Abstract
When biomedical research investigates the human surgical situation in the need of a chronic course, it is more often possible to do so using large animal models. The use of farm animals always poses special challenges for the institution conducting the research in terms of infection prevention and occupational safety. Especially for the zoonotic disease coxiellosis it is important to be aware of the constant risk of pathogen introduction by small ruminants and to take appropriate precautions. In this way, personal injury should be avoided or at least be kept to a minimum in the event of infection since then sustainable zoonosis control can be immediately initiated. Using the example of a Q fever outbreak at a research facility, we want to share with this extended case report the importance of central emergency structures, provisions and the inclusion of relevant experts and disciplines in a crisis team. Its primary purpose is to support the affected facility and coordinate the implementation of necessary cleaning, disinfection and decontamination measures in close contact with the responsible local authorities. The aim is to inactivate the pathogen in a systematic and controlled manner in few steps of action only and to keep the interruption of the facility’s operations as short as possible.
Introduction
Outbreaks of Coxiella burnetii (Cb) infections in research institutions using sheep as a surgical model have been repeatedly reported in the past.1 –4 From the perspective of an affected institution, this paper derives a catalogue of measures that enable such institutions to deal with the precautionary protection of personnel, the elimination of the pathogen and the resumption of research operations in a structured and sustainable manner.
The zoonotic disease Q fever is caused by the Gram-negative, obligate intracellular bacterium Cb and has a wide range of hosts in addition to ruminants (cattle, goats, sheep) as the main reservoir.5 –7 In the small ruminant, which was the experimental animal in the present case, infection with the pathogen is usually asymptomatic and results mainly in excretion of the pathogen in vaginal mucus, in birth products, and in milk, faeces and urine.6,8 –10 When symptoms occur, they manifest as stillbirths, abortions or the birth of weak lambs.9,11 –13
Since Cb is a zoonotic pathogen, infected humans may show symptoms of acute infection such as fever, headache, chills and limb pain after an incubation period of 2–3 weeks, some of which may progress to pneumonia, hepatitis or even myocarditis 14 and chronic fatigue (Q-fever fatigue syndrome). 15 Infection occurs through contaminated secretions, foetal membranes or afterbirth associated with lamb birth or through contaminated carriers such as clothing, wool or animal faeces.4,9,11 In the presence of immunosuppression or pre-existing disease in human patients, chronic infection with organ manifestation may occur in about 1% of cases.14,16 Infection with the pathogen in early pregnancy can increase the risk of miscarriage and in late pregnancy the risk of premature birth or intrauterine foetal death.5,17 However, 50% of infections are asymptomatic. 14 Direct infection with desiccated pathogen, inhaled via contaminated dust particles or aerosols, is the main source of infection.9,11
Cb can exhibit high environmental resistance owing to its ability to create persistent forms and can thus remain infectious for many months under unfavourable conditions.11,18 It has been reported to spread for several kilometres around an outbreak event.19,20 The incubation period is inversely proportional to the infectious dose. 14
Persons with occupational exposure primarily to small or large ruminants, such as laboratory animal technicians, veterinarians, slaughterhouse personnel, farmers, shepherds and sheep shearers, may have evidence of passed-through infections on a larger scale.2,4,21 –23
Several rules and regulations must be considered when dealing with Cb in Germany. The direct or indirect detection of the pathogen, which indicates an acute human infection, is notifiable according to §7 clause 1 and §8 clause 2 of the Infection Protection Act (Infektionsschutzgesetz (IfSG), Germany 24 ). 14 The pathogen Cb is a biological agent of risk group 3 (Annex III of Directive 2000/54/EC) according to the Biological Substances Ordinance (BioStoffV, Germany 25 ), §3 Classification of biological agents in risk groups.
From 2001 to 2020, between 55 and 416 Q fever cases per year were reported in Germany according to IfSG (average 196 cases per year). 26
In this extended case report, we describe a zoonotic outbreak with Cb in a course of an experiment on pregnant sheep in 2021 that occurred in the preclinical research department of Charité-Universitätsmedizin Berlin and its clinical manifestation of Q fever in several individuals.
Animals
For a gynaecological research project performed in accordance with the German Animal Welfare Act and the EU Directive 2010/63/EU to be reported elsewhere, 12 pregnant Merinoland sheep (Sheep farm, Brandenburg, Germany) were placed in an animal husbandry facility.
