. The data on pup numbers per litter were collected from all litters produced by all breeding females within a defined period. This analysis was conducted independently of the number of litters each female produced and without considering the sequence of the litters. Both SPF and GF females were maintained in permanent breeding pairs (1:1). Breeding started at 8 weeks of age for SPF females and 7–9 weeks for GF females. The average age for breeding reassembly was 46 weeks for SPF females and 41 weeks for GF females. GF: germ-free; SPF: specific pathogen free.
Visceral morphology
It is important to differentiate between sterile visceral parts of the body and organs with close microbial contact. Sterile organs in germ-free animals closely resemble those in colonized rodents, such as the heart, liver, and kidneys. 4 However, the absence of microbial presence affects the visceral morphology of compartments usually colonized by microorganisms. These changes are primarily observed in the gastrointestinal tract and involve reduced lamina propria and gut-associated lymphoid tissues, 4 increased mucus layer penetrability, 11 and slower intestinal transit. 12 However, these changes are reversed upon microbial colonization. In the absence of microbes, the intestinal mucosa remains specialized for absorption, while in their presence, it develops a dual function of absorption and defense. 4 The major difference between colonized and germ-free rodents is an enlarged cecum, which is influenced by the accumulation of water-retaining macromolecules and varies across different genetic backgrounds of germ-free mice.4,13 Our recent study found that germ-free mice on a B6 background have larger ceca than those on C3H or BALB/cJ backgrounds, resulting in more pronounced abdominal dilatation in aged B6 mice. 13 In early gnotobiology, cecal dilatation hindered reproduction and could cause cecal torsion,4,14 leading to intestinal obstruction similar to that observed in humans. 15 Initially, reproduction of germ-free rodents was facilitated by reducing cecal enlargement through dietary adjustments such as limiting food intake, adding fiber 4 or anion exchange resin. 16 Moreover, cecal enlargement is reversible, as microbial colonization of germ-free rodents induces cecum shrinkage. 17
Today, cecal torsions in germ-free animals are rare, with less than 0.2% of cases occurring in our facility. This improvement is partially due to advancements in housing conditions and care. At our facility, germ-free animals are housed in plastic film isolators (Metall + Plastik GmbH, Radolfzell-Stahringen, Germany) within a controlled environment with a 14:10-h light/dark cycle. They receive pelleted, 50 kGy gamma-irradiated feed (Complete feed for mice – breeding (M-Z), V1124-927, Ssniff Spezialitäten GmbH, Soest, Germany) and autoclaved ultrapure water ad libitum. Annually, the facility produces around 3600 germ-free animals for scientific projects and line maintenance, typically up to 12 months of age.
Immune system
The maturation and development of the immune system rely on environmental stimuli, including dietary and microbial components. Consequently, the absence of microbes and reduced antigen exposure lead to adjustment in the immune system. Germ-free rodents exhibit differences in lymphatic organ morphology and size, reduced numbers of both innate and adaptive immune cells as well as immunoglobulins. 18 This outcome is expected, since energy investment in unnecessary defense systems would be a wasteful resource allocation in a microbe-free environment. Similar immune phenomena occur in laboratory rodents under specific pathogen free (SPF) conditions compared to those with a wild microbiota. Due to high hygienic standardization, the immune system of SPF mice is different and does not reflect the immunological status of wildling mice. 19 In humans, a similar effect was observed during the COVID-19 pandemic. Here, the application of non-pharmaceutical interventions such as social distancing, facemasks, and hygienic interventions has been associated with increased population susceptibility to respiratory infection due to immunological adaptation to low antigen exposure. 20 Nevertheless, most immunological changes associated with germ-free status can be reversed by microbial colonization. Exposure to microbes enhances immunological fitness by promoting the production of immune cells, lymphoid tissues, and defense molecules.18,21
Nutrition and metabolism
The enzymes from intestinal microbiota provide a significant advantage in efficiently utilizing dietary components. Commensal bacteria metabolize nutrients into products like secondary bile acids, short-chain fatty acids, and vitamins K and B, enhancing host accessibility to these vital compounds. Conversely, their absence reduces dietary requirements for vitamins A, C, and lysine.4,22,23 It is important to note that a standard laboratory rodent diet is sufficient to fulfill the nutritional requirements of germ-free rodents. However, sterilization processes such as irradiation or high temperatures destroy nutrients, and their loss in the diet must be adequately supplemented. Thus, using fortified diets to account for nutrient loss is crucial.
