Abstract
The use of germ-free (GF) isolators for microbiome-related research is exponentially increasing, yet limited by its cost, isolator size and potential for trans-contamination. As such, current isolator technology is highly limiting to researchers engaged in short period experiments involving multiple mouse strains and employing a variety of mono-inoculated microorganisms. In this study, we evaluate the use of positive pressure Isocages as a solution for short period studies (days to 2–3 weeks) of experimentation with GF mice at multiple simultaneous conditions. We demonstrate that this new Isocage technology is cost-effective and room-sparing, and enables maintenance of multiple simultaneous groups of GF mice. Using this technology, transferring GF mice from isolators to Isocage racks for experimentation, where they are kept under fully germ-free conditions, enables parallel inoculation with different bacterial strains and simultaneous experimentation with multiple research conditions. Altogether, the new GF Isocage technology enables the expansion of GF capabilities in a safe and cost-effective manner that can facilitate the growth, elaboration and flexibility of microbiome research.
Keywords
During recent years, the use of gnotobiotic animal experimentation in the study of host–microbial interactions has grown dramatically. This evolving interest reflects the realization of the importance of the interrelationships of microbes and animal health in multiple respects. This has led to the concept of the ‘superorganism’ or ‘metaorganism’, a functional entity encompassing the host and its symbiotic microbiota, which is dependent for its proper physiological function on both eukaryotic and prokaryotic partners for an optimal co-existence. 1 As such, research involving gnotobiotic animals may have an important role in the study of diseases, as these ‘sterile’ mice can reflect the host as a germ-free (GF) animal which is devoid of all demonstrable associated forms of life, and can then be left to its own resources. GF mice can be modified by mono- or poly-inoculation with known microbial organisms (gnotobiote) for the study of inter-microbiota relationships within the host. Finally they may be used in the mechanistic study of any external or endogenous factor that modulates or has modulated the mammalian microbial milieu (immune reaction, metabolism, cancer and more).
Based on this concept, gnotobiotic animals have been used in a wide variety of models demonstrating the critical importance of various microbiota partners and their interaction with the mammalian host such as, among others, metabolic diseases,2,3 autoimmune diseases,4–7 cancers 8 and central nervous system (CNS) diseases.9,10
Gnotobiotic animals are usually reared and continuously maintained using GF techniques under isolator conditions to prevent microbial contamination. Performing biological experimentation within GF isolators is technically challenging, and so is long-term animal handling for the purposes of breeding and colony maintenance within a GF environment. Furthermore, the use of conventional GF isolators has even greater disadvantages when research necessitates the simultaneous use of multiple mouse strains or parallel inoculation of GF mice with a variety of microorganisms. In order to prevent any cross-contamination of study groups, current isolator technique mandates that each group be individually housed in a different isolator, which occupies space and creates handling difficulties (often requiring gavage and repeated handling using thick plastic gloves with limited field of view). Simplifying the complexity of GF work in such experiments will enable multi-group experiments to be carried out in a cost-effective and efficient manner. In this article we demonstrate a new simple, room-sparing and easy-to-handle method for maintaining groups of GF mice for short periods of time using GF Isocages.
Materials and methods
GF mouse validation studies were performed independently by two different laboratories using distinct methods (a biological experimental group, in parallel with the Veterinary Resources Laboratory). Our major endpoint was to validate the Isocage technique for short period studies, and in addition we tested other aspects of the use of these cages, including:
Maintenance of stock mice in GF condition in the Isocage for at least 6 months. Performing the procedure with more than one technician (successful repeatability of the procedures). Attempting to facilitate some breeding activity in the Isocage. Experimental/research activity using three different groups of mice, each inoculated simultaneously with different microorganisms using the Isocage technology (with scientists and students involved with the standard procedures).
Animals
Newborn GF Swiss–Webster (with parental origins from Taconic, Germantown, NY, USA) male or female mice were maintained in semi-rigid isolators (Park Bioservices, LLC, Groveland, MA, USA) using classical GF techniques. At weaning age, the mice were transferred to irradiated Isocages (Tecniplast SpA, Buguggiate, Varese, Italy) with Sani-chips wood shavings (Teklad Aspen Sani Chips 7090A; Harlan Madison Wisconsin, USA), fed ad libitum irradiated and autoclaved chow diet (2018 irradiated global 18% rodent diet; Harlan Laboratories, Harlan Madison Wisconsin, USA), and given autoclaved water.
For conventionalization, the GF mice were orally administered by sterile gavage with freshly collected fecal matter from specific pathogen-free (SPF) donor mice, suspended in sterile phosphate buffered saline (PBS). Alternatively, the GF mice were co-housed with female SPF mice for four weeks. All the procedures were performed in accordance with the approved protocols using the Institutional Animal Care and Use Committee (IACUC) guidelines of the Weizmann Institute of Science.
