Abstract
Hyperpolarized noble gas (HNG) magnetic resonance imaging (MRI) has been shown to be useful for studying rodent models of lung disease. Image quality can be substantially degraded by signal loss from molecular oxygen entering the airway, requiring invasive surgery to ensure a good seal between the endotracheal (ET) tube and trachea. A modified Foley catheter having an inflatable cuff near the tip provides a novel approach for ensuring image quality for HNG MRI, thereby enabling longitudinal studies and reducing animal numbers. A Foley catheter was modified for rodent intubation and to minimize dead space. Three pairs of age-matched male Sprague Dawley rats 400 (30) g were used. Two pairs were intubated using the Foley and the third with an intravenous catheter. Leak rates were measured from pressure versus time curves within each animal. The pairs were euthanized immediately or six days postrecovery to assess the effects of the procedure on animal health, as reflected by histological examination. The Foley catheter resulted in minimal leak rates (−0.20 (0.03) versus −0.16 (0.05) cmH2O/s), and were shown to be well below upper-limit leak rates of −0.5 and −0.7 cmH2O/s. Tracheal samples from rats in a separate Foley group (not mechanically ventilated) showed superficial damage six days postextubation (grade = 0). 3He imaging performed using the Foley showed good image quality. Though some technical issues remain to be solved, a modified Foley catheter used as an ET tube offers the potential to enable longitudinal studies in rodents and reduce animal numbers.
Chronic respiratory diseases including chronic obstructive pulmonary disease, emphysema, asthma and lung fibrosis are the fourth largest contributors to mortality worldwide and are expected to account for one-third of all deaths by 2030. 1 Magnetic resonance imaging (MRI) following administration of hyperpolarized noble gases (HNGs), helium-3 and xenon-129 hereafter referred to as 3He and 129Xe, has been shown to be sensitive to changes in the lung caused by disease in humans. 2 Furthermore, HNGs can provide quantitative anatomical and functional information within the lungs of ventilated rodents such as ventilation and ventilated volume, perfusion, apparent diffusion coefficients (ADCs) and diffusing capacity, 3 including rodent models of emphysema, 4 asthma 5 as well as lung injury due to bleomycin, 6 radiation-induced lung injury 7 and gastric aspiration. 8
Typically, special MRI-compatible ventilators are used in conjunction with intubation to ensure consistent and timely HNG breath-holds in rodents; however, this limits the method to endpoint studies requiring larger numbers of animals due to the need for surgery to ensure an airtight seal between the endotracheal (ET) tube and the trachea. An airtight seal prevents the interaction of molecular oxygen with the HNGs which would otherwise destroy the observable MRI signal due to T1 relaxation. Furthermore, an airtight seal allows precise control of lung pressure and volume which are important for quantitative measurements of ADC, absolute ventilated lung volume (|VLV|) 9 and diffusing capacity of xenon (D Xe). 10,11 Typically, this is achieved using a combination of the well-known Seldinger technique 12 followed by a surgical tracheotomy, involving exposure of the trachea and the securing of ligatures 3 around the trachea containing the ET tube.
Unfortunately, these invasive surgical methods require euthanization of the animal at the completion of the imaging study. A less invasive technique that could replace such surgical interventions would allow the trachea of the animal to heal and thereby permit the study of the time course of lung diseases in an individual rodent. This would allow the individual rodent to serve as its own control, eliminating variability arising between different animals within a cohort. More importantly, fewer animals would be required. Ultimately, the ability to measure changes in a given animal will prove helpful for understanding human diseases.
Though the proposed Foley catheter method is the method of choice for this work, it is not uniquely less invasive. Other methods described in the literature refer to the use of soft sealant adhesive putty 13 or rolled tape attached to the ET tubes 14 to provide a seal without the use of surgery. The putty is placed on the external end of the ET tube and moulded to the shape of the animal's mouth and held in place by the adhesive. The rolled tape method uses tape rolled around the outer diameter of the ET tube to increase its diameter enough to create a seal with the trachea. To our knowledge, there is no literature describing careful measurements of the breath-hold quality using these methods or their associated leak rates.
In this work, a Foley catheter with an inflatable cuff near the tip is modified into an ET tube and used for ventilation and breath-holds using a mechanical ventilator in rats. Tracheal pressure is measured and used to assess the quality of breath-hold with the Foley catheter compared with a conventional tracheotomy technique and an upper limit leak rate tolerance derived from previous 3He work using the standard method. 9 Rats were studied both immediately following extubation of the Foley catheter and six days after in order to measure the healing of the trachea over time. Microscopy was performed on tracheal sections obtained from rats intubated using the Foley catheter to assess safety and repeatability. It is also demonstrated that the Foley catheter technique can be used to perform breath-hold MRI using hyperpolarized 3He in a representative rat lung.
