Abstract
This study aims to determine the ability of laboratory animal bacteria to resist desiccation and inactivation by hydrogen peroxide vapour (HPV) on paper bedding pieces. Bedding pieces were saturated with bacterial suspensions in water or 2% (w/v) bovine serum albumin (BSA) in water, and held in a mouse facility. Viable counts showed variable survival rates over time for the bacterial species used ([
The microbiological quality of experimental animals is a prerequisite for obtaining valid in vivo results. Room disinfection and microbiological monitoring represent two practices required to achieve and maintain the specific pathogen-free (SPF) status of laboratory animals. Microbiological monitoring of the animals housed in individually ventilated cages is usually carried out on sentinel mice exposed to soiled bedding. There are several studies that have investigated the transmission of microbes to sentinels via soiled bedding.1–4 One critical factor that may influence the transmission of bacterial agents via soiled bedding is their ability to survive for a sufficient time on bedding particles. However, there are only a few studies documenting the environmental stability of such microorganisms. 5
The successful application of hydrogen peroxide vapour (HPV) decontamination in hospital settings encouraged its use for the disinfection of laboratory animal rooms or equipment used in SPF areas as an alternative to formaldehyde fumigation. 6 Hydrogen peroxide (H2O2) is a potent oxidizing agent. Its microbicidal activity is thought to be mediated by free hydroxyl radical production, which has a deleterious effect on membrane lipids, DNA and proteins.7,8
Although microbiological monitoring by sentinel animals using soiled bedding and HPV room disinfection are routine procedures in modern animal facilities, no data are currently available on the ability of bacterial pathogens to survive on bedding particles or to resist HPV disinfection in animal room settings. For these reasons, the main goal of the present trial was to investigate the ability of selected bacteria, relevant to laboratory animals such as rodent
Materials and methods
Bacterial isolates
Bacterial strains used in this study.
Quantification of bacterial survival on paper bedding pieces within the animal room
Shepherd’s™ ALPHA-dri® bedding pieces (width/height/depth approximately 5/5/1 mm; Shepherd Speciality Papers, Kalamazoo, MI, USA) (Figure 1), made of alpha cellulose, which are used as bedding for mice housed in individually ventilated cages in our facility, were used as carrier for the bacterial cells in order to simulate natural contamination. To prepare the test bedding pieces, bacteria were cultured aerobically on Columbia blood agar plates (bioMérieux, Nürtingen, Germany) for 24 h at 37℃. Subsequently, the bacteria were suspended at OD600 = 0.5 in sterile distilled water (SDW) or SDW containing 2% (w/v) bovine serum albumin (BSA) (Sigma-Aldrich, Taufkirchen, Germany), to simulate biological soiling. Approximately 40 bedding pieces per bacterial strain were introduced into the bacterial suspensions and allowed to absorb bacteria for 5 min at room temperature. The bedding pieces were then collected with sterile forceps and placed into Petri dishes which were closed and kept during the experiment in a populated mouse room, at 22 ± 2℃, relative humidity of 45–65% and light/dark light cycles of 12:12 h, in order to test the resistance of bacteria to desiccation over time. To determine the viable bacterial counts, one bedding piece was introduced into a 1.5 mL Eppendorf tube containing 1 mL of SDW and shaken for 10 min at 1000 rpm at room temperature to allow bacterial re-suspension into water. Subsequently, serial 10-fold dilutions with 20 µL volumes were performed within a 96-well plate (Rotilabo®-microtest plates; Carl-Roth, Karlsruhe, Germany). Ten-microlitre suspensions from each dilution were then placed on Columbia blood agar and incubated for approximately 24 h at 37℃. The counts of viable bacteria on the bedding pieces were determined immediately and after 2, 6, 24 and 48 h for the Form and size (cm) of the bedding pieces used in this study.
Quantification of bacterial inactivation by HPV
To test bacterial resistance to HPV, three bedding pieces inoculated 24 h before testing with
HPV was generated from 30% liquid H2O2 with the help of the HPV Generator Bioquell Q-10 (Bioquell, Andover, UK) which was placed in the centre of the room. The HPV generator delivered HPV into the room and included an instrumentation unit which measured the concentration of H2O2, the relative humidity and temperature of the room air. These parameters were monitored by a computer system placed outside the room. During HPV exposure, the ventilation and air-conditioning system of the room were disabled to prevent the removal and dilution of HPV. When the cycle was completed the catalytic converters R-30 (Bioquell) broke down the HPV into oxygen and water vapour. Access to the room was permitted when the HPV concentration in the room was <1 ppm, as recommended by the manufacturer. The samples were then collected and processed immediately as described above.
