Abstract
Non-alcoholic fatty liver disease (NAFLD) is a common problem with a wide variety of phenotypes. While its pathogenesis is still not fully understood, several risk factors for disease progression have been identified. Therefore, defining adequate animal models may serve to unreveal the pathogenesis in NAFLD. We studied Lewis and Sprague-Dawley rats of both genders (n = 6) fed standard (Std) or high-fat (HF) diet for three weeks. Disease stage was assessed by haematoxylin–eosin, Azan Heidenheim and Oil-Red staining, apoptosis by single-stranded DNA (ssDNA) detection and liver regeneration by Ki-67 staining. Serum markers of liver injury and lipid metabolism including adipocytokines were analysed. Livers of both strains and genders fed with HF diet demonstrated evidence of steatosis. Lewis rats developed microvesicular steatosis whereas Sprague-Dawley rats presented macrovesicular steatosis accompanied by pronounced fibrosis. Female gender of both strains was associated with lower steatosis grade and higher proliferation rate (P < 0.05). Gender-specific differences were most prominent in Lewis rats on a HF diet, where females showed lower alkaline phosphatase, cholesterol, triglyceride and leptin levels and a more favourable low-density lipoprotein/high-density lipoprotein ratio than males (P < 0.05). Reverse transcriptase-polymerase chain reaction analysis was performed to demonstrate changes in expression of various genes important for liver regeneration, fibrosis and steatosis. HF diet induced downregulation of proangiogenic genes such as vascular endothelial growth factor receptor 1 and 2 (P < 0.05) in males was not present in females. In conclusion, strain and gender served major roles in disease progression. These differences should be considered when designing studies and may offer new ways to advance therapeutic strategies.
The increasing burden of non-alcoholic fatty liver disease (NAFLD) has resulted in greater attention towards its associated rise in morbidity and mortality. Even children can be affected. Among the various risk factors identified so far, a high-fat (HF) diet is one major prerequisite. 1 The spectrum of NAFLD ranges from bland fatty infiltration to nonalcoholic steatohepatitis (NASH), fibrosis, cirrhosis and can eventually lead to end-stage liver disease with an increased risk of hepatocellular carcinoma. 2 Our current understanding of the pathogenesis may be explained by the two-hit hypothesis, where steatosis induced by increased fatty acid synthesis and hepatic uptake is followed by oxidative stress and release of proinflammatory and profibrogenic mediators. 3
NAFLD has been associated with several conditions such as ‘metabolic syndrome’ (including obesity, insulin resistance and dyslipidaemia), metabolic diseases, surgical interventions, medications, age, ethnicity and gender. 2 Gender-specific differences in prevalence, degree of liver injury and adipocytokine levels of adolescents with NAFLD have been reported. 4 In general, NAFLD is more common in men than in women and a reversal in gender distribution following menopause suggests an oestrogen-mediated protective effect on liver fat accumulation. 5 The higher prevalence of NASH in morbidly obese men compared with women indicates gender differences even in the spectrum of NAFLD. 6 However, animal studies showed conflicting results regarding oestrogen-mediated protection against the development of steatosis.7–9 Additionally, strain seems to be an important factor for the induction of fatty liver also, but mechanisms causing interstrain differences in the development of NAFLD are unknown.10,11 In the present study the impact of gender and strain on the degree of hepatic steatosis, lipid metabolism and gene expression was investigated in a model of HF diet induced steatosis. Gender and strain-specific differences may have translational implications for the evaluation of novel therapeutic strategies for the treatment of liver disease.
Materials and Methods
Animals and study design
All experimental procedures were conducted in accordance with the German Animal Welfare Law and with the approval of the district administrative authority of Muenster. Animals were accommodated in air-conditioned rooms with a 12 h light/darkness cycle and received water and food ad libitum. The study was performed in 24 Lewis and 24 Sprague-Dawley rats (Charles River, Sulzfeld, Germany) at the age of five weeks and weighing 90–120 g, one-half each female or male, respectively. Six animals per gender and strain were regarded as being sufficient for statistical analysis. Animals were fed with either standard nutrition (11% kcal fat, 24% kcal protein and 65% kcal lipids; Diet 1320, Altromin GmbH & Co KG, Lage, Germany) or HF diet (71% kcal fat, 18% kcal proteins, 11% kcal saccharides; Highfat Lieber-DeCarli Diet, Altromin GmbH & Co KG) containing a high proportion of fat calories for a three-week period. 12 Body weight was recorded regularly at three-day intervals. At the end of each experiment blood samples and liver biopsies were taken for analysis.
Serum chemistry
Bilirubin, aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (AP), pseudocholine esterase (PCHE), cholesterol, triglycerides, low-density lipoprotein (LDL) and high-density lipoprotein (HDL) were determined at 37°C photometrically or by enzymatic absorption photometry in a Vitros 250-Analyzer (Ortho Clinical Diagnostics, Neckargemünd, Germany). Leptin and adiponectin concentration was assessed by enzyme-linked immunosorbent assay (ELISA) using the following commercial kits: mouse-leptin ELISA kit (R & D, Minneapolis, MN, USA) and Assay Max Human adiponectin kit (Assay Pro, St Charles, MO, USA).
Histological analysis
Sections of 10 μm thickness were prepared from liver specimens fixed in 4% formalin and embedded in paraffin followed by haematoxylin/eosin staining. Oil-Red staining was performed in cryostat sections of 6–8 μm thickness from native cryopreserved tissues. Sections were stained with fresh filtrated Oil-Red solution for 10 min followed by Mayer's haematoxylin staining and mounted with Kaiser's glycerine–gelantine at 40°C. Furthermore, Azan Heidenheim staining was performed in liver sections of all animals to assess the fibrosis stage. Specimens were evaluated by an experienced pathologist using a modified scoring system originally developed by Kleiner and Brunt. 13 Size of fat vacuoles (microvesicular, macrovesicular), percentage of affected cells by fat vacuoles, fibrosis stage, lobar inflammation and cell damage were assessed as described. 14 The pathologist was blinded to gender, strain and diet used.
To determine whether apoptosis was related to steatosis grade sections were stained with primary antibodies against single-stranded DNA (ssDNA) (IBL Co Ltd, Gunma, Japan). Rabbit primary antibodies were detected using DAKO anti-rabbit EnVision-HRP (DAKO Cytomation) and NovaRed substrate kit (Vector Laboratories, Burlingame, CA, USA). Sections were counterstained with Ehrlich haematoxylin and mounted with Kaiser's glycerinegelantine. ssDNA-positive hepatocytes and non-parenchymal cells were counted in consecutive microscopic areas under ×400 magnification. For assessment of liver regeneration an antibody (MIB-5) specific for the rat homologue of the Ki-67 proliferation associated antigen (DAKO Cytomation) was used as described previously. 15 For semiquantitative analysis, Ki-67 positive hepatocytes and non-parenchymal cells were counted under ×400 magnification.
