Abstract
Regardless of the microbiological status of an animal facility, research animals may experience health problems, leading to pain, suffering and distress. Simple and efficient tools are needed to collect data systematically, allowing researchers to react and resolve animals’ health issues. We have developed a real-time notification method for recording clinical observations, which caretakers can input into the ELLI record-keeping system, accompanied by a picture or video. A browser-based interface system sends alerts using a three-tier scale (+, 120 hours; ++, 72 hours; +++, 24 hours) by email and/or SMS. The percentage of animal health notifications for rodents was 1.31% in 2016, 1.33% in 2017 and 1.58% in 2018, with 34–44% for coat and skin conditions (wounds, bites and scratches). All other notifications, including environment and behaviour, procedure-specific indicators (weight loss, bleeding and abnormal secretions) and other abnormalities such as eye and teeth malformations, ranged from 5% to 10% during the three-year period. Researchers displayed good compliance by reacting to the notifications within the expected time frame. Most health notifications concerned genetically modified (GM) animals without a predetermined harmful phenotype, regardless of being on project licence or maintenance licence. Health notification records may be useful retrospectively not only to review the health and welfare issues of new GM lines but also to evaluate the actual severity of procedures. The health notification system described here provides valuable information to the veterinarian and the animal welfare body by helping to address specific health conditions and to improve animal welfare and implement the 3Rs.
Introduction
To mitigate health problems resulting in animal pain and distress, establishments use clinical observation, scoring sheets or computerised data management systems to assist the recognition of adverse effects. 1 Recording and reporting health problems in animal facilities should be part of routine operating procedures. Data regarding clinical signs collected systematically from all research animals in the facility may help to identify any background causes and improve animal welfare. 2
Regular health monitoring programmes often detect pathogens and help to maintain a bio exclusion programme with a defined microbiological status. 3 Spontaneous or induced genetic mutations may lead to health issues at any age. Immunodeficient or gnobiotic animals are at high risk of contamination from opportunistic agents. Any animals subjected to irradiation or immunosuppressive treatments are also at risk from infectious diseases. These animals may exhibit clinical symptoms even without any infection. Following experimental procedures, animals may suffer from known or unexpected complications. Behavioural issues due to housing are a common cause of adverse health conditions. It is well known that skin and other symptoms are commonly observed in male mice due to fighting.4,5
EU Directive (2010/63/EU) and national and federal laws dictate documentation of the care and treatment of all laboratory animals. Animal welfare bodies are obliged to maintain records on welfare issues. 6 All methods to reduce suffering, including accurate record keeping, not only serve to ensure good animal welfare and research integrity, but this is also the ultimate aim of the legislation. 2
Animal technicians have a critical role in the daily care of the animals. It is essential to educate and train these caretakers to recognise species-specific clinical signs. Ultimately, recognising and alleviating pain in animals requires teamwork consisting of at least animal care staff, research personnel and a veterinarian or other qualified expert.
Simple and efficient tools are needed that enable researchers to react and resolve animals’ health issues. We have developed a real-time notification method for recording and alerting clinical observations, which caretakers input into the ELLI record-keeping system, accompanied by a picture or video. Here, we report the analysis of health notifications and researchers’ reaction times collected from all rodents in the facility during 2016, 2017 and 2018.
Methods
We retrospectively collected data regarding health notifications from 2016, 2017 and 2018. The data collected concerned all rodents, regardless of genetic status, age or sex, in the facility, with approximately 95% of the animals being mice and the rest being rats. Rodents were either purchased from commercial breeders or bred in the small mice or rat colonies at the University of Turku, Central Animal Laboratory (UTUCAL). The health notification system was developed as an integral part of the record-keeping system, called ELLI, which is used in our animal facilities. All animals at UTUCAL are maintained either under project licences approved by the National Animal Experiment Board of Finland or by internal licences granted by UTUCAL for maintaining animals for breeding purposes or for GM animals that do not exhibit any adverse phenotype.
