Abstract
Gnotobiotics or gnotobiology is a research field exploring organisms with a known microbiological state. In animal research, the development of gnotobiotics started in the late 19th century with the rederivation of germ-free guinea pigs. Cutting-edge achievements were accomplished by scientists in the Laboratories of Bacteriology at the University of Notre Dame (LOBUND). The primary goals of gnotobiotics were not only the development of the equipment required for long-term husbandry but also phenotypic characterization of germ-free animals. The first isolators were designed by Reynolds and Gustafsson as rigid-wall stainless steel autoclave-like chambers, which were subsequently replaced by Trexler’s flexible-film polyvinyl plastic isolators. Flexible-film or semi-rigid isolators are commonly used today. The long-term maintenance of gnotobiotic rodents is performed in positive-pressure isolators. However, to facilitate gnotobiotic experimental procedures, short-term husbandry systems have been developed. Gnotobiotic animal husbandry is laborious and requires experienced staff. Germ-free animals can be rederived from existing rodent colonies by hysterectomy or embryo transfer. The physiology and anatomy of germ-free rodents are different from those of specified pathogen-free (SPF) rodents. Furthermore, to guarantee gnotobiotic status, the colonies need to be regularly microbiologically monitored. Today, gnotobiotics provides a powerful tool to analyse functional effects of host-microbe interactions, especially in complex disease models. Gnotobiotic models combined with ‘omics’ approaches will be indispensable for future advances in microbiome research. Furthermore, these approaches will contribute to the development of novel therapeutic targets. In addition, regional or national gnotobiotic core facilities should be established in the future to support further applications of gnotobiotic models.
Introduction
The mammalian gut harbours approximately 1014 microorganisms (consisting mostly of bacteria but also including some protozoa, fungi, archaea and viruses) that coevolved over millions of years together with their host.
1
The entire collection of microorganisms inhabiting a specific niche is called the microbiota.
2
The two major bacterial phyla dominating the gut are
An interest in developing a detailed understanding of host-microbe interactions and their impact on host health and disease established a focus on gnotobiotic techniques and methods within the scientific community. Gnotobiotics or gnotobiology explores organisms, whose microbial state is strictly defined and controlled. The term is derived from the Greek words for ‘know’ (
The rise of gnotobiotics
The development of gnotobiotics started in the late 19th century. In 1885 and 1886 life scientists were confronted with two opposing hypotheses proposed by Louis Pasteur and Marceli Nencki. While Pasteur hypothesized that life is impossible without commensal bacteria, Nencki suggested that the absence of germs would prolong life and health. 11 To resolve this controversy, scientists started to develop techniques and equipment to isolate living animals as much as possible from their inner and outer environments.
Pioneering work was done in the period from 1885 to 1896 by George H. F. Nuttall and Hans Thierfelder at the University of Berlin. They delivered germ-free guinea pigs by Caesarean section and fed them sterile milk. This accomplishment demonstrated that life without bacteria is possible. However, when sterile vegetables were used as a diet, these animals gained less weight than the controls, indicating the importance of bacteria for digestion.12–14 Several years later, Ernst G. F. Kuester designed the first isolator prototype, in which he maintained germ-free goats for over a month. 15
In the middle of the 20th century several institutions dedicated to gnotobiotic work were opened. At the University of Lund in Sweden, Gösta Glimstedt established a gnotobiotic department and worked with germ-free guinea pigs. In the USA, James A. Reyniers founded The Laboratories of Bacteriology at the University of Notre Dame (LOBUND) and, together with Philip C. Trexler, became the world’s leader in germ-free research. Furthermore, a gnotobiotic centre was also established by Masasumi Miyakawa in Japan several years after those in the USA and Europe.11,14 Gnotobiotic institutions focused not only on the technical aspects of long-term maintenance and housing of germ-free animals but also on the phenotypic characterization of germ-free animals. Although many species such as chickens, rabbits and non-human primates were successfully raised to be germ-free, germ-free rodent colonies were still not established at that time due to difficulties in hand feeding. 11
In the 1940s, Bengt E. Gustafsson joined Glimstedt at the University of Lund and started to develop germ-free rat colonies. He first reported on hand-reared and weaned germ-free rats in 1946. 16 Later in 1946 Reyniers published the first detailed report on the technique used for rearing germ-free rodents.17,18 Hand feeding germ-free mice was even more challenging than hand feeding germ-free rats. Finally, germ-free mice were successfully weaned and have been available as reproducing colonies at the LOBUND institute since 1954.18,19 After germ-free rodent colonies were established, hand feeding was replaced with cross-fostering of neonates by germ-free foster mothers, which made the rearing of germ-free rodents easier and more accessible for scientists.
