Abstract
Health monitoring is essential for ensuring animal health and reliable research results. Each animal facility should establish adequate health monitoring methods, and microbiological quality control should be implemented through regular health surveillance. Recently, specific pathogen free (SPF) mice have been housed in individually ventilated cage (IVC) racks in the majority of mouse facilities globally, and health monitoring is implemented using a soiled bedding sentinel (SBS). Even though SBS monitoring is a standard method, it has a limitation in that some pathogens are not sufficiently transmitted to the sentinel housed in the IVC. The exhaust air dust polymerase chain reaction (EAD PCR) method has been reported to be a reliable complementary method to SBS monitoring based on research findings. In Korea, health monitoring programs using EAD PCR have not yet been applied to laboratory animal facilities. The microbiological status of mouse colonies housed in the two IVC racks was compared using SBS and EAD PCR monitoring in our SPF mouse facility. Except for
Introduction
Laboratory mouse health monitoring ensures animal health and quality of biomedical research results.
1
The use of individually ventilated cage (IVC) rack systems in specific pathogen free (SPF) mouse facilities has lately increased. SPF mice are housed in IVC racks in the majority of mouse facilities globally, and health monitoring is implemented using a soiled bedding sentinel (SBS). A sentinel cage is usually placed every 50–80 IVCs. Because of several factors such as housing circumstances, immunological status of resident animals and a limited number of animals for direct sampling, health monitoring using SBS is the most standard monitoring method for indirect testing of IVC-housed mouse colonies.
2
Even though SBS monitoring is a standard method, it has the limitation that some pathogens are not sufficiently transmitted to sentinels housed in IVCs. Respiratory pathogens including
In previous studies, exhaust air dust polymerase chain reaction (EAD PCR) monitoring was found to be a suitable detection method for several murine pathogens including
In Korea, health monitoring programs using EAD PCR have not yet been applied to laboratory animal facilities. Using SBS and EAD PCR monitoring in an SPF mouse facility, we compared the microbiological status of mouse colonies housed in the two IVC racks.
Materials and methods
Animal protocol
This study was reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of the Institute for Basic Science (IBS). IBS IACUC followed the Laboratory Animal Resources Institute guidelines (approval number: IBS-2021-025).
Animals
BALB/c mice (four weeks old, female) were used as sentinels under SPF conditions, introduced from OrientBio (Seoul, Korea), a registered laboratory animal commercial vendor. In accordance with animal welfare principles, this animal protocol followed humane end point criteria.
Husbandry
In each mouse room, a relative humidity of 40–60%, a temperature of 22 ± 1°C and a 12:12-h light:dark cycle were maintained. In each sentinel cage, two BALB/c mice were housed in IVCs. The irradiated mouse diet (Teklad global 18% protein 2018S, USA) and autoclaved reverse osmosis water were freely available to the mice. Sterilized aspen bedding (LAS bedding PG3, Germany) and enrichment materials (Ancare, USA) were provided to each cage. Used cages were replaced on a weekly basis with new ones. After autoclaved forceps were briefly dipped into a disinfectant (Virkon-S, Lanxess, Germany), mice were transferred from cage to cage.
Facility
The IBS Laboratory Animal Resource Facility has 17 mouse rooms with IVC systems. All mouse strains including genetically engineered mice were housed in a SPF barrier. Mice from licensed vendors were quarantined before they were introduced into the main mouse colony. In vitro fertilization has been used to rederive mice from various research institutes.
IVC rack
A micro-ventilation cage system rack (Three-Shine Inc., Korea) can accommodate up to 80 cages and is connected to a standing air control unit (ACU) capable of providing 60 ventilations per hour. Three-Shine IVC is solid bottom polycarbonate (20.0 cm × 32.0 cm ×14.5 cm). Air enters the cage through a supply valve linked to the cage and exits through the top filter of the cage and the exhaust valve on the lid. Before being delivered to the cage, the indoor air was filtered with a pre-filter and a HEPA filter, and the air exhausted from the cage was likewise filtered with a pre-filter and a HEPA filter before entering the mouse room exhaust system (Figure 1).