Realtime quantitative reverse transcriptase-polymerase chain reaction (RT-PCR) analysis
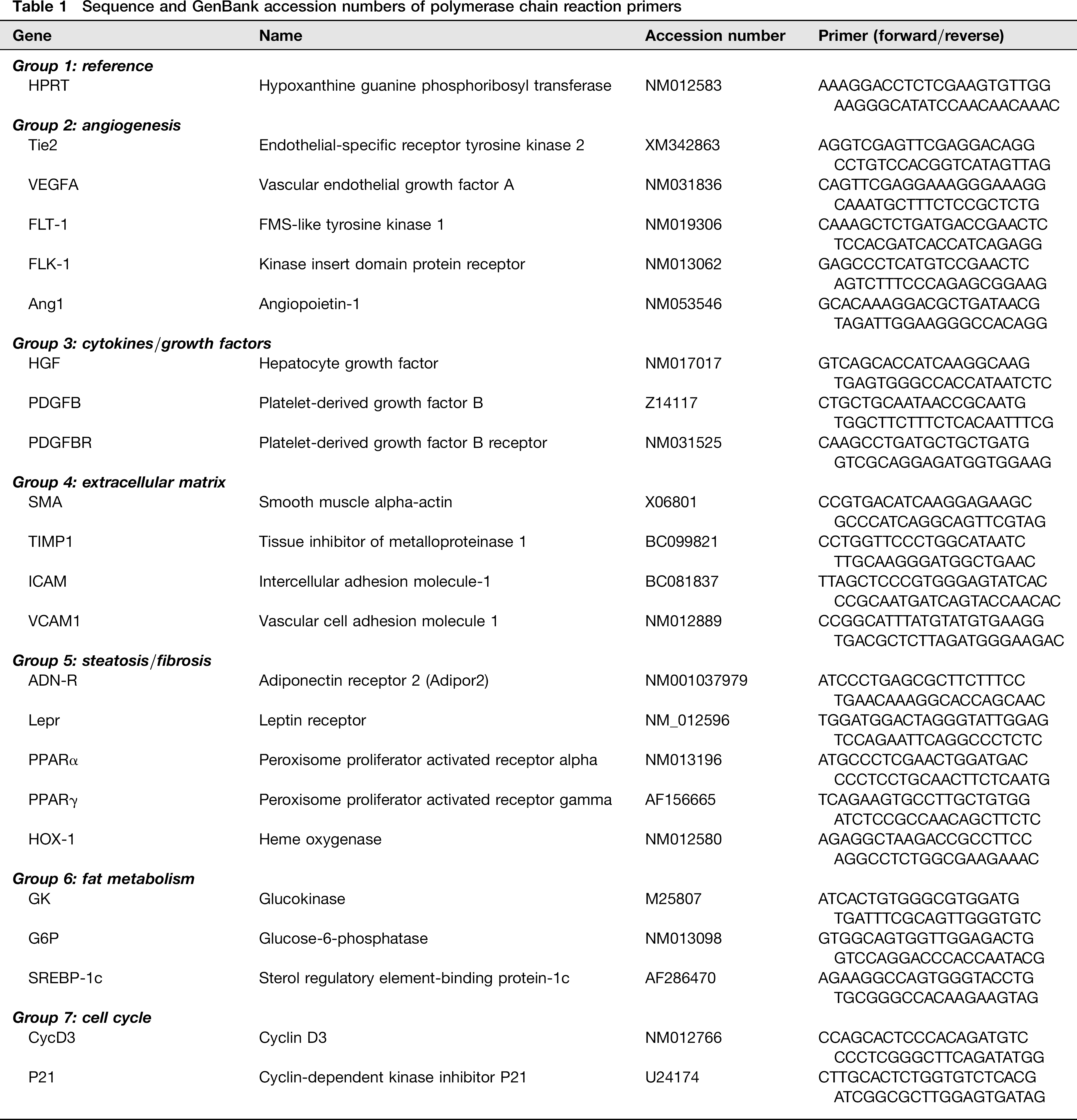
Snap-frozen liver tissue was homogenized and applied to RNeasy columns (Qiagen, Hilden, Germany) for total hepatic RNA extraction. RNAs were reverse transcribed using SuperScript II (Invitrogen, Karlsruhe, Germany) according to the manufacturer's instruction. PCR primers (Table 1) were designed using Primer 3 Express Software (Whitehead Institute for Biomedical Research, Cambridge, MD, USA) and purchased from TIB Molbiol, Berlin, Germany. Primers were located such that exon–intron boundaries of the respective genes result in PCR amplicons of 90–120 bp. Specificity of products was confirmed by ethidium bromide-stained agarose gel. For quantitative PCR analysis, an ABI Prism 7900 HT sequence detection system (PE Applied Biosystems, Foster City, CA, USA) was used. Three-stage program parameters were as follows: 2 min at 50°C, 10 min at 95°C and then 40 cycles of 14 s at 95°C, and one minute at 60°C. Data were normalized to the housekeeping gene HPRT and fold gene expression differences were determined using the ΔΔ ct method.
Sequence and GenBank accession numbers of polymerase chain reaction primers
Statistical analysis
Data are expressed as means±SD. Significances were analysed by t-test, Mann-Whitney rank sum tests or analysis of variance (ANOVA), including Holm-Sidak pairwise comparisons with SIGMASTAT 3.1 (Systat Inc, Point Richmond, CA, USA). P < 0.05 was considered significant.
Results
Gender and strain-specific morphometric differences
All animals fed with a standard diet or a HF-diet survived up to the end of the experiment. In some animals daily food intake was monitored for analysing differences in daily caloric intake. Daily caloric intake was significantly higher in the HF diet group (46.0 ± 7.7 kcal/100 g body weight) than in the standard nutrition group (28.7 ± 2.5 kcal/100 g body weight). In general, Sprague-Dawely rats became significantly heavier than Lewis rats and males heavier than females in both strains (P > 0.05; Figure 1). Within six days male Lewis rats fed with a HF diet showed a significant higher body weight gain than their controls fed with standard nutrition. Female Lewis as well as Sprague-Dawley rats of both genders did not show those diet-dependent differences during the three-week feeding period.