The first germ-free rodents showed growth retardation and premature death due to lack of appropriate nutrition. These phenotypes prompted studies addressing the cause of the nutritional deficiencies in germ-free animals. After years of research, which improved knowledge of important nutritional components, the diets of germ-free animals were fortified with supplements such as vitamins K and B, resulting in the capability of providing long-term housing under germ-free conditions. 11 By overcoming these obstacles, gnotobiotics pursued two important goals: providing a nucleus of pathogen-free animals for biomedical research and studying anatomical and physiological particularities in germ-free animals.
With advances in microbiome research, the interest in gnotobiotics has once again increased. Since 2000, the number of publications obtained using the PubMed search engine including terms microbiome, microbiota or microflora exponentially increased. The number of publications including these terms increased from five in the year 2000 up to more than 2000 published yearly since 2015. Together with methodological progress and the availability of various mouse models, gnotobiotics provides insights into the host processes regulated by the presence and composition of the intestinal microbiota. Furthermore, gnotobiotics allows the establishment of standardized conditions in which the functionality and causality of host–microbe–host interactions can be addressed to reveal important genetic, microbial and environmental factors shaping homeostasis or pathological conditions.
Gnotobiotic rodent husbandry
Long-term housing of gnotobiotic rodents is possible in positive pressure isolators (Figure 1(a)). Before plastics became broadly available, isolators were made from stainless steel and were large, heavy and difficult to handle. The first stainless steel isolators were developed by Reyniers and Trexler and were designed as autoclave chambers. Rigid steel isolators with steam-sterilized locks were also developed by Gustafsson and were used for decades. In the late 1950s, Trexler designed the first transparent and flexible-film plastic polyvinyl chloride (PVC) isolator.
20
These types of isolator as well as semi-rigid isolators are commonly used today.
Gnotobiotic rodent housing units.
Isolators are designed as closed, impermeable barriers with a sterile inner environment. They can be rigid- or flexible-wall isolators, and the material most commonly used today is clear PVC. Additionally, in gnotobiotic husbandry air pressure within an isolator is kept positive. This can prevent the entry of microorganisms through microscopic punctures on the isolator, but it also keeps a flexible-film isolator inflated without the need for a frame to support it. The main components of isolator construction have not changed much in the last 50 years, and include an isolation chamber, a port system, an air inlet and outlet filter, a blower that maintains positive pressure by pushing air into the isolator chamber and arm-length gloves.
Complete gnotobiotic husbandry of gnotobiotic animals is performed without direct contact between the outside environment and the animals. The critical parts of the isolator are the gloves and the transfer port. All procedures in the isolator are performed through arm-length gloves. The gloves can be made from different materials such as nitrile rubber, latex or polyurethane. 11 Furthermore, it is very important to inspect the gloves for any damage before each use to prevent possible contamination. The second critical point is the transfer port, which connects the sterile isolator chamber to the outside environment. The port is usually designed as a plastic or stainless-steel cylinder bordered by double doors. 11 Isolators fitted with a single-door port are also available; however, these isolators must be connected to a transfer isolator or specific cylinder for transferring materials from and into these isolators. All materials needed for animal maintenance as well as for the experimental setup are introduced into the isolator through the transfer port. Supplies are usually introduced by using a supply cylinder made from stainless steel and wrapped in a filter medium that allows steam penetration during the autoclaving cycle. 11 The cylinders are loaded with autoclave-resistant supplies such as bedding, food or nesting material, closed with biaxially-oriented polyethylene terephthalate film (such as Mylar® or Hostaphan®) and autoclaved. 11 Autoclaving protocols need to be optimized in each facility and adapted to particular needs and material properties. 21 Furthermore, all sterilization processes, including the autoclave cycles, need to be validated. After sterilization is confirmed, the cylinder can be docked onto the isolator port by using a PVC transfer sleeve that was previously sterilized by a highly effective chemical sterilant (such as peracetic acid, chlorine dioxide or 10% bleach) 22 to create a sterile tunnel between the cylinder and the isolation chamber. In our facility, 2% buffered peracetic acid is used as the main sterilant.