Enumeration of bacteria by experimental group
Fecal matter was collected from conventionalized mice and their GF counterparts (five mice in each group), 1–30 days post conventionalization, and snap-frozen in liquid nitrogen. Samples were then homogenized in cold PBS using a tissue homogenizer. At the indicated time points, the mice were sacrificed by CO2 asphyxiation; and colons, ceca and spleens were harvested into cold PBS, followed by tissue homogenization. Fecal and tissue homogenates were then plated on LB agar (Difco) or Trypticase soy agar with 5% sheep blood (BD Biosciences, Franklin Lakes, NJ, USA), and cultured in liquid LB broth or chopped meat carbohydrate broth (BD Biosciences). Samples were cultured in a 37℃ incubator for up to 96 h under aerobic or anaerobic conditions (75% N2, 20% CO2, 5% H2), followed by enumeration of colonies on plates or optical density measurement.
Microbiological quality control by Veterinary Resources Laboratory
Initially,
After three months of negative results a decision to extend the testing of used cages was made using the following method.
Sample collections
Cages with 3–4 GF mice were changed every 14 days in a decontaminated bio-safety laminar hood. Upon cage changing, soiled bedding and water from the used water bottle was collected and placed in a sterile vial. From some Isocages, a sentinel animal was taken for cecum and oropharynx cultures. The sample vials and sentinels were taken to the lab directly. A control sample was taken from the SPF mice.
Incubation and testing
Sample handling was performed inside a bio-safety hood. A small amount of nutrient broth (just enough to moisten the sample) was added to the sample. Sterile swabs were used in order to mix the sample and to break down the fecal pellets. The same swabs were used to plate the sample on agar plates as listed below. Plates were incubated for seven days. More nutrient broth was then added to the sample (20 mL). Sample vial (loose cap) was incubated at 37℃. Re-plating was done on the next day and on days 4–5.
Testing of animals
Testing media and incubation condition.
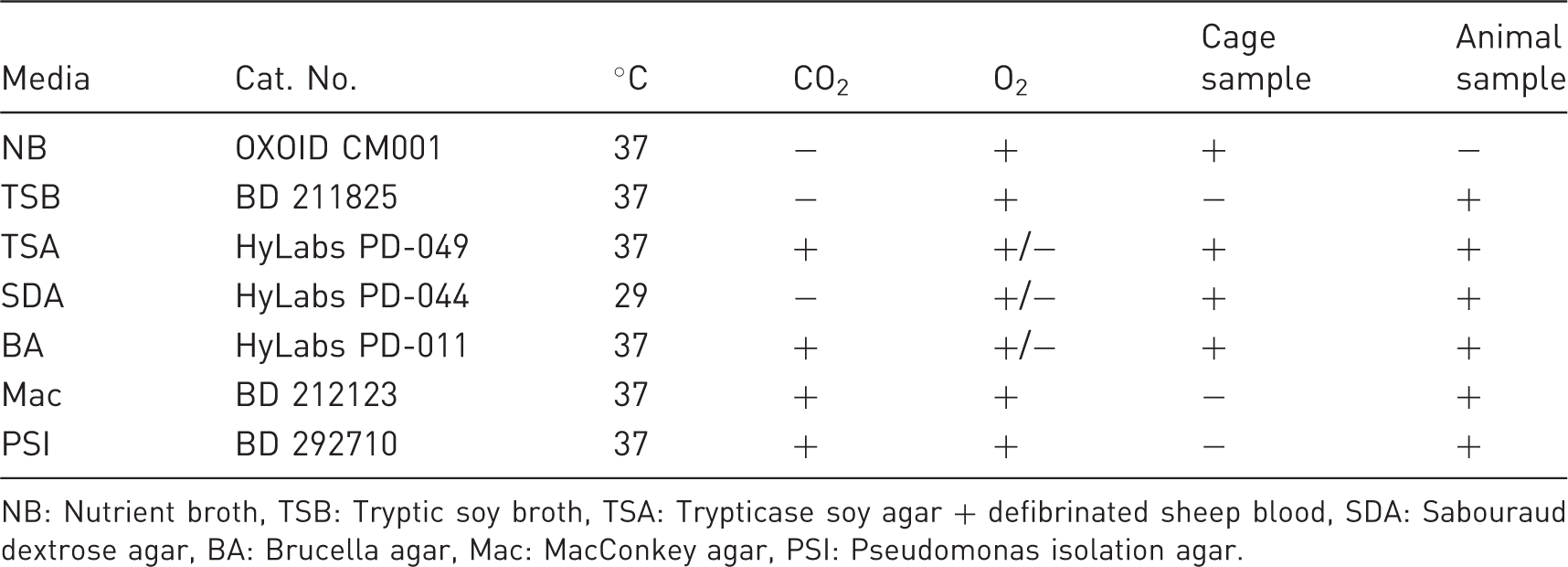
NB: Nutrient broth, TSB: Tryptic soy broth, TSA: Trypticase soy agar + defibrinated sheep blood, SDA: Sabouraud dextrose agar, BA: Brucella agar, Mac: MacConkey agar, PSI: Pseudomonas isolation agar.
