Abstract
In mice, burrowing is considered a species-typical parameter for assessing well-being, while this is less clear in rats. This exploratory study evaluated burrowing behaviour in three rat strains during training and in the direct postoperative phase after complex intracranial surgery in different neuroscience rat models established at Hannover Medical School or Aachen University Hospital. Male Crl:CD (SD; n = 18), BDIX/UlmHanZtm (BDIX; n = 8) and RjHan:WI (Wistar; n = 35) rats were individually trained to burrow gravel out of a tube on four consecutive days. Thereafter, BDIX rats were subjected to intracranial injection of BT4Ca cells and tumour resection (rat glioma model), SD rats to injection of 6-hydroxydopamine (6-OHDA) or vehicle (rat Parkinson’s disease model) and Wistar rats to endovascular perforation or sham surgery (rat subarachnoid haemorrhage (SAH) model). Burrowing was retested on the day after surgery. During training, BDIX rats burrowed large amounts (mean of 2370 g on the fourth day), while SD and Wistar rats burrowed less gravel (means of 846 and 520 g, respectively). Burrowing increased significantly during training only in Wistar rats. Complex surgery, that is, tumour resection (BDIX), 6-OHDA injection (SD) and endovascular perforation or sham surgery for SAH (Wistar) significantly reduced burrowing and body weight, while simple stereotactic injection of tumour cells or vehicle did not affect burrowing. Despite the training, burrowing differed between the strains. In the direct postoperative phase, burrowing was reduced after complex surgery, indicating reduced well-being. Reduced burrowing was accompanied with postoperative weight loss, a validated and recognised quantitative measure for severity assessment.
Introduction
Burrowing behaviour in mice and rats was first described by Deacon as species-typical behaviour and rewarding activity.1,2 In mouse models, burrowing is considered an indicator of general well-being, as it decreases due to acute pain after laparotomy, 3 colitis 4 and lesions and infections of the central nervous system.1,5–7 Reduced burrowing may not only indicate pain but has also been described as a sensitive parameter for changes in the affective state or cognition.1,6–8 In rats, burrowing decreases in chronic inflammatory and neuropathic pain states9–12 and the chronic state after status epilepticus.13,14 However, so far, burrowing has not been tested in the direct postoperative phase, especially after intracranial surgery of different complexity as applied for neuroscience models.
Due to inherent species differences, comparisons of burrowing behaviour between rats and mice should be made with caution. 15 While mice burrow anything right away out of a tube, rats prefer to dig earth-like substrates and are often trained before the actual testing.2,16 Since inter-and intra-individual variability in burrowing is high, 15 it has been suggested only to test rats that reliably burrow at least more than 500 g at baseline after training.2,9,12,15
When implementing burrowing for severity assessment in scientific experiments, it must be taken into account that the use of specific rat strains has been established over many years in different working groups. In addition, certain settings may only allow the use of a specific rat strain, for example immunodeficient rats,17,18 or rats that are syngeneic to a certain tumour cell line. 19 Furthermore, certain requirements of the animal facility may only allow rats of particular breeders due to different health certificate standards.20,21 Also, despite the rigorous implementation of high standards, even in established and large-animal facilities, environmental conditions may slightly differ, that is, actual handling by different caretakers, housing (e.g. open cages vs. individually ventilated cages) and maintenance conditions. Finally, the actual procedure for burrowing in rats differs between working groups concerning the test duration, the number of training days and baseline testing, the kind and amount of material, the tube design or the time of day, all of which may cause variation in burrowing data.2,11,15
We evaluated burrowing behaviour systematically in neuroscience models established and used for years with certain rat strains in two different locations. At the Hannover Medical School (MHH), BDIX/UlmHanZtm (BDIX) rats are used as a model for intracranial glioma,19,22–24 whereas Crl:CD (SD) rats are used as a model for Parkinson’s disease (PD).25–28 At the Aachen University Hospital (RWTH), RjHan:WI (Wistar) rats are used as a model for subarachnoid haemorrhage (SAH).29,30
We first tested whether BDIX, SD and Wistar rats would show different burrowing behaviour during four training days. After that, the effect of intracranial surgery on burrowing behaviour was tested after (a) intracranial tumour cell injection and resection as a model for intracranial glioma in BDIX rats, (b) injection of 6-hydroxydopamine (6-OHDA) or vehicle as a model for PD in SD rats and (c) endovascular perforation or sham surgery as a model for haemorrhagic stroke in Wistar rats. These different approaches comprise intracranial surgery of varying complexity, ranging from a simple vehicle solution injection to removing a solid tumour via a large craniotomy. Burrowing was performed according to a previously published standard operating procedure (SOP; https://severity-assessment.de/group-area/).
Methods
All investigations were conducted in line with the EU directive 2010/63/EU and the German Animal Welfare Act. Animal health monitoring was carried out according to FELASA recommendations. 21
The studies were approved by the respective authorities as follows: intracranial tumour in BDIX rats, licence number AZ 33.12-42502-04-14/1533; PD model in SD rats, licence number AZ 33.8-42502-04-16/2315 (both at MHH); and SAH model in Wistar rats, licence number AZ 81-02.04.2017.A457 (at RWTH). In addition, burrowing behaviour was tested in rats allocated to ongoing scientific projects at MHH and RWTH. Therefore, an a priori sample size calculation was performed for these projects but not for the explorative burrowing study. The study was not preregistered.