Isolator housing is optimal for long-term maintenance and breeding of gnotobiotic rodent colonies. Furthermore, it is also advisable to perform long-term gnotobiotic experiments in the isolators. However, because a single isolator represents one hygienic unit, it is only possible to keep experimental groups identical in their microbiological status within one isolator. Furthermore, because isolators offer open-cage housing, an introduced contaminant will rapidly spread to all cages. Moreover, at the end of the study or upon contamination the entire isolator needs to be disassembled, sterilized and made germ-free again. This procedure is time consuming and expensive. Therefore, several systems for short-term gnotobiotic rodent housing have been developed. These systems include static gnotocages such as Hannover gnotocages (Han-gnotocages), or airtight sealed individually-ventilated cage (IVC) systems such as the Tecniplast IsoCage P system. This cage technology represents a mini isolator at a cage level and allows animal handling in a class II biosafety cabinet.21,23 Nevertheless, the sterile handling procedure requires well-trained staff and strict adherence to the operating procedures to maintain sterility or unchanged microbiological status of the gnotobiotic animals. In our laboratory, the operating procedure involves cold sterilization of the biosafety cabinet and cage surfaces with an effective sterilant. The work is performed by two persons: a ‘sterile’ partner and a ‘semi-sterile’ partner. Both persons wear personal protective equipment including a sterile coat, sterile surgical gloves, a face mask and a hair bonnet. The semi-sterile partner is responsible for opening the cage and assists the sterile partner by providing sterile materials. The sterile partner is only allowed to touch items within the cage and sterile materials provided by the semi-sterile partner. However, due to the handling of materials and animals within the biosafety cabinet, the risk of contamination is higher than in the isolator. Moreover, each cage represents a separate hygienic unit. Therefore, these techniques enable work with multiple different gnotobiotic groups simultaneously with separation at the cage level. The Han-gnotocage is built from autoclavable rigid plastic sealed with a metal lid filled with a filter medium (Figure 1(b)). Air flow through the filter medium occurs naturally and is not driven by a ventilation system. In the isocage-system, the air flow is driven mechanically and enters the cage by passing through a high-efficiency particulate air (HEPA) filter (Figure 1(c)). Furthermore, when the cage is removed from its position, the air inlet and outlet paths are sealed, and no air circulation in or out of the cage is possible.