Material and methods
Animal health monitoring
The preliminary animal examinations at the farm of origin one week before and the clinical examination upon delivery were unremarkable and fulfilled all requirements for placing the animals in the facility. A group of 12 sheep were transported to the facility between the 129th and 134th day of pregnancy in accordance with Regulation (EC) 1/2005. According to daily obstetrical examinations, the lambs were born between the eighth and the 17th day after arrival. The ewes were transported back to the farm of origin 14 days after delivery, except one ewe showing reduced general condition and increased recumbency over the next two weeks, which therefore remained at the facility for 10 more days under veterinary supervision until recovery allowed transport.
Contact examination
Nineteen days after the animals’ arrival, respectively 11 days after the birth of the first lamb, the first individuals from the group directly involved in the experiment (scientists, animal caretakers, veterinarians) developed flu-like symptoms, such as mild headache, fatigue and malaise, up to several days of high fever and other clinical symptoms. Three people required hospitalization for one to several days.
Because of the clustering of cases with similar symptoms a presumptive diagnosis of Q fever was made seven days after the first case. The blood sera of the contacts were tested by enzyme-linked immunosorbent assay (ELISA) and immunofluorescence technique in the Q fever conciliar laboratory (Landesgesundheitsamt Baden-Württemberg (LGA), Stuttgart, Germany) (Supplementary material supplement 7 online).
Based on facility access documentation, a total of 110 possible contact persons were identified and made an offer to participate in an occupational health screening performed by a company doctor according to §5(2) of the Ordinance on Preventive Occupational Health Care (ArbmedVV, Germany 27 ). Persons with possible direct contact with the animals or premises where the animals were kept were prioritized and serologically tested first, followed by all others.
Monitoring of titres was offered and carried out by the Occupational Medicine Centre in accordance with the recommendations of LGA. Those recommendations were based on the initial titre results. If the initial tests resulted in positive titres, the borderline cases were retested some weeks later, but most people were retested three and six months after the incident to rule out residual titres or a chronic course of the disease.
Examination of the environmental samples from the rooms
The facility’s large animal holding rooms were subject to annual routine examination for Cb by a polymerase chain reaction (PCR 28 ) (Supplement 5) method from environmental dust samples prior to the infection event. According to a standardized scheme (Supplement 1) the rooms were swabbed and tested by the reference laboratory of Lower Saxony State Office for Consumer Protection and Food Safety (LAVES, Hannover, Germany).
Environmental dust samples of the first sampling (Supplement 1) were collected from animal rooms and analysed by LAVES (PCR 28 ) (Supplement 5) (Figure 1).
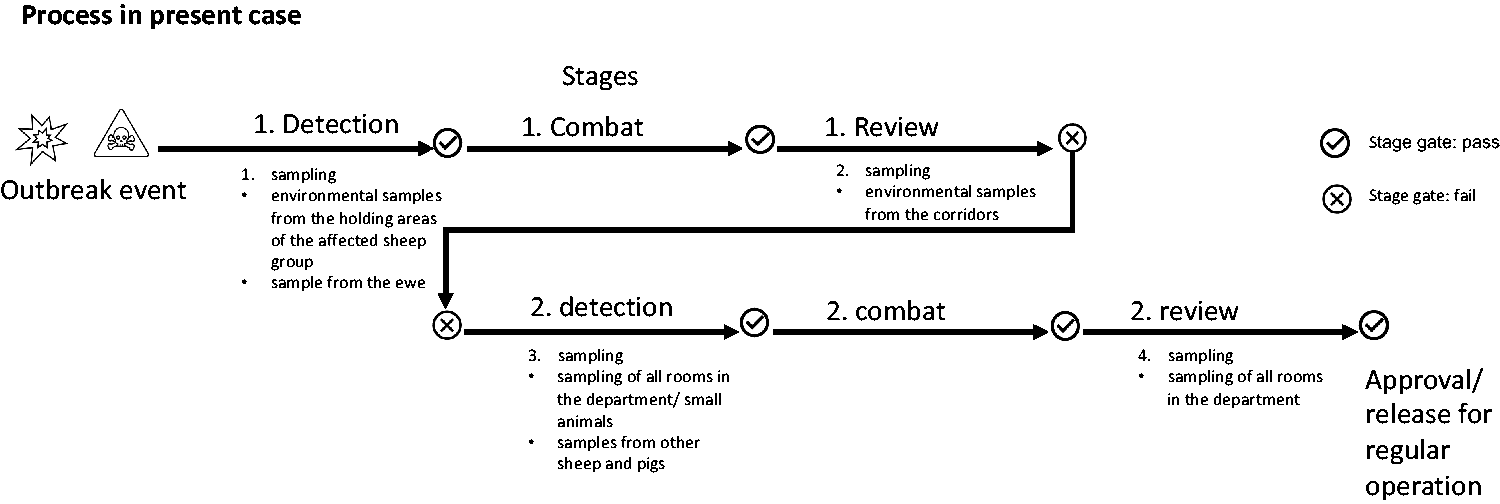
Process in the present case in the form of a flow diagram.
