Abstract
Open chest surgery in rodents requires assisted breathing and the most common approach for ventilation is via an endotracheal tube. Even with well-trained operators the endotracheal intubation is technically challenging and may lead to prolonged procedures and endotracheal intubation complications. Nose cone ventilation is a simpler procedure compared to endotracheal intubation and has the potential to improve animal welfare by reducing procedure time and endotracheal intubation associated complications. Rats are obligate nose breathers, and therefore replacing intubation with air supply from a nose cone would be an advantage and a more natural way of breathing. Here, we compared the values for several blood gases, blood pressure and heart rate from rats that were nose cone ventilated with rats that underwent endotracheal intubation at 12 timepoints equally distributed across three surgical stages: baseline, open chest and closed chest. Throughout the monitoring period the hemodynamic and blood gas values for both methods of ventilation were within published, normal ranges for the rat and were biologically equivalent (equivalence test p value ≤ 0.05). Our data showed that nose cone ventilation-maintained blood gases and hemodynamic homeostasis equivalent to endotracheal intubation. Nose cone ventilation can be recommended as an alternative to endotracheal intubation in rat experiments where investigators require airway control.
Introduction
Open chest surgery is commonly used in animal models for cardiovascular research, such as myocardial infarction (MI), transverse aortic constriction (TAC) and intramyocardial injections. 1 During open chest surgery it is important that the animal is well ventilated and properly anesthetized. The use of mechanical ventilation is critical to prevent lung collapse, optimize experimental outcomes, improve survival and is consistent with good anesthesia practice.
Tracheal intubation is the primary technique used for securing airways. This technique requires highly skilled operators to avoid endotracheal intubation complications such as: laryngeal damage and laryngeal oedema, oral pharyngeal and/or tracheal laceration due to multiple intubation attempts; intubation of the esophagus leading to abdominal distention; endobronchial intubation leading to hypoxia; and airway perforation that may result in subcutaneous emphysema and pneumothorax. 2
The use of a mask for mechanical respiration in rats was first presented by Shuer et al. in 1978 and has been followed by several mask variations.3–6 A simple Bain circuit can be used which delivers a strong flow of cool, dry anesthetic gas via a loose-fitting mask. The Bain system lacks a closed exhalation circuit and therefore a large volume of oxygen and anesthetic gas must be delivered to maintain anesthesia. This method significantly increases the risk of hypothermia and dehydration and provides little recourse in the event of airway collapse. 7 These mask ventilation techniques share a fundamental problem of high dead space which can only be mitigated by increased airflow. In addition to increasing the cost of surgery, high gas flow also increases the risk of human exposure to waste gas.
The nose cone ventilation technique developed by Rindfield and McBrian 8 has overcome the high dead space problem without increasing gas flow. Their work shows that it is feasible to use assisted ventilation without intubation and that nose cone ventilation maintains appropriate anesthesia throughout the surgical procedure without fatal incidents. It is important to establish whether there are any differences between nose cone ventilation and endotracheal intubation that could affect surgical outcomes in animal models of cardiovascular disease. To our knowledge, no comparative studies have been published before. To compare these two ventilation techniques, we measured blood gases and hemodynamic parameters over three surgical stages (baseline, open chest and closed chest) in rats ventilated either via endotracheal intubation or nose cone.
Materials and methods
Animals and housing
All animal work was performed in accordance with the National Institute of Health (NIH) guidelines for use of experimental animals and the study protocol was approved by the Animal Ethics Committee at Gothenburg University (Gothenburg Ethical Review Board number 1047-2017). Six animals were housed together in each rat cage system from Scanbur (40 × 60 × 84.5 cm3) in a temperature controlled (20–23°C) facility with a 12 h light/dark cycle, relative humidity from 40 to 60% with 20 air changes/hour, in an American Association for the Accreditation of Laboratory Animal Care (AALAC)-approved facility. The animals had free access to pelleted food (R70, Lantmännen, Sweden) and water. Cages that contained hardwood bedding (J. Rettenmeier & Söhne GMBH, Germany), shredded paper (Papyrus AB, Sweden), a plastic house and gnawing sticks (Tapvei, Estonia) were changed weekly. A total of 12 male, naïve Lewis rats (Crl:LE, Charles River, Germany) weighing 300–400 g were used in this study. The animals were acclimatized for a minimum of 5 days before the start of the study.