The major risk associated with gnotobiotic maintenance is contamination by environmental microorganisms; therefore, all aspects of husbandry need to be strictly monitored for sterility. Depending on the type of material that needs to be sterilized, different sterilization methods can be used such as autoclaving, irradiation, gas sterilization or cold sterilization by using liquid chemical sterilant.21,22 Furthermore, all sterilization methods need to be controlled and validated.11,21 In addition, experienced users need to perform preventative maintenance on equipment to avoid a breach of isolator integrity. Moreover, gnotobiotic animals need to be regularly tested, and the monitoring frequency needs to be adapted based on the number of transfers or material introduction. 21 The goal of microbiological monitoring of germ-free animals is to confirm the complete absence of microbes, whereas monitoring of defined colonized animals is needed to confirm the presence of the introduced species and the absence of all other microorganisms. The main contaminants are environmental bacteria or fungi; however, human skin commensals can enter isolators through holes in the gloves. Therefore, samples from the animals’ environment are highly relevant for hygienic monitoring. Microbiological monitoring of gnotobiotic animals is performed on collected bedding, food, swabs from various isolator positions as well as faecal samples. 21 Gnotobiotic mice should also be monitored for specific pathogens, for example those listed by the Federation of Laboratory Animal Science Associations (FELASA).21,24 Viruses, except those included in the FELASA recommendations, are not specifically excluded from germ-free animals. Therefore, it is recommended that the gnotobiotic colony be tested whenever a new virus has been discovered. 21 Furthermore, the probability that the gnotobiotic colony will be infected with pathogenic bacteria or fungi is very low. Based on the current published recommendations for monitoring germ-free rodents, bedding, food and fresh faecal samples should be collected and tested by culture every 4 weeks, and animals should be tested by full necropsy (including testing for the pathogens from the FELASA recommendation list) every 3–6 months. 21 Some facilities report that they monitor their germ-free colonies every 2 weeks or even weekly. The diagnostic techniques include macroscopic and microscopic examination as well as culture-based and molecular methods. 21 The presence of metazoan and protozoan parasites can be assessed by analysing cecal content or by using a tape test when animals are available for necropsy. For successful microbial monitoring, it is crucial that the samples are obtained under sterile conditions to prevent false-positive results. Detailed procedures and protocols for the microbiological monitoring of gnotobiotic rodent colonies can be found elsewhere.21,22
Rederivation and characteristics of gnotobiotic animals