Individually ventilated cage (IVC) racks. (a) The MVCS IVC rack (Three-Shine, Korea) includes a standing air control unit (ACU) and supports 80 cages with 60 air changes per hour and (b) The Lab & Bio IVC system (Lab & Bio, Korea) includes a standing ACU and can accommodate 80 cages with 70 air changes per hour.
A mouse cage system standard IVC system (Lab & Bio, Korea) can hold 80 cages, which are linked to a stand type ACU that provides 70 ventilations per hour. Lab & Bio IVC is solid-bottom polysulfonate (19.0 cm × 37.2 cm × 13.0 cm). Air was provided and exhausted at lid height. Air dust exhausted from the cage can descend to the horizontal air-exhaust plenum via the vertical plenums. Before entering the cages, room air was filtered through the HEPA and pre-filter. After filtration with the HEPA and pre-filter, the exhaust air entered the room exhaust air system. Negative pressure was maintained in both the exhaust plenum (Three-Shine) and exhaust filter box (Lab & Bio) to prevent unfiltered air from being released (Figure 1). The average occupancy percentage in each IVC rack was 80%.
Selection of pathogen
Based on the FELASA recommendations
2
and the prevalence of pathogens in laboratory mouse facilities in Korea,
18
16 mouse pathogens, including
Sample collection, EAD PCR
EAD samples were taken from the IVC rack’s vertical air-exhaust plenum (Three-Shine; Figure 2) and beneath the ACU’s pre-filter (Lab & Bio; Figure 2) using sterile collecting media. For optimal dust accumulation, the EAD collection medium was exposed for three months. All of the samples were delivered to Charles River Laboratories (Wilmington, USA) for analysis by RT-PCR.

Exhaust air dust (EAD) sample collection. EAD samples were taken from MVCS IVC rack’s (Three-Shine, Korea) vertical air exhaust plenum (a) and beneath the ACU’s pre-filter of a mouse cage system standard individually ventilated cage (Lab & Bio, Korea) and (b) using sterile collecting media.
SBS test