Animal experiment
The experiments were conducted using excess male animals therefore a formal power calculation was not conducted. Six animals per group is recommended for pilot studies. Blinding of treatment groups was not possible due to the nature of the study. Attention was paid to reduce time of day effects by ensuring an equal distribution of both types of ventilation in morning and afternoon experiments (blocking). Three animals from each group were included in experiments in the morning and three animals from each group were included in experiments in the afternoon. The animals were anesthetized in an anesthetic induction chamber with isoflurane (5%). An air (1000 ml/min) and oxygen (100 ml/min) mixture was used as the carrier gas. Anaesthesia was maintained by isoflurane throughout the experiment. After anesthesia induction the animals were connected to the ventilator (Ventilator, UGO BASILE S.R.L, model 7025, SN 0723U09) breathing through a nose cone or an endotracheal tube.
Ventilation via intubation
After induction of a surgical plane of anaesthesia, each rat, in the intubation group, was placed on the intubation board (Figure 1). With the aid of a laryngoscope (HEINE F-22.804 with blade Classic + Miller00 FO, the blade was adjusted in our workshop to better fit the rat) the endotracheal tube (c. 5 cm polyethylene tubing, internal diam. 1.40 mm, outside diam. 1.90 mm, Intramedic Item nr: 427441) was inserted into the trachea (Figure 1) and was connected to the adapter on the rat ventilator tubing. Accurate endotracheal intubation was confirmed by observing chest movements coordinated with the ventilator and absence of abdominal distention. The water pillar on the ventilator was monitored for the appearance of water bubbles at the start of ventilation. Absence of water bubbles, or presence of abdominal distension indicated that a second attempt at intubation should be performed. The rat was placed in the anaesthesia chamber again to be re-anesthetised before making a second attempt at intubation.
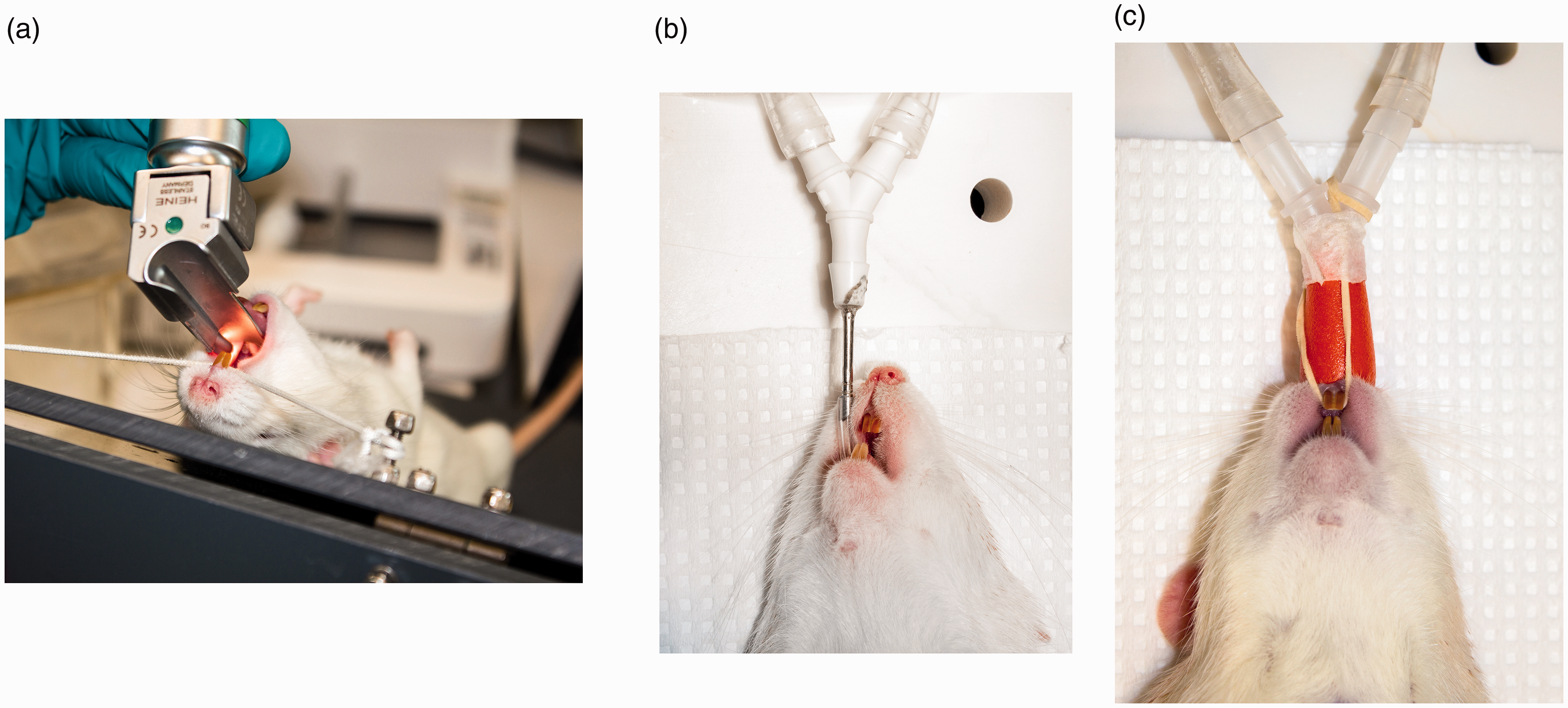
(a) Intubation procedure in a rat on a tilted table with the aid of a laryngoscope. (b) A rat ventilated by intubation and (c) ventilated by nose cone.
Ventilation via nose cone