A designated veterinarian, available for consultation, visits the facilities regularly. UTUCAL operates according to the OECD principles of Good Laboratory Practice and inspected regularly by the Finnish Medicines Agency, FIMEA. The animals were maintained in either open-top cages or individually ventilated cages under controlled conditions at a temperature of 21±3°C and a relative humidity of 55±15% on a 12-hour light/dark cycle. The rodents were fed ad libitum with project-specific diets, and water was provided ad libitum.
As a part of their daily work, animal caretakers input any clinical observations to the health notification tool of the record-keeping system, accompanied with a picture or video. All animals are observed regularly twice daily during weekdays and once a day during weekends and official holidays. Additionally, all animals are also observed in detail during cage-changing practices at least once a week. The caretakers select terms for observations of environmental conditions and clinical signs, including behavioural observations, and they can fill in free-text boxes. In order to ensure that clinical observations are standardised and objective, the animal caretakers’ monthly training programme includes clinical observations as a topic at least once a year. The system uses a browser-based interface to send alerts according to a three-tier scale (+, ++, +++) by email and/or SMS. In addition to the researcher, the facility manager and the veterinarian receive the same messages. Depending on the urgency, researchers must react to the animal’s status within 120, 72 or 24 hours (see Table 1). In 2018, we added an option button for extremely urgent cases: ‘Emergency! Animal will be euthanised if investigator doesn’t react in 4 hours’ (tick box), which can be clicked to confirm the urgency of the messages. These additional urgency messages were sent for eight animals in 2018.
Classification of health notifications.

Symptoms reported in the + and ++ urgency categories were similar, but classification to either category depended on the animal caretaker’s assessment. These symptoms included fur condition, scratches and wounds, abnormal faeces, body secretions, abscesses, tumours, and eye and teeth abnormalities. More severe symptoms such as hunched posture, seizures, severe respiratory conditions, paralysis, severe dermatitis, malformations, significant loss of body weight and dehydration were mostly reported in the +++ urgency category.
The system alerts the researcher until the animal’s health problem is solved, although UTUCAL may issue an invoice for a veterinary visit. Researchers react to notifications using the computer or mobile device or by checking their animals in person at the facility followed by instructions on the animal’s fate. These include decisions for euthanasia, analgesia, follow-up and isolation. The veterinarian and the animal caretakers ensure that the researcher responds to a notification in an appropriate way, ensuring that the best action is taken in terms of welfare. If researchers are not able to check the animals, they can ask for additional details from the technicians or require a veterinarian consultation (Figure 1).

Process flow chart demonstrating the health notification system.
Data extracted from the ELLI record-keeping system included the number of animal welfare notifications of genetically modified (GM) and non-GM animals that were on maintenance licence (ML) and project licence (PL) for the three-year period from 2016 to 2018. Health notifications were also categorised as follows: GS1, not genetically altered; GS2, genetically altered without a harmful phenotype; GS3, genetically altered with a harmful phenotype. Each category was further classified into animals on ML or PL. Animals on ML included rodents bred to maintain the colony or animals used to establish GM lines. Notifications were categorised for appearance and body functions (coat and skin conditions, wounds, bites) and body conditions; environment and behaviour (moving abnormalities/tremors); procedure-specific indicators (weight loss, bleeding, abnormal secretions); and other abnormalities (e.g. eye or teeth malformations). 7
Results
Number of animals and health notifications for GM and non-GM animals
During the three-year data-collection period, the yearly number of rodents in our facilities was 60,520 in 2016, 56,339 in 2017 and 46,619 in 2018. Of the all rodents, the percentage of rats was 5–6%, with the rest being mice. Most of the animals were GM animals with no harmful phenotype (GS2), followed by non-GM GS1 animals. Health notification percentages ranged from 0.1% to 2.0% for GS1 and GS2 categories. In 2016 and 2017, none (or a minor number) of the GM animals were reported to have any harmful phenotype (GS3). In 2018, there were 1036 GS3 animals recorded, for which 4.2% health notifications were sent (Table 2).