Statistical analysis
Two-tailed Student’s
Results
Survival of the bacterial clinical isolates on bedding sheets within the animal room
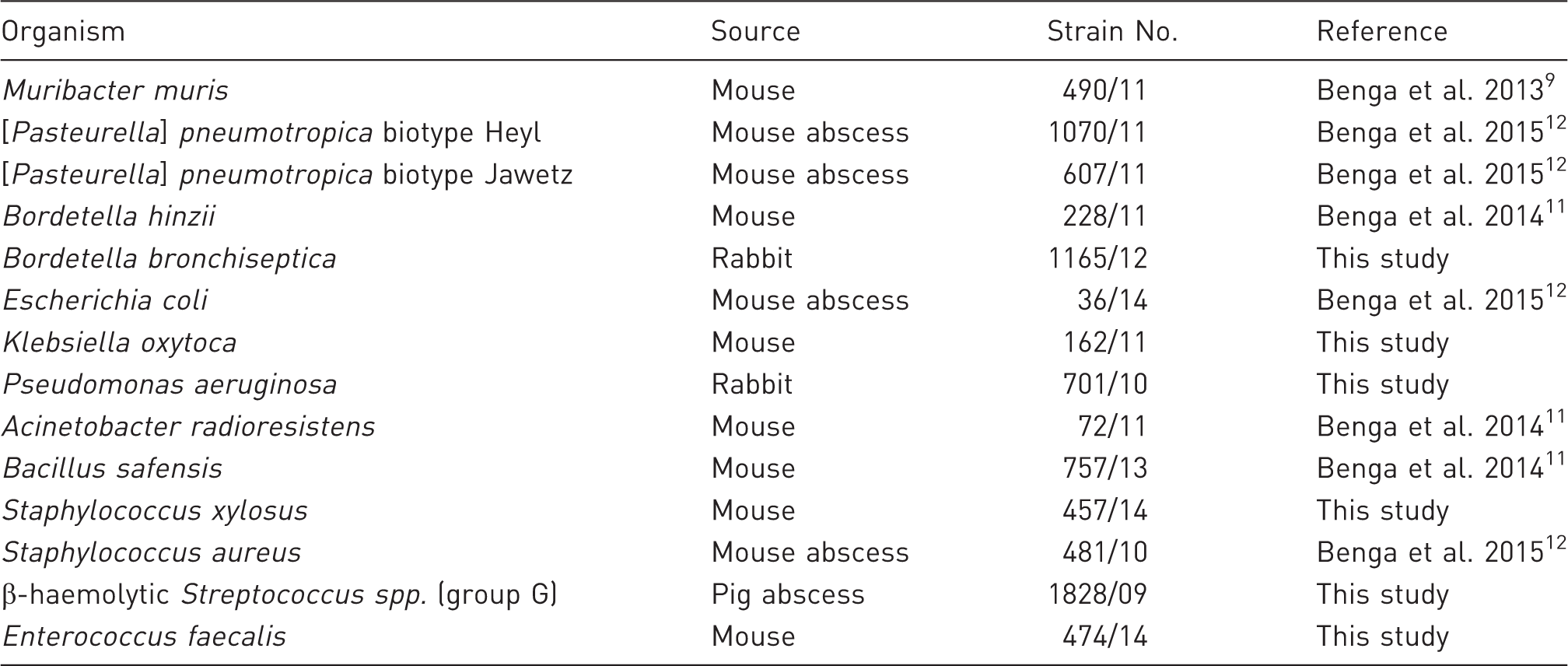
Relative survival rates of the bacteria on bedding pieces under controlled environmental conditions of the animal room varied considerably depending on the bacterial species and the presence/absence of BSA. Generally, for all bacterial species tested the presence of 2% BSA resulted in a significant increase in the survival rate on the bedding pieces, compared with the water-suspended bacteria. The least resistant bacterial species proved to be the three rodent Survival of rodent Survival patterns of Survival kinetics of 
 ), [
), [ ), [
), [
 ) (A),
) (A),  ) (B),
) (B),  )/
)/ ) (D), and
) (D), and  )/
)/
Survival of the bacterial clinical isolates on bedding pieces after exposure to HPV
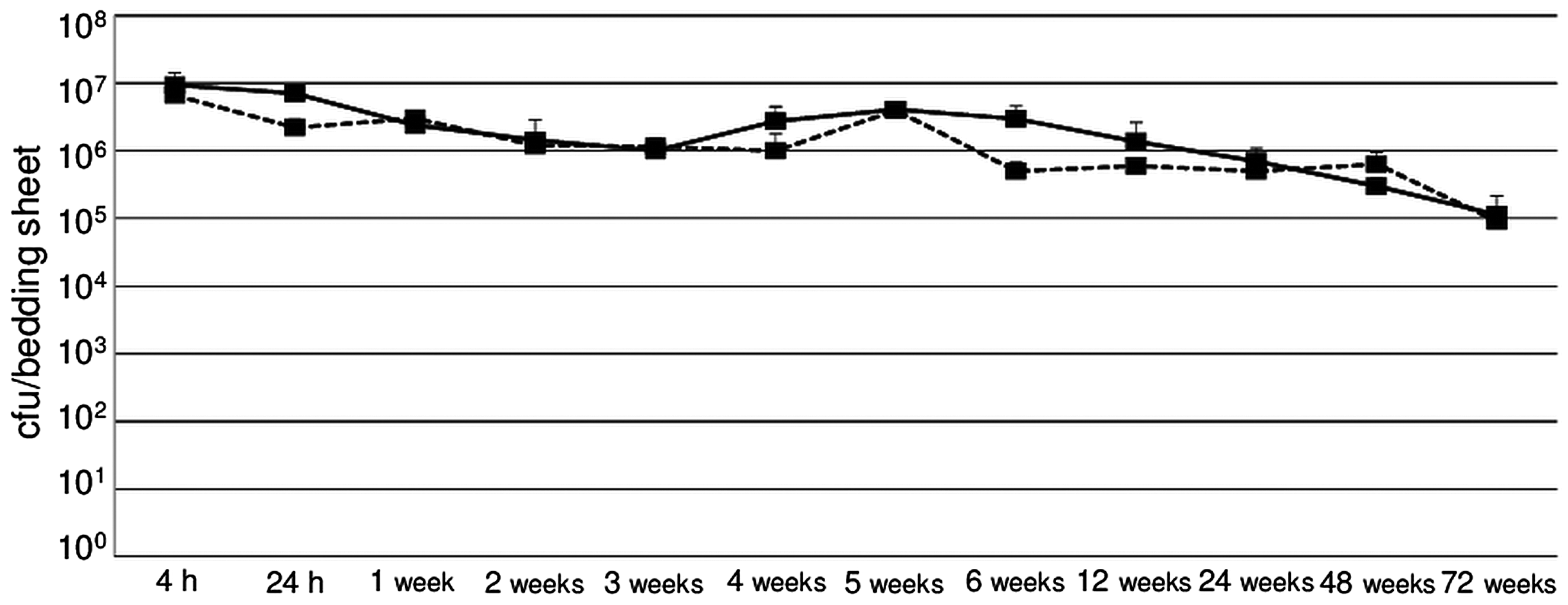
The cycles used for the decontamination of the room with HPV consisted of an injection phase followed by a dwell phase of 20 min each. Subsequently, the HPV was converted to oxygen and water vapour during the aeration phase which lasted until the HPV concentration dropped to below 1 ppm. The HPV decontamination was also accompanied by changes in temperature and relative humidity. The parameters recorded were similar among the three cycles performed. The data from a representative HPV treatment cycle are shown in Figure 5.
Kinetics of data recoded during a representative hydrogen peroxide vapour (HPV) cycle. Hydrogen peroxide (H2O2) concentration, relative humidity and temperature were registered by the instrumentation unit of the HPV generator and displayed on the control computer system placed outside the room.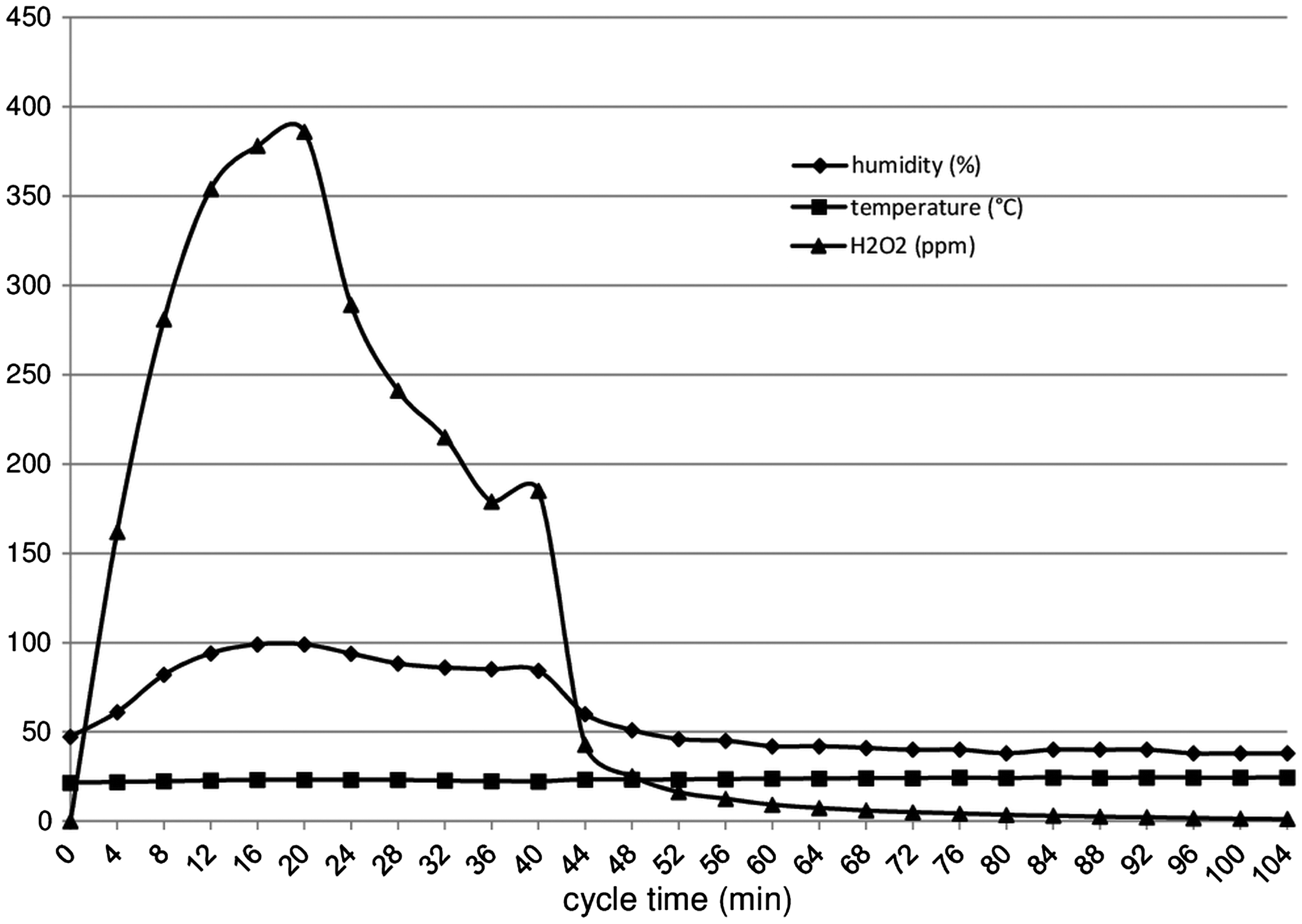