To confirm the initial suspicion, an additional litter sample collection (Supplement 2) was examined.
For the second sampling (Supplement 1) in the first review phase (Figure 1), the animal housing corridors walls were equally swabbed.
In a third sampling (Figure 1), all other rooms of the facility (small animal husbandry, storage, office rooms) were sampled (Supplement 1).
According to the requirements of the competent authority, State Office for Occupational Safety, Health Protection and Technical Safety (Landesamt für Gesundheit und technische Sicherheit (LaGetSi)), all disinfected rooms had to test negative twice by PCR to prove the effectiveness of the procedures used and to obtain official approval for the resumption of operations.
Personal protective equipment (PPE) under enhanced safety measures
In addition to the standard PPE (dedicated clothing and shoes for the area, gloves, FFP2 mask), a disposable protective overall (Tyvek®-500Xpert-Overall, DS SafetyWear GmbH, Lohmar, Germany) with battery-powered respiratory filter device and head hood (SR500, SR 561, Sundström Safety AB, Lagan, Sweden) with a combined particle/chemical filter was used. Sampling after the disinfection was performed using standard PPE.
Examination of large animals (sheep, pigs)
The relevant ewe was identified during the initial sampling (Figure 1) and examined for Cb by PCR detection 28 (LAVES) (Supplement 5) using a vaginal swab.
In the third sampling (Figure 1), samples were taken from remaining separated pigs by fur swabs in the spinal region of the animals and from sheep by vaginal swab.
Examination of small animals (mice, rats, guinea pigs, rabbits)
After positive findings of the second sampling (Figure 1), the entire small animal population was also examined for further risk assessment via collective fur swabs (spinal region) of the animals and dust swab samples from cages, exhaust air ducts of the individually ventilated cage (IVC) housing systems and the airflow (LAF) housing cabinets.
Blood samples from existing sentinel animals (mice, rats) were sent to FLI (Nationales Referenzlabor Friedrich-Löffler-Institut, Jena, Germany) for testing for Q fever antibodies by means of a modified ELISA 29 (Supplement 4) and, after painless killing of the animals, also spleen samples were sent to FLI for PCR detection (Supplement 6). In parallel, pooled spleen, lung and liver samples from the sentinel animals were sent to LAVES for PCR examination 28 (Supplement 5).
Investigation of the ventilation and air conditioning system of animal husbandry
To exclude pathogen spreading through the air conditioning system into the environment, dust swab samples were taken (Supplement 3) for PCR detection 28 (LAVES) (Supplement 5).
Carrying out the cleaning, disinfection and decontamination work
The entire facility was cleaned and disinfected in several steps and under increased safety precautions (see section on PPE above).
Cleaning of livestock rooms
Animal bedding and excrement were collected as infectious waste and disposed of separately. Animal rooms were mechanically cleaned using a high-pressure cleaner (Hochdruckreiniger, Alfred Kärcher Vertriebs-GmbH, Winnenden, Germany) and then surface-disinfected using a foam disinfection device (WOFA-MAT®, Kesla Hygiene AG, Bitterfeld-Wolfen, Germany). A German Veterinary Society-listed antimicrobial disinfectant with the active ingredients peracetic acid with acetyl hydroperoxide, hydrogen peroxide and acetic acid (Wofasteril® 2%ig, Kesla Hygiene AG) was used.