Materials and methods
Animals
All procedures followed animal care protocols approved by the University of Western Ontario Animal Care and Veterinary Services (ACVS) and were conducted in accordance within the guidelines of the Canadian Council on Animal Care (CCAC). In total, 20 rats were used to accomplish the sub-objectives of this work. Six Sprague Dawley rats (400 (30) g) were used in this study, with three in the standard group and three in the Foley catheter group. A separate group of three animals was used to assess the survivability of Foley catheter intubation six days postextubation. Seven additional rats were used, with four of these rats added to the Foley catheter group (n = 7 total 6 days postextubation) and the remaining three comprised a new group for which the Foley catheter was introduced and removed without the use of mechanical ventilation. A single rat was used to demonstrate the feasibility of an imaging experiment with 3He. Three rats were also used to assess normal trachea. The rats were anaesthetized by infusion of a mixture of propofol (Astra Zeneca, Mississauga, Canada) and ketamine (Bioniche, Belleville, Canada) (10:1) via the tail vein at rates between 2.0 and 2.4 mL/h using a drug-delivery pump. Three rats were prepared using the Seldinger technique followed by tracheotomy (hereafter referred to as the standard method)
9,15
and the remaining rats were intubated with the Foley catheter as described above. The standard method of intubation consisted of insertion of a 14–16-gauge intravenous (Beckton Dickinson, Mississauga, Canada) catheter of length 5–6 cm into the trachea followed by tracheal exposure and securing of three ligatures around the trachea using 0 silk suture material (Ethicon, Somerville, NJ, USA). The ET tube was then attached directly to a small animal custom ventilator (GEHC, Malmo, Sweden) compatible with HNG MRI.
15
Insertion of the Foley catheter required no surgery. The Foley cuff was inflated using the assembly shown in Figure 1. The insertion length was measured to be equivalent to the length of a standard IV catheter and inserted orally, up to the manibrium of the rat. The syringe pump was set to deliver 0.4 mL of water to the cuff in less than one second. The pressure gauge read 4 psig (281.3 cmH2O) in the water-filled line connected to the inflation airway. It was observed that the cuff volume was not able to hold pressure when inflated using air for periods longer than 20 min. This was likely due to repeated use and porosity of the cuff which also introduces cuff asymmetry requiring replacement of the catheter. To overcome this limitation, expansion was achieved using water which did not leak from the cuff, maintaining the cuff volume indefinitely. During ventilation manoeuvres, if free breathing was apparent (e.g. gasping) then a dose of 1 mg/kg