One BALB/c mouse from the sentinel cage placed in each single-sided IVC rack was used for SBS testing (Figure 3). Sentinel mice were used for health monitoring after three months of exposure to dirty bedding collected during cage changes. Every week, each sentinel mouse cage set (cage, wire, bottle, bottle nozzle) was changed. Sentinel mice were exposed to soiled bedding and fecal materials of the resident mouse colony in the same IVC rack during cage changes. Two teaspoons of soiled materials were collected from each cage on the two rows (16 cages) of the same rack on a weekly rotating system. The sentinel cage was filled with the collected soiled materials mixed with the same amount of fresh bedding. Sentinel mice were sent to Korea Research Institute of Bioscience and Biotechnology (KRIBB, Korea) for health monitoring. Serological testing for mouse hepatitis virus (MHV), mouse parvovirus (MPV), murine noro virus (MNV), minute virus of mice (MVM), pneumonia virus of mice (PVM), mouse encephalomyelitis virus (GDVII), rotavirus (EDIM) was implemented using an ELISA kit (Charles River Laboratories, USA).
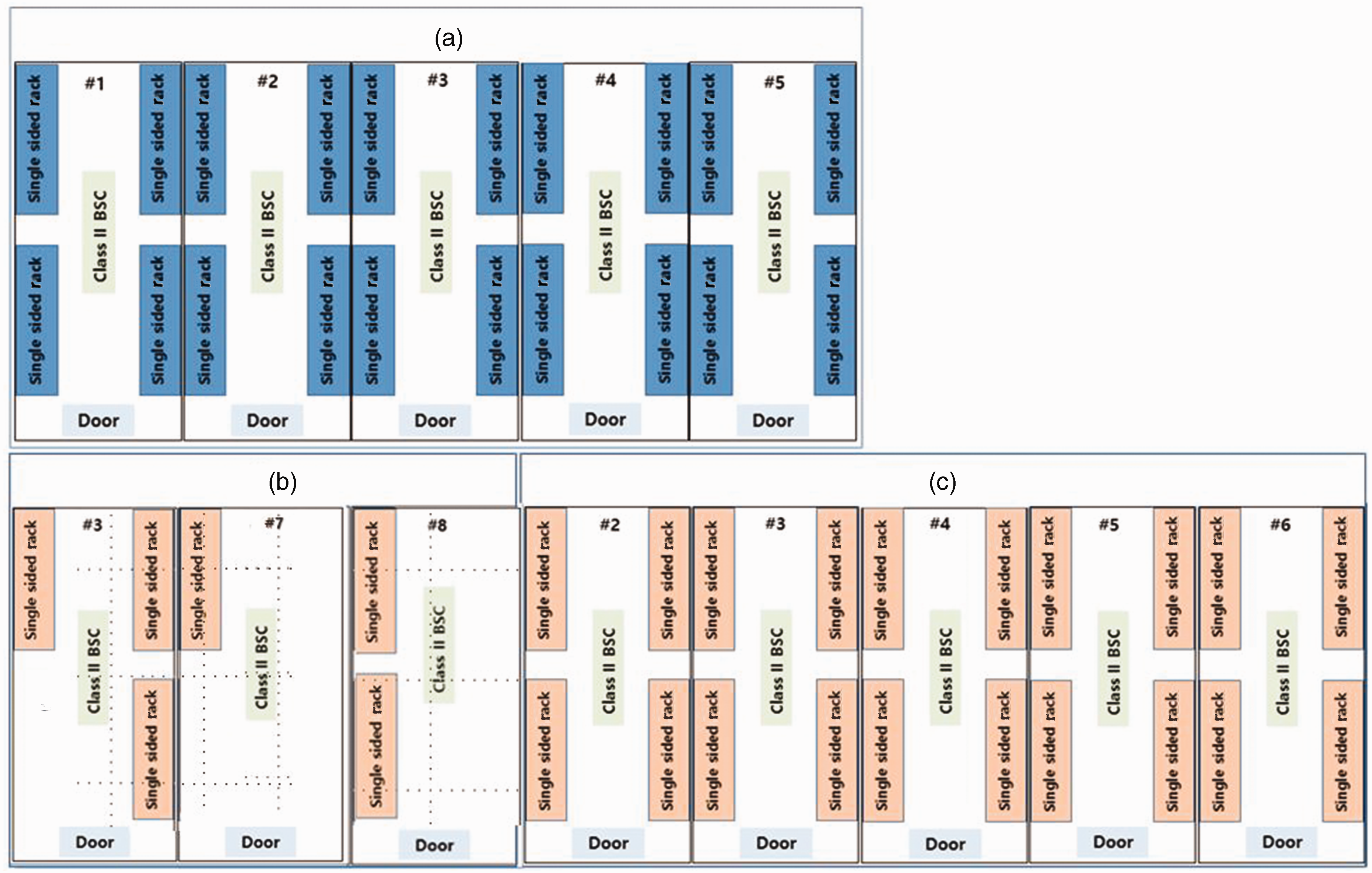
Arrangement of individually ventilated cage (IVC) racks in mouse rooms. Three-Shine IVC (a), Lab & Bio IVC ((b) and (c)); sentinel cage was placed in each single-sided IVC rack. BSC: bio safety cabinet.
Pooled fecal PCR test
A maximum of 10 fresh fecal pellets were pooled from each cage in the SPF mouse room. All samples were delivered to Charles River Laboratories for analysis of 16 pathogens.
Results
In our institution, two types of racks were used for housing mice: Three-Shine IVC racks (Area A) and Lab & Bio IVC racks (Areas B and C), which allowed us to evaluate microbiological status using EAD PCR testing.
Sentinel mice from 13 mouse rooms in three SPF areas and EAD samples from the IVC rack were tested for 16 pathogens.
Prevalence of pathogen in specific pathogen free mouse colony by health monitoring method.
