Abstract
Q fever is a worldwide zoonosis caused by Coxiella burnetii that can lead to abortion, endocarditis, and death in humans. Researchers utilizing parturient domestic ruminants, including sheep, have an increased risk of occupational exposure. This study evaluated the effectiveness of our screening protocol in eliminating C. burnetii–positive sheep from our facility. From August 2010 to May 2018, all ewes (N = 306) and select lambs (N = 272; ovis aries) were screened twice for C. burnetii utilizing a serum Phase I and Phase II antibody immunofluorescence assay (IFA). The first screen was performed by the vendor prior to breeding, and the second screen was performed on arrival to the research facility. Ewes that were positive on arrival screening were quarantined and retested using repeat IFA serology, enzyme-linked immunosorbent assay, buffy coat polymerase chain reaction (PCR), and amniotic fluid PCR. The overall individual seroprevalence of C. burnetii in the flocks tested by the vendor was 14.2%. Ewes with negative Phase I and Phase II IFA results were selected for transport to the research facility. Upon arrival to the facility, two (0.7%) ewes had positive Phase I IFA results. Repeat testing demonstrated seropositivity in one of these two ewes, though amniotic fluid PCR was negative in both. The repeat seropositive ewe was euthanized prior to use in a research protocol. No Q fever was reported among husbandry, laboratory or veterinary staff during the study period. Serologic testing for C. burnetii with IFA prior to transport and following arrival to a research facility limits potential exposure to research staff.
Introduction
Q fever is a worldwide zoonosis caused by the intracellular bacterium Coxiella burnetii. In humans, C. burnetii infection may be asymptomatic, or it may present as an acute, febrile, influenza-like illness. 1 , 2 Untreated, however, Q fever can have severe sequelae, including endocarditis and even death. 1 During pregnancy in women, Q fever has been associated with obstetric complications, including intrauterine growth restriction, premature birth, and intrauterine fetal demise leading to abortion.3–5 Emphasis has been placed on decreasing the risk of transmission, as there is no vaccine for humans commercially available in the USA. 2
Occupational exposure to parturient domestic ruminants (largely cattle, sheep, and goats) remains the primary source of transmission from animals to humans. 6 , 7 This transmission occurs via inhalation of aerosolized bacteria, which can persist in the environment for months to years following excretion in urine, feces, and uterine tissue from infected animals. 2 , 6 Farmers, veterinarians, husbandry staff, and abattoir workers are at a high risk of exposure. 2 In addition, laboratory personnel working with domestic ruminants for research purposes are at an increased risk.8–10
Sheep remain the most utilized large animal for studying in utero procedures and treatments due to the large fetal size, long gestation, tolerance of human handling, and ability to sustain pregnancy after multiple uterine surgeries. 11 Yet, the nature of the in utero research means that research and husbandry staff are in contact with the amniotic fluid as well as uterine and placental tissue that carries the highest bacterial concentration of C. burnetii in infected animals. 1 Outbreaks of Q fever have occurred in staff at research facilities where sheep are utilized. 9 ,12–15 Further, because C. burnetii can be aerosolized, the occupational risk extends to those working near or visiting the animal facility. 14
With seroprevalence of C. burnetii in sheep ranging from 2.7% to 16.5% in the USA, animal and personnel screening are recommended to limit occupational exposure to Q fever. 2 , 16 , 17 The US Centers for Disease Control (CDC) published guidelines in 2013 for occupational personnel screening, facility cleaning, and education in biomedical health research facilities with animals at risk for C. burnetii infection. 2 While the CDC suggested that an animal surveillance program requiring two negative serologic tests would help to “reduce the likelihood of infection,” no specific protocol was put forth. The Office of Research: Campus Veterinary Services at the University of California, Davis (UCD), has partnered with multiple research investigators to care for pregnant ewes through in utero surgeries and subsequent parturition. We have developed and implemented a protocol to minimize the risk of Q fever exposure to research and husbandry personnel. The primary objective of this retrospective study was to evaluate the effectiveness in eliminating C. burnetii positive sheep from our facility. The secondary objectives were to assess the sensitivity of serologic immunofluorescence assay (IFA) testing for C. burnetii and to determine the seroprevalence of C. burnetii in sheep in Northern California.