Wipe disinfection
Outside the animal holding rooms, all ancillary, operating and office rooms were disinfected by wiping with surface disinfectants containing sodium carbonate peroxyhydrate and 1h-benzotriazole as active ingredients (Incidin™Active, 2%ig, 60 min contact time, Ecolab Deutschland GmbH, Monheim am Rhein, Germany) respectively; in the case of sensitive surfaces, a double wipe disinfection with Incidin OxyWipe-S wipes containing hydrogen peroxide and glycolic acid as active ingredients (Ecolab Deutschland GmbH) took place as well as post-cleaning with surfactant-containing water to remove disinfectant residues, all performed by state-certified disinfectors of Charité Facility Management GmbH.
Gassing
For complete pathogen destruction to allow safe cleaning of the rooms, the facility was disinfected via H2O2 fumigation by a certified service provider (HIB, Berlin, Germany) based on a decontamination concept specially prepared for this purpose and a risk assessment for the execution of the decontamination work in accordance with the Biological Substances Ordinance and the IfSG (Biosafety Consultant, MLT Medizin- und Labortechnik, Frankfurt am Main, Germany), Technical Rules for Biological Agents No. 120 for ‘Laboratory Animal Science’ and recommendations of the Committee on Hygiene of the Society for Laboratory Animal Science (GV-SOLAS), for disinfection of animal rooms using formaldehyde or hydrogen peroxide (Supplement 8).
Establishment of a crisis team
Immediately after the zoonosis quality of the event became obvious, an operational and an extended crisis team were established. With both teams, it was possible to react to new developments in a timely manner and to deal with various parallel strands of action. This operational crisis team comprised an internal, operationally oriented group of people, consisting of the facility management, the person responsible for breeding and husbandry, the supervisor for animal care and employees of the veterinary service to make organizational preparations for containment, quarantine, diagnostics, hygiene, cleaning and disinfection measures, animal welfare, operational adjustments including the necessary employee briefings and direct communication with authorities. Depending on the facility size, an extended crisis team is to include authorized staff from the areas of facility hygiene, occupational medicine, facility management, occupational safety, biological safety, disaster control, legal department. Corporate communications approved the measures, provided legal and external resources, supervised official coordination with the supervisory authorities, press communications and informed the facility’s management.
Time sequence of the outbreak event
To streamline crisis management all actions taken during the event were assigned to three defined stages of action: ‘detection’, ‘combat’ and ‘review’ (Figure 2).
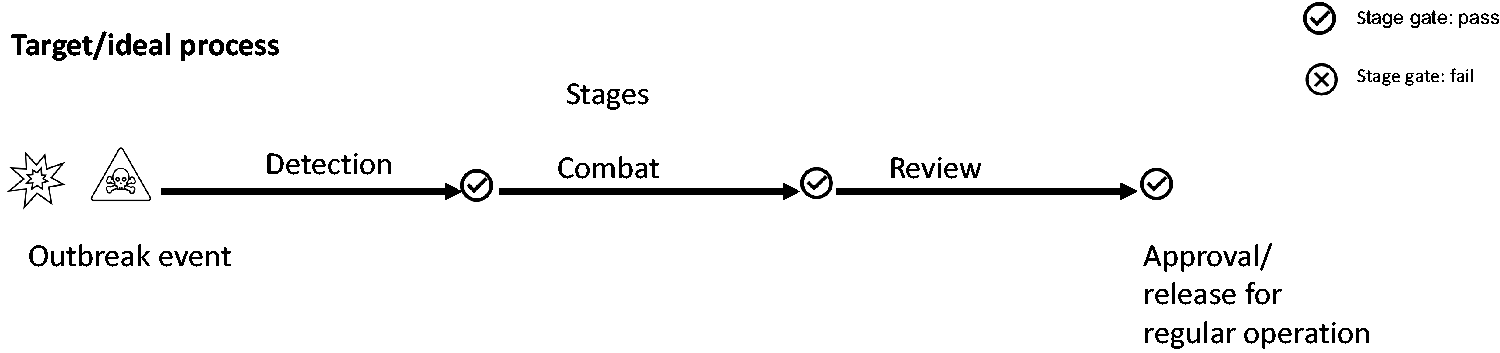
Target/ideal process of an outbreak in the form of a flow diagram.
‘Detection’ comprises activities related to the detection of pathogens in livestock and among contact personnel. In the ‘combat’ stage all disinfection measures for inactivation of infectious pathogens are summarized, including necessary euthanasia of livestock. The ‘review’ stage includes activities to verify prior stage actions and prepare the restart of operations, including follow-up detection.