Animals and experimental design
The burrowing behaviour of BDIX, SD and Wistar rats was evaluated during four training days and after intracranial surgery. Animals were group housed during one week of acclimatisation and training. After surgery, the rats were single housed but had visual and olfactory contact with their conspecifics. Blinding was not possible during training because of the different coat colours of the rats and the different test locations, that is, brown BDIX and albino SD rats at MHH, and albino Wistar rats at RWTH. Wistar and SD rats were randomly allocated to sham surgery and to surgery. The persons performing the perioperative burrowing tests were blinded in terms of the actual treatment. Randomisation for BDIX rats was not possible, since tumour resection was done in all rats eight days after glioma cell injection, that is, after developing a solid tumour. At MHH and RWTH, animals were handled similarly, starting on the days before the training, by speaking to them, placing the hand in the cage and gently lifting them by holding them under the chest. No tail lifting was performed. In particular, BDIX and SD rats were kept under the same environmental conditions at MHH, including handling by the same person.
At MHH, eight adult male BDIX rats (>200 g, ∼9–12 weeks old) bred in the Central Animal Facility of MHH were used to train the burrowing behaviour. In these rats, burrowing was also tested before and after local intracranial injection of glioma cells for brain tumour formation and, one week later, for tumour resection. Also, at MHH, 18 adult male SD rats (>200 g, ∼6–9 weeks old) from Charles River Laboratories (Sulzfeld, Germany) were used for training. In these rats, the effect of intracranial injection of 6-OHDA (n = 11) or vehicle (n = 7) as a model for PD was tested.
At RWTH, 35 adult male Wistar rats (>250 g, ∼6–9 weeks old) from Janvier Labs (Le Genest Saint Isle, France) were used for training. In these rats, the effect of the endovascular filament perforation (n = 16) or sham surgery (n = 19) as a model for SAH was tested (please see table 1.).
Additional information regarding animal husbandry and study design is provided in the Supplemental Material.
Burrowing procedure
For burrowing, a plastic tube (length=32 cm; diameter = 10 cm; entrance raised 6 cm above the floor, back end sealed) was filled with 2.5 kg of gravel (2–4 mm in diameter) and placed outside the home cage in a standard open rat cage (RWTH: Tecniplast Type 2000P, l × w × h 612 mm×435 mm×216 mm; MHH: Eurostandard Type IV, l × w × h 598 mm × 380 mm ×200 mm).
During the light phase of the light/dark cycle, burrowing was carried out between 9:00am and 2:00pm. The day before the first training, rats were habituated to the cage with an empty burrowing tube for 60 minutes. For training on four consecutive days, each rat was placed in the empty cage for 30 minutes, after which the gravel-filled burrowing tube was added for 60 minutes. Four BDIX rats were only trained for three days, since these rats burrowed all the gravel from the first day of training. The last training day was taken as a baseline to assess the effect of different complex intracranial surgeries on the status before the operations. The rats were then retested the day after the surgery. The term ‘before’ operation refers to one or two days before surgery. The term ‘after’ operation was the day (i.e. about 24 hours) after surgery. Finally, the gravel was washed with water and acetic acid (0.1%) to remove any confounding odours.
Intracranial surgeries
General procedure for all rats: throughout all experiments, the general well-being of the rats was closely monitored by daily body weight measurements and model-specific clinical score sheets (for details, see Supplemental Material).
MHH: For intracranial tumour formation, BT4Ca rat glioma cells were stereotactically injected via a small burr hole into the right frontal cortex of BDIX rats under general anaesthesia. A large craniotomy was made for tumour removal that radially extended around the original burr hole by about 2 mm (for details, see Supplemental Material).
In the PD model, 6-OHDA was stereotactically injected through a small burr hole into the right medial forebrain bundle of SD rats to lesion the nigrostriatal dopamine pathway. For sham lesioning, rats received vehicle injections (for details, see Supplemental Material).
At the MHH, all rats received carprofen intraoperatively and o.d. on the first two postoperative days (8:00am; 5 mg/kg, s.c.). More details concerning the surgery, anaesthesia and intra- and postoperative care are given in the Supplemental Material.
RWTH: For SAH, a small filament was introduced into the common carotid artery and advanced via the internal carotid artery for vessel perforation at the circle of Willis in Wistar rats under general anaesthesia. For sham operation, the filament was advanced without perforation of the vessel. A craniotomy and a thinned skull bone window were prepared for all rats to measure intracranial pressure and cerebral blood flow. Also, a catheter in the tail artery was used for blood pressure monitoring and blood gas analysis. After 30 minutes of post-SAH recording, vessels were ligated, and wounds were closed. Intraoperative analgesia was provided via fentanyl and buprenorphine injections. For postoperative analgesia, buprenorphine was administered b.i.d. on the first three postoperative days (7:00am and 7:00pm; 0.03 mg/kg, s.c.). More details concerning the surgery, anaesthesia and intra- and postoperative care are given in the Supplemental Material.
Analysis of burrowing, including statistics
The main parameter was the amount of gravel burrowed in grams within 60 minutes. GraphPad Prism v9.0.0 (GraphPad Software, San Diego, CA) was used for statistical analysis. Data were tested against the hypothesis of normal distribution using the Shapiro–Wilk test.