of pancuronium bromide (Sandoz, Quebec, Canada) was administered to allow the ventilator to completely control the animal's respiration. Three additional rats were intubated using the Foley catheter and recovered for histological assessment after six days of recovery to compare with the day 0 group. Three tracheas were also excised from similar rats (650 (50) g) to serve as controls for comparison between uncatheterized trachea, trachea intubated using the standard method and trachea intubated with the Foley catheter.
Experimental set-up including rat, Foley catheter, inflation apparatus and mechanical ventilator. After intubation, the cuff of the Foley was quickly expanded within less than one second by 0.4 mL of water delivered by a syringe pump connected by a series of tubing. A pressure gauge in-line prevented over-pressuring the trachea and the cuff by inflating to pressures no greater than 4 psig. The Foley was connected to a mechanical ventilator to facilitate ventilation of the animal while under anaesthesia and tracheal pressure (P
Tr) was monitored with an in-line pressure transducer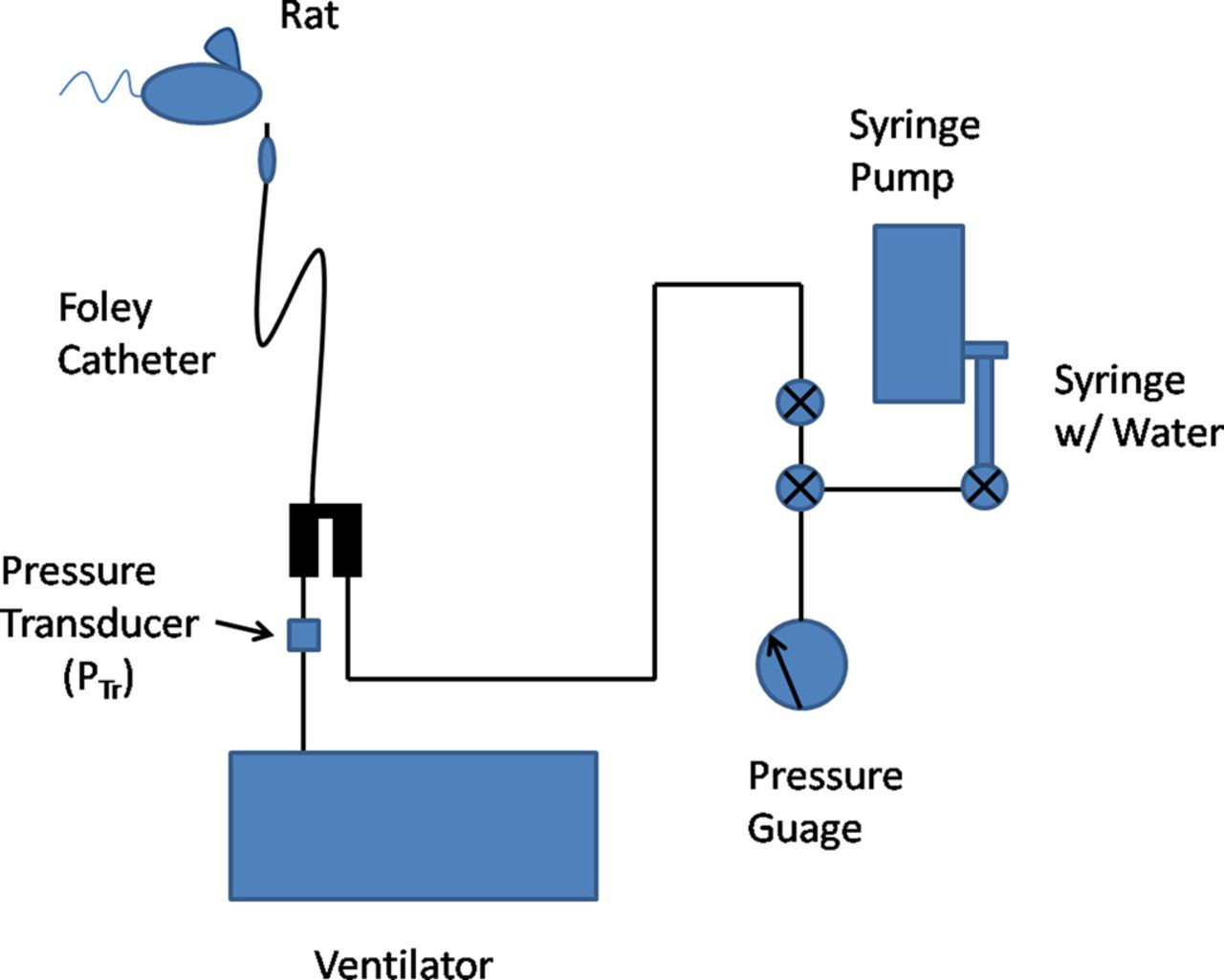
ET tube (Figure 2) shows the two-way Foley catheter used in this study. This standard 6 Fr paediatric Foley catheter (Rusch, Teleflex Medical, Kamunting, Malaysia) was modified to permit rodent intubations and minimize dead space. The two-way Foley catheter possessed two parallel airways to facilitate gas delivery through the tube, and to expand the cuff, respectively. Modifications included shortening of the ventilator attachment port as well as tapering and dulling the catheter tip to resemble a standard ET tube and reduce dead space. The Foley catheter had an inner diameter of the airway lumen of 1.5 mm and had a total length of 30 cm. The total inflatable volume of the cuff was 1.5 mL. To ensure controlled expansion of the cuff, the inflation port was fixed with a three-way stopcock (Smiths Medical, Markham, Canada) and sealed to prevent any leaks. This end was then connected to a system of Tygon tubing (Saint Gobain North America, Valley Forge, PA, USA) as shown in Figure 1, consisting of a pressure gauge (Winters, Toronto, Canada), 10 mL syringe (Beckton Dickinson) and infusion pump (Univentor, Zejtun, Malta). The lines were filled with water and connected to an infusion pump to deliver precise volumes of water to the cuff both quickly (i.e. less than 1 second) and reliably. This speed ensured animal survival during preparation due to the comparative size between the inner diameter of the trachea (2.6–2.7 mm) and largest outer diameter of the Foley catheter (2.3 mm) which would otherwise occlude the airway. Rapid inflation of the cuff mitigates the unwanted occlusion allowing for resumption of ventilation as quickly as possible and controlled by the ventilator.