Without prior knowledge and consistent application of this stage system, further sampling and decontamination measures were carried out at different times in the present case, which must be clearly separated from one another spatially and are associated with additional time and cost (Figure 1).
Results
Environmental samples
Routine sampling of animal rooms for Cb DNA was negative four months prior to the outbreak.
In environmental samples from the holding areas of the affected sheep group, Cb-specific nucleic acid sequences were detected in seven of seven submitted dust samples (Ct values 24.81 to 36.08) and in the submitted litter sample (Ct value 29.73). For polymerase chain reaction (PCR) detection of DNA, a Ct value up to 45 is considered positive. Since cultivation of the pathogen is lengthy and uncertain, the pathogen was detected in coordination with state reference laboratories in further course, mainly via DNA from environmental samples.
In the second environmental sampling (Figure 1), from the corridors, five of 12 dust samples were positive (Ct values 35 to 39). Subsequent sampling of all rooms in the department showed positive results in 25 of 84 samples distributed over the entire department (Ct values 28.96 to 41.12).
Contact persons
Twenty-one per cent (23/110) were positive with positive IgG/IgM antibody titres and acute infection confirmed by further follow-up. This group was directly exposed to the source of infection, that is, they had direct animal contact or were in the immediate vicinity of affected animals.
In 34.5% (38/110), positive IgG antibody titres had been found, with no relevant change in titre during the course, indicating previous infections, mostly unknown to the individuals, interpreted as serum scars. The proportion of the contacts who had no antibodies and remained seronegative was 44.5%.
All persons who tested positive were suspected by the LGA of having an acute infection and were advised to consult an infectious disease specialist to begin treatment as soon as possible. For this purpose, an offer was made to present oneself directly in-house. This was done in collaboration between the Occupational Medicine Centre, the Tropical Institute and the Infectious Diseases Clinic.
Large animals
In the first sampling of the experimental animals (Figure 1), the vaginal swab sample from the ewe tested positive for Cb (Ct value 33.59).
In the third sampling (Figure 1), no pathogen could be obtained by means of fur swab (pigs) and vaginal swab samples (sheep).
Small animals
In more extensive studies of the small animals in the third sampling (Figure 1), Cb DNA (Ct values 34 to 38) was detected in two of seven dust swab samples from the IVC systems and LAF enclosures and in eight of 29 pooled fur swabs (one pooled fur swab from all animals on an animal housing shelf) from the entire small animal population (mice, rats, guinea pigs and rabbits). Neither Q fever antibodies nor Cb-specific nucleic acid sequences were detected in the 52 blood samples and 16 spleen samples from the sentinel animals (FLI). Similarly, no detection was possible in 22 organ pool samples of spleen, lung and liver (LAVES).
Organizational measures after an outbreak of infection
To prevent pathogen spread, affected animal rooms were subjected to relatively negative air pressure compared with surrounding areas controlled by the ventilation system. Samples taken from filter stages in the exhaust air duct of the ventilation and air-conditioning system provided evidence that the pathogen had not spread to the outside environment.
Spread within the building through recirculation in the ventilation system (circulating air operation) could also be excluded, since owing to hygienic standards these systems are 100% fresh air systems.
Even before results were available the veterinary authority ordered that the sheep in question be returned to the farm of origin, as Q fever is a purely surveillance and notifiable animal category E disease (according to the categories of listed diseases under Regulation 2018/1882) without animal-related need for action.
After confirmation of the suspected Q fever (LAVES), the holding rooms of the affected sheep in the large animal husbandry area were quarantined by the responsible health department and the rooms were then cleaned and disinfected as described.
In coordination with animal welfare and occupational safety monitoring authorities, the remaining small animal population was euthanized due to the residual risk of infection to humans following department-wide detection of Cb DNA.
Cleaning, disinfection work
An effective room fumigation was demonstrated for all rooms by bioindicators and chemical indicators. The effective process was confirmed for all foam- or wipe-disinfected rooms by a subsequent, twice-negative examination of dust samples. One complete cleaning round, with sampling and processing samples in the lab averaged out to 3–4 weeks. In the case of positive results, this time-consuming process required variable consecutive rounds of cleaning and sampling of the rooms concerned to achieve two consecutive negative results. Due to the great heterogeneity of surfaces to be cleaned, which in many cases were not smooth, but rough and fissured, inactive residual DNA of the pathogen was to be expected, which further led to positive PCR results. All of this made up to eight consecutive rounds in the worst case of cleaning, sampling and testing necessary.