Training: Training data were analysed using a linear mixed-effects model with restricted maximum likelihood (REML) on the amount of gravel as the dependent variable, with the subjects as random effect. The categorical variable strain (with the levels BDIX, SD and Wistar) and the continuous variable time were modelled as fixed effects with their interaction in this model. In the case of multiple comparisons, the p-values were adjusted with the Bonferroni criterion.
The burrowing parameter was assigned to three categories to characterise the burrowing behaviour during training further and to capture the possible range of individual rat performance in this variable: category 1, the rat burrowed on the last training day (measured before OP); category 2, the rat burrowed once or twice during training but not on the last training day; and category 3, the rat did not burrow at all.
Surgery: The effect of cell injection or tumour resection on the amount burrowed in BDIX rats (MHH), the effect of 6-OHDA or vehicle injection in SD rats (MHH) and the effect of sham surgery was determined with a paired t-test. The effect of the endovascular perforation in Wistar rats (RWTH) was determined with a one-sample t-test. Additionally, paired t-tests were applied to pre- and postoperative body weight. All t-tests were two-sided.
Data are shown as mean values with 95% confidence intervals, including all data points, or median values with interquartile range in case of non-Gaussian distribution. Asterisks indicate significant intra-strain differences during training or before/after surgery, and a curly bracket with a circle indicates significant inter-strain differences during training compared to BDIX rats.
Results
Comparison of burrowing of BDIX, SD and Wistar rat strains during training
Overall, a clear strain difference between BDIX versus SD and Wistar rats was found for burrowing. During training, BDIX rats constantly burrowed high amounts of gravel out of the tubes (mean d1 vs. d4: 1899 vs. 2370 g), while SD and Wistar rats either did not engage at all or only burrowed small amounts (SD mean d1 vs. d4: 496 vs. 846 g; Wistar mean d1 vs. d4: 165 vs. 520 g). Performances increased significantly during training only in Wistar rats.
The statistical analysis of the burrowing training was performed with a linear mixed-effect model. It showed a significant effect for the fixed effects strain (F(1.368, 46.51) = 63.28, p < 0.001) and time (days) (F(2.293, 77.96) = 3.359, p = 0.034), but not for the interaction between the fixed effects strain and time (F(3.01, 14.04) = 2.400, p = 0.111). Post hoc analyses, after Bonferroni adjustment, showed that SD and Wistar rats burrowed significantly less material than BDIX rats (both strains p < 0.001), with no difference between SD and Wistar rats (p = 0.366). The analysis of differences between days across all strains revealed significantly higher amounts of gravel burrowed on day 4 compared to the first two training days (d1 vs. d4, p = 0.001; d2 vs. d4, p = 0.009). Although the interaction between strain and day did not reach the level of significance, we additionally analysed the effect of training in different strains with a mixed-effect model. This analysis showed that burrowing performance increased significantly on the third and fourth training day compared to the first training day only in Wistar rats (p < 0.013 and p < 0.008, respectively; Figure 1(a)).
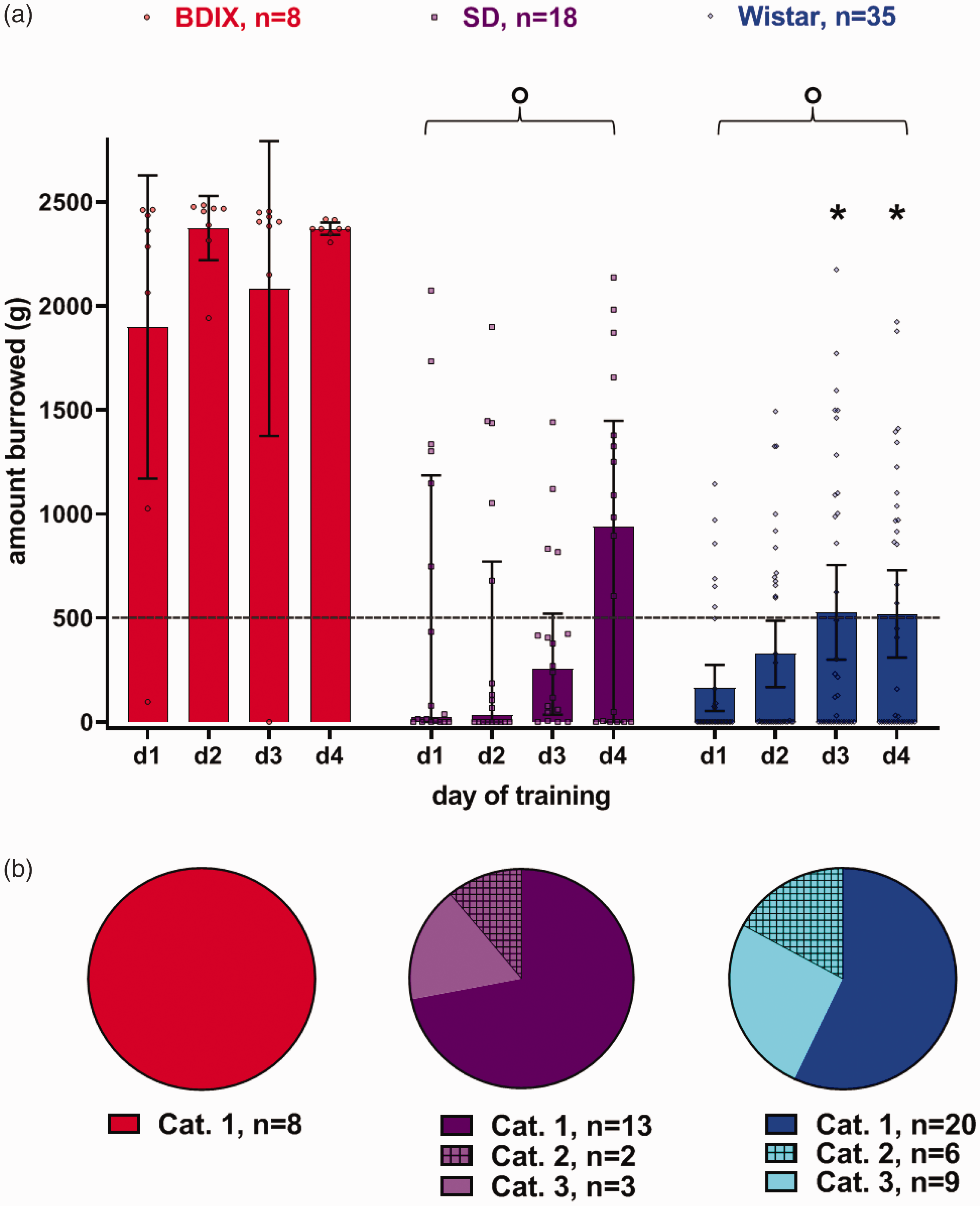