(a) Photograph of the Foley catheter with inflatable cuff near the tip. Arrows indicate the region of the catheter where the cuff resides. (b) The deflated cuff at the end of the catheter. (c) The cuff inflated to a maximum volume of 1.5 mL for effect. A volume of 0.4 mL was used during the study. (d) The two-way end has been modified to allow coupling with a tubing system to control the inflation of the cuff
Ventilation
Ventilation was carried out in a manner described previously. 9,15 Animals were ventilated with pure oxygen at a rate of 20 breaths per minute with a tidal volume of approximately 8 mL/kg based on the mass of the rat. The inspiration/expiration ratio (I/E) was adjusted between 0.4 and 0.6 to facilitate proper tidal volume delivery as well as to allow time for passive exhalation due to the higher airway resistance of this long narrow Foley catheter. Oxygen was provided by the ventilator from a cylinder and regulated down to a range of between 12 and 16 cmH2O. To simulate HNG breath-holds, Tedlar bags (Jensen Inert, Coral Springs, FL, USA) filled with 4He placed inside a gas delivery reservoir were pressurized to 15 cmH2O to deliver gas at the same rates and tidal volumes as above for 10 s durations which were consistent with the timescales of typical lung MRI acquisitions. 9 3He was delivered in an identical fashion during the imaging experiment.
Quality of breath-hold
Breath-hold quality was assessed by measuring tracheal pressure (P Tr) versus time during a 10 s breath-hold using an in-line pressure transducer (Sensor Technics, Puchheim, Germany) (Figure 1), following five washout breaths of 4He for both standard and Foley methods. These washout breaths were necessary to remove residual oxygen from the lung which would otherwise be taken up by the blood and affect the P Tr measurements. P Tr values were recorded as a function of time at a rate of 10 samples per second for the entire breath-hold duration and repeated five times for each animal. The animal was ventilated with pure oxygen between breath-hold measurements for a duration of 5 min to maintain normal physiological conditions, as assessed by manually monitoring heart rate and checking for signs of cyanosis.
Recovery and histological review
Recovery at zero and six days postextubation was assessed by histological examination of animals undergoing Foley intubation only. Animals were intubated and ventilated using the Foley catheter in a manner similar to the breath-hold experiments above but for a duration of one hour while under anaesthesia to simulate the typical length of time that the catheter would be in place during an imaging experiment. After one hour, propofol anaesthesia was switched to saline and the animal was allowed to recover. Animals were ventilated with pure oxygen until signs of free breathing and alertness were apparent. At this point, the cuff of the Foley catheter was deflated and extubated from the animal. A pure oxygen nose cone was used for the remainder of the recovery. Animals were then euthanized or returned to cages for a period of six days. At the conclusion of days 0 and 6, animals were anesthetized and euthanized using direct injection of sodium pentobarbital (Bimeda-MTC, Cambridge, Canada) at a dose of 0.5 mL using the cardiac puncture technique. The trachea of each rat was carefully extracted and fixed in 10% buffered formalin for at least 24 h. Each trachea was then sectioned 16 times in order to have an adequate sampling along the length of the tracheas and then haematoxylin and eosin (H&E) stained and transferred to a veterinary pathologist for analysis. For comparison, three control tracheas and three tracheas intubated with the standard method were excised, fixed and graded following the analysis described below.
3He Imaging
To demonstrate image quality using the Foley catheter, 3He MRI in a single rat (442 g) was performed at 3.0 T (Discovery MR750, GE Healthcare, Waukesha, WI, USA) using a transmit–receive bird-cage coil (Morris Instruments, Ottawa, Canada) tuned to the 3He resonance (97.3 MHz) and a high-performance insert gradient coil (G = 17 G/cm, slew rate = 1500 mT/m/s) as described previously. 16 Single-slice two-dimensional (2D) images were obtained in the coronal plane using a fast 2D gradient-echo method (TE = 0.5 ms, TR = 2.4 ms, 128 × 128, field of view (FOV) = 5 cm2, bandwidth = 31.3 kHz). In order to minimize signal variations and image artefacts due to the radiofrequency (RF) depolarization, RF pulses were applied with a variable flip angle trajectory. Three breaths of 3He were administered by the mechanical ventilator prior to initiating a breath-hold. The P Tr versus time curves were used to determine when to acquire images after waiting 2.5 s postairway occlusion. 3He gas was polarized to levels in excess of 40% using a spin-exchange optical pumping system (Helispin®, GE Healthcare, Durham, NC, USA). 3
Analysis
To assess quality of breath-hold, slopes of the P Tr versus time curves during a breath-hold were determined using linear least squares fitting for time-points during the period occurring 2.5 s postairway occlusion where slight changes in pressure over 10 s were assumed to be caused only by leaks and not animal respiratory motion. Slopes corresponding to the breath-hold regions were calculated for each data-set and averaged for each rat as an indication of the leakage rate. Average P Tr and pressure drops during the breath-hold were also quantified. Although an acceptable leak rate is not stated in the literature, a total change of less than 10% over a breath-hold no greater than 4 s was considered acceptable, as this has been reported to minimize errors in volumetric imaging using 3He MRI and xenon-computed tomography (CT). 9 That work demonstrated that for a 2 s breath-hold at typical inflation pressures (10–14 cmH2O) for which 3D 3He imaging can be performed, leak rates between −0.5 and −0.7 cmH2O/s are tolerable, and therefore were used here as an upper limit threshold for the application of a non-inferiority test to validate measured leak rates between the standard method and Foley catheter.