To further support pathogen DNA degradation for negative PCR detection, all rooms were treated by robotic ultraviolet treatment (UVD ROBOTER MODEL C, UVD Robots® ApS, 5260 Odense S, Denmark).
Revision of the operating protocols of the department
The following innovations were included in the department’s revised operating protocols:
A project-specific risk assessment and an assessment of required protection levels for large animal projects involving farm animals. For example, projects with direct access to the gravid ovine reproductive tract are excluded from further implementation at this site if there is a zoonotic risk within the animal population. Such projects refer to facilities with a higher protection level. This does not apply to minipigs and small animals, since these are sourced from controlled and standardized laboratory animal breeders. The review of operating instructions and risk assessment in accordance with the BiostoffV for targeted/non-targeted activities in laboratories, laboratory animal husbandry and biotechnology (§4 BioStoffV), the bio substances inventory as part of the documentation of the risk assessment (§7 para. 2 BioStoffV) and the matrix for combating an operational malfunction (§13 para. 1 BioStoffV) agreed with the supervisory authority have been supplemented in the form of a flow chart for the facility, in which the levels of action and dependencies are clearly shown (Figure 3).
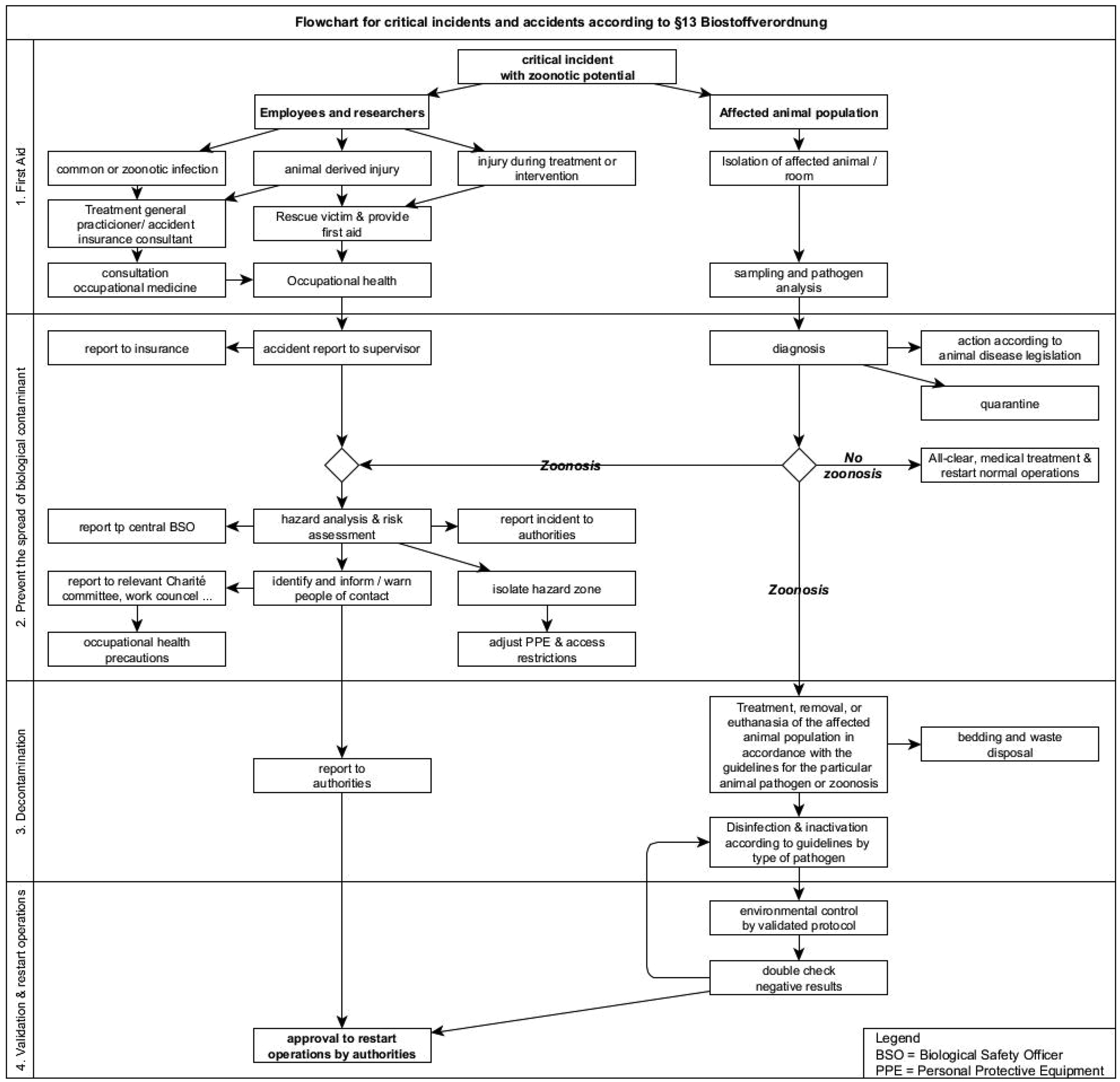
Flowchart of critical incidents and accidents according to §13 para. 1 Biostoffverordnung. BSO: Biological Safety Officer; PPE: personal protective equipment
Sourcing requirements for obtaining and maintaining farm animals for use in animal experiments were expanded following the outbreak:
Sourcing of large animals is restricted to the animal husbandry department from approved farms of origin only. Criteria for approval include species-appropriate husbandry, delivery in compliance with animal welfare requirements, possible boarding of laboratory animals with on-site veterinary care, controlled purchase of individual, tested breeding animals and thus traceable supply chains without contact with external animal stocks. Herd diagnostics including testing of faeces and dust samples from holding areas for Cb twice a year. A health certificate is required prior to delivery and placement for each individual animal, including serology for caseous lymphadenitis, Maedi-Visna, Chlamydia spp., Brucella spp., Leptospira spp., Salmonella spp. and Mycoplasma agalactiae and Cb, plus a general examination with pregnancy exclusion and an anthelmintic treatment by veterinary herd management. Quarantine for seven days after visual inspection on arrival by veterinary services to reduce stress after transportation followed by thorough general veterinary examination and immediate veterinary action in the event of any abnormalities.
Follow-up
Following two negative pathogen tests and submission of the updated operating protocol, which, in contrast to the previous situation, provides for a strict separation between the large and small animal areas, permission to restart operations was granted by the competent authority.