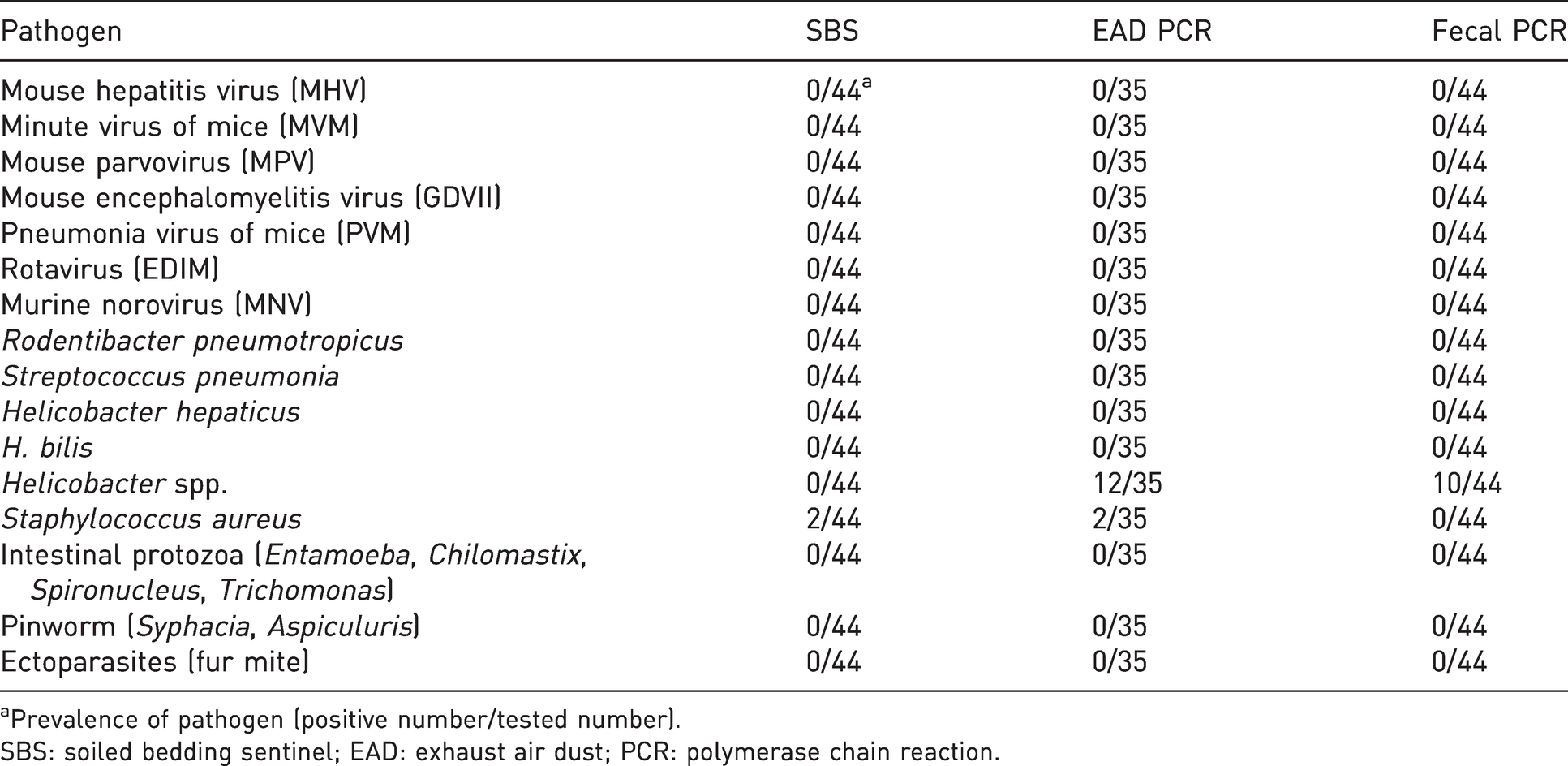
aPrevalence of pathogen (positive number/tested number).
SBS: soiled bedding sentinel; EAD: exhaust air dust; PCR: polymerase chain reaction.