Body weight gain (in %of baseline weight). Lewis (LEW) and Sprague-Dawley (SD) rats of both sexes were randomized to either standard nutrition (Std) or high-fat diet (HF) at five weeks of age. *P < 0.05 versus ♀ fed with corresponding diet; **P < 0.05 versus standard nutrition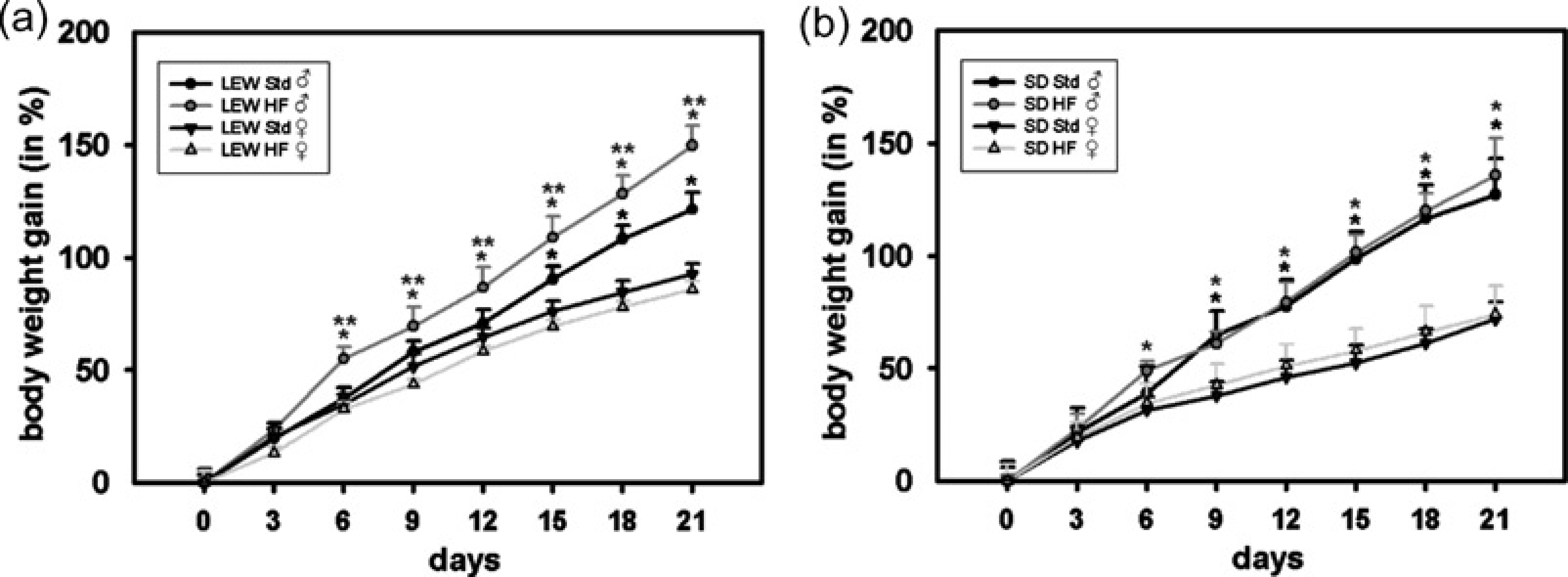
Livers of both strains and genders fed with a HF diet showed evidence of steatosis. In all specimens of control animals fed with standard nutrition no noticeable morphological abnormalities were observed (Figure 2a). In Lewis rats being fed a HF diet caused a microvesicular fatty vacuolation. Male Lewis rats developed a grade 3 steatosis with small intracytoplasmic fat vacuoles in >66% of liver cells whereas in female individuals much less hepatocytes (33–66%) were affected (Figure 2b). Further histological analysis revealed no noticeable signs of fibrosis, inflammation or hepatocyte damage. Livers of Sprague-Dawley rats on a HF diet exhibited a macrovesicular steatosis with widepread deposition of large lipid droplets. Although the gender-specific differences were only minor it was noteworthy that male rats showed a tendency toward larger vacuole size and higher steatosis grade. In general, Sprague-Dawley rats showed more architectural distortion with scattered fibrosis, neutrophil infiltration and hepatocyte ballooning than Lewis rats. Signs of fibrosis, lobar inflammation and hepatocyte damage were more pronounced in male than in female rats (Figure 2b).
Assessment of hepatic steatosis. (a) Representative histological sections of male and female Lewis (LEW) and Sprague-Dawley (SD) rats fed with either standard (Std) or high-fat (HF) diet for three weeks are shown. Staining and original magnification: haematoxylin and eosin (H&E) (×100); Oil-Red (×200); insets show magnified views of hepatic lipid vesicles (LEW: microvesicular; SD: macrovesicular). Panel b shows evaluation of histological sections using a modified scoring system originally developed by Kleiner and Brunt
13
(*P < 0.05 versus standard nutrition). Histological changes in the development of NAFLD were more pronounced in male than in female rats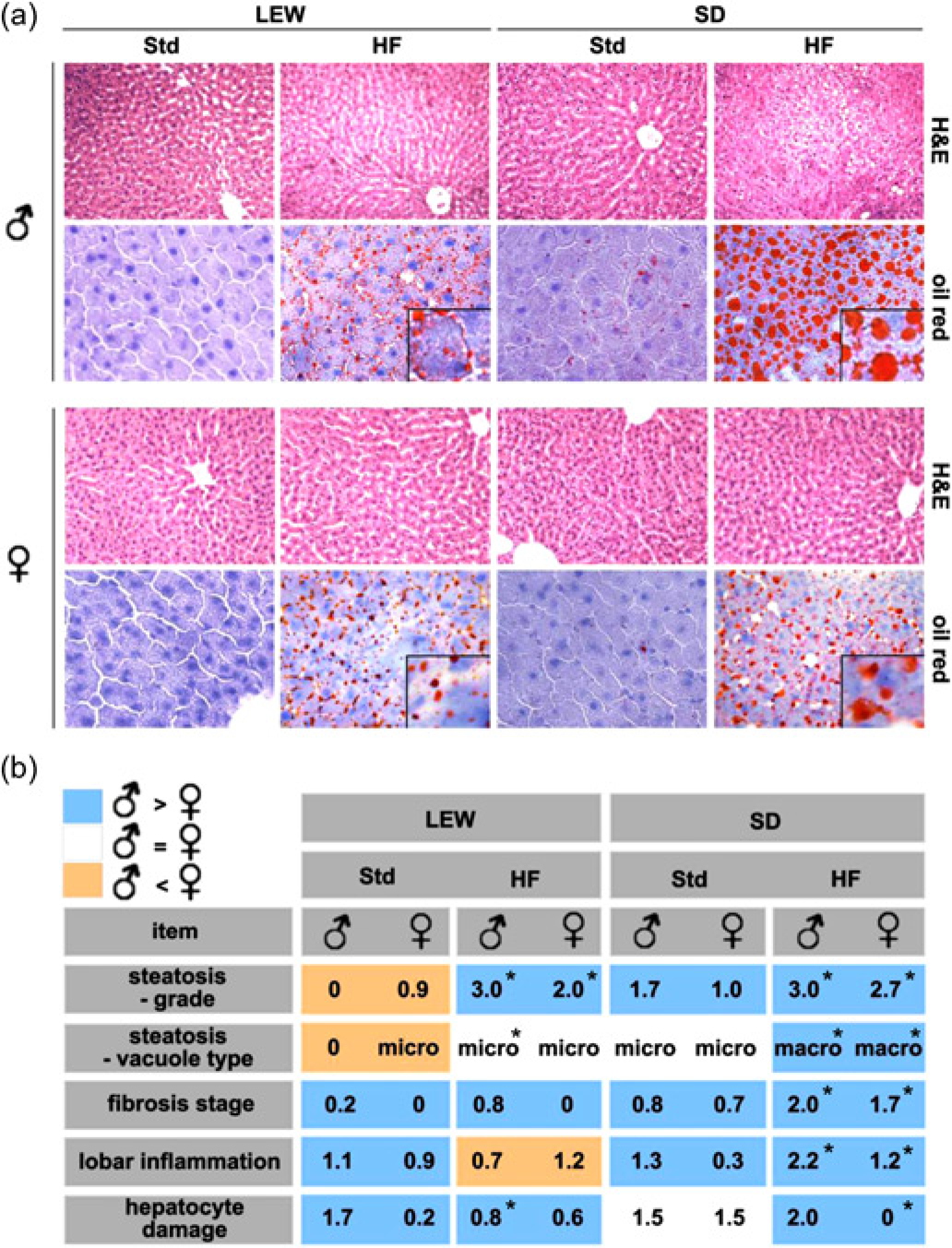
To determine whether differences in the development of steatosis were associated with proliferation and apoptosis we performed Ki-67 and ssDNA immunostains. In male Lewis as well as male Sprague-Dawley rats fed with a HF diet a two to three-fold higher proliferation rate of hepatocytes was detected compared with animals on standard nutrition (P < 0.05; Figure 3a). The number of Ki-67 positive non-parenchymal cells did not differ in those animals. In female rats of both strains proliferation was not affected by diet. However, female Lewis rats showed a 2- to 10-fold higher proliferation rate of hepatocytes and non-parenchymal cells than their male counterparts in general (P < 0.05). In Sprague-Dawley rats this was just the case for non-parenchymal cells (P < 0.05). Apoptotic cells were mainly localized in the centrilobular regions of the liver. Apoptosis rate of hepatocytes and non-parenchymal cells was not affected by diet, strain or gender (Figure 3b). A tendency toward higher apoptosis rates could be observed in female rats and animals on a HF diet.
Cell proliferation and apoptosis. Gender-specific comparison of Lewis (LEW) and Sprague-Dawley (SD) rats fed with standard (Std) or high-fat (HF) diet for three weeks. Panel a shows quantitative analysis of Ki-67 positive hepatocytes (Hep) and non-parenchymal cells (NPC) (identified by histochemistry, ×200, red color). (b) Single-stranded DNA immunhistology of apoptotic cells (×200; brown colour). *P < 0.05 versus ♂ fed with corresponding diet; HPF = high power field (magnification ×200), *P < 0.05 versus standard nutrition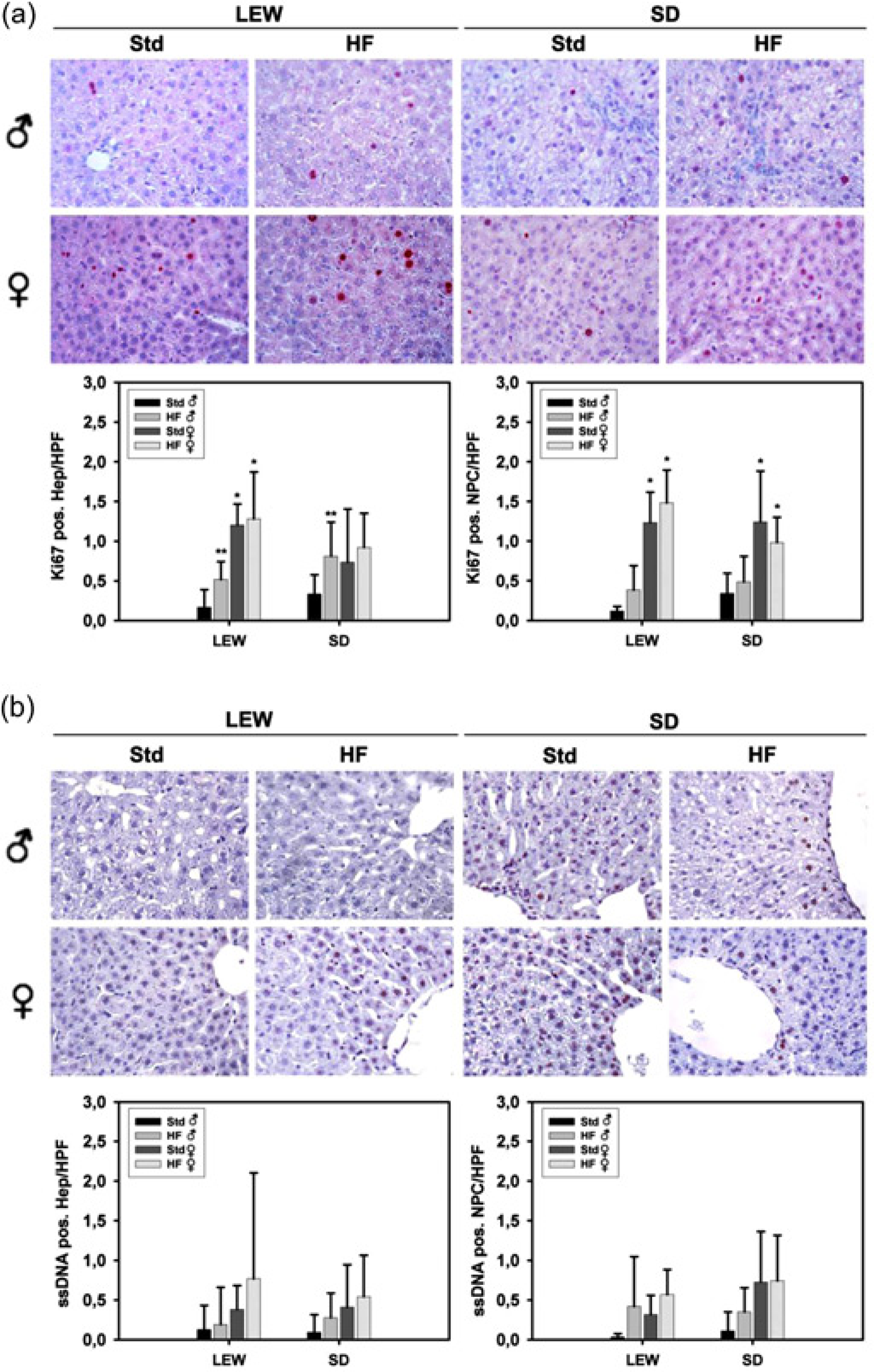
Serum markers of liver injury
Bilirubin levels were within normal ranges in all animals. Induction of steatosis by a HF diet did not correlate with serum levels of transaminases as markers of haepatocellular injury within this short-term exposure to a HF diet (Figure 4). AST levels did not significantly differ. Female Lewis rats on a HF diet showed higher ALT levels compared with their corresponding controls fed with standard nutrition (P < 0.05). In male Lewis rats ALT levels tended to be higher also without reaching statistical significance (87.5 ± 17.0 U/L versus 70.7 ± 5.2 U/L). By contrast, male Sprague-Dawley rats fed with a HF diet showed even lower ALT levels than their controls (55.8 ± 9.4 U/L versus 78.7 ± 11.4 U/L, P < 0.05). But in general, absolute differences in transaminase levels were only minor and therefore negligible. Animals of both strains and genders on a HF diet had a significant increase in AP levels by approximately two-fold compared with controls on standard nutrition (Figure 4, lower left panel, P < 0.05). It was noteworthy, that the female gender in both strains, Lewis and Sprague-Dawley, was associated with significantly lower AP levels than the male (P < 0.05). Furthermore, female rats of both strains presented with significantly higher serum PCHE levels than their male counterparts irrespective of diet used (Figure 4, lower right panel, P < 0.05). HF diet induced an increase in PCHE levels in male and a decrease in female Sprague-Dawley rats (P < 0.05). This parameter was not affected by diet in Lewis rats.
Serum markers of liver injury. Aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (AP) and pseudocholine esterase (PCHE) levels measured in serum of Lewis (LEW) and Sprague-Dawley (SD) rats receiving either standard (Std) or high-fat (HF) diet for three weeks. *P < 0.05 versus ♂ fed with corresponding diet; **P < 0.05 versus standard nutrition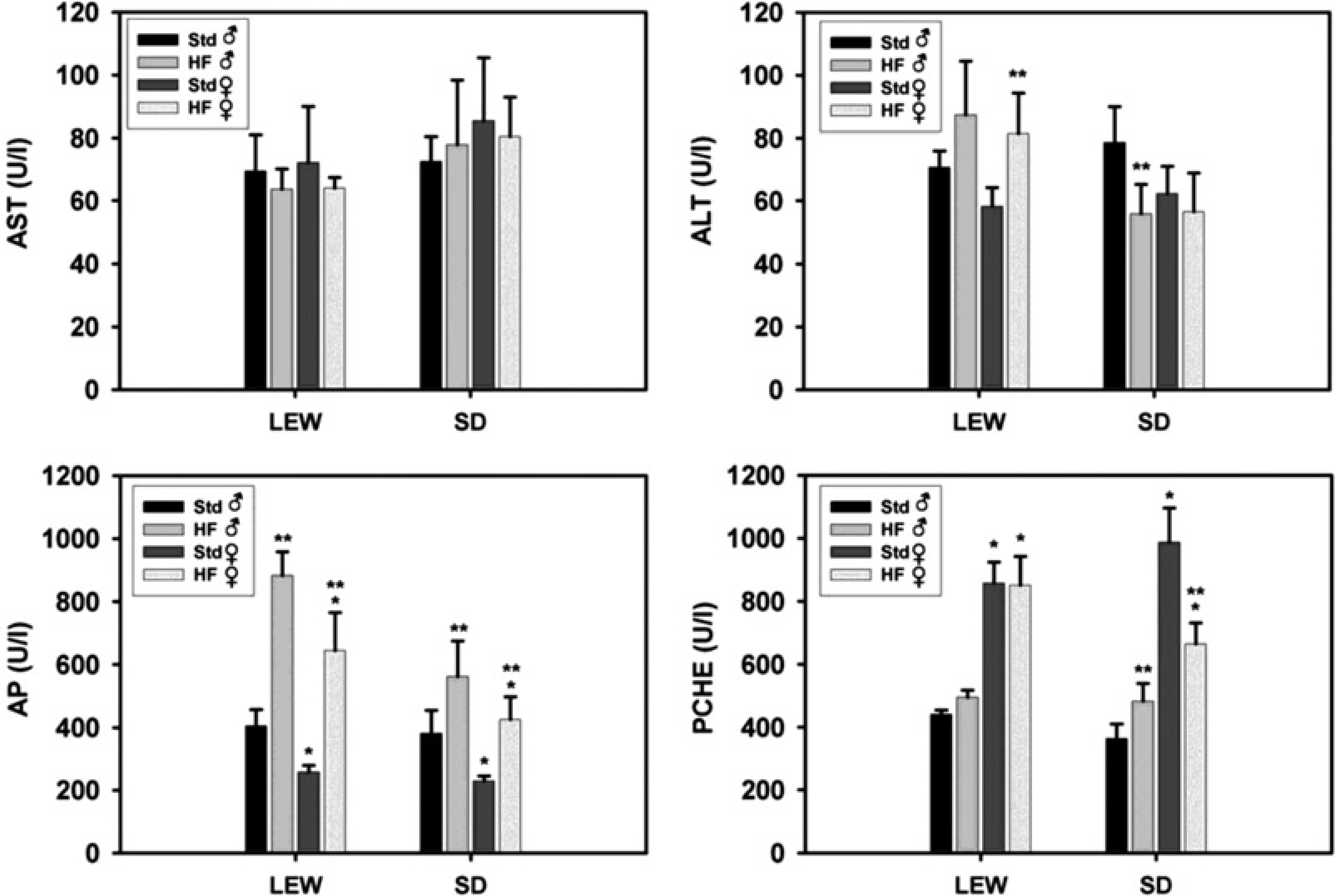
Serum markers of lipid metabolism
To establish whether gender and strain-specific differences were reflected by abnormalities of lipids and adipocytokines we measured cholesterol, triglycerides, LDL, HDL, leptin and adiponectin. In male Lewis rats, a HF diet led to elevated triglyceride, LDL and leptin levels as well as decreased HDL levels compared with controls fed with standard nutrition (P < 0.05, Figure 5). This effect was only partially observed in female Lewis rats, where the HF group showed higher leptin and lower HDL levels than their control group, also (P < 0.05). Triglycerides were not affected and significant decreases were recorded for cholesterol, LDL and adiponectin. For Lewis rats consuming the HF diet, male rats had significantly higher cholesterol, triglyceride, LDL, leptin and lower HDL levels than females (P < 0.05). In Sprague-Dawley rats no significant differences regarding gender or diet in serum levels of cholesterol, triglycerides, HDL or adiponectin were noted. Triglycerides tended to be higher in the HF group, but this was not significant. HF diet resulted in increased LDL and leptin levels in male, but not female, Sprague-Dawley rats (P < 0.05). Moreover, leptin levels in female Sprague-Dawley rats on a HF diet were significantly lower than in males.
Serum markers of lipid metabolism. Serum levels of cholesterol, triglycerides, low-density lipoprotein (LDL), high-density lipoprotein (HDL), leptin and adiponectin (AND) in Lewis (LEW) and Sprague-Dawley (SD) rats receiving either standard (Std) or high-fat (HF) diet for three weeks. *P < 0.05 versus ♂ fed with corresponding diet; **P < 0.05 versus standard nutrition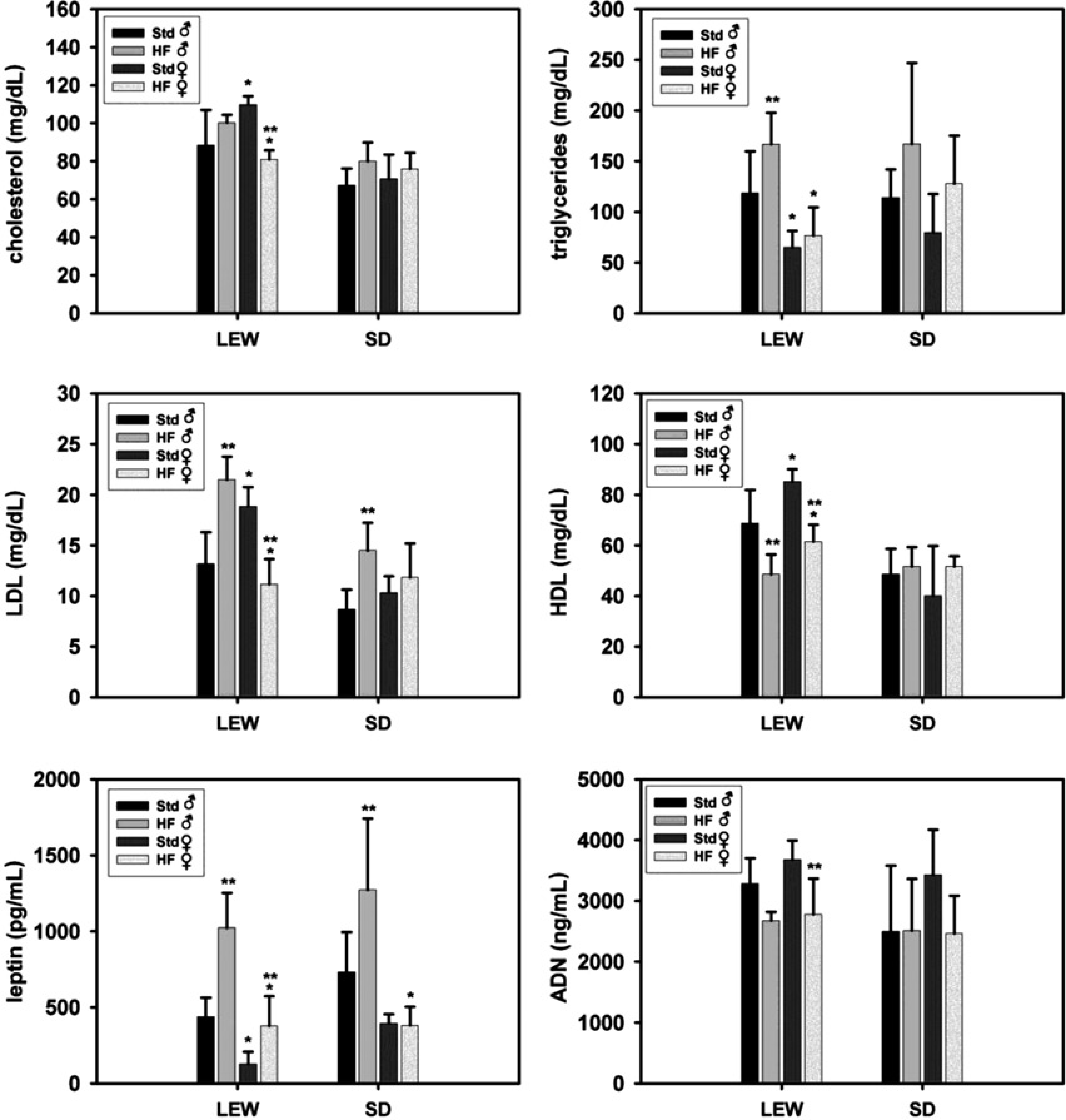
Gender and strain-specific differences in gene expression
To demonstrate HF diet induced changes in the expression of various genes important for angiogenesis, cell-to-cell signalling (cytokines and growth factors), extracellular matrix deposition, steatosis, fibrosis, fat metabolism and cell cycle quantitative RT-PCR analysis was performed (Figure 6). Changes in gene expression of three-fold or greater with P values <0.05 were considered significant. We examined liver gene expression profiles of Lewis and Sprague-Dawley rats on a HF diet compared with rats on standard nutrition. In male Lewis rats HF diet induced significant downregulation of three relevant genes including two proangiogenic genes, FLT1 and FLK1, also known as vascular endothelial growth factor (VEGF) receptor 1 and 2, and platelet-derived growth factor B (PDGFB), an important growth factor for liver regeneration (P < 0.05, Figure 6a). Hepatocyte growth factor (HGF) and cyclin D3 (CycD3) showed a trend toward downregulation without reaching statistical significance. By contrast, gene expression profile of female Lewis rats was different. HF diet induced significant upregulation of angiopoietin 1 (Ang1), a ligand for TIE2 (endothelial-specific receptor tyrosine kinase 2) promoting vessel maturation and stability (P > 0.05). Furthermore, CycD3 and p21, both involved in cell cycle regulation, were upregulated, although this was not significant. No other significant changes were noted in expression of selected genes.
Changes in expression of selected gene groups. Cumulative changes in gene expression of Lewis (a) and Sprague-Dawley (b) rats fed with a high-fat diet for three weeks compared with corresponding rats fed with standard nutrition. Changes in gene expression of three-fold or greater (dotted line) with P values <0.05 were considered significant. **P < 0.05 versus standard nutrition; na = not available (due to widespread of values in the data set)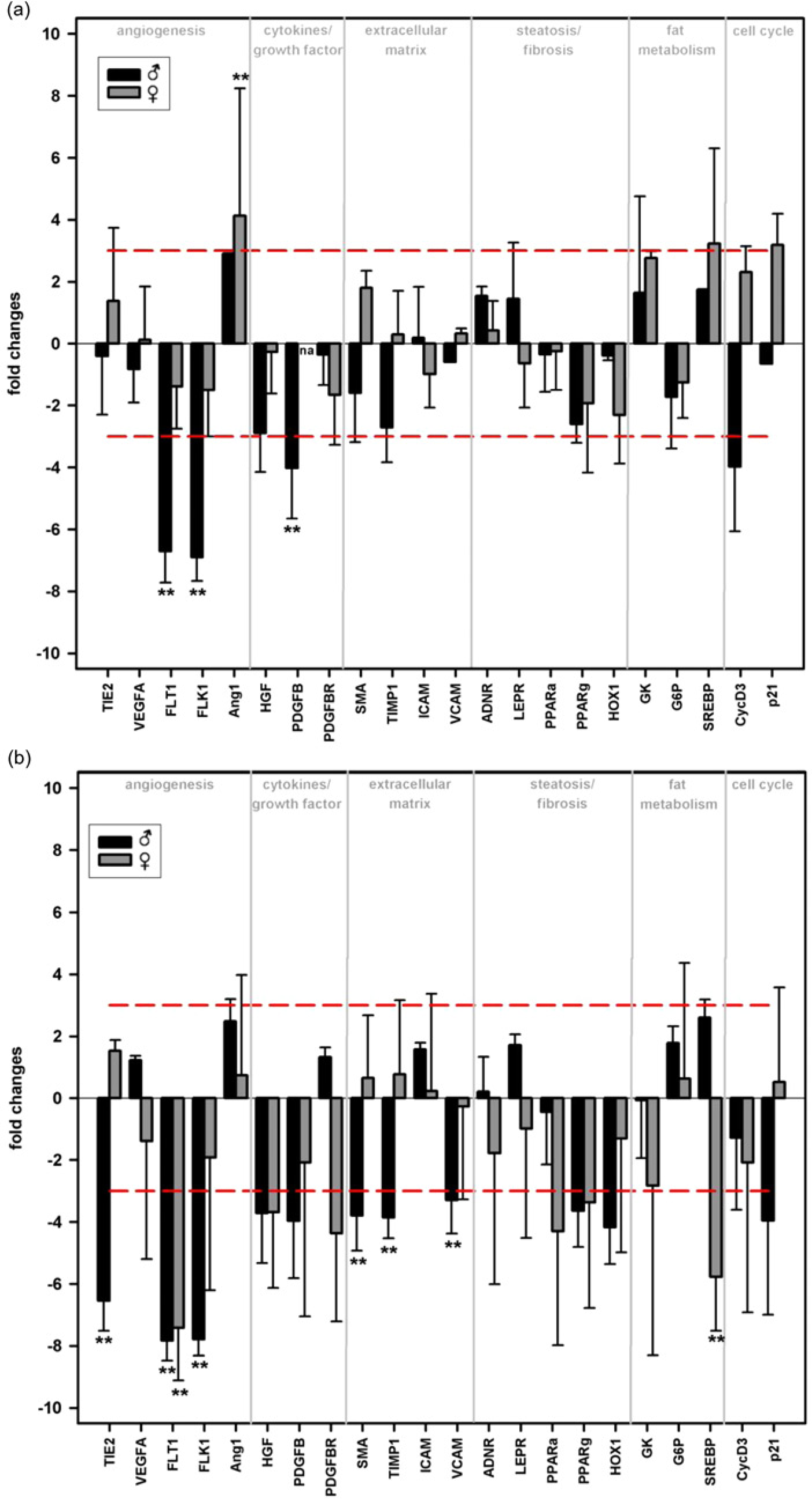
Male Sprague-Dawley rats on a HF diet showed significant downregulation of six relevant genes, including three proangiogenic genes, TIE2, FLT1 and FLK1, and three genes involved in extracellular matrix deposition, namely smooth muscle alpha-actin (SMA), tissue inhibitor of metalloproteinase 1 (TIMP1) and vascular cell adhesion molecule 1 (VCAM1, P < 0.05, Figure 6b). However, in female Sprague-Dawley rats HF diet induced downregulation of only two genes, FLT1 and SREBP-1c (sterol regulatory element-binding protein-1c), was observed (P < 0.05). SREBP-1c is associated with fatty acid synthesis and the development of hepatic steatosis. 16 In male as well as female Sprague-Dawley rats peroxisome proliferator activated receptor alpha and gamma (PPARΑ, PPARΓ), whose inhibition may contribute to the developement of fatty liver disease, were downregulated more than three-fold, but this was not signficant. 17
Discussion
In the present study, strain and gender in rats play a pivotal role in the development of liver steatosis, using the nutritional model developed by Lieber et al. 12 Since childhood obesity along with paediatric NAFLD is becoming a growing problem, pubertal rats correlating to human childhood and puberty were used for experiments. 18 The model of HF diet induced steatohepatitis was chosen because it may probably best resemble the clinical situation while reproducing the key features of human NASH in rats. 12 Clinical relevance of genetic (e.g. leptin-receptor deficiency) and other experimental models such as methionine-choline-deficient (MCD) diet is limited.19,20 The administered diet relates to our contemporary diet with HF content. Results must be interpreted with care, because a human diet with 71% of energy derived from fat would be considered extreme.