Isocage
The Isocages© (Tecniplast SpA) were packed into two nylon bags with a
Results
Veterinary Resources Laboratory
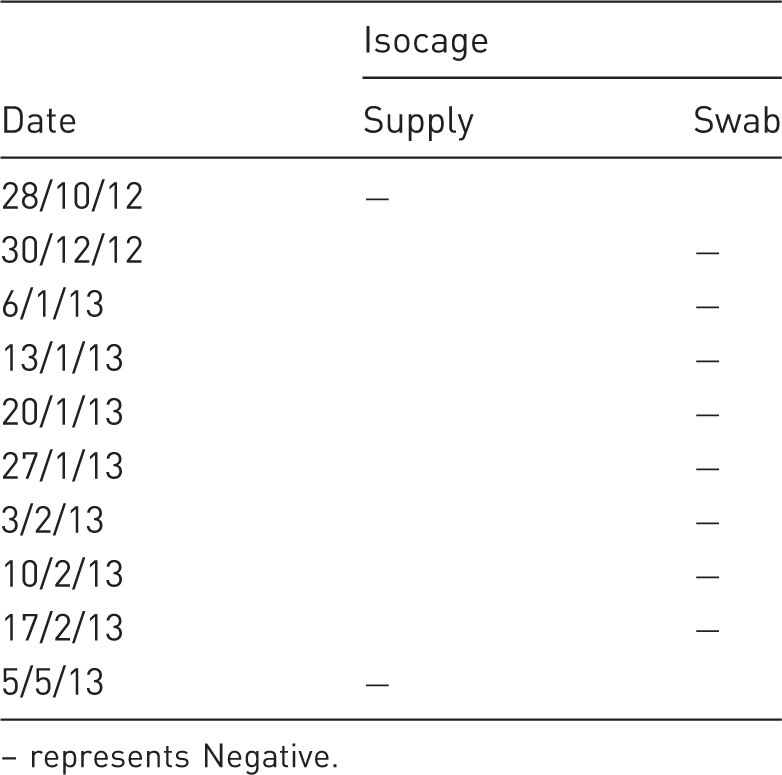
represents Negative.

represents Negative.
Sentinel animals (born in Isocages) cecum and oropharynx cultures from Isocages.
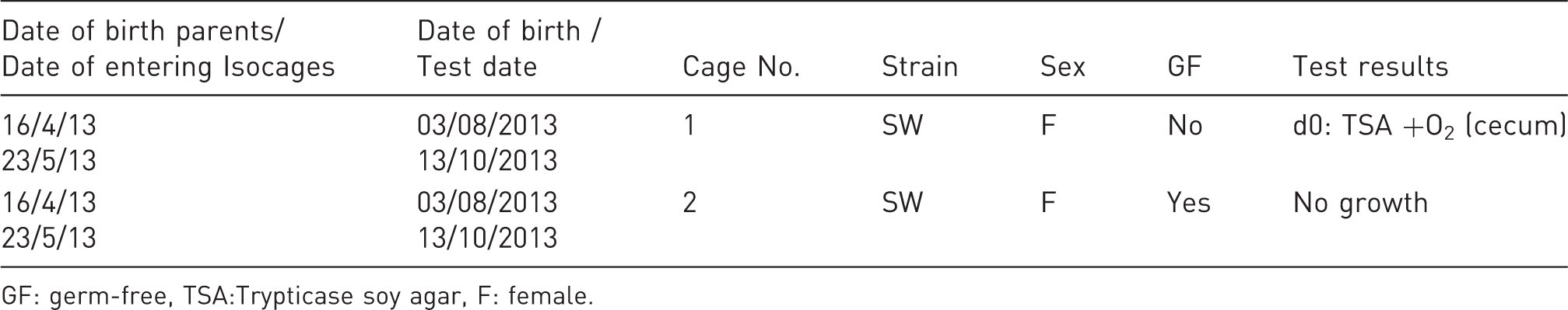
GF: germ-free, TSA:Trypticase soy agar, F: female.
Soiled bedding, water and fecal sample from the cages and animal cecum and oropharynx cultures from six different Isocages.

DOB: date of birth, GF: germ-free, M: male, BA: Brucella agar, TSA: Trypticase soy agar.