Number of rodents per year and the percentages of health notifications sent for GS1, GS2 and GS3 animals.
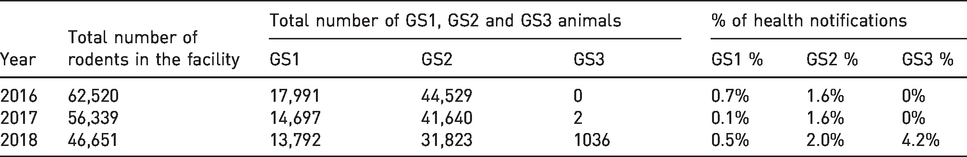
The percentage of animal health notifications from the total number of animals was 1.31%, 1.33% and 1.58% in 2016, 2017 and 2018, respectively (Table 3). The total number of notifications ranged from 818 in 2016 to 718 in 2018, and corresponded with the number of animals (Table 3). GS1, GS2 and GS3 animals were further qualified as being on ML or PL. For GS1 animals, the total percentage of health notifications was similar between animals on ML and PL, ranging from 3.4% to 10.3%. GS2 animals displayed the highest percentage of notifications consistently from 2016 to 2018. Animals on ML and PL received a similar amount of notifications, ranging from 33.4% to 51.5% in the GS2 category. Surprisingly, only 6% of the notifications were sent in the GS3 category for 1036 animals, that is, GM animals under PL in 2018 (Table 3). In 2016, no animals were registered under the GS3 category, and in 2017, only two such animals were registered (Table 2).
Percentage of welfare notifications for genetically modified (GM) and non-GM rodents that were on maintenance licence (ML) and project licence (PL).
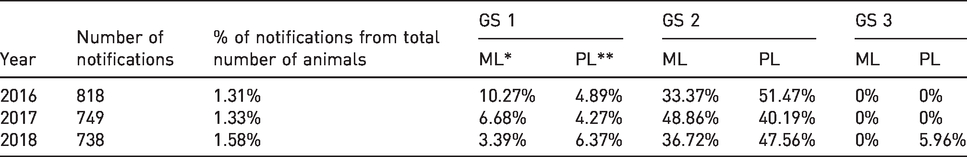
Number of notifications for each urgency category and reaction times of researchers
For most of the animals, more than one message was sent (see Table 1) in order to ensure that the researcher received the message. The average number of health notifications per animal was approximately four, and this number was similar during the three-year period.
The total number of health notification messages was highest in the + category, being highest in 2018. The + category indicates the messages sent were not for severe symptoms. The lowest number of messages was sent for animals that had more severe symptoms requiring prompt attention in the +++ category (Figure 2).