Six-month follow-up control of all rooms for Cb DNA (LAVES) produced only one positive result out of 109 samples (Ct value 35.89) and was interpreted as inactive residual pathogen signal.
Discussion
Infection with a zoonotic pathogen in animal research is an extremely challenging event for all involved, and experience has shown that only a structured and coordinated approach can keep the situation under control.1,2,4,21
A zoonotic outbreak requires immediate and targeted coordination of infection control measures. Therefore, the first step should be to establish a crisis team that includes all the expertise required for the situation. The involvement of the legal team and the corporate communications team is crucial, as proactive communication through early press releases and responding to press inquiries should be the gold standard to provide transparent information to the public to avoid speculation. A Frequently Asked Questions catalogue should be prepared at a very early stage to provide expert information as broadly and ad hoc as possible to corporate communications, which should be closely linked to the management of a facility. Within the operational crisis team, a dedicated contact person should be appointed to communicate and provide mandatory information to the authorities. This person should document the chronology of all actions, correspondence and results of all meetings for any post-event debriefings or possible legal proceedings. Finally, involvement of legal counsel is highly recommended to document an ‘event file’ for possible future litigation due to conflicting interests of occupational safety and animal welfare.
In retrospect, the outbreak event could be structured into three action stages: ‘detection’, ‘combat’ and ‘review’ (Figure 2), allowing to define milestones, set appropriate communication times with, for example, representatives of the authorities, and agree on essential decisions for the next phase in the presence of all relevant experts. While the ‘detection’ stage is initially concerned with the detection of the infection and its spread among the animal population and affected personnel, the measures for the ‘combat’ stage are derived from the detection. With detection of coaxial infection, all husbandry tasks that would normally be performed at level 1 must now be performed under appropriately heightened safety conditions. This includes preparatory measures such as bedding removal and pre-cleaning of surfaces prior to decontamination, particularly in large animal housing. The central element of the ‘combat’ phase is the controlled and meticulously documented inactivation of infectious pathogens in rooms, technical systems and existing user-specific room equipment by certified service provider specialists. In our case, a combination of repeated wipe disinfection followed by room and air-conditioning system fumigation was used and proven effective by biological and chemical indicators. Decontamination may also require the euthanasia of affected animal stock if health risks to personnel persist. At the end of this stage, the facility should be safe to re-enter.