Methods
From August 2010 to May 2018, all ewes and select lambs (ovis aries) that were part of multiple research protocols at the UCD were tested for C. burnetii.18–31 No additional animals were obtained specifically for this study. Sheep were of various breeds, including Dorper, Merino, and Polypay, and were all female due to use of parturient animal models for all of the protocols. All research was approved by the UCD Institutional Animal Care and Use Committee (protocols 19676 and 19722). The UCD is accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International, and all animal care is in compliance with the Guide for the Care and Use of Laboratory Animals. All procedures were performed in the large-animal core surgical unit within the UCD animal program. All animals were housed in an enclosed barn within the UCD large-animal facility, and maintained in pens partially bedded with pine shavings on a textured concrete floor. The maximum number of sheep in each pen was five. Animals were kept on a 12:12 light/dark cycle. However, environmental lighting was visible through the barn windows that varied based on the season. Temperatures were subject to environmental changes, but at times where the temperature was <55°F, additional heating lamps were activated, and at temperatures >85°F, an internal facility fan and individual pen misters were activated. Water was provided via automatic troughs with city tap water. Alfalfa hay, pellets, and sheep grain were available as a twice-daily feed based on reproductive status. Food was provided in an open trough, and sheep had access to this throughout the entire day. Broom brush heads were provided as enrichment. Euthanasia was performed at completion of the protocol with a lethal dose of intravenous pentobarbital sodium and phenytoin sodium solution (Euthasol Euthanasia Solution; Virbac Animal Health, Carros, France) and bilateral thoracotomies.
Two vendors supplied time-mated ewes during the study period. The vendors bred procedure-naïve sheep via artificial insemination and controlled internal drug release (CIDR) synchronization. Ewes underwent serology testing prior to transportation to the research facility. Following confirmation of seronegativity, breeding proceeded, and pregnant ewes were transported to the UCD large-animal facility at 60–70 days of gestation where they were quarantined for an intake assessment that included a physical exam, complete blood count, ketone testing, and a repeat serology test. The majority of vendor serology (97%) and all of the arrival serology was performed at the UCD Veterinary Public Health Laboratory. The time interval between the vendor serology tests and the repeat serology test at the UCD ranged from 2 weeks to 3 months, depending on the timing of transport. Serology testing at the UCD was performed using Phase I and Phase II antigen-detecting IFA that were validated in humans (Fuller Laboratories, Fullerton, CA). A 1:128 dilution was used to determine the presence of antibodies against either Phase I antigens, which are associated with high virulence and chronic Q fever, or Phase II antigens, which are avirulent and present in acute infection. Such a dilution was selected to reduce the risk of Phase II false-positive tests, as seen using a lower (1:64) dilution. 32
Seronegative ewes were selected for the planned research protocol. To handle the ewes, staff and investigators changed into scrubs and shoes that were used only at the UCD large-animal facility. There was no additional required personal protective equipment to handle the ewes such as masks or respirators. Operating investigators used the sterile attire required for surgery, including a surgical face mask.
Seropositive ewes were quarantined and underwent repeat IFA serology, enzyme-linked immunosorbent assay (ELISA), buffy coat C. burnetii polymerase chain reaction (PCR), as well as amniocentesis for amniotic fluid PCR starting in 2018. ELISA was performed at the California Animal Health & Food Safety Laboratory System (IDEXX BioAnalytics, Westbrook, ME). Serologic testing with IFA is the most commonly used serologic method of diagnosis in the USA. 2 IFA testing kits are commercially available and allow more clear cutoffs for positive results compared to ELISA. PCR (proprietary primer; UCD Comparative Pathology Lab) is best for detection in cases of acute infection in humans. 2 Ewes that were positive on any single repeat serology testing or with PCR were either returned to the vendor or euthanized with disposal equipment using appropriate biohazard safety (Figure 1).