Burrowing performance during training. Burrowing data are shown as the amount of gravel in grams burrowed by BDIX (n = 8), SD (n = 18) and Wistar rats (n = 35) during four training days. Data are shown as mean values with 95% confidence intervals, including all data points (a). Training data were analysed using a linear mixed-effects model on the amount of gravel as the dependent variable, with the subjects as random effect. The categorical variable strain (with the levels BDIX, SD and Wistar) and the continuous variable time were modelled as fixed effects with interaction. In the case of multiple comparisons, the p-values were adjusted with the Bonferroni criterion. Asterisks (*) indicate significant differences (p < 0.05) compared to the first training day within rat strains; a curly bracket with a circle indicates significant inter-strain differences during training compared to BDIX rats. The dotted line shows the 500 g threshold used by other working groups, which serves as an inclusion criterion of rats that reliably burrow at the baseline. The pie chart shows the grouping of training data into three categories (b): category 1, rat burrowed on the last training day before OP; category 2, rat burrowed once or twice during training but not on the last training day; and category 3, rat did not burrow at all. Performances of the individual rats within each strain were assigned to these three categories, and the percentage was calculated.
All BDIX rats (100%; 8/8) burrowed gravel out of the tubes before the OP, whereas in SD rats, only 72% (13/18 rats) burrowed at least on the last day of training (i.e. category 1). Of the remaining SD rats, two burrowed only once or twice during training but not on the last training day (category 2), and three rats did not burrow at all during training (category 3). About half of the Wistar rats (57%; 20/35) burrowed at least on the last training day (category 1), whereas of the remaining Wistar rats, six only burrowed once or twice but not on the last training day (category 2), and nine rats showed no burrowing behaviour at all during training (category 3; Figure 1(b)).
Effect of intracranial surgery on burrowing and body weight
No intraoperative complications occurred, and all animals recovered from anaesthesia uneventfully. After surgery, clinical scores did not deteriorate after tumour cell injection or tumour resection in the intracranial glioma model (BDIX rats) or after vehicle or 6-OHDA injection in the PD model (SD rats). After surgery for endovascular perforation in Wistar rats in the SAH model, scores were mildly elevated.
To assess the effect of different intracranial surgery on burrowing in each rat strain, only rats that burrowed on the last training day (i.e. measure before OP; category 1) were included in the analysis.
Cell injection or tumour resection in BDIX rats at MHH
All rats (n = 8) burrowed on the last training day (i.e. before surgery) and were included in the statistical analysis of surgery. A paired t-test showed that burrowing after cell injection did not differ from preoperative data (p = 0.939). Data of surgery for tumour resection were not normally distributed. Therefore, the Wilcoxon signed rank test for matched pairs was used. The analysis showed that burrowing in BDIX rats decreased after tumour resection (p = 0.016). Qualitative analysis showed that three out of eight rats did not burrow at all after tumour resection. Notably, these three rats did not experience any intraoperative complications such as excessive bleeding. The remaining five rats almost burrowed the total amount of gravel after tumour resection (90% of gravel; Figure 2(a) and (b)).