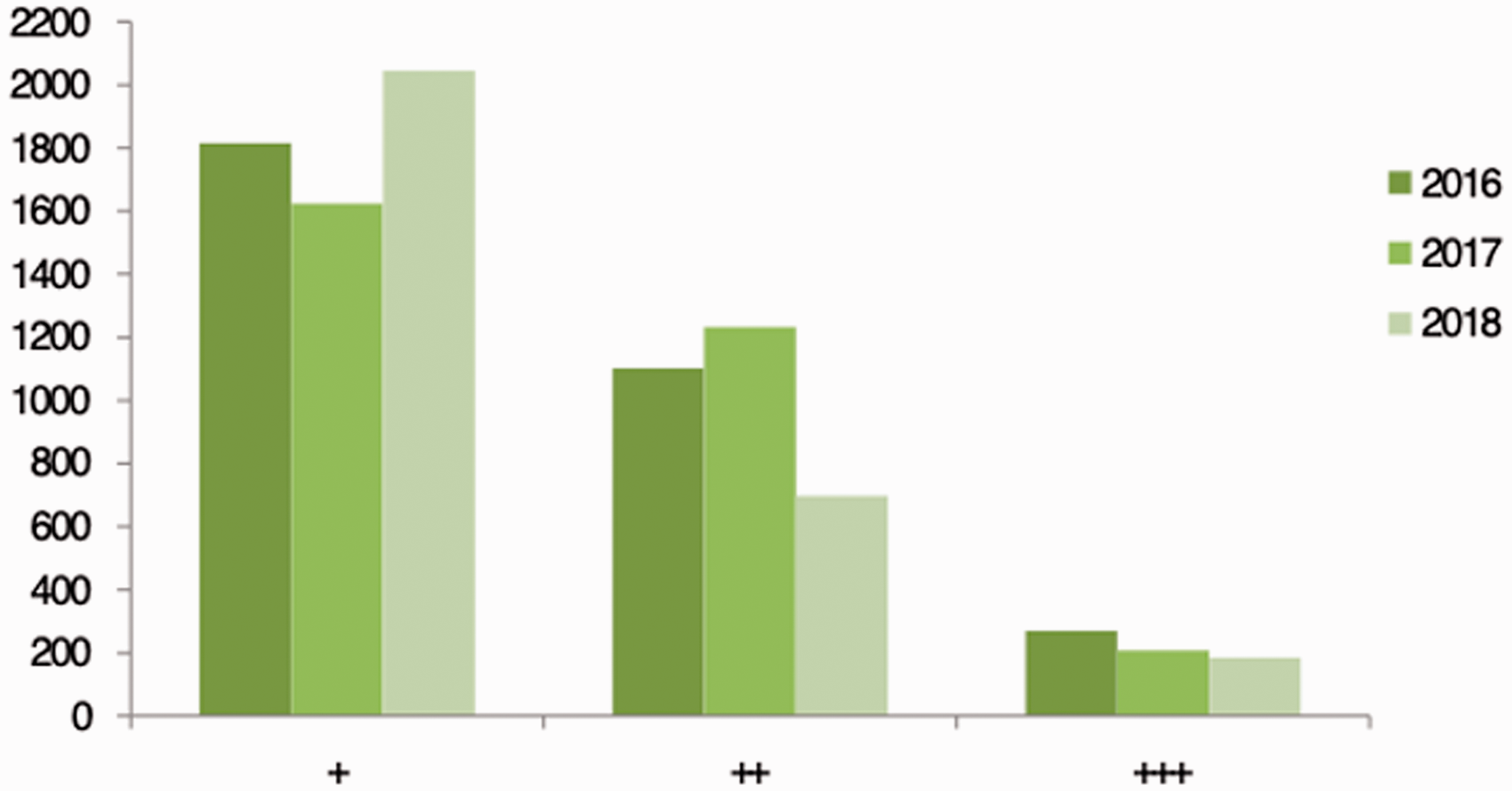
Total number of health notification messages for each urgency category.
The average reaction times to different notification categories remained within the maximum allowed: for +, 26–36 hours; for ++, 23–43 hours; for +++, 5–9 hours (Figure 3). In 2017, the researchers’ reaction times were quicker for each urgency category.