The degree of tracheal healing was assessed quantitatively using a histological grading scale as determined by a veterinary pathologist. After determining the range of damage from a few representative data-sets, a scale of between 0 and 3 was chosen. The grading scale corresponded to the amount of denuded respiratory epithelium normally present on the mucosal surface area of the trachea. A grade of 0 corresponded to normal trachea mucosa with no pathological loss of epithelial cells. Grades 1–3 corresponded to mild, moderate and severe damage equating to losses of 25–50, 50–75 and 75–100% of denuded epithelium, respectively. The remaining sets of slides were randomized and analysed using this scale. A grade of 0 after six days postextubation was interpreted as full healing of the trachea.
The signal-to-noise ratio (SNR) was calculated from the acquired image by using the mean signal within the left lobe divided by the standard deviation of the noise. This SNR value was used to assess image quality using the Foley, by comparing with SNR values of images acquired previously using the standard method.
Results
Quality of breath-hold
A representative Foley breath-hold curve is shown in Figure 3. Results are mean plus or minus one standard deviation calculated from five repeated breath-holds within the same animal. The leak rates measured were −0.16 (0.05) and −0.20 (0.03) cmH2O/s for the standard method and the Foley, respectively. Using a non-inferiority test with a 95% confidence interval, the worst case leak rate given these data is −0.27 cmH2O/s. These leak rates are well below the upper limit leak rates of −0.5 and −0.7 cmH2O/s. Average tracheal pressures during a breath-hold were measured to be 10.8 (0.8) and 6.7 (0.4) cmH2O, respectively. Breath-hold end pressure drops were 12 (3)% and 22 (4)%, respectively, which are directly related to the leak rates during 10 s. A full listing of these results can be seen in Tables 1 and 2.
Tracheal pressure (P
Tr) versus time curve depicting the entire prescribed breath-hold in a representative rat with the Foley in place. Data are mean, plus or minus one standard deviation. Linear least squares fitting (dotted line) was performed for the interval between 4 and 14 s in order to estimate both the leak rate of gas from the trachea as well as the average tracheal pressure for the duration of the breath-hold. The dashed line represents the mean held-breath pressure during the imaging period of 10 s Leak rates from three rats prepared using