Abstract
Various animal models are available to study cystic fibrosis (CF). These models may help to enhance our understanding of the pathology and contribute to the development of new treatments. We systematically searched all publications on CF animal models. Because of the large number of models retrieved, we split this mapping review into two parts. Previously, we presented the genetic CF animal models. In this paper we present the nongenetic CF animal models. While genetic animal models may, in theory, be preferable for genetic diseases, the phenotype of a genetic model does not automatically resemble human disease. Depending on the research question, other animal models may thus be more informative.
We searched Pubmed and Embase and identified 12,303 unique publications (after duplicate removal). All references were screened for inclusion by two independent reviewers. The genetic animal models for CF (from 636 publications) were previously described. The non-genetic CF models (from 189 publications) are described in this paper, grouped by model type: infection-based, pharmacological, administration of human materials, xenografts and other. As before for the genetic models, an overview of basic model characteristics and outcome measures is provided. This CF animal model overview can be the basis for an objective, evidence-based model choice for specific research questions. Besides, it can help to retrieve relevant background literature on outcome measures of interest.
Introduction
Cystic fibrosis (CF) is a debilitating disease, with a changing epidemiology. 1 The life expectancy of CF patients has increased from 0.5 to more than 30 years. 2 While this has been called ‘one of the biggest success stories in medicine’, 3 relatively little is known on the developing CF pathology in older patients. This warrants studies on the developing pathology and on new treatments to counteract unmet patient needs, specifically in older patients.
CF animal models may further enhance our understanding of CF pathology and contribute to the development of new treatments.4,5 When planning an animal experiment to answer a specific research question, investigators need to select the most appropriate model. All previously described models should be considered, and the selection should be based on model validity for the specific research question.
We started a systematic review to create a complete and structured overview of the available CF models to aid animal model choice. As a basis for decision making, systematised reviews are preferred over narrative reviews, 6 because they transparently describe their methods and limitations. We retrieved more references than expected (12,303), and could not conduct our full review protocol in the way we planned. We therefore decided to convert our systematic review into a systematic mapping review of all published animal models for CF (797 references). Mapping reviews (or systematic maps) are high-level overviews with broad research questions, which comprise a full comprehensive systematic search, and present global results of the relevant studies in a user-friendly format. 7
The results of our mapping review were split into two parts; we previously published the genetic CF animal models, 8 and describe the other CF animal models in this publication. For genetic diseases such as CF, genetic models may have a higher construct validity (i.e. resemblance in aetiology and pathophysiology between experimental animal and human)9,10 than other animal models. However, differences between animal and human phenotypes with similar genotypes11–14 have been a reason for concerns on face validity (i.e. resemblance in symptoms between experimental animal and human)9,10 of the genetic CF models. Thus, the non-genetic models may still be relevant.
Combined with our preceding paper, this paper should aid scientists in the evidence-based selection of the most appropriate CF model for their studies. Besides, it can help to retrieve relevant background literature on outcome measures of interest.
Methods
The methods for our review, and the deviations from the preregistered protocol (www.SYRCLE.nl, 2015, Leenaars et al.), have been fully described in the first publication. 8 In brief, we performed a comprehensive search in PubMed and Embase. Retrieved references were screened in two distinct phases (title–abstract and full text) by two independent reviewers (out of a team of seven; AH, CH, CP, DH, DV, JR and WB). We included primary studies describing a mutation or intervention inducing CF-like symptoms in live animals, with the authors’ intention to study CF.
For each reference, one reviewer (AH, CL, DH, DV, JE, JR or RK) extracted the following data: language; animal (species); strain/stock (not analysed, see part 1); 8 sex; type of model induction; percentage survival; and outcome measures. If a negative non-model control group was described, the direction of significant changes in untreated model animals compared to non-model control animals was also extracted. Also, in principle, only those outcome measures analysed in vivo from otherwise untreated model animals were extracted. Analyses on materials derived from animal models and only used for further experiments ex vivo, that is cell and tissue cultures or perfused organs, did not meet our original inclusion criteria, and corresponding outcome measures were not systematically extracted. Moreover, several papers measured responses to pharmacological stimulation (other than pharmacological model induction), for example, pancreatic secretion after pilocarpine, dopamine and caerulein, 15 or salivary excretion after acetylcholine. 16 These outcomes were not systematically tabulated. However, extractors’ tendencies towards caution may have resulted in occasional extraction of other types of outcomes. If multiple models were described in a single reference, the data were extracted for each model separately.