After induction of a surgical plane of anesthesia, each rat in the nose cone group, was placed directly on the surgical table and the nose cone (No-Intubation Nose Cone, VetEquip Item nr: 921465) was placed over the rat’s nose and secured with a rubber band behind its upper incisors (Figure 1). Accurate positioning of the cone was confirmed by observing chest movements coordinated with the ventilator and absence of abdominal distention. The water pillar was monitored for the appearance of water bubbles at the start of ventilation. Absence of water bubbles, or presence of abdominal distension indicated that the nose cone should be repositioned.
All rats were ventilated at c. 60 breaths/min, c. 2.5 ml tidal volume for intubated rats and c. 3.5 ml tidal volume for rats with nose cone. 9 The isoflurane vaporizer (Ohmeda Isotec 5, BOC Health Care) was set to 2.5–3% for maintenance of surgical anesthesia with carrier gas of air 1000 ml/min and oxygen c. 100 ml/min through the whole experiment. The animals were placed in a supine position on a ventilated table. Core body temperature was maintained at 37.0 ± 0.1°C by a heated table and a heating lamp controlled by a rectal thermometer.
Surgery –carotid artery
The animals were shaved and surgically scrubbed with Descutan 4% (Fresenius Kabi, Vnr 498402) and Chlorhexidine 1 mg/ml (Fresenius Kabi, Vnr 537993) at the sites of surgery. Eye ointment (Viscotears 2 mg/g, Thea Laboratories Vnr:569173) was applied. A skin incision was made on the right side of the neck of the rat and blunt dissection was performed to locate the carotid artery. A catheter (PE 50, Intramedic® polyethylene tubing, Becton Dickinson, Sparks, MD, USA) was inserted in the right carotid artery for monitoring of mean arterial pressure (MAP) and heart rate (HR). Rate pressure product (RPP) was calculated as MAP × HR. The catheter was also used for sampling blood for blood gas analyses (ABL825, Triolab Sweden). The arterial catheter was flushed with 10 μl/min saline (9 mg/ml, Fresenius Kabi AG, Bad Homburg, Germany) throughout the experiment to maintain patency and to compensate for blood samples collected.
An electrocardiogram (ECG) was recorded from three lead skin electrodes. Signals from MAP, HR, ECG and body temperature were recorded using computer and software (PharmLab V6.0, AstraZeneca R&D Mölndal, Sweden) every 10th second and averaged over 1 min to represent one measurement. After the surgical prep with the arterial catheter the animals were stabilized to 37 ± 0.1°C body temperature. Baseline blood samples were collected (Clinitubes ref.942-878, Radiometer) at 0, 10, 20 and 30 min. After baseline recordings a thoracotomy was performed.