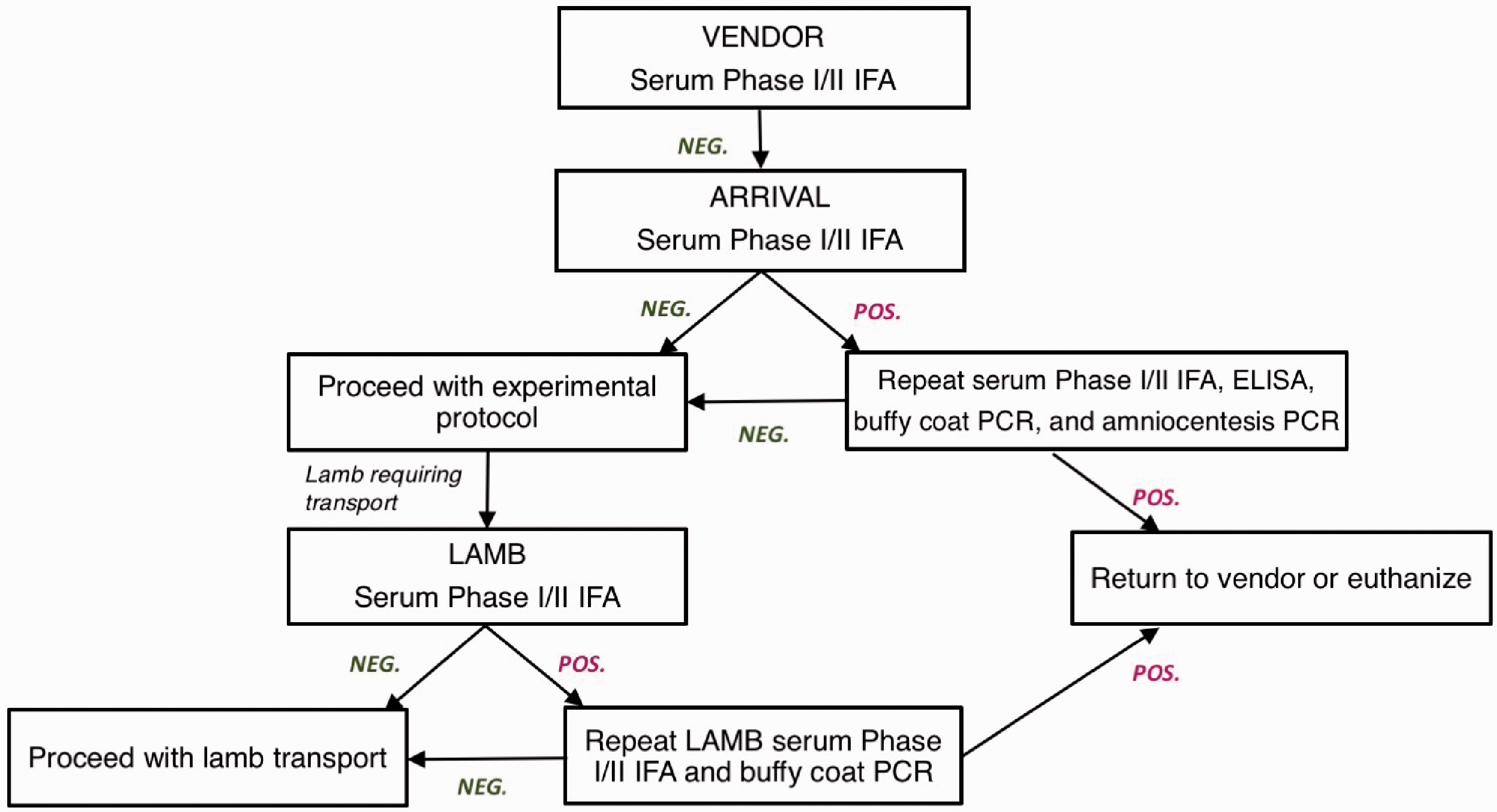
Coxiella burnetii testing protocol. Ewes undergo serology screening prior to transfer to the research facility (by vendor) and upon arrival at the facility. If positive, ewes undergo repeat serum Phase I and Phase II immunofluorescence assay (IFA), enzyme-linked immunosorbent assay, buffy coat polymerase chain reaction (PCR), and amniocentesis for amniotic fluid PCR. Following the experimental protocol, lambs requiring transport to another facility have serum Phase I and Phase II IFA testing. If positive, the serology is repeated with additional buffy coat PCR. If repeat ewe or lamb testing remains positive, the animal is returned to the vendor or euthanized.