Germ-free animals are rederived from existent rodent colonies. Once a new strain is rederived germ-free, the colony can be bred and maintained in the isolator. Furthermore, this colony can be used as a source of new pathogen-free colonies or be transferred to isolators or microisolator cages dedicated to experiments (Figure 2). Today, germ-free rodents are rederived by hysterectomy/Caesarean section or by embryo transfer. Detailed descriptions of these methods can be found elsewhere.22,25 In short, the hysterectomy method includes timed mating of the donor and recipient strain. During the procedure, the gravid uterus of the donor strain is removed and transferred into the isolator under sterile conditions using a dip-tank filled with disinfectants, for example iodine or 10% bleach.
22
In the isolator, the neonates are fostered by a germ-free foster mother until weaning.
22
During the embryo transfer method, two-cell embryos of the target strain are implanted into the oviduct of a pseudopregnant, germ-free female.
25
For this technique we recommend the use of a dedicated sterilized laminar flow surgical area that is connected to an isolator to ensure a fully sterile work process.
Application of the concept of gnotobiotic rodent husbandry.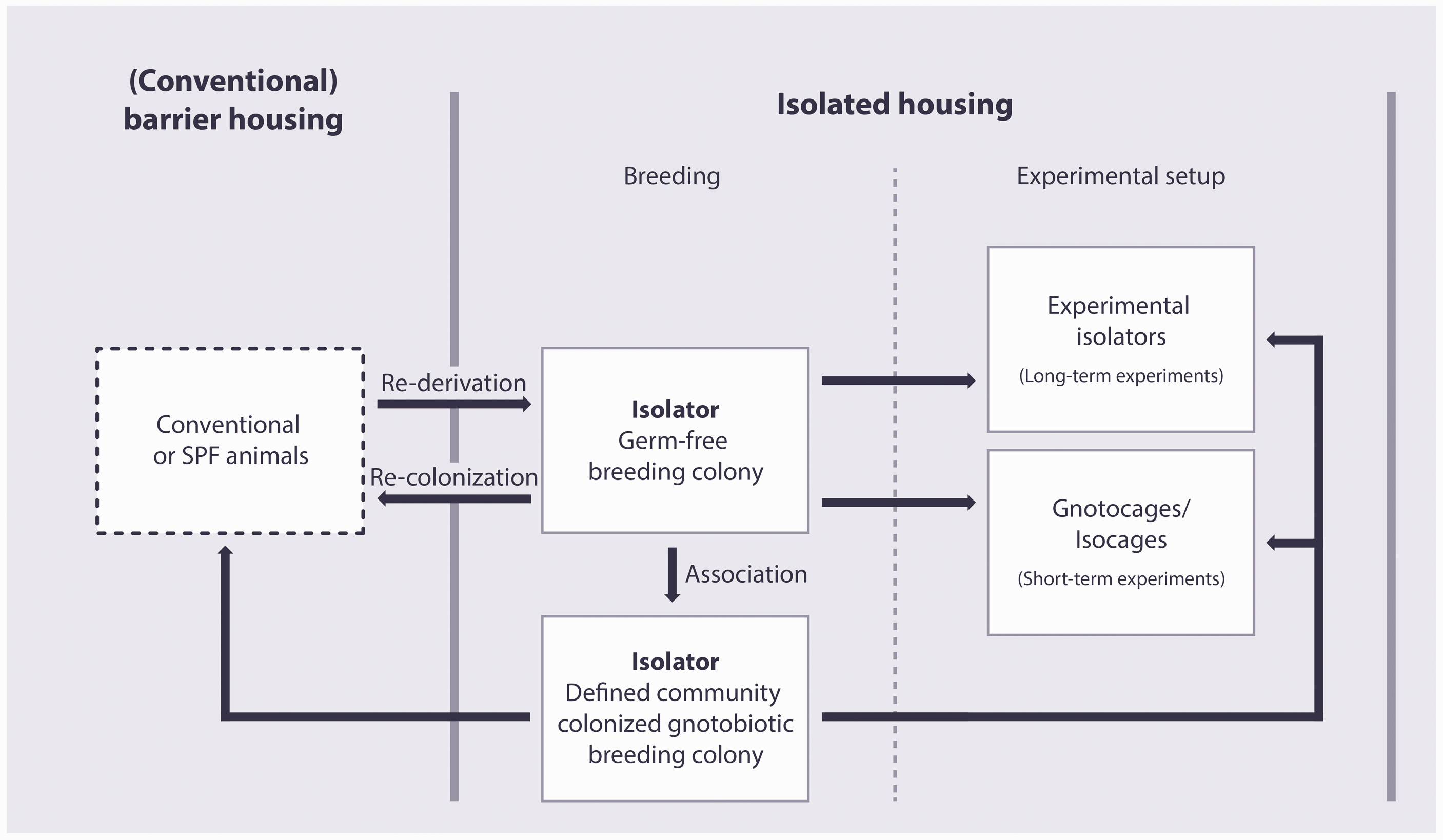
Life in the germ-free environment induces marked anatomical and physiological changes in rodent models. Germ-free animals have smaller hearts, lungs and livers. 26 However, the most prominent characteristic of germ-free animals is the cecum size, which can be up to five times larger than that of SPF animals. This cecal swelling occurs due to undegraded mucopolysaccharides, which attract water, and intestinal atonia. 26 Even though an enlarged cecum could affect the reproduction of germ-free mice, in our experience, the litter size of mice bred under germ-free conditions is similar to that of mice kept under SPF conditions. Furthermore, various mucosal parameters such as epithelial cell renewal, bowel motility and mucosa thickness are decreased in the absence of microbiota. The faecal pellets of germ-free animals contain more water and are softer. Moreover, the intestinal microbiota plays a role in the metabolism of bile acids and in the production of SCFA and vitamins. 26 The absence of microbial stimulation also has a prominent impact on the development of the immune system. 27 Germ-free mice display reduced numbers of gut-associated lymphoid tissues, poorly formed T-cell and B-cell zones in the germinal centres, reduced numbers of intestinal T cells and decreased IgA production.27–29 Detailed characteristics of germ-free immunological phenotypes are reviewed elsewhere.27–30
Gnotobiotic animals as a tool in microbiome research
Gnotobiotic animal models have become a powerful tool for investigating the functional effects of host-microbe interactions at the level of single, defined, or complex microbial communities (Figure 3). Furthermore, these strategies allow mechanistic approaches under highly standardized conditions. The protective or adverse nature of host-microbe interactions depends on the respective bacteria and the host susceptibility. Additionally, the disease phenotypes of various animal models may depend on the composition of their microbiota.31,32 Moreover, immense interfacility differences in microbiota abundance and composition have been reported.