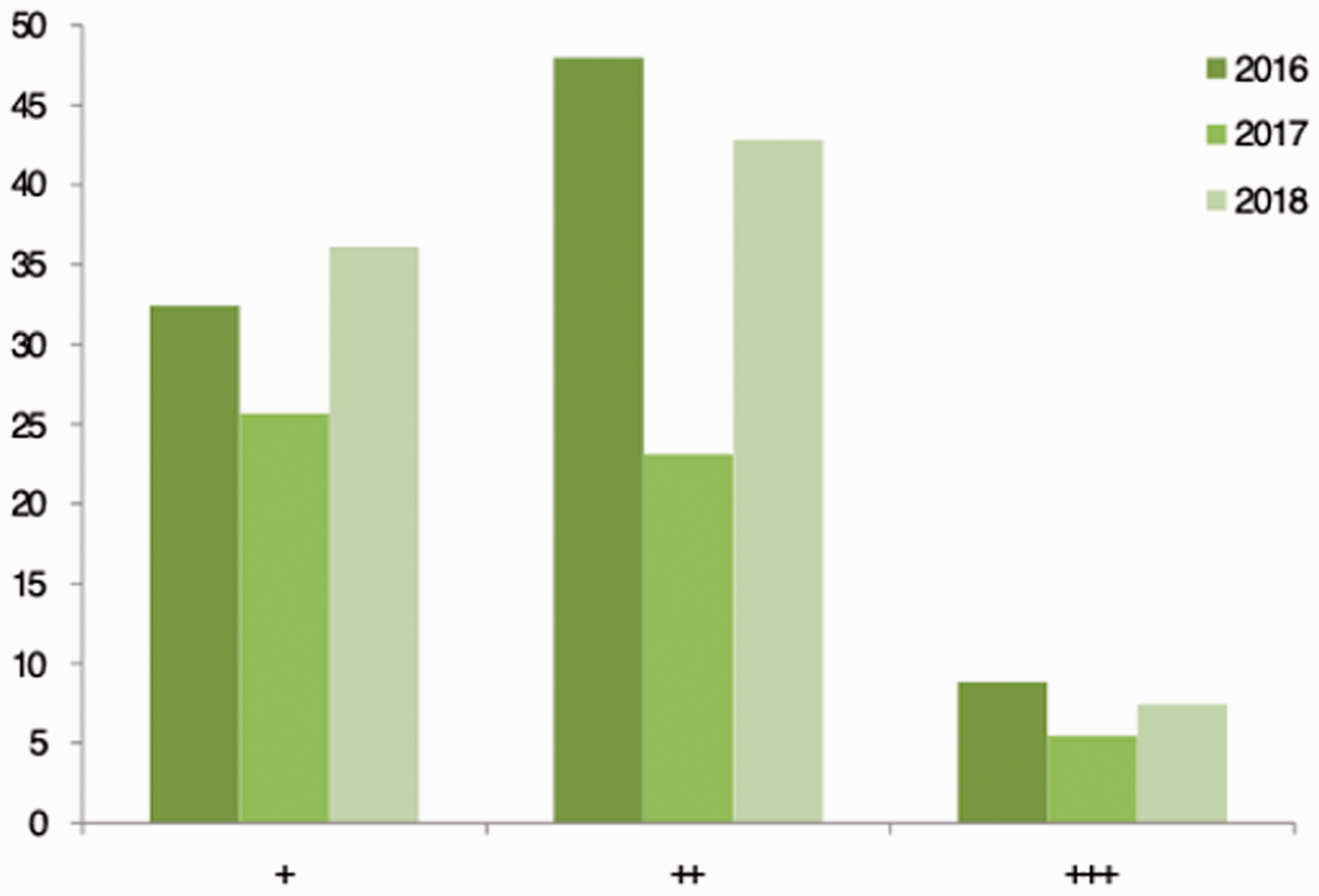
Researchers’ average reaction times (hours) to health notifications for each urgency category.
The most notifications were sent for coat and skin conditions (wounds, bites, scratches), ranging between 34% and 44%, and for appearance and body conditions, ranging between 38% and 42%. Breathing abnormalities and vocalisation comprised about 1% of the notifications. All other notifications regarding environment and behaviour, procedure-specific indicators (weight loss, bleeding, abnormal secretions) and other abnormalities such as eye and teeth malformations ranged from approximately 5% to 10% during the three-year period (Figure 4).