the standard method which were shown to be equivalent compared with the Foley and to the upper-limit leak rates Average pressures, pressure percentage drops and leak rates are tabulated and their mean and standard deviations calculated for each Leak rates from three rats prepared using the Foley which were shown to be equivalent when compared with the standard method and upper-limit leak rates Average pressures, pressure percentage drops and leak rates are tabulated and their mean and standard deviations calculated for each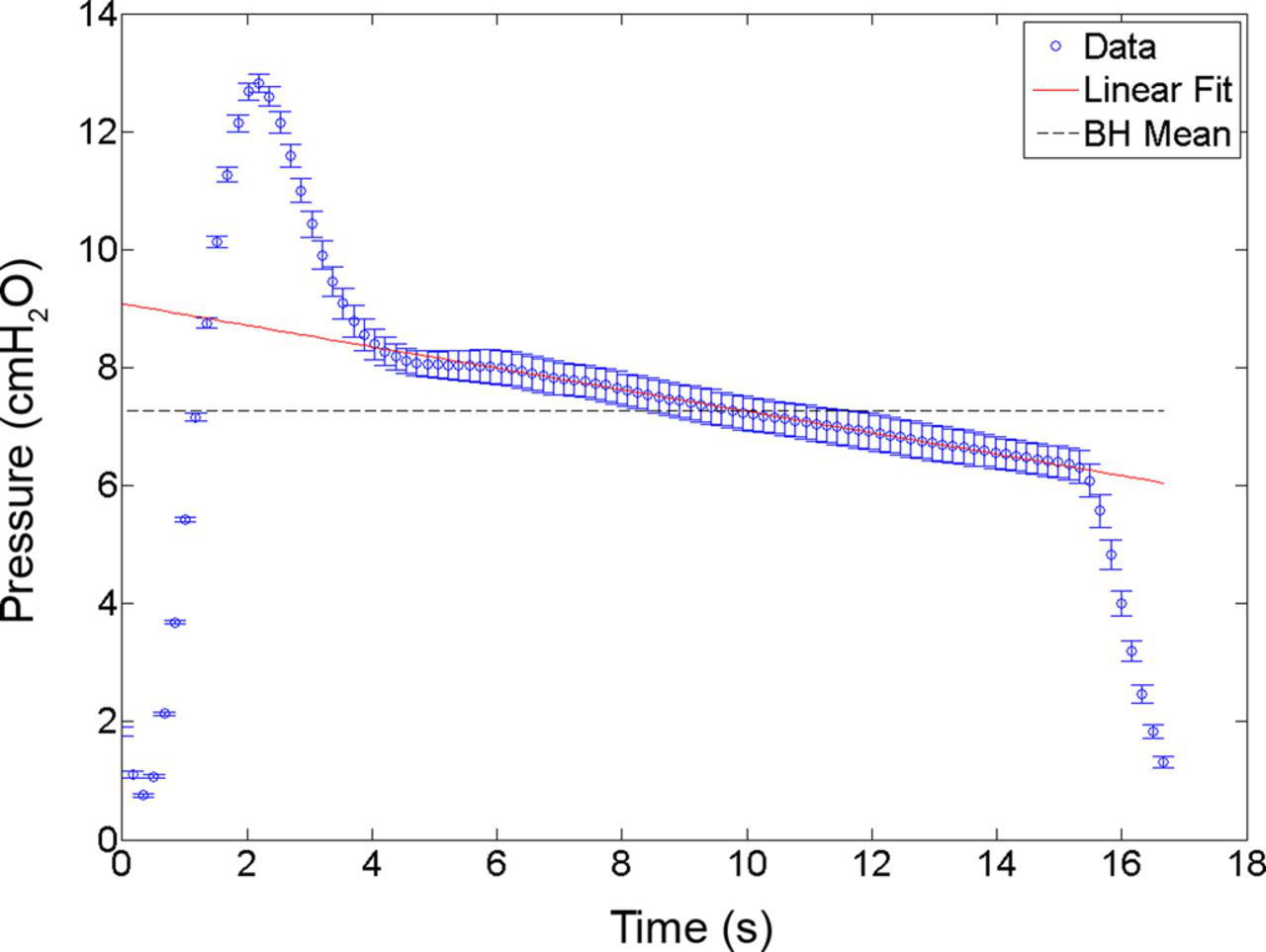
Histology and survivability
Figure 4 shows micrographs at ×5 and ×40 magnification of the trachea from three representative rats depicting normal tracheal sections from a control animal (Figures 4a and d), sections from a rat prepared using the Foley at zero days postextubation (Figures 4b and e) and at six days postextubation (Figures 4c and f). Average scores across each of the 16 tracheal slices for each experimental group were 2.3 (0.4), 1.4 (0.2) and 0 for the Foley, standard method and control groups, respectively. A high degree of damage (i.e. values of 2 and 3) caused either by pressure from the cuff or more likely, stripping of the respiratory epithelium during extubation as determined by the grading scale can be seen in Figures 4b and e (arrows). The average grade score for rats surviving six days of recovery was 0. The grading scale confirms that after about a week of recovery time (day 6) the animals were fully healed (i.e. mean grade of 0 for respiratory epithelium) from the trauma and were ready for imaging once again (Figures 4c and f).
Photographs of sections of tracheal tissue. Panel a depicts a healthy undamaged trachea, corresponding to a histological grading scale of 0. Panel b shows a tracheal section showing significant denuding of respiratory epithelium (arrow). This corresponds to a histological grading scale of 3 and represents damage postextubation from one representative section of animal trachea from the Foley group. Panel c shows a tracheal section which was intubated and extubated using the Foley and allowed six days recovery time. The grading scale for this image corresponds to a score of 0. Panels d, e and f are versions of a, b and c magnified to a power of ×40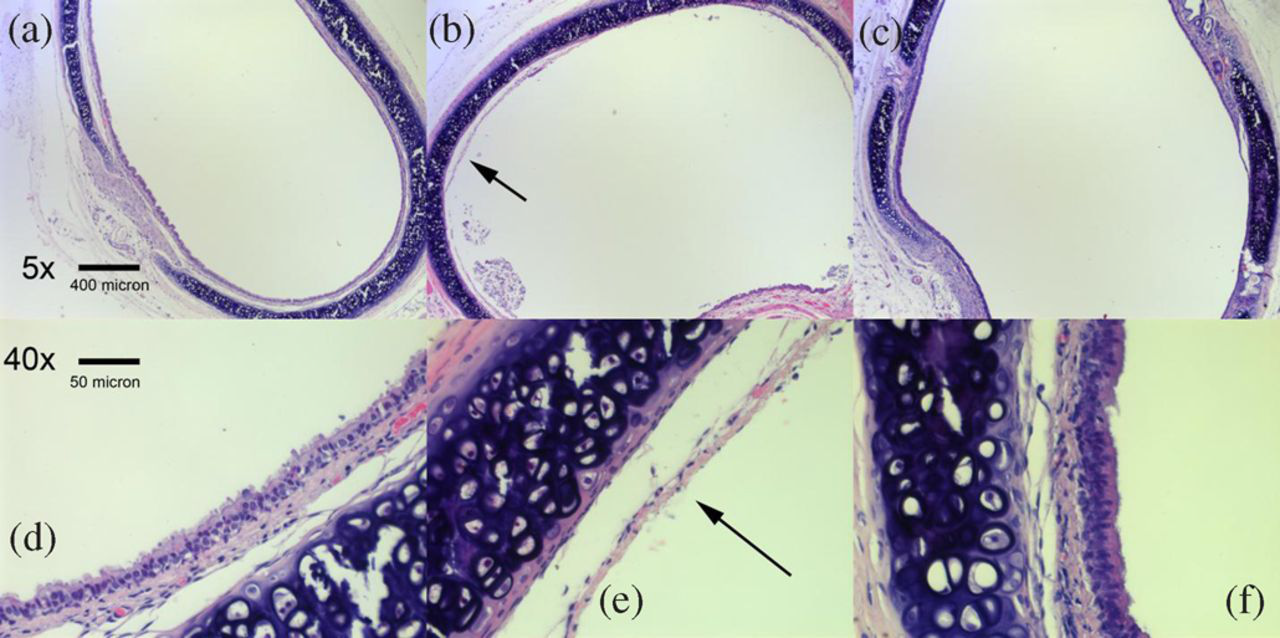
Imaging
Figure 5 shows a 3He image that was obtained with the Foley catheter in place. The image SNR was measured to be 260 in the lower left lobe of the lung. The SNR measured is comparable with other 2D images acquired using the standard method after a single breath of 3He with an SNR of 185.