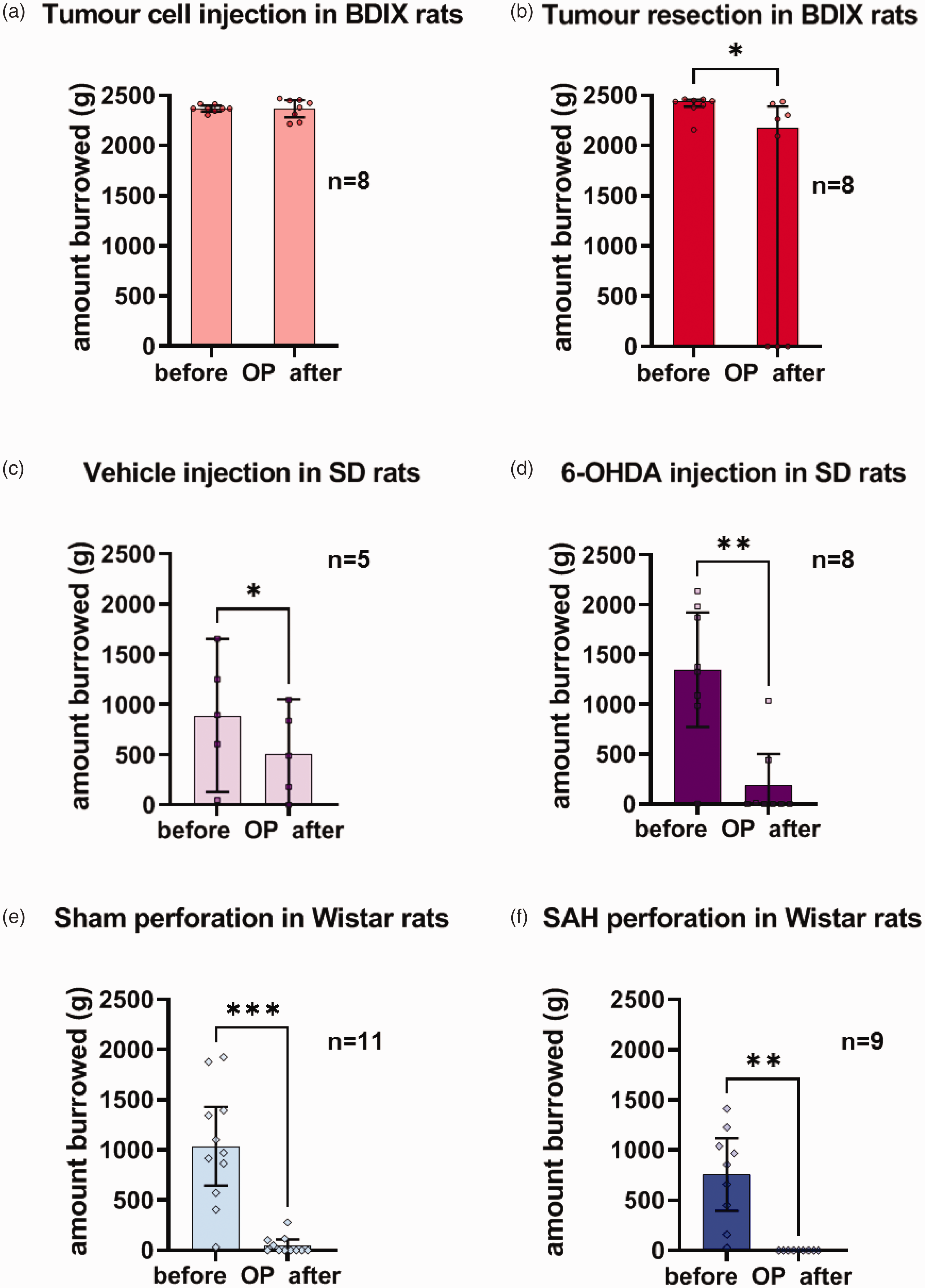
Burrowing performance before and after the operation. Burrowing performance (amount burrowed in grams) before and after the OP in BDIX, SD and Wistar rats that burrowed before the OP ((a)–(f); category 1; see legend to Figure 1(b)). Data are shown as mean values with 95% confidence intervals, including all data points, or median values with interquartile range in the case of non-Gaussian distribution (BDIX rats undergoing tumour resection). The effect of surgery on burrowing behaviour was determined with a paired t-test or a one-sample t-test (Wistar rats undergoing subarachnoid haemorrhage perforation). The difference in gravel amount burrowed before and after the OP was tested. All t-tests were two-sided. The Wilcoxon signed rank test for matched pairs (BDIX rats undergoing tumour resection) was used if the burrowing data were not normally distributed. Significant differences between the measures before and after the OP within a strain and type of surgery are indicated by asterisks (*).
With respect to body weight, a paired t-test showed no change in body weight after tumour cell injection compared to preoperative weight measures (p = 0.158), whereas after tumour resection, rats lost body weight (p = 0.016; Figure 3(a) and (b)).