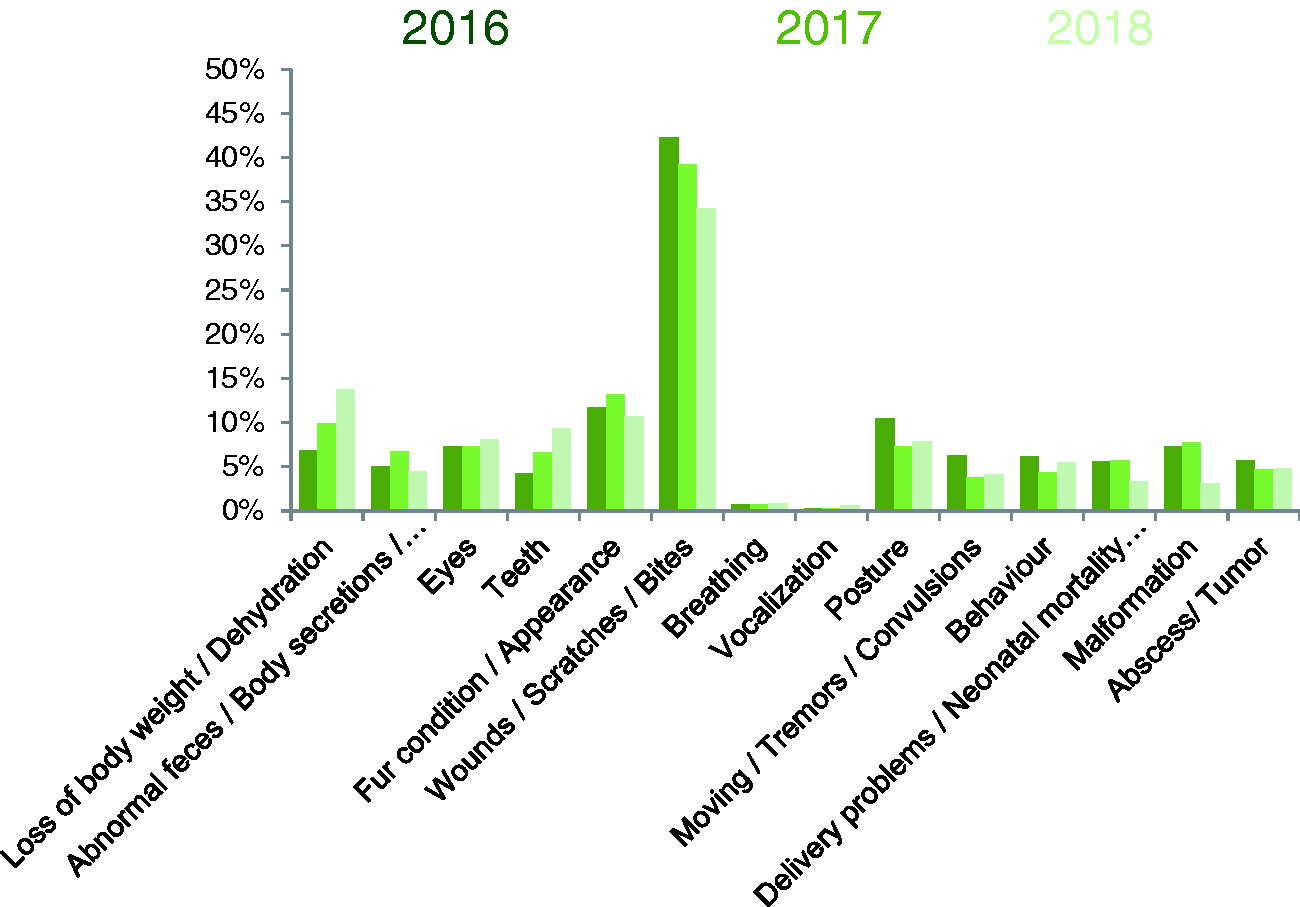
Percentage of rodent health notifications for each symptom category.
Discussion
Clinical symptoms in experimental animals can be caused by genetic background, experimental procedures, environmental factors or disease due to pathogenic agents. There is a general lack of reporting of scheduled or unscheduled observations or clinical signs in scientific publications. 2 Data on clinical signs can be used to assess pain, suffering and distress in experimental animals. In safety and toxicology studies, reporting of clinical signs is usually done as a part of evaluating the clinical pathology (OECD guidance document 19). 7
We analysed the health notifications sent by the ELLI record-keeping system and reaction times to the notifications during 2016, 2017 and 2018. The percentage of animal health notifications concerned 1.35%, 1.32% and 1.59% of all rodents in our facility during these three years, respectively. Of those, the percentages for the +, ++ and +++ categories were 55–70%, 23–36% and 7–9%, respectively. The + category refers to mild clinical signs that require the attention of the investigator within 120 hours. Indeed, most of the health notifications concerned signs related to skin and fur conditions such as wounds, scratches, abscesses or tumours. Consistent with the low percentage of health notifications in the +++ category, the percentage of conditions such as neurological symptoms or delivery problems remained relatively low.
Anorexia, body-weight loss and teeth abnormalities tended to increase towards 2018, while fewer wounds and posture-moving abnormalities were reported in 2018. This might reflect a real change between 2016 and 2018. It is possible, however, that the differences seen between 2016 and 2018 might also be due to changing attitudes on when to report symptoms. During these years, animal technicians were continuously trained to recognise symptoms and the importance of reporting them. In some cases, animals were euthanised as agreed among the researchers without sending a health notification, possibly affecting the number of reported cases.
It is expected that animal technicians are usually the first to detect any abnormalities or signs of suffering or distress in animals. Clinical signs can be objective (measured body weight and temperature, heart and respiration rates, food and water consumption, species or experimental ethogram) or subjective (appearance of coat, eyes, movement, quality of respiration). Our animal technicians do not use objective signs based on specific measurements or clinical examination when reporting clinical signs. Instead, the health notification system provides a list of a myriad of ready-made clinical symptoms, as well as a free-text box to record any other observations. The researcher and the veterinarian may comment or specify the clinical signs either based on the pictures or videos posted by the animal technicians or after examining the animals. Health notifications provide data and tools for assessment of the experience during the project.
The average reaction times for different notification categories remained within the maximum hours allowed, that is, 26–36 hours for category +, 23–43 hours for category ++ and 5–9 hours for category +++. This indicates good compliance by the researcher in reacting to the health notifications. It also demonstrates that repeated notifications by email or SMS are efficient in attracting the researchers’ attention. In the +++ category, the reaction time was nine hours in 2016. This decreased to five hours in 2017, and increased in 2018 to seven hours. Ideally, the reaction times should be faster for severe symptoms, such as hunched posture, seizures, severe respiratory conditions and paralysis. In order to speed up the reaction times to the notifications even further, especially for the +++ category, we added the ‘Emergency! 4 hours’ option button to confirm the urgency of the messages. An urgent need may arise to euthanise animals that are severely suffering if the researcher’s response time is not quick enough. Killing the animal(s) without consulting the researchers may result in the loss of the project due to inadequate statistically robust numbers remaining, or animals nearing the end of long-term studies may have undergone previous suffering in vain if the data are now not useable because they are killed early. The issue of killing an animal in an ongoing experiment in order to prevent further suffering obviously presents an ethical dilemma. Therefore, any possible urgent euthanasia decision by the care staff or the veterinarian should be based on mutual agreement between the researcher and animal facility staff. Project start meetings or continuing communication between the researcher and the care staff is important for agreeing such decisions beforehand and will also contribute to fostering a culture of care. It remains to be seen how the reaction times of researchers will develop in subsequent years and whether they will be influenced by the ‘Emergency! 4 hours’ button. Since all PL and personal information is recorded in the ELLI record-keeping system, it not uncommon to call an inattentive researcher by phone when necessary. In addition to the researcher, the facility manager and the veterinarian also receive the same messages, ensuring that the notifications are not ignored. Even though it is possible to issue a bill for a veterinary visit if the researcher does not react to the health notification, no such need has arisen so far. In practice, the veterinarian, depending on the amount and type of notifications, can schedule an animal welfare body meeting. Indeed, health notifications and other welfare issues led to approximately 20 animal welfare body meetings per year during the three-year period.