There were multiple investigators at the facility during the study period who followed the established protocol. However, one of the investigators (Investigator A) performed additional testing because the lambs were transferred to another university following delivery. For Investigator A’s protocol, amniotic fluid samples were obtained from the ewe at the time of fetal intervention and sent for PCR testing. In addition, after completion of the research protocol, ewes underwent repeat (final) serum IFA testing, and the lambs underwent serology testing with IFA. If either the ewe or lamb serology results were positive, the IFA serology was repeated, and buffy coat PCR was performed. Buffy coat PCR was performed starting in 2016 (Figure 1).
Results were obtained from the UCD Veterinary Public Health Laboratory, UCD Comparative Pathology Lab, or UCD Campus Veterinary Services and cross-referenced to investigator-provided lists of animals participating in research during the study period in order to minimize missing results. All ewes had IFA testing. However, ewes with missing original result reports were excluded from analysis. Specificity of the serologic assays was determined in the group of animals where the C. burnetii PCR was performed on amniotic fluid samples. Sensitivity was not performed because no cases of C. burnetii were identified. Analysis was performed using SAS v9.4 (SAS Institute, Inc., Cary, NC).
Results
Initial screening of all eligible ewes in the main vendor’s flocks was performed with an overall individual seroprevalence of C. burnetii in the flocks of 14.2% (120/844 samples). C. burnetii Phase I and Phase II IFA seroprevalence rates were 13.3% and 5.0%, respectively, with 4.0% of ewes seropositive for both.
From August 2010 to May 2018, 306 ewes that had negative initial vendor Phase I and Phase II IFA screening were utilized in our research protocols. In addition, 272 lambs were tested for C. burnetii after birth at the UCD research facility. Upon arrival of the ewes at the research facility, C. burnetii Phase I IFA was positive in two (0.7%) animals, though Phase II IFA was never positive (Table 1). One of these two positive Phase I IFA ewes in Investigator A’s protocol had negative repeat arrival serology and therefore proceeded with the experimental protocol. Amniotic fluid PCR was negative for C. burnetii. After birth, the final Phase I IFA for both this ewe and its lamb were positive. However, repeat lamb Phase I and Phase II IFA serology was negative, and according to protocol, the lamb was transported to the receiving university. The second ewe with positive Phase I IFA on arrival had a positive repeat Phase I IFA, negative repeat Phase II IFA, negative ELISA, and negative buffy coat PCR and amniotic fluid PCR. The negative PCR suggested it was a false-positive serology result, but the animal was euthanized due to the positive repeat Phase I IFA.
Coxiella burnetii serology and PCR results.
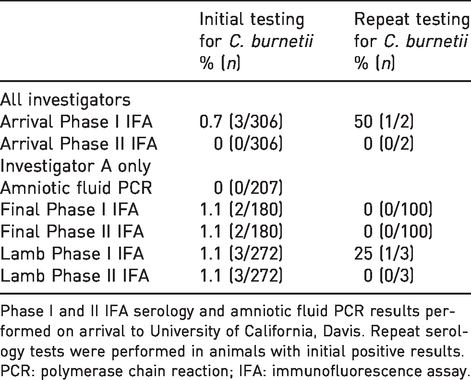
Phase I and II IFA serology and amniotic fluid PCR results performed on arrival to University of California, Davis. Repeat serology tests were performed in animals with initial positive results.
PCR: polymerase chain reaction; IFA: immunofluorescence assay.
In Investigator A’s additional tests, all 207 amniotic fluid PCR results were negative. However, Final Phase I and/or Phase II IFA was positive in two (1.1%) ewes. In this cohort of 207 ewes in which the PCR was performed on amniotic fluid, the specificity of both Phase I and Phase II IFA was 99%.
Of the 272 tested lambs in Investigator A’s additional tests, four (1.5%) were positive for Phase I IFA or Phase II IFA results. Two of the four lambs were positive on both Phase I and II IFA tests. The repeat serology tests for the lambs were negative, except for one positive Phase I IFA. All repeat Phase II tests and buffy coat PCR in these four lambs were negative. Buffy coat PCR testing was performed on 36 of Investigator A’s lambs (due to additional testing requirements starting in 2017), and all were negative.