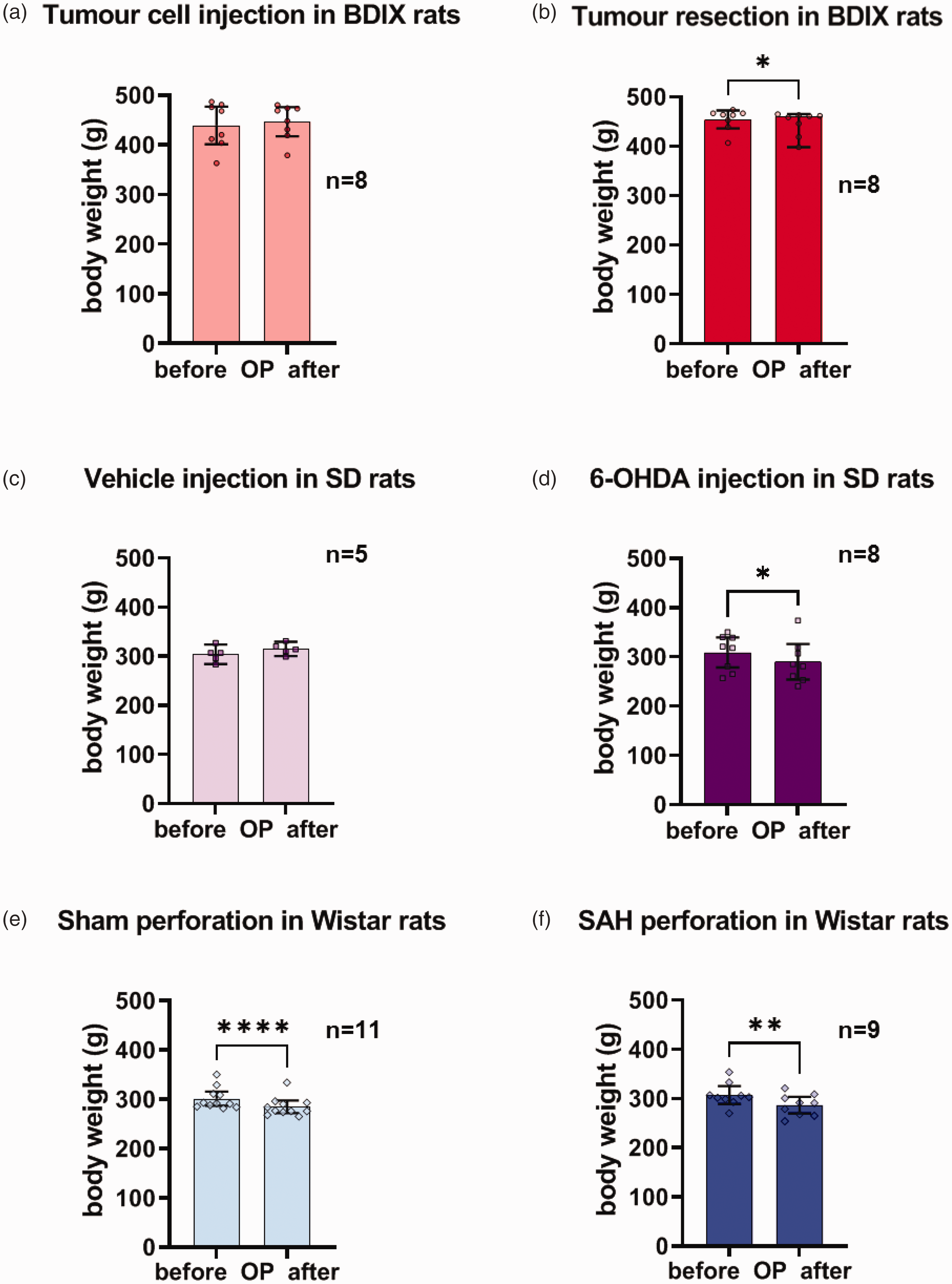
Body weight before and after the operation. Body weight (in grams) before and after the OP in BDIX, SD and Wistar rats that burrowed before the OP ((a)–(f); category 1; see legend to Figure 1(b)). Data are shown as mean values with 95% confidence intervals, including all data points. The difference in body weight before and after the OP was tested with a paired t-test. All t-tests were two-sided. Significant differences between the measures before and after the OP within a strain and type of surgery are indicated by asterisks (*).
6-OHDA- or vehicle injection in SD rats at MHH
For analysis of 6-OHDA or vehicle injection in SD rats, only rats that burrowed before the OP (i.e. category I: n = 8 for 6-OHDA; n = 5 for vehicle) were included in the statistical analysis. A paired t-test showed reduced burrowing after vehicle injection compared to the status before the OP (p = 0.045; Figure 2(c)). However, the overlapping 95% confidence intervals indicate a lack of evidence due to the small sample size (n = 5). Therefore, the significant result of the paired t-test is ambiguous. After 6-OHDA injection, a paired t-test showed reduced burrowing behaviour compared to the status before the OP (p = 0.002; Figure 2(d)).
With respect to body weight, a paired t-test showed a significant reduction after 6-OHDA injection (p = 0.036) but not after vehicle injection (p = 0.214; Figure 3(c) and (d)).
SAH or sham surgery in Wistar rats at the RWTH
For SAH or sham surgery analysis in Wistar rats, only animals burrowing before the OP were included in the statistical analysis (i.e. category I: n = 9 for SAH; n = 11 for sham). Burrowing decreased after surgery in both SAH (one-sample t-test; p = 0.001) and sham-operated rats (paired t-test; p < 0.001; Figure 2(e) and (f)).
With respect to body weight, a paired t-test showed a significant reduction after endovascular perforation (p = 0.002) and sham surgery (p < 0.001; Figure 3(e) and (f)).