Because of the size of the overall review, we did not retrieve information referenced in preceding publications (as common for descriptions of the methods). For data not described in the full-text publication with the corresponding supplements, we registered ‘na’ (not available). Furthermore, outcomes had to be summarised, which results in a loss of information. For example, specimens used for analyses were not fully described, and results were pooled if the authors presented both the crude and corrected (e.g. expressed as a percentage of body weight) data.
The non-genetic models were categorised into infection models (administration of pathogens), pharmacological models (administration of a specific chemical), models based on the administration of human body fluids derived from CF patients, xenograft models and other models.
Data analysis
Extracted data were tabulated by experimental model category. A specific reference may thus occur more than once in the tables (e.g. Hjelte et al., 1990 17 , a publication describing both a pharmacological and a dietary (‘other models’ category) model, is tabulated twice). Reporting frequencies of specific experimental details (as an indication of reporting quality) were calculated separately for the different model categories. To provide an indication of the relevance of the animal model for CF and the aspects of the human disease that are modelled, we tabulated the outcome measures from all studies, with the direction of any potential difference between model animals and controls.
Results
Search, selection and model categories
As described previously, 8 we retrieved 7976 references from Pubmed and 9403 from Embase. After duplicate removal, 12,303 unique references remained. In the screening of titles and abstracts, 9700 references were excluded, in screening of the full texts another 1806. Of the remaining 797 references, 189 described nongenetic animal models which are presented in this paper. Compared to our preceding publication, one duplicate was identified post hoc, and one reference (Abraham, 2008) 18 was post-hoc excluded (based on lacking primary data). The flow of the retrieved references is provided in Figure 1. Note that papers describing both genetic and non-genetic animal models (e.g. Duranton et al., 2012) 19 are included in both parts of our review, where the applicable models are described.
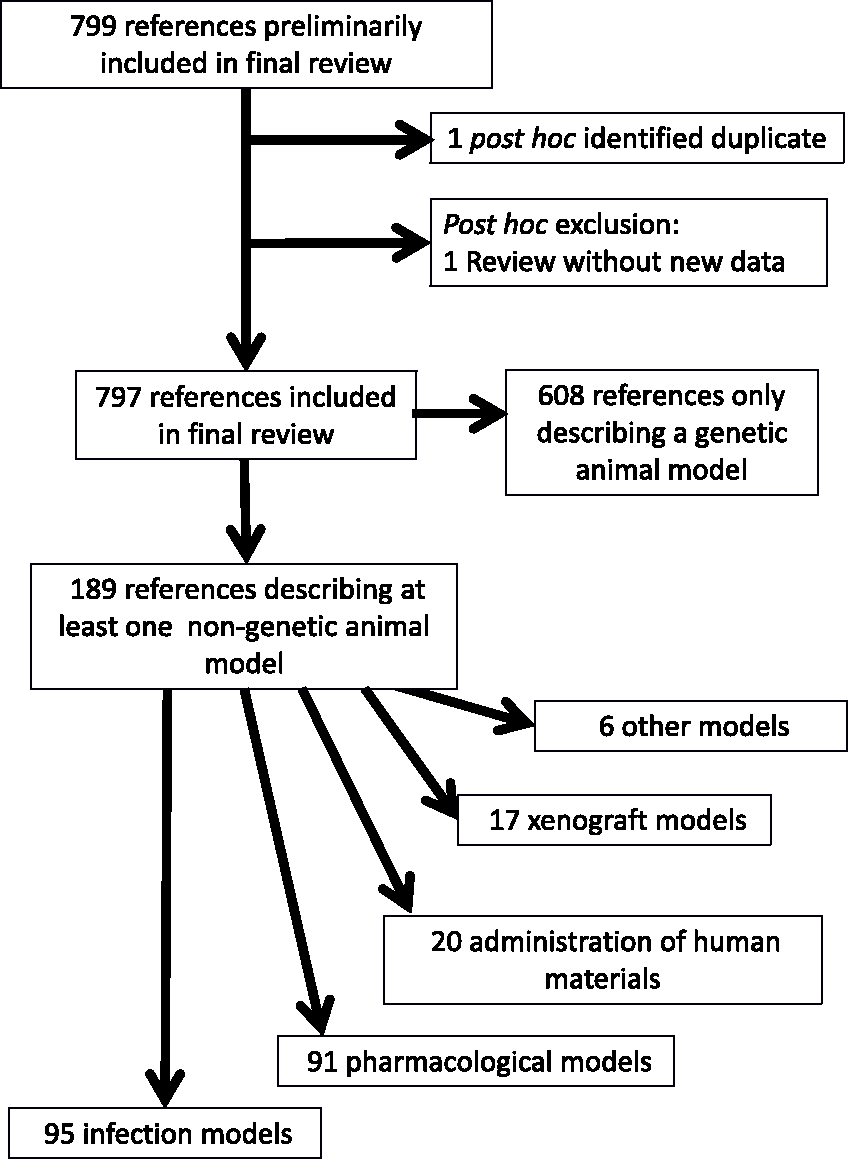
Reference flow.