15
A 2D coronal MRI of a representative rat lung acquired using a breath-hold of hyperpolarized 3He with the Foley catheter in place and connected to a mechanical ventilator. Acquisition parameters include, 128 × 128 matrix size, 5 cm2 FOV and three washout breaths of hyperpolarized 3He prior to imaging. The signal-to-noise ratio of this image was 260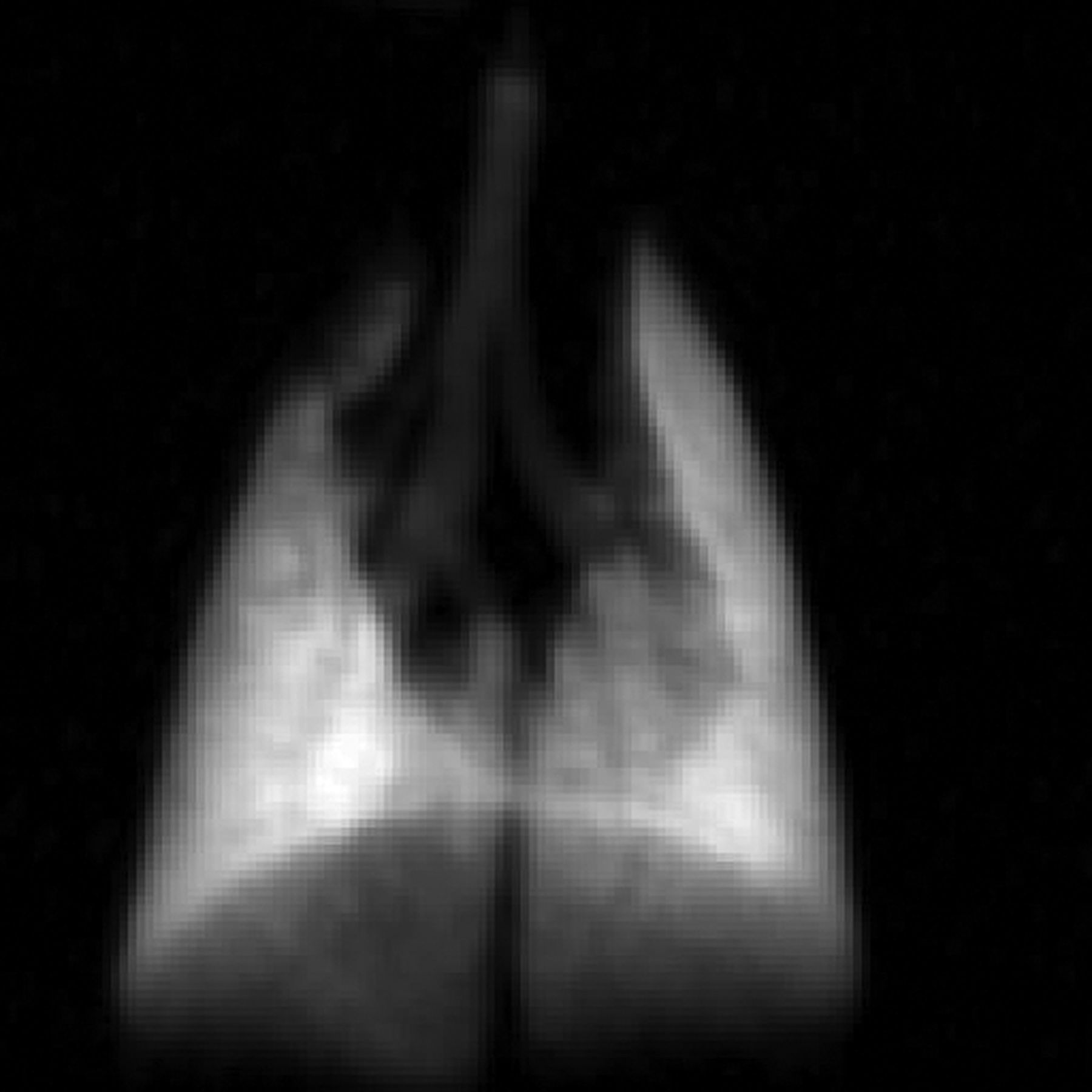
Discussion
In this study it has been shown that the Foley catheter is capable of achieving a breath-hold quality equivalent to that obtained using the standard method as is evidenced by the slopes of the P Tr versus time curves and percent drops during the stable regions of the breath-hold curves (Figure 3). Figure 3 and Tables 1 and 2 confirm that the Foley is as effective at minimizing leakages during a breath-hold as the standard method with the Foley catheter performing no worse than −0.27 cmH2O/s, which is well below the adopted leak rate thresholds. For an imaging experiment involving 3D volumetric imaging using MRI, a pressure drop of 10% or less is preferred. 9 From the average leak rate in a Foley catheter set-up, this is equivalent to a 10% drop over a 4 s breath-hold. 3D volumetric imaging has been done for acquisition times as low as 2 s 9 giving confidence that the leak rates observed using the Foley are acceptable for imaging and quantitative image analysis.
The effects of over-inflation of ET tube cuffs is well known. The pressure exerted on the trachea by the cuff acts to reduce in size or completely occlude capillaries in the walls of the trachea leading to ischaemia of the tracheal mucosa or cartilage. 17 For this reason a cuff pressure of no greater than 30 cmH2O is considered acceptable. 18 The cuff pressure used in this study was measured to be 281.3 cmH2O. This is abnormally high when viewed conventionally; however, most of the work is spent on expanding the low compliance cuff at the end of the Foley catheter. As a result, the full force caused by this high pressure was not entirely transmitted to the trachea. Though the actual force transmitted to the trachea was not measured in this study, it was expected to be substantially less given the histological results after six days of healing.