The ‘review’ stage includes all activities that prepare for the resumption of operations. This includes follow-up detection and cleaning activities until, as in this case, indirect pathogen detection by PCR to a residual signal was requested that allowed to distinguish a new infection from a residual pathogen signal in the event of reinfection.
With prior knowledge, consistent application of this staging system, milestones regarding the biology of the pathogen defined early in the sampling phase for appropriate risk assessment, and straightforward infection management, setbacks and duplication of effort at various stages could have been avoided, as well as the extension of sampling to up to three rounds.
For higher safety level pathogens, such as Cb, appropriate disinfection equipment and PPE, as well as qualified, trained personnel, should always be available in large animal facilities to allow for ad hoc risk reduction measures. For example, a battery-powered respiratory filter device keeps breathing resistance low for the physically demanding work in large animal husbandry and prevents impaired vision behind the mask.
The chosen activated oxygen-based agents recommended by the German Veterinary Society used in a foam disinfector appear to have contributed to the reduction of the PCR-detectable pathogen signal in addition to pathogen inactivation in this case. However, up to eight consecutive disinfections were required in some rooms before a double-negative test for Cb could be obtained.
As mentioned above, a repeated negative PCR test was required by the supervisory authority for the renewal of the operating permit due to the complex method with low sensitivity of direct pathogen detection via cultivation.30 –32
It is important to note that despite correct inactivation of the pathogen, Cb-specific nucleic acid sequences may still be detectable in the affected premises. If mechanical removal or DNA-degrading effects of other measures are not feasible with reasonable effort, the goal should be to reduce the PCR signal to a level that reliably distinguishes future pathogen introduction from residual signals. A differentiated consideration of the Ct value together with additional criteria should be defined in an operational protocol to provide the necessary confidence to the authorities to distinguish a re-infection event.
An animal facility’s contingency plan should therefore always include a quarantine concept in the event of infection, including supply and disposal routes and staff access to the affected areas. The associated change to a higher level of protection should be considered from the outset. In accordance with §14 of the BioStoffV (Germany 25 ), the supervisory authority was provided with an adopted matrix of operational incidents (Figure 3), together with an optimized operational protocol for large animal husbandry with a focus on prophylactic and proactive exclusion of pathogens, analogous to the situation for small laboratory animals, which is currently not common for sheep under experimental conditions. The latter has been coordinated in close cooperation with the Q-GAPS research network (Q fever GermAn interdisciplinary Program for reSearch 7 ), which has itself published an updated Q fever guideline 33 covering recommendations for animal experiments on ruminants.
Finally, for animal experiments for which it is difficult to create the necessary conditions within a research institution, external collaborators capable of creating the necessary conditions should be identified to avoid incalculable risks and always comply with existing legal regulations.
Supplemental Material
sj-pdf-1-lan-10.1177_00236772241271028 - Supplemental material for Management of zoonoses in research institutions – lessons learned from a Coxiella burnetii outbreak case
Supplemental material, sj-pdf-1-lan-10.1177_00236772241271028 for Management of zoonoses in research institutions – lessons learned from a Coxiella burnetii outbreak case by Pia KS Ostach, André Dülsner, Anne Keil and Stefan Nagel-Riedasch in Laboratory Animals
Footnotes
Acknowledgements
We would like to thank all Charité staff involved, such as the FEM ARF team and all participants in the extended crisis team and supporting departments, who provided outstanding support under these difficult conditions. We would like to thank Prof. Runge from LAVES for advice and timely PCR analyses, Dr Mertens-Scholz from Friedrich-Loeffler-Institute and Prof. Fischer from Q fever conciliar laboratory for the support.
Data availability statement
Data is available from the corresponding author.
Declaration of conflicting interests
The author(s) declares no conflict of interest with respect to the research, authorship and/or publication of this article.
Ethical statement
Our study did not require an ethical board approval because it did not contain human or animal trials.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