The animal models were categorised into the following groups: genetic (n = 636, previously described), 8 infection-based (n = 95), pharmacological (n = 91), administration of human materials (n = 20), xenografts (n = 17) and other (n = 6). Note that a paper describing multiple animal models could be included into several categories. All references describing non-genetic CF models are listed in the Supplemental material, Appendix Table 1, tabulated by model category, and listing the species and the outcome measures. Refer back to the Supplemental material, Appendix Table 7 of Leenaars et al. (2019) 8 for the full bibliographic details.
Infection-based CF animal models
Overall, 86 references described 95 CF animal models based on infection. Most of these models reproduce infections commonly seen in CF patients. They were published from 1980 to 2015. Of the 95 models, 60 were in mice, 29 in rats, two in rhesus monkeys, two in fruit flies, one in cats and one in sheep. The sex of the animals was reported for 70 models; 34 were restricted to male animals, 29 to female animals, and seven used both sexes. The percentage of the model animals surviving to a certain time point or experimental stage was described for 39 of the models (survival range: 0–100%).
The most common species of pathogen used is Pseudomonas aeruginosa, which was used in 86 of the models. Only eight models used other bacteria: Burkholderia ce(noce)pacia (n = 4) Staphylococcus aureus (n = 1), Stenotrophomonas maltophilia (n = 1), Escherichia coli (n = 1) and Haemophilus influenzae (n = 1). The study in fruit flies compared P. aeruginosa with various oropharyngeal isolates containing several species. 20
Data on pathogen strain, pathogen concentration, administration route and administration medium were inconsistently reported by the authors of the included studies. Data extraction on these study characteristics added further dissimilarities. Thus, these data could not be analysed. This does not mean that these variables are irrelevant. Several studies compared the response to different strains of P. aeruginosa within the same animal model and observed differences in the severity of the response to different strains.21–27 Many studies used agar(ose), alginate and mineral oil beads to administer the pathogens.28–31 Some of the studies specifically analysed the effects of variations in the administration medium, for example the size of the alginate beads. 29
A full overview of the included infection models with the measured outcomes is provided in the Supplemental material, Appendix Table 1. Of note, the table lists CF animal models primarily based on infection, but infection has also been used to induce lung pathology resembling CF in genetic and other models (e.g. Guillemot et al., 2014, 21 Cowley et al., 1997, 31 Hector et al., 2015 32 and Hoffmannn et al., 2007 33 ).