In germ-free rodents, the absence of microbial contribution prompts metabolic adaptations to optimize digestive efficiency. Consequently, germ-free rodents exhibit prolonged gastrointestinal transit. 12 Moreover, they balance reduced nutritive digestion capacity by increasing food intake. 24 Although germ-free animals excrete higher dry fecal masses with lower energy density, they exhibit no apparent daily energy consumption differences from those of colonized mice.24,25
The regulatory perspective of adaptation
Understanding the nature of changes under germ-free conditions is important for determining the legal status of maintaining these animals. German authorities classify germ-free maintenance in isolators as a “procedure” requiring project authorization, assuming the residual risk of contamination could lead to harmful phenotypes. This is attributed to the underdeveloped immune system of germ-free rodents, leading to increased susceptibility to infections. Additionally, morphological changes, particularly the enlarged cecum, are considered harmful, as they are cited as a cause of cecal torsions and reproductive problems due to reduced abdominal space.
An organism reacts to environmental stimuli by either adaptation or compensation. Adaptation involves a physiological change in function or structure that is advantageous. Conversely, compensation occurs when an impaired organ function is counteracted by another change, which may result in dysfunction. Decompensation, however, results in a pathological state and illness.
Studies have demonstrated that rodents adapt to the germ-free status without compensation or decompensation. When exposed to microorganisms, intentionally or unintentionally, gnotobiotic animals do not generally fall ill or die but adapt to the new conditions. High-efficiency barriers, like isolators, can keep gnotobiotic animals free from pathogenic and non-pathogenic microbes over extended periods. Even in rare contamination events, these microbes are part of the natural microbiome or environment without pathological consequences.26,27 Additionally, advancements in housing, such as the integration of positively pressured, individually ventilated caging systems, further reduce the number of animals affected when contamination occurs.
Furthermore, we have not observed a relevant risk of cecal torsions due to cecum enlargement. In contrast, overall disease and spontaneous mortality rates are lower under germ-free conditions, rather underscoring the reduced risk of harmful phenotypes. Additionally, the reproductive performance of germ-free mice is in the range of that of SPF-maintained mice.
Gnotobiotic animals are invaluable biological tools for dissecting microbial contributions to the host phenotype and vice versa. Specialist knowledge is required to maintain these animals, comparable to the expertise necessary for keeping different animal species, a simple example being guinea pigs that require vitamin C supplementation. This expertise is mandated by EU-Directive 2010/63 and ensured by the authorization and registration of breeders, suppliers, and users. In our opinion, there is no evidence that the germ-free status results in anything more than adaptation or that it causes burden (pain, suffering, distress or lasting harm) to the animals. Thus, it does not meet the requirement for project authorization under the directive.
Footnotes
Acknowledgements
We thank Anna Smoczek and the animal caretakers of the gnotobiotic unit at the Central Animal Facility of the Hannover Medical School for their excellent technical support. The illustration in figure 1 was created using BioRender.com.
Data availability
The data reported in this study are available within the article. The raw data are available on request from the corresponding author (
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical statement
This study did not require ethical board approval because it did not involve animal trials. The animal husbandry procedures were performed in accordance with the German Animal Welfare Legislation and covered by the permission of the local veterinary authorities, Lower Saxony State Office for Consumer Protection and Food Safety (reference no. 42500/1H).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the German Research Foundation (DFG) – CRC1371, project no. 395357507 awarded to MB, and FOR2591, BL 953/10-2 and BL 953/10-3 awarded to AB.