33
Therefore, researchers should be aware that differences in the microbiota composition can impact the outcomes of their studies or modify disease pathologies.31,34
Gnotobiotic animals as a tool for analysing the effects of the microbiome.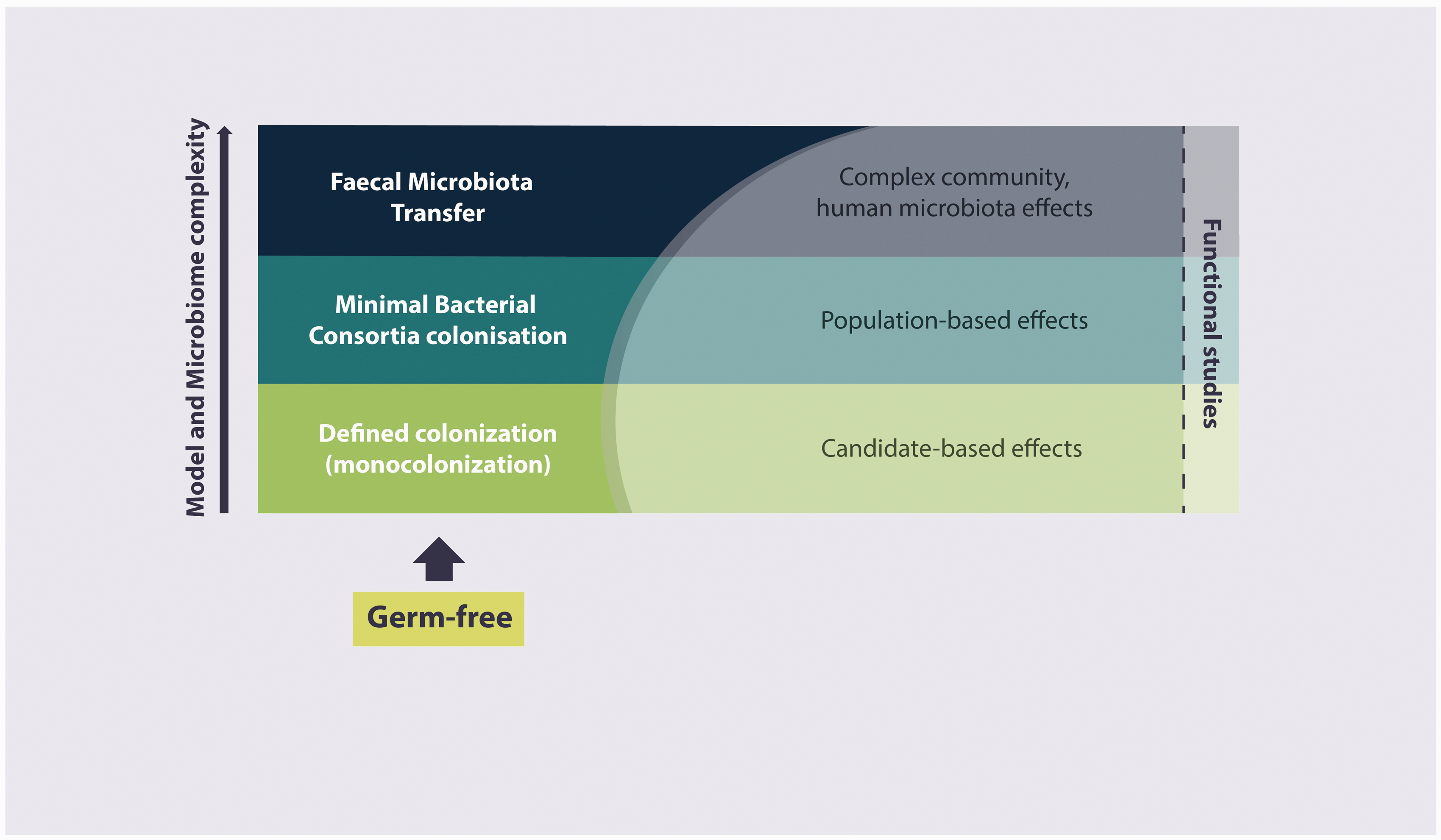
Introduction of one or a few microorganisms at a time provides valuable information on the causative effect of a particular microbe on the model phenotype. While this strategy might also be applied in conventional models, extraction of the molecular mechanism is usually hindered in animals with complex microbiome. For example, in mouse models of inflammatory bowel disease, association with
Associating mice with simplified microbial consortia contributes to the standardization of the intestinal microbiota and reduces interindividual variabilities and the complexity of the microbiota. Furthermore, mice associated with defined microorganisms can be morphologically and functionally similar to mice associated with complex microbiota.