Surgery – thoracotomy
A skin incision from the rat’s left axillary space to midline was made with care to avoid the left mammary vein. The pectoral muscle was gently lifted and the ventral edge of the underlaying latissimus dorsi muscle was separated from the intercostal muscle towards the dorsal aspect. Immediately cranial to the fifth rib an incision c. 1 cm was made through the fourth/fifth intercostal space to access the thoracic cavity. A retractor was inserted into the intercostal incision to keep the chest open. Care was taken not to trap lung tissue behind the retractor. The blood sampling was repeated at 0, 10, 20 and 30 min after thoracotomy. To close the chest two single sutures (3-0 Vicryl, Ethicon ref. V393G) were placed around the fourth and fifth rib and gently tightened until the ribs were in normal approximation. To re-establish negative pressure in the thoracic cavity the ventilator was used to hold the rat’s breath for 3–4 s before tightening the last suture. The pectoralis minor and latissimus dorsi muscle was repositioned to their normal position. The skin incision was closed using an interrupted pattern (3-0 Vicryl, Ethicon ref. V393G). Once again, the blood sampling was repeated at 0, 10, 20 and 30 min after closure of the chest. Adequacy of anesthesia and surgical depth was assessed by continuously monitoring mean arterial pressure, heart rate and ECG. Continuous monitoring of blood gases including pH, bicarbonate (HCO3–), carbon dioxide (pCO2), oxygen (pO2) and lactate was also performed. Hemodynamic measurements were sampled every 10 s and each measurement represents 1 min. The clinical instrument ABL825 (Triolab, Sweden) was used for analysis of blood gases. After the last blood sample and still under surgical anesthesia the rat was euthanized by opening the chest and cutting the heart.
Statistical analysis
All blood gas and hemodynamic parameters were inverse normal rank transformed so that they were normally distributed and were expressed in units of SD (RNOmni package R statistics). 10 A linear mixed effects model (LMM) in combination with the emmeans package was used to calculate means and standard deviations for each parameter by type of ventilation and surgical stages (R package Imer).11,12 The LMM took into account that measures and time points were not independent of each other as the same cohort of rats were measured multiple times. 13 We also accounted for the effect of body weight on the measured parameters by including it as a covariate in the LMM.
The potential similarities and differences of blood gas and hemodynamic measures at each surgical stage was assessed by an equivalence test between ventilation groups. 14 An equivalence test is an extension of a t-test, which sets an upper and lower bound on the size of the difference which returns a significant result if the difference falls within these bounds. In this study any group differences within three SDs of 0 was considered too small to be considered a potentially meaningful ‘biological’ difference between the two ventilation types. Therefore, a p ≤ 0.05, after adjusting for multiple testing, indicated that the parameters were ‘biologically’ equivalent for the different ventilation types. 15
Results
Ventilation procedure
Successful endotracheal intubation was achieved in all six rats, but a second attempt was needed in two animals (Figure 1). In contrast, nose cone ventilation was easily performed on the six rats with no repositioning of the nose cone required (Figure 1). Of the six endotracheal intubated rats, one rat was excluded due to missing values from equipment malfunction.
Hemodynamic parameters and blood gases
Hemodynamic parameters and blood gases were measured at baseline, during open chest and after closing of the chest. We found that hemodynamic features for MAP, HR and RPP were very similar between ventilation techniques and within physiological ranges 15 for both techniques of intubation (Figure 2 and Supplementary Table 1).