Discussion
Burrowing behaviour substantially differed between BDIX, SD and Wistar rats, despite strictly following SOPs at MHH and RWTH. For example, while BDIX rats burrowed high amounts of gravel from the first day of training, about one quarter of the SD and half of the Wistar rats burrowed just once or twice during training or did not burrow at all, even on the last day of training. In BDIX and SD rats, housing and maintenance procedures unlikely account for the differences, since both strains were kept under identical conditions, including the same caretaker at MHH. However, Wistar rats were kept at RWTH and with high-end maintenance conditions for laboratory animals according to FELASA and DIN ISO 9001-2015 standards. Notably, all animals were handled similarly by gently lifting them under the chest.
Moreover, during individually housing and burrowing, rats always had visual and olfactory contact with their conspecifics. Thus, the impact of social isolation should be the same for all rats and may play a minor role as a potentially confounding factor. A recent study also showed no influence of social isolation compared to social housing on rat-related burrowing behaviour. 31
It has been reported that the outcome of behavioural testing can be affected by many factors and may therefore differ between laboratories but should not extensively vary when instructions and SOPs are rigorously followed. 8 Nevertheless, specific mouse and rat strains/stocks have been found to burrow more than others. 2 Different outcomes of behavioural testing, emotional reactivity, susceptibility to neuroscience disease models among different rat strains and even from rats of the same strain but other breeders have been reported before.32–35
In the present study, the inbred BDIX rat strain, which is used as a model for an intracranial tumour because of its syngeneicity to the BT4Ca rat glioma cell line, burrowed the most. Instead, SD and Wistar rats, which are the most frequently used outbred rat stocks worldwide, burrowed less. This finding corroborates previous studies demonstrating highly variable inter- and intra-individual burrowing behaviour in Wistar and SD rats.10,11,15 However, some researchers reported that SD and Wistar rats burrowed more than 1400 g gravel.9,10 An explanation may be that outbred stocks are genetically heterogeneous and thus show more variability in burrowing.33,34
Most working groups expose rats to two or three training sessions before the effect of an experimental intervention on burrowing behaviour as a measure for welfare is tested.11,15 In the present study, however, four days of training only marginally improved burrowing in Wistar rats while not affecting SD rats. Different strategies have been proposed to deal with rats that do not show burrowing behaviour: (a) excluding animals that burrow <500 g, which, however, leads to the exclusion of about 10% of rats;9,11–13 (b) training together with a ‘good burrowing’ social partner;2,9,10,36 or (c) testing burrowing behaviour before or during the dark cycle, since rats are nocturnal. 2
However, the exclusion of poor burrowing rats, as done by several working groups,9,11–13 would increase the initial number of rats exposed to the burrowing training procedure to generate a group of reliable burrowers for severity assessment in the actual scientific experiment. If the criterion of 500 g had been applied in the present work, this would have led to the exclusion of one more Wistar and five more SD rats. Nevertheless, all the suggestions above would be worth the effort if burrowing was a robust measure for severity assessment in specific scientific settings.
It has been reported that burrowing is reduced in the chronic state of rat disease models associated with pain, including mucositis, 15 neuropathic pain,9,10,36–38 inflammation9–11,15,39–41 and arthritis.12,42 In a recent study, burrowing also detected the burden of rats that display spontaneous epileptic seizures in the chronic state after status epilepticus.13,14 For acute postoperative pain, the suitability of burrowing has only been investigated after laparotomy in mice.2,3,43–45 We specifically investigated burrowing behaviour in the direct postoperative phase after intracranial surgery with different complexity. Notably, these intracranial surgical approaches are exemplary for most surgical techniques used in neuroscience research, have been established for many years and are routinely applied to rats in our and other laboratories to investigate pathophysiology and treatment of neurological disorders, that is, brain tumour,19,22,23,46 PD25,47 and stroke.29,30,48–50
All rats that did not burrow at all on the last training day (i.e. the measure before the OP) were excluded in assessing the effect of intracranial surgery.
In BDIX rats, intracranial stereotactic tumour cell application via a microinjection cannula through a small burr hole did not affect burrowing behaviour. In contrast, after tumour resection, via a large craniotomy, burrowing was significantly reduced. It has been reported before that surgery for tumour resection imposes more burden to the rat than inert fluid/cell injection in brain regions via injection through a burr hole. 51 However, detailed data analysis showed that five of eight rats continued with normal or slightly reduced burrowing after tumour resection. In comparison, three BDIX rats completely stopped burrowing after tumour removal. No intraoperative complications, such as excessive bleeding or other confounds, occurred during surgery in these rats. In that regard, it has been shown that BDIX rats with intracranial glioma formation still vigorously burrow until reaching the humane end-point criterion. Notably, they already show other apparent signs of burden at this stage, for example reduced clinical scoring and weight loss. 24 Interestingly, after tumour resection, body weight was reduced, while cell injection did not affect body weight.
In SD rats, burrowing was reduced after 6-OHDA microinjection, while the effect was ambiguous after vehicle injection. However, the low sample size, attributed to the fact that only about three quarters of the SD rats burrowed at all, renders an effective statistical analysis ineffective. With regard to the reduced burrowing after 6-OHDA injection, it remains open to which extent motor disturbances contribute to the reduced burrowing, as in the rat PD model, reduced motor activity has been described before.51,52 Interestingly, SD rats also lost body weight after 6-OHDA microinjection. Notably, anaesthesia and perioperative analgesia and the procedure of stereotactic surgery did not differ between BDIX and SD rats at MHH. Furthermore, we did not observe any apparent differences in the recovery of SD and BDIX rats after anaesthesia.