10
A well-known example of a defined bacterial microbiota consisting of bacteria isolated from the murine gut was developed by Russell W. Schaedler.
44
This community was refined and standardized by Roger P. Orcutt, resulting in the generation of the ‘Altered Schaedler Flora’ (ASF), a model community of eight bacterial species that was mainly used by commercial rodent vendors.45,46 However, the ASF consortium is also widely used in biomedical research. This model community can be utilized for a wide range of studies including microbiological, immunological and metabolic studies.
46
As the ASF community represents a normal murine gut microbiota, this community can be used to gain knowledge about the gut ecology and spatial distribution of commensal representatives.
47
This model community can also be used to assess perturbations in the enteric microbiota upon infection with pathogens or colonization by other commensals.46,48 Furthermore, the ASF community can be used to evaluate the organism-specific impact on the host immune system.49,50 Recently, several novel human and mouse model communities were developed. The ‘Oligo Mouse Microbiota’ is a new model community composed of 12 bacterial species isolated from mice that represent members of the major bacterial phyla in the murine intestine. This microbiota provides partial resistance to colonization by
Furthermore, gnotobiotic mice can be associated with complex human or rodent microbiota. Though alterations in the composition may occur during xenogenic microbiome transfer, 58 the transfer of complex communities from healthy or diseased donors into gnotobiotic animal models can reveal pathophysiological effects of the respective microbiota. Several human or human-like disorders such as obesity, irritable bowel syndrome, Crohn’s disease-like inflammation or resistance to cancer therapy were shown to be transmissible by the transfer of dysbiotic microbial communities.59–64 This approach allows the impact and function of microbiota in human diseases to be studied using a model organism that recapitulates the human disease phenotypes and aetiology.
The future of gnotobiotics
Gnotobiotic animal models provide an excellent
As advances in the microbiome research field are tightly bound to the use of gnotobiotic animals, we believe that gnotobiotic facilities will be confronted with increased demand for the use of these models. Furthermore, as building and maintaining a gnotobiotic facility is expensive and requires a well-trained staff, we think that the establishment of regional or national core gnotobiotic centres is advisable. These core centres could serve as central repositories of animal models, rederive new models and provide training for scientists who want to use gnotobiotic models. For research groups that do not have suitable infrastructure for conducting gnotobiotic experiments, these experiments could be performed in a collaborative manner at the core facility. Furthermore, for researchers who have available isolators or short-term housing systems, germ-free or gnotobiotic animals could be sent to their facilities. Our facility successfully implemented this model in collaborative projects within the Germany-wide Priority Program ‘Intestinal microbiota’. Moreover, development of national funding programmes that would support gnotobiotic facilities as resource centres would be valuable in the future. In addition, the creation of a network of regional, national and international gnotobiotic facilities would allow the facilities to share their expertise and protocols as well as increase the availability of different models.
Conclusion
Gnotobiotic animal husbandry is laborious, expensive and requires well-trained staff. However, it provides a fundamental tool for analysing the impact of the microbiome on host physiology and disease development under highly standardized conditions. Association of gnotobiotic animal models with single microorganisms or defined ‘allogenic’ or ‘xenogenic’ communities will help unravel the mechanistic pathways driving human microbiota-dependent diseases. Therefore, the use of gnotobiotic animal models will probably grow continuously over the next few years, likely causing inundation of current gnotobiotic sites with increasing numbers of service requests. An approach to handling this demand in facilities lacking gnotobiotic capabilities might be through collaboration with existing gnotobiotics centres or through the local use of individualized maintenance systems (gnotocage/isocage systems) for experiments.
Footnotes
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the German Research Foundation (DFG) Priority Program SPP1656 [grant number BL 953/5-2].