Survival of
CFU: colony forming units; HPV: hydrogen peroxide vapour.
Discussion
Several of the bacterial species are important in the quality assurance of the SPF status of laboratory rodents. With an increasing number of genetically-engineered animals available, even agents which have recently been regarded as non-pathogenic for wild-type mouse strains have become problematic with some animal strains.
13
Detection and environmental sanitation of such microbes are essential in obtaining and maintaining microbiologically standardized laboratory rodents. In the present investigation we analysed the ability of bacteria relevant to laboratory rodents to survive on bedding particles in the animal room environment, which is essential for microbiological monitoring using soiled bedding sentinels. As expected, we noticed considerable differences in the survival rates of the bacterial strains tested (Figures 2–4). Overall, the presence of 2% BSA, used to simulate biological soiling, led to increased survival rates on bedding pieces for all vegetative bacterial species tested compared with their corresponding water-suspended samples. Our findings are in agreement with those of Coughenour et al. who have found that the presence of BSA enabled MRSA to survive significantly longer on five different environmental surfaces.
14
Rodent
Numerous studies tested the efficiency of HPV to inactivate relevant human bacterial pathogens on hard surfaces such as stainless steel.15,17 Nevertheless, only a few studies have investigated porous surfaces, such as cotton or carpet, as carriers for the bacteria.18,19 From this perspective, our study on the efficacy of HPV to inactivate bacteria on cellulose bedding is new and has relevance for both laboratory animals and hospital settings, where cellulose/paper materials can be contaminated by such bacteria. The resistance of bacteria to HPV can also vary with the strain, as has previously been shown,
15
where a reference strain of
HPV readily inactivated
HPV disinfection should be used after rooms are cleaned. However if the cleaning is suboptimal, microbes embedded in animal secretions and excretions may contribute to the failure of HPV disinfection. From this perspective, future experimental HPV disinfection studies should consider more natural aspects regarding bacterial soiling and carriers, and should not be limited to artificial conditions of water-suspended bacteria that had dried on hard surfaces such as stainless steel.
In summary, our findings show that bacterial species relevant to laboratory animals can survive for different periods of time on bedding pieces under animal room conditions, and that BSA soiling favour bacterial survival in the environment and their resistance to HPV when placed on bedding. Our results indicate that an extensive room cleaning is a pre-requisite for a successful HPV disinfection. Overall, such aspects must be considered when developing standard operating procedures for laboratory rodent colonies.
Footnotes
Acknowledgements
We gratefully acknowledge Sonja Green, Andrea Grunwald, Theresa Ohly and Manuela Stockhausen for their excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