A health monitoring test was implemented in 10 mouse rooms in three SPF areas. In Area A, no pathogens were detected in 19 sentinels and 19 fecal samples from the five mouse rooms. Four out of 20 EAD samples from Area A were detected as
Detection of
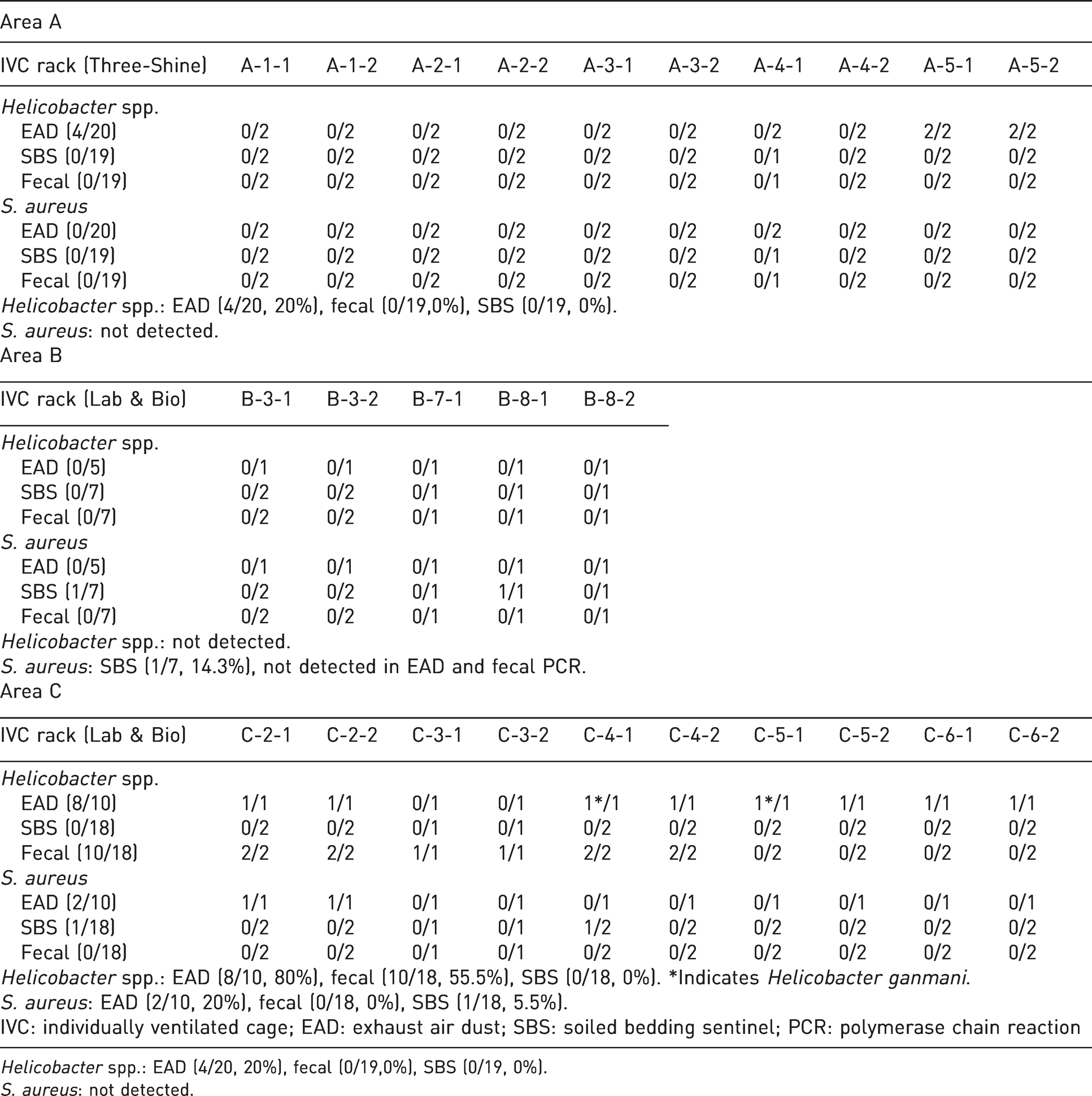
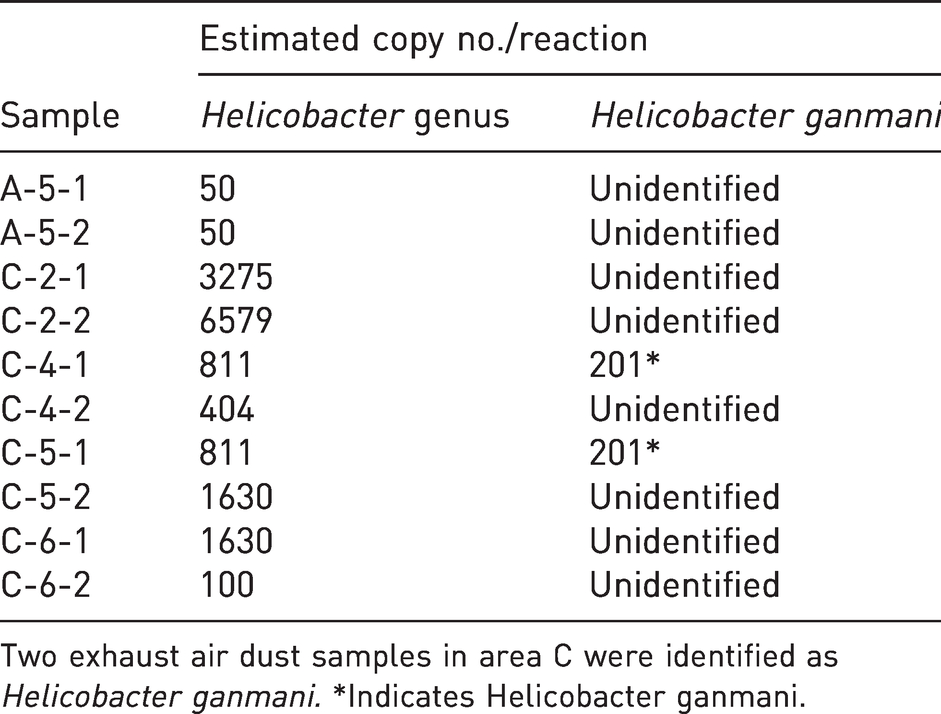
Two exhaust air dust samples in area C were identified as
Discussion
SBS and EAD PCR monitoring for 16 pathogens in three SPF areas were performed in this study. An appropriate site for collecting EAD samples was chosen based on the air flow of each IVC rack, and EAD samples were collected for three months. In the Lab & Bio IVC rack, a medium for the EAD sample was placed under ACU pre-filter, while a holder was added to the vertical exhaust port of the Three-Shine IVC rack to collect EAD samples. To prevent the emission of unfiltered air, the EAD sampling sites were kept at a negative pressure. Some IVC racks were designed to optimize EAD surveillance using a specially developed medium consisting of filters and filter holders. 6 The optimized EAD sample medium is available with Techniplast and Allentown’s IVC. It is expected that more accurate testing will be possible if a customized EAD medium is developed for the two types of IVC systems used.
In this study, MPV, MNV, MHV, mouse rotavirus, Theiler’s murine encephalomyelitis virus,
Regular health surveillance using EAD PCR is required for mouse studies, such as hepatitis and inflammatory bowel disease affected by
Two out of 44 sentinels tested positive for
Some authors have reported that EAD PCR analysis has limitations due to cross-reactivity with the PCR assay used and false positives.
21
Furthermore, the capability to detect contaminated trace DNA fragments or inactive substances in the environment has been identified as a result of the high sensitivity of EAD PCR surveillance.3,4 A PCR test can result in false-positive results owing to sample contamination or non-specific amplification of DNA.