Pharmacological CF animal models
Overall, 64 references described 91 pharmacological CF animal models. They were published between 1966 and 2015. Of the 91 models, 67 were in rats, 17 in mice, three in cats, three in rabbits and one in hamsters. The sex of the animals was reported for 68 models; 48 were restricted to male animals, 14 to female animals, and six used both sexes. The percentage of the model animals surviving to a certain time point or experimental stage was described for two of the models (0–100% and 100% respectively).
The early pharmacological models were based on the clinical observation of abnormal secretions, and strove to replicate these observations with drugs known to affect the salivary glands. The drugs most used are reserpine (n = 47), isoproterenol (n = 8) and pilocarpine (n = 8). Of note, combinations of reserpine and isoproterenol were also tested,34–36 but data for these were not specifically extracted in line with excluding secondary treatments. Also, the dose, frequency and duration of administration were not specifically analysed. Drugs were mostly administered intraperitoneally (IP, n = 40), orally (PO, n = 11), subcutaneously (SC, n=10) or via unspecified injections (n = 6). Oral administration comprised gavage, 37 but also voluntary intake via food38–40 or drinking water. 41
Other drugs and administration routes comprise, for example, intratracheal elastase administration 42 and intraileal and intranasal perfusion of N-(2-naphthalenyl)-[(3,5-dibromo-2,4-dihydroxyphenyl)methylene]glycine hydrazide (GlyH-101). 43 Besides the classical pharmacological models, we also categorised a paper describing the effects of intratracheal extracts of P. aeruginosa on glycoconjugate release in this group. 44
A full overview of the included pharmacological models with the measured outcomes is provided in the Supplemental material, Appendix Table 2.
CF animal models based on the administration of human body fluids
Overall, 19 references described 20 CF animal models based on the administration of human materials. They were published between 1968 and 2007. Of the 20 models, eight were in oysters, seven in rats, two in mussels, two in rabbits and one in shrimps. The sex of the animals was reported for seven models: four were restricted to male animals, two to female animals and one used both sexes. The percentage of the model animals surviving to a certain time point or experimental stage was explicitly described for only one model: all shrimps survived the treatment. 45
The most frequently administered human materials were serum, sputum and saliva from CF patients. The most common outcome measures were related to cilia movement. A full overview of the included models using administration of human materials, with the measured outcomes, is provided in the Supplemental material, Appendix Table 3.
CF animal models based on xenografts
Overall, 17 references described 17 xenograft CF animal models. They were published between 1987 and 2010. All xenograft models were in mice. The sex of the animals was reported for six models: four were restricted to male animals and two to female animals. The percentage of the model animals surviving to a certain time point or experimental stage was described for none of these models, but most experiments ended in euthanasia.
The xenografts comprised various pulmonary cells and tissues from both humans and rats. When rat tracheas were used, they were populated with human CF cells. The most common outcome measures were related to the cellular immune response. Only few studies reported more functional CF-related outcomes such as, for example, the transepithelial potential difference.46–48 A full overview of the included xenograft models with the measured outcomes is provided in the Supplemental material, Appendix Table 4.
Other non-genetic CF animal models
Overall, six included references described six other CF animal models. These models are listed with the analysed outcomes in the Supplemental material, Appendix Table 5. They were published between 1982 and 2015. Of the six models, four were in rats, one in rabbits and one in chickens. The sex of the animals was reported for four models: two were restricted to male animals and two to female animals. The percentage of the model animals surviving to a certain time point or experimental stage was explicitly described for only one of the models (range: 58–60%).
Four references described dietary interventions to induce CF-like pathology, notably essential fatty acid depletion. The other references described induction of CF-like pathology by heat exposure (intratracheal dry air preheated to 50°C) 49 and nerve stimulation to induce saliva. 50
Note that other interventions resulting in CF-like symptoms could also be combined with models classified in one of the preceding categories and are then not listed here. Examples are hypoxia (8% O2) 19 and fasting. 51
A full overview of the other included models with the measured outcomes is provided in the Supplemental material, Appendix Table 5.