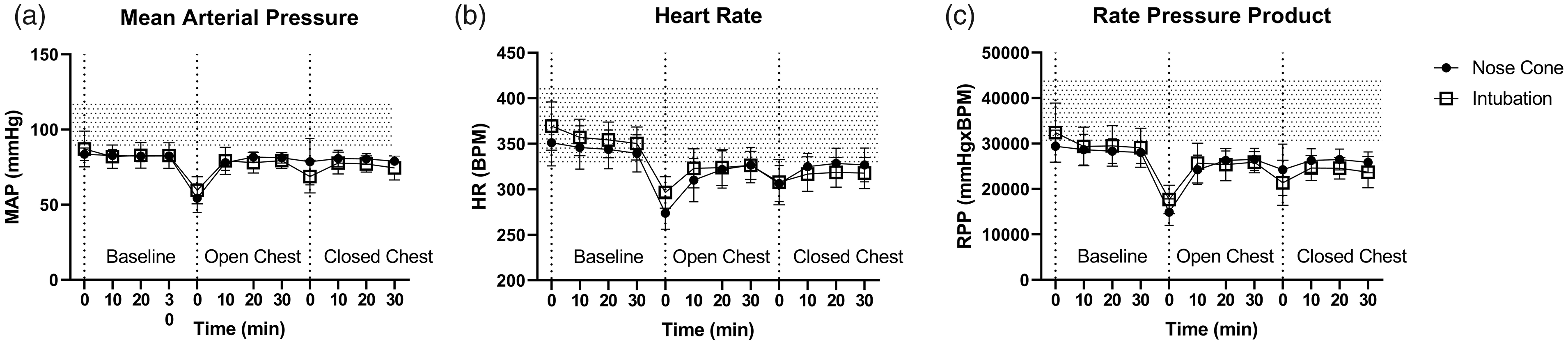
Hemodynamic parameters in anesthetized rats during baseline, open chest and after closing of the chest: (a) Mean arterial pressure (MAP); (b) heart rate (HR); and (c) rate pressure product (RPP). Filled circles, ventilated via nose cone; open squares, ventilated via endotracheal intubation. Shaded area indicates normal range in literature and during historical closed chest rat experiments in our lab. Data shown are mean values ± SD; n = 6 nose cone, n = 5 endotracheal.
Similarly, no deviations from physiological ranges16–18 were observed for arterial blood pH, pO2, pCO2, lactate and HCO3
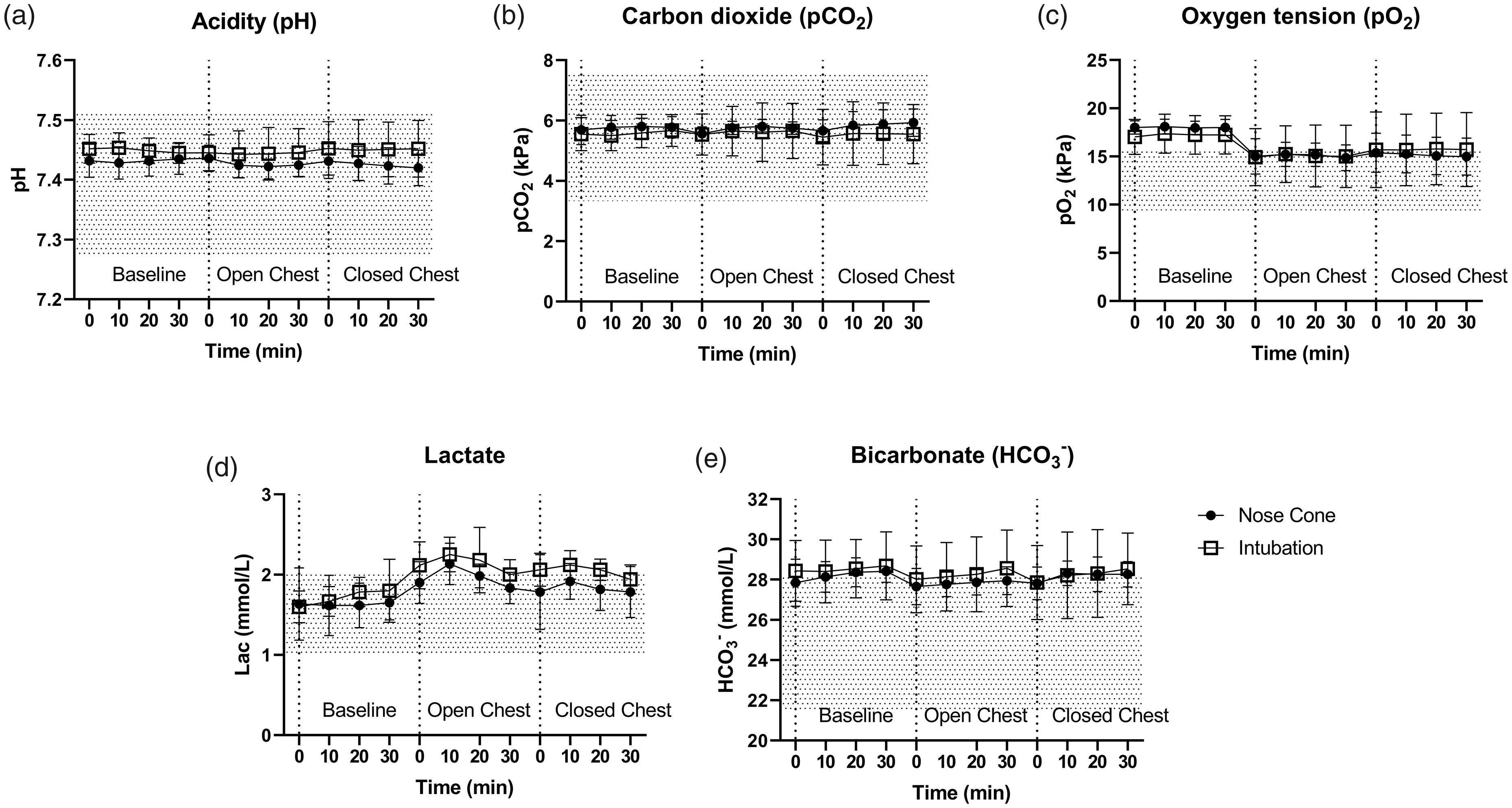
Blood gases in anaesthetized rats during baseline, during open chest and after closing of the chest: (a) pH; (b) pCO2; (c) pO2; (d) lactate; and (e) HCO3–. Filled circles, ventilated via nose cone; open squares, ventilated via intubation. Dotted grey area show published range. 11 Data shown are mean values ± SD; n = 6 nose cone, n = 5 endotracheal.
Discussion
Our data showed equivalent blood gases and hemodynamic parameters during different stages of surgery when comparing nose cone and endotracheal tube ventilation in male rats. While different stages of surgery (baseline, open chest and closed chest), may change hemodynamics and blood gases, it is clear from our experiments that the impact of the ventilation type is equivalent. Indicating that you could use nose cone or endotracheal tube ventilation interchangeably. We chose to use isoflurane as the anesthetic agent as it has favorable effects on hemodynamics and the ability to dose titrate the depth of anesthesia and shows good recovery after dosing. 19
We have shown that using nose cone ventilation during anesthesia, it is possible to maintain: adequate blood circulation presented by heart rate, mean arterial pressure and rate pressure product; normal acid–base status as indicated by that pH, pCO2, HCO3– and lactate that were within normal respective reference intervals; and normal oxygen uptake in the lungs assessed by pO2 blood oxygenation.15–18
The learning curve for this technique is shallow and no special instruments except the nose cone are necessary. Even with experience in endotracheal intubation technique, the nose cone ventilation is considerably easier to perform. Therefore, animal surgeons or technicians will require less time and fewer animals to train for this technique. While we did not observe any stomach dilation in our study (indicating that the nose cone was misplaced or the esophagus had been intubated), the consequences are different for each ventilation type. The animals in the intubation group would need to be reintubated increasing the risk of complications and thus animal suffering. Rinfield and MacBrian reported a few occasions where the nose cone was mispositioned resulting in a dilated stomach minutes after nose cone placement. 8 A quick adjustment of the nose cone resolved the issue on every occasion. Hence, the nose cone procedure is well aligned with principles relating to international laboratory animal welfare principles – 3Rs (replace, reduce, refine), since it is a refinement that will reduce animal suffering and that the number of laboratory animal needed in science can be reduced. 20
In conclusion, these experiments show that nose cone ventilation is similar to intubation in all physiological parameters that were measured. Quality of oxygenation, acid–base status, blood pressure and heart rate were maintained, while simplifying surgery preparation and increasing animal welfare by avoiding endotracheal intubation complications. In our experience nose cone ventilation is easier and quicker to perform also for experienced animal surgeons/technicians and can be recommended in rat experiments where investigators require respiration control as an alternative to endotracheal intubation.
Supplemental Material
sj-pdf-1-lan-10.1177_00236772211031039 - Supplemental material for Ventilation via nose cone results in similar hemodynamic parameters and blood gas levels as endotracheal intubation during open chest surgery in rats
Supplemental material, sj-pdf-1-lan-10.1177_00236772211031039 for Ventilation via nose cone results in similar hemodynamic parameters and blood gas levels as endotracheal intubation during open chest surgery in rats by Nina Krutrök, Susanne Pehrsson, Natalie Van Zuydam, Karin Jennbacken and Johannes Wikström in Laboratory Animals
Footnotes
Acknowledgements
The authors acknowledge Ann-Christine Nordkam for the photos.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