2
In addition, nucleic acids of pathogens may remain in all environments within the laboratory animal facility. Some authors have reported that MNV and
In previous studies, the EAD PCR test was found to be comparable to the SBS test or more effective for certain pathogens.6,9 When compared with the SBS test, the EAD PCR test showed similar results in this study. In particular, the EAD PCR test had a higher sensitivity to
Some authors have compared the financial aspects of health monitoring between SBS and EAD PCR. According to them, EAD PCR was 26% less costly than SBS monitoring. To compare the cost benefit of two methods, a detailed expense analysis for the overall test scale of individual mouse facilities, calculation of additional costs for both animal tests in the event of an infection outbreak and further tests for the potential false positive results of EAD PCR are required. Aside from financial benefits, the working time spent on health monitoring was lowered by EAD. Furthermore, no sentinel mice were used in the EAD PCR. 24
Thus, EAD PCR can supplement SBS monitoring, enhancing microbiological quality control in animal facilities. EAD PCR can reduce the number of animals used, making it a 3R-consistent method. If further validation for several pathogens such as
Conclusion
We compared SBS and EAD PCR monitoring in an SPF mouse facility, with respect to detection of microbiological status of mouse colonies housed in two IVC racks. In the detection of
Footnotes
Acknowledgment
The authors would like to acknowledge appreciation of staff for providing dedicated animal care during this study.
Data availability
The original data for this study are available; please contact the corresponding author for further information.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by The Institute for Basic Science (IBS) (grant number IBS-R901-H2-2021-a00).