Histological data (Figure 4) confirm that damage to the respiratory epithelium caused by the Foley catheter is healed within six days as revealed by a histological grade of 0. Though the degree of tracheal damage was greater using the Foley (2.3 (0.4) versus 1.4 (0.2) points), the surgical damage caused by the standard method is non-recoverable. Furthermore, the Foley catheter allowed successful recovery of a cohort of animals to the state of free breathing after extubation, where they were monitored and subsequent histology was performed six days later in order to characterize the physiological response to inflation of the cuff in the trachea and its extubation. Histology showed a marked decrease in the degree of tracheal damage present in the last day in comparison with the first day of recovery (grade 2.3 (0.4) for first day compared with grade 0 for day 6). This indicates that the animals are able to recover and could likely tolerate this procedure on a weekly basis for imaging purposes to follow the time course of a disease model by HNG MRI.
The failure rate of animal recovery postextubation of the Foley and mechanical ventilation was high (6 of 7 animals). In order to decouple the causes of death potentially caused by the Foley and/or the ventilator, additional Foley experiments were performed independently of the ventilator. Three animals were intubated using the Foley for a short time and recovered since without a ventilator it was not possible to maintain a one-hour exposure to the inflated cuff. After six days, the animals were anaesthetized and their tracheas extracted for histological review using the grading scale as described above. The degree of inflammation was shown to be similar to the rat that survived the six-day recovery post-ventilation, all having grades of 0. This implies that the combination of the Foley catheter used (i.e. high airway resistance) with mechanical ventilation is not capable of life support for long periods of time perhaps due to difficulty of carbon dioxide clearance. Though death did not occur during ventilation, this may imply that there is a maximum time limit for how long an animal can sustain ventilation using these techniques without improvement to the apparatus. An animal monitoring system to monitor heart rate and oxygen saturation should be used during ventilation in future experiments to better ensure animal health. Blood gas samples collected before intubation and ventilation, during and postextubation will also aid in monitoring animal health by confirming the levels of arterial paO2 and paCO2. 3He image quality with the Foley catheter was acceptable and comparable with image quality previously