Discussion
We performed a mapping review; a high-level review of publications on animal models for cystic fibrosis. As common for mapping reviews, 7 it comprises a full comprehensive systematic search of a broad field and presents the global results of the relevant studies. Because of the large size of mapping reviews, data extraction is generally limited to key study characteristics and outcomes. Also, in mapping reviews, risk of bias cannot be evaluated against a specific hypothesis. Other recent examples of mapping reviews including animal studies focused on late-life cannabis use 52 and corticosterone measurements. 53
We grouped the included publications by model type: genetic; infection-based; pharmacological; administration of human materials; xenografts; and other. Many included references described two or more models; 28 papers described a genetic and at least one other model, and 29 papers described multiple non-genetic models, mostly within the same category. In this second publication, we provide an overview of the published nongenetic animal models for CF up to December 2015. The Supplemental material lists the basic nongenetic CF model characteristics and the outcome measures, with the direction of a potential difference between model and control animals.
A limitation inherent to mapping reviews is that the high-level data extraction from a large number of papers does not allow for in-depth evaluations of the individual models.7,8 This means that questions on which model is optimal for a specific study remain open. However, this mapping review can provide a solid base for evidence-based model selection. When an animal study is considered, a literature review should be performed to prevent unnecessary duplication and to optimise experimental design. While we do not provide data on tested treatments, we do provide the measured outcomes, which are not always listed in the abstract. Finding the relevant studies for a specific outcome in a (group of) species from our appendices, as described below, will save time compared to starting from scratch.
Together with Part 1, 8 this overview can thus be used to aid the design of future experiments, keeping in mind that more recent primary studies have become available since. Models in which a specific outcome measure has been reported can be found by a text search (including synonyms and overarching terms) of the Supplemental material Appendix Tables 1–5 from both Part 1 and Part 2. Specific types of models can be found by screening the relevant Appendix Table(s) line by line. These strategies should be supplemented with searching at least one literature database such as EmBase to retrieve more recent publications on the topic. Our CF search strings (Table 1 in Part 1) 8 and the SYRCLE animal filters54,55 can be combined to limit to CF animal studies. Screening of the retrieved references can be restricted to those published since 28 December 2015.
The main strengths (systematic methodology, size) and limitations (search date, lack of reference list screening, non-standardised data extraction) of our review were discussed in the first publication. 8
While the oldest genetic animal model for CF dates back to 1974, 56 the second part of this review shows that many different animal models were used before and after the introduction of mutant mice. We classified the CF animal models into six categories: genetic; infection-based; pharmacological; administration of human materials; xenografts and other. We recognize that more detailed and other classifications would also have been possible. However, the team of extractors found these categories straightforward to work with and they were suitable for the goal of this review.
In contrast to the papers on genetic models, most studies included in this part only analysed animals of a single sex, and mainly male animals were used. Testing animals of a single sex is only defendable in rare cases, based on sound argumentation. Examples included in this review are treating only pregnant females to analyse the effects of prenatal drug exposure39,57 and studying only males to analyse the male reproductive system. 58 While it is common to analyse both male and female animals within a single genetic study (probably because of the overall limited availability of animals), there clearly is room for improvement in other fields of animal research.
As discussed before, 8 one of the limitations of this work is the time needed for completion; we performed our searches on 28 December 2015. New animal models for CF have become available since then. For example, at our search date, the only described CF model in sheep was infection-based, 59 and genetic models were restricted to mice, pigs, rats, ferrets and zebrafish. 8 A genetic CF sheep model has become available since then, 60 which is promising because of similarities in lung development and anatomy between sheep and humans. Another example is the development of a second G542X cystic fibrosis mouse model 61 by another group compared to the one included in our review. 62
Besides the development of new models, the characterisation and validation of models has progressed. For example, a genetic rat model was included in our review, 63 but the development of airway mucus abnormalities in this model has been further described since then. 64 Also, in genetic mouse models (C57bl/6 F508del and R117H), the respiratory system has been characterised further since our search. 65 As explained before, 8 an update of our search was not feasible.
Of note, besides these and other new animal models, there also have been great developments in in-vitro methods, comprising, for example, cell lines, primary cells, air–liquid interface cultures and organoids, 66 which will allow for replacement of at least part of the animal experiments.
While some of the animal experiments may soon be replaced by alternatives, and while some of the older models described in this publication can be considered somewhat outdated, one of the powers of systematic reviews is to bring to light rare and ‘forgotten’ experiments and relevant animal models. Older models can be forgotten when newer techniques become more interesting, and it may even be easier to acquire funding for newer models, but newer models do not necessarily result in increased understanding. For the review authors, the models using administration of patient materials to muscles and oysters (1969–1981) are the main example of forgotten models within this review. While these are still animals and thus not a real alternative to an animal experiment, some scientists would consider the use of invertebrate species a good alternative. To what extent the cilia movement in these species is a valid model for cilia movement in humans remains to be determined.
On the other hand, a full systematic review can also retrieve models that are currently considered old-fashioned. All models have limitations, but mainly for the pharmacological models, which are now mostly considered outdated, several authors addressed the limitations of their models. For example, chronic treatment with diuretics was considered of little use to study CF liver disease. 39 The limited usefulness of reserpine- and isoproterenol for modelling the basic CF airway defects was noted in 1990. 67
Based on the current dataset, we cannot properly evaluate described models and studies. In a non-random sample of the included papers, we analysed the reporting of several study design parameters (results described in our preceding publication). 8 Reporting of experimental details was poor. While study quality may exceed reporting quality, poor reporting of experimental detail is a common problem encountered in systematic reviews.
Three types of validity are generally distinguished in the evaluation of animal models: construct, face and predictive validity. Construct validity refers to concordance in disease pathology and aetiology between model animals and patients.9,10 Thus, construct validity should be high for the genetic animal models carrying the same mutation as human CF patients. Several studies indeed show that genetic rat, pig and ferret CF models have mucociliary clearance resembling that in human CF patients, 68 indicating construct validity for these models. However, most CF mice do not develop spontaneous lung disease. 69 Face validity refers to phenomenological concordance, that is similarity in symptoms.9,10 While the face validity for the genetic CF mouse models is low concerning lung disease, most of the studies included in this review have face validity for at least some CF symptoms. Predictive validity is defined as the success of predictions made from the model. 10 We previously argued that actual numbers for predictiveness are more informative than qualitative, subjective analyses of similarities between animal models and human pathology. 70 We are unaware of any quantitative analyses of CF model predictivity. Of note, validation studies are scarce, we included only one formal validation study self-identifying as such in the title. 71
Together with our preceding publication, 8 this paper provides a full overview of the published CF animal models up to the end of 2015. Researchers initiating new CF animal experiments can save time by searching our tables for relevant models and outcomes of interest in relation to their research, and restricting their further literature searches to publications after 2015.
Supplemental Material
sj-pdf-1-lan-10.1177_0023677221990688 - Supplemental material for Animal models for cystic fibrosis: a systematic search and mapping review of the literature. Part 2: nongenetic models
Supplemental material, sj-pdf-1-lan-10.1177_0023677221990688 for Animal models for cystic fibrosis: a systematic search and mapping review of the literature. Part 2: nongenetic models by Cathalijn HC Leenaars, Rob BM de Vries, Joey Reijmer, David Holthaus, Damian Visser, Anna Heming, Janneke Elzinga, Rosalie WM Kempkes, Wouter Beumer, Carine Punt, Franck LB Meijboom and Merel Ritskes-Hoitinga in Laboratory Animals
Footnotes
Acknowledgements
The authors would like to acknowledge Alice Tillema (information specialist) and Peter Mercus (clinician) for reviewing our protocol.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: WB works for ProQR Therapeutics, a company developing an investigational product (eluforsen) for treating CF. CP was involved in this project when she also worked for ProQR Therapeutics. She currently works for BunyaVax BV, a company developing vaccines against neglected viral pathogens that affect animal and human health. In our opinion, these affiliations do not result in competing interests for a systematic review of animal models not describing therapeutic interventions.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NWO [grant number 313-99-310], R2N, Federal State of Lower Saxony, and the DFG [grant number FOR2591, BL 953/11-2].
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