Experimental group
Enumeration of bacteria in fecal matter.
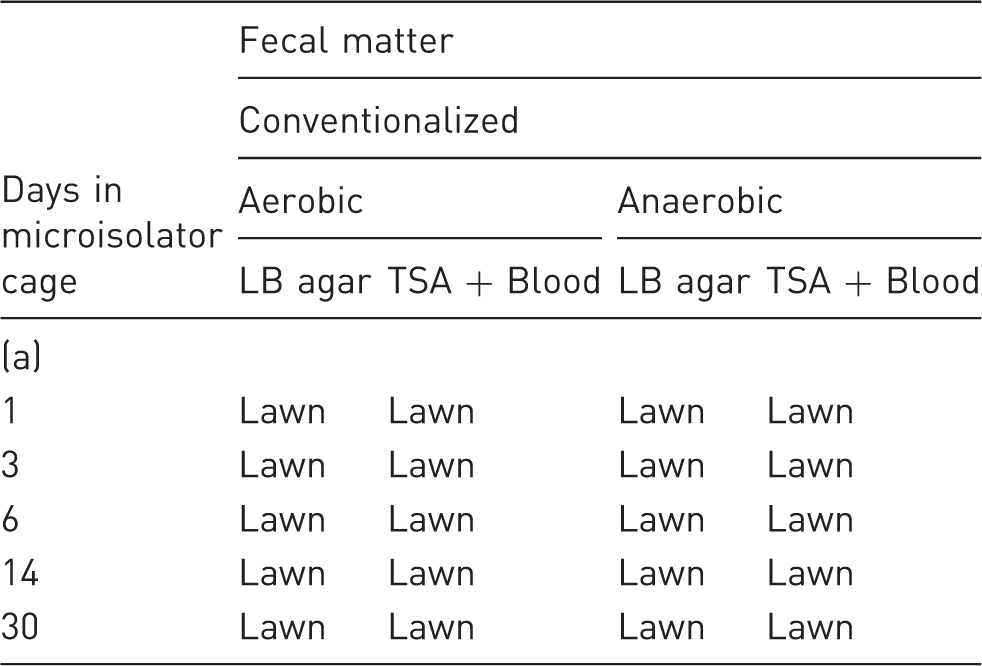
Fecal samples were collected aseptically and homogenized in the indicated time points. Plates and liquid media were grown at 37℃ under aerobic or anaerobic conditions. A lawn of bacteria was observed after overnight growth from conventionalized mice, but no colonies or change in optical density was observed in any fecal sample homogenate from germ-free (GF) mice. LB: liquid media (Luria-Bertani), TSA: trypticase soy agar, ND: no colonies were detected after 96h of incubation.
Discussion
Gnotobiotic animals are usually reared and continuously maintained with GF techniques under isolator conditions. While this is the best, and probably the only, method for breeding and maintenance of lines of GF mice for long periods of time, this technique is problematic in short time research studies, especially when the scientific need mandates multi-group and multi-condition experimentation with individual bacterial colonization of mice. The new possibilities described in this study, involving the use of individual Isocages, enable the use of multiple mutated strains of mice and simultaneous mono-inoculated animal groups. We found this technology to be reliable with an excellent rate of maintenance of germ-freeness when experiments were performed for a period of one month or less.
Although we tested the possibility of breeding and maintaining GF mice for longer periods in the Isocages (up to six months), we believe that at present this option is too complicated, as frequent changing of the cages for transfer of mice from dirty to clean cages, as well as additions of food or water, constantly endanger the GF condition, resulting in non-consistent rates of long-term Isocage sterility.
Enumeration of bacteria in tissues.
Colon, cecum and spleen samples were collected aseptically and homogenized in the indicated time points. Plates and liquid media were grown at 37℃ under aerobic or anaerobic conditions. LB: liquid media (Luria-Bertani), TSA: trypticase soy agar, CMCM: Chopped Meat Carbohydrates Medium, ND: no colonies were detected after 96 h of incubation.
Footnotes
Acknowledgments
We would like to thank Joana Bom, Instituto Gulbenkian da Ciencia, Lisbon, Portugal, for sharing with us all her knowledge regarding GF mice; Dr Inbal Biton for technical support; James Vitale, Taconic, USA and Frank Razzaboni, Park Bioservices, USA. CAT is the recipient of a Boehringer Ingelheim Fonds PhD Fellowship. EE is supported by Yael and Rami Ungar, Israel, Abisch Frenkel Foundation for the Promotion of Life Sciences, the Gurwin Family Fund for Scientific Research, Leona M and Harry B Helmsley Charitable Trust, Crown Endowment Fund for Immunological Research, Estate of Jack Gitlitz and Estate of Lydia Hershkovich.