Morphologically, hepatic steatosis can be classified as macro- or microvesicular, depending on the size of the lipid vacuoles. 21 Different hypothesis for developing micro- or macrovesicular steatosis have been discussed so far. The microvesicular type may be more likely related to mitochondrial dysfunction, the macrovesicular type may be originated from disturbed protein synthesis. 22 When fed with a HF diet for three weeks, Lewis rats revealed a solely microvesicular steatosis whereas Sprague-Dawley rats presented with a pure macrovesicular steatosis accompanied by pronounced fibrosis. The impact of the type of steatosis on liver injury remains controversial and there is not sufficient evidence to define the significance of fat droplet size. Experiments in mice fed with MCD suggest that microvesicular steatosis is a primary form of hepatic steatosis. 23 The increased surface area of multiple microvesicular droplets may be associated with increased lipid efflux due to the amount of lipid that is accessible to enzymes. 24 Small lipid vacuoles may fuse to become a large droplet with progression of disease. 17 In our experiments microvesicular steatosis induced by a HF diet was associated with less signs of fibrosis, lobar inflammation and hepatocyte damage than the macrovesicular type supporting this hypothesis. Our previous studies indicated that hepatic stellate cells (HSC), known to be involved in the fibrogenic response to chronic liver injury, were more activated in Sprague-Dawley rats on a HF diet than in Lewis rats. 14 Interestingly, PCR analysis revealed that in male Sprague-Dawley rats developing the greatest degree of macrovesicular steatosis and fibrosis on a HF diet, SMA, TIMP1 and VCAM1 were downregulated. We do not have a definitive explanation for these results, transcriptional but also post-transcriptional regulation might play a role. Clinically, microvesicular steatosis due to genetic or toxin-induced abnormalities in mitochondrial and peroxisomal β-oxidation of fatty acids tends to be rapidly progressive and more severe. 17 A recently published cross-sectional study demonstrated that microvesicular steatosis correlated with more advanced histology of NAFLD. 25 These conflicting findings could be attributed to differences in fatty liver induction by inherited/acquired defect in β-oxidation of fatty acids or HF diets. However, others showed a trend to a correlation between microvesicular droplets and less fibrosis in patients with NAFLD. 26 Microcirculatory disturbances may contribute to liver injury and oxidative stress. The mere presence of lipid droplets in hepatocytes can impair hepatic microcirculation by sinusoidal compression, which in turn is related to the size of fat vesicles. 14 Animal experiments demonstrated that macrovesicular steatosis has a decreased tolerance to ischaemic injury correlating with impairment in hepatic blood flow after reperfusion. 22 For patients with macrosteatosis the risk of postoperative complications following liver resection or liver transplantation is increased compared with those with microsteatosis.27–29 However, Oleszczuk et al. raised concerns about the regenerative capacity of microvesicular steatosis. 30 In the present study, in male Lewis as well as male Sprague-Dawley rats fed with a HF diet, a two- to three-fold higher proliferation rate of hepatocytes was detected compared with animals on standard nutrition. Since leptin may promote progression of hepatocytes into the cell cycle via control of cyclin D1 expression elevated leptin-levels in male rats fed with a HF diet may account for increased proliferation rates. 31 Interestingly, Sydor et al. 32 reported recently that a Western diet induced steatosis may even enhance liver cell proliferation after partial hepatectomy via increased HGF and leptin signalling as well as Erk1/2 phosphorylation. On the other hand, higher proliferation rates may represent a need for more liver regeneration due to more severe injury. We did not find differences in apoptosis rates between micro- and macrosteatotic rat livers being in concordance to the study by Oleszczuk et al. 30 In fact, apoptosis rates and hepatocyte damage of animals on a HF diet were comparable to those on standard nutrition. According to the second hit theory an additional source of oxidative stress or a longer feeding period might have shown different results.
It was quite remarkable, that the female gender of both strains was associated with decreased susceptibility to liver injury. Steatosis and fibrosis were less pronounced and proliferation rates were higher even on standard nutrition. Experimental evidence implies that oestrogen can protect from adipose tissue oxidative stress and inflammation, development of liver steatosis and insulin resistance.9,33 Furthermore, the portal/fatty acid flux theory can partially explain the higher risk of males developing NAFLD through increased accumulation of visceral fat. 5 In the present study, gender-specific differences in lipid metabolism were most prominent in Lewis rats on a HF diet. Female Lewis rats showed lower cholesterol and triglyceride levels and a more favourable LDL/HDL ratio than males. It has been described, that the severity of liver injury is strongly associated with a more atherogenic profile in NAFLD. 34 Furthermore, the lower leptin levels of female rats on a HF diet may be related to histological changes also, since leptin has a proinflammatory role and is considered to be an essential mediator of liver fibrosis. 35 It was noteworthy, that realtime PCR analysis of liver tissue samples from female Sprague-Dawley rats showed downregulation of SREBP-1c, one of three known transcription factors regulating liver de novo fatty acid synthesis that may result in steatosis. 17 The decreased hepatic expression of this lipogenic gene could be attributed to an oestrogen-mediated effect, as described before. 36
HF diet in male rats of both strains resulted in decreased expression of several genes involved in angiogenesis such as VEGF receptor 1 and 2 (FLT1, FLK1) whereas female rats showed a more favourable gene expression profile. These changes in gene expression were in agreement with higher proliferation rates of females as shown by Ki-67 immunostains. VEGF-mediated signalling can protect hepatocytes and improve the impaired regeneration of steatotic livers.37–39 Therefore, those gender-specific differences in the expression of proangiogenic genes should be relevant and may offer therapeutic options for preservation of organ function.
In summary, strain and gender should be considered when exploring the induction of steatosis in models on a HF diet. Among the rats studied, male Sprague-Dawley rats developed the greatest degree of steatosis on a HF diet and may represent a useful animal strain for studying advanced stages of NAFLD. Female gender was associated with an alleviated disease phenotype and less changes in gene expression, e.g. of VEGF receptor 1 and 2. Therefore, strain and gender in our model of NAFLD is of high relevance and should be taken into account once pathogenesis or therapeutic targeting is evaluated.
Footnotes
Acknowledgement
The authors wish to express their thanks to K Cebulla for technical assistance in the conduct of this study.