Discussion
Utilizing an animal screening protocol requiring two negative serologic tests, we have markedly reduced the risk of C. burnetii occupational exposure and transmission. There were a handful of possible false-positive results from the IFA serologic testing. However, the not-infected status could be proven by reflex testing with buffy coat and amniotic fluid PCR. Importantly, no false-negative serology results were identified in the large number of animals that had routine amniotic fluid PCR testing.
Serodiagnosis of C. burnetii with IFA is simple and accurate, and it can be performed using commercially available products. 33 IFA serodiagnosis is preferred over ELISA because it can detect both Phase I and Phase II antigens, and cutoffs for positive results can be established more easily. 33 The commercially available IFA from Focus Diagnostics (Cypress, CA) reports a relative sensitivity and specificity (compared to state IFA and complement fixation) of 100% and 99%, respectively, for Phase I antigens, and 100% in both measures for Phase II antigens. 34 Of note, the commercially available IFA used at the UCD Veterinary Public Health Laboratory does not have published rates sensitivity or specificity, but is likely similar to the results reported by Focus Diagnostics. In the current study, the sensitivity could not be calculated without cases of C. burnetii. However, the specificity was 99% when compared to amniotic fluid PCR.
Despite the high specificity of the test, we had suspicion of cross-reactivity in our testing. The ewe and lamb in Investigator A’s protocol that had a positive Phase I IFA test initially and a negative test on recheck had weak positivity (weak immunofluorescence on IFA compared to strong immunofluorescence in the positive control). Both the mixed positive and negative testing results and the weak immunofluorescence suggest a possible cross-reactivity with another bacterial pathogen of sheep such as Bartonella melophagi rather than a true positive.
Quantitative PCR tests have been developed against multiple gene targets, and can be performed rapidly, though sensitivity and specificity vary by target and laboratory. 2 , 33 , 35 , 36 PCR has an advantage over serology in that early infections can be detected before the development of antibodies. While both IFA and PCR are rapid and easy to perform, IFA is performed due to lower cost and ease of obtaining serum. IFA serologic testing requires 2.5 hours, and the cost is <$20 per sample. In contrast, PCR testing requires 3 hours, and the cost is approximately $50. As discussed in the CDC Q Fever Recommendation for Research Facilities, antibody titers (IFA) do not correlate with active shedding of Coxiella organism. Therefore, PCR and IFA testing cannot completely ensure an animal is not infected. 2 While a theoretical risk remains, we have had no reported cases of human Q fever at our institution or in the serodiagnosis laboratory. Occupational health services at the UCD monitors employees for infection, with initial serologic testing prior to work with sheep or in the laboratory and then annually for the duration of employment.
The effectiveness of two negative IFA screening results in our protocol suggests that routine PCR testing is not necessary. However, in this study, it was performed for another university’s protocol that required negative amniotic fluid PCR, lamb serology, and lamb buffy coat PCR prior to transport. In contrast, we have identified that vendor screening for C. burnetii prior to transfer plays a crucial role in preventing exposure to research staff by minimizing transport of infected animals to the facility. Further, vendor screening of sheep likely reduced the number of animals needed for the research protocols, since C. burnetii–positive animals did not have to be euthanized.
Seroprevalence of C. burnetii in flocks of ewes tested by our main vendor was 14%, consistent with the literature. 16 , 17 Though lower than goats, C. burnetii seropositivity is more common in sheep flocks than in cattle herds (3.4%) in the USA. 17 Despite the high seroprevalence in potential animals used in our medical research, our protocol essentially eliminated the risk of transmission to research and husbandry personnel.
Conclusion
Serologic testing of domestic sheep (ovis aries) for C. burnetii with IFA prior to transport and following arrival at a research facility, in combination with reflex testing of amniotic fluid using PCR, limits potential exposure to research staff.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to research, authorship, and/or publication of the article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by funding from the California Institute of Regenerative Medicine (grants #PC1-08103, #CLIN1-11404), Shriners Hospitals for Children (grants #85120-NCA-16, 85108-NCA-19), the NIH (grants #5R01NS100761-02, #1R03HD091601-01, #R01HL 061284), and March of Dimes Foundation (grant #5FY1682). L.G. was also supported by the National Center for Advancing Translational Sciences, National Institutes of Health, through grant number UL1TR001860. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