In our experience, data collected systematically helped the animal welfare body to integrate the team approach efficiently, consisting of researchers, care personnel and the attending veterinarian, and helped resolve several health issues. 8 The benefits of reporting clinical signs also include determining and following humane end points more precisely. Additionally, a copy of the approved PL, including all details about the procedures as well as humane end points, is available in the ELLI record-keeping system. When necessary, the animal technicians or the veterinarian can check the humane end points from the PL, and these can be used to terminate the studies or euthanise the animals without prolonging pain and suffering.
Approximately two thirds of the rodents were GM animals without a harmful phenotype (GS2). Of the GS2 animals, most of the health notifications concerned the animals on PL. However, a significant percentage of GS2 animals on ML also received health notifications. Overall, the GS2 animals had significantly more health notifications, indicating that GM animals are more prone to displaying clinical signs compared to non-GM animals. Surprisingly, only 6% of the notifications were sent for 1036 GS3 animals, that is, GM animals under PL in 2018 (Table 3). Our results indicate that predetermined classification of GM animals as being in GS2 or GS3 does not necessarily reflect the actual clinical signs and suffering experienced by those animals. Usually, when GM lines are generated, there is no phenotype information or experience as to what kind of health issues the GM line may demonstrate. Health notification records help to determine whether the GM line possesses a harmful phenotype and may help in phenotyping. This information can be used to transfer animals that have a harmful phenotype to a PL. Alternatively, these records can be used to switch animals from a PL to a ML.
The EU Directive requires reporting the actual severity experienced by an animal when used in a procedure. Recording the actual severity of the pain, suffering, distress or lasting harm experienced by the animal should be taken into account rather than the predicted severity at the time of the project evaluation.
In the latest animal use statistics from the EU from 2015, 2016 and 2017, 8–11% of uses were assessed as severe. 9 In our study, the percentage of health notifications sent for GS3 category animals was 6%. This indicates that the animals classified and maintained under PL with expected health issues might not always display severe clinical symptoms. In fact, some of these health notifications possibly concerned mild clinical symptoms. If these animals are maintained under PL and are classified as severe, care should be taken not to report them to the authorities as severe automatically without performing the actual severity evaluation. Actual severity is recorded based on the lifetime experience of animals. Health notifications provide useful medical history records for individual animals that could be instrumental in evaluating the actual severity. This may prevent over- or underestimating the actual severity of the procedures.
Our results demonstrate that most of the health notifications concerned GM animals without predetermined harmful phenotypes (GS2). Furthermore, we observed that the number of health notifications was not dependent on whether the animals were on ML or PL, and thus were not dependent on the performed procedures. Compliance of researchers in responding to health notifications indicates that such tools may improve animal welfare. The health notification system described here has become a significant tool for facilitating communication and directing efforts to solve certain health conditions by implementing the 3Rs.
Footnotes
Acknowledgements
The authors thank the personnel of Central Animal Laboratory, University of Turku, for their dedication to reporting health issues and improving animal welfare. We greatly appreciate the work of Caroline MacFarlane for language editing.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: Jukka Maaranen is the owner and Aija Saukkonen is the employee of Eliö Software, LLC, Helsinki, Finland, owning the ELLI record-keeping system. Personnel of UTUCAL and researchers from the University of Turku contributed to the development of the ELLI record-keeping system and the health notification tool as users without any financial compensation. Ulla-Marjut Jaakkola, Nina Kulmala and Emrah Yatkin declare that there is no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