Wistar rats, however, burrowed significantly less gravel, after both endovascular perforation for SAH and sham surgery. Reduced burrowing was accompanied by postoperative weight loss, which is widely used and recommended as a parameter for severity assessment. The complex and invasive surgical procedure in this model, that is, craniotomy for intracranial pressure detection and the filament insertion in the neck region, most likely account for the decline in burrowing. However, actual rupturing of the artery with bleeding into the subarachnoid space may not add further burden to the animals compared to the sham surgery alone. This is different from human patients, where an initial severe headache accompanies SAH due to increased intracranial pressure. 53 In the SAH rat model, pain relief using buprenorphine may be sufficient to block this additional burden. On the other hand, after surgery for SAH, rats completely stopped burrowing, which may also indicate a floor effect in this rat strain. In addition, cognitive or motor dysfunctions after SAH might add additional burden to the animal.49,53,54
A potentially confounding factor is the use of different anaesthetics and analgesics 8 in the other rat models. At MHH, however, BDXI and SD rats are both treated with carprofen. In both models, simple intracranial surgery, that is, vehicle injection in SD and tumour cell injection in BDIX rats, did not lead to reduced burrowing, indicating that carprofen does not affect burrowing behaviour. However, at the RWTH, burrowing was decreased in Wistar rats after surgery and sham surgery, which may be related to buprenorphine treatment. Nevertheless, burrowing was not affected in Wistar rats that received only anaesthesia and analgesia (data not shown). These findings are also supported by Liles and Flecknell as well as by Roughan and Flecknell. They found no changes in the behaviour of rats induced by the analgesics alone except for increased motor activity in buprenorphine treated rats.55–57
So far, body weight and clinical scoring have been widely used for severity assessment in laboratory animals, but other parameters are increasingly introduced and discussed as valuable. However, the analysis of facial expressions, 58 for example, is limited by the need for extensive observer training and particular subjectivity, although there have been attempts for automation recently. Ultrasonic vocalisation requires special expensive equipment, 59 and for body postures, the analysis is very labour intensive if not fully automated. 60 Burrowing in rodents has been proposed as valuable for assessing well-being, as it may be a species-typical behaviour and is cheap, easy, observer independent and objective. 2
However, rats need to be trained to burrow gravel out of the tubes, and the burrowing procedure is very labour intensive. Notably, in the present study, weight loss after surgery was accompanied by a reduction in burrowing. Therefore, burrowing may not add further information for assessing well-being in the direct postoperative phase after intracranial surgery.
Conclusion
At least in the rat strains used in this study, we demonstrated major differences in burrowing behaviour, with inbred BDIX rats genuinely burrowing high amounts of gravel, while outbred SD and Wistar rats showed less and more variable burrowing behaviour. Overall, reduced burrowing after surgery can be related to the different complexity of the intracranial interventions. While simple cell or fluid injection had only minor effects, SAH led to complete loss of burrowing behaviour, indicating impaired well-being after complex intracranial surgery. Interestingly, reduced burrowing in the SAH model, after 6-OHDA injection and after tumour resection, was accompanied by postoperative weight loss – a validated and recognised quantitative measure for severity assessment. Nevertheless, as already shown in chronic epilepsy models, burrowing may be a valuable measure in the chronic state of neuroscience models. In that regard, cognitive dysfunction or anxiety have been shown to reduce burrowing behaviour and may gain more importance in chronic disease states but have not been behaviourally addressed in the present study.
Overview of animals and experimental design.

At MHH, adult male BDIX rats were used to assess burrowing before and after local intracranial injection of glioma cells for brain tumour formation and tumour resection. Also, at MHH, adult male SD rats from Charles River Laboratories (Sulzfeld, Germany) were used to assess the effect of intracranial injection of 6-OHDA or vehicle as a model for PD. At RWTH, adult male Wistar rats from Janvier Labs (Le Genest Saint Isle, France) were used to assess the effect of endovascular filament perforation or sham surgery as a model for SAH.
MHH: Hannover Medical School; RWTH: Aachen University Hospital; PD: Parkinson’s disease; SAH: subarachnoid haemorrhage.
Supplemental Material
sj-pdf-1-lan-10.1177_00236772211072977 - Supplemental material for Burrowing behaviour of rats: Strain differences and applicability as well-being parameter after intracranial surgery
Supplemental material, sj-pdf-1-lan-10.1177_00236772211072977 for Burrowing behaviour of rats: Strain differences and applicability as well-being parameter after intracranial surgery by Ann-Kristin Riedesel, Annika Bach-Hagemann, Arif Abdulbaki, Steven R Talbot, René Tolba, Kerstin Schwabe and Ute Lindauer in Laboratory Animals
Footnotes
Acknowledgements
The authors thank Ekaterina Harder for her excellent support in carrying out the burrowing tests in the Wistar rats.
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was funded by the German Research Foundation (Deutsche Forschungsgemeinschaft – DFG, grant number SCHW1176/7-1, SCHW1176/7-2 to KS, LI588/5-1, LI 588/5-2 to UL, and TO542/5-1, TO542/6-1 to RT) as part of the German research unit ‘Severity assessment in animal based research’ FOR 2591. The funder had no role in study design, data collection and analysis, decision to publish or manuscript preparation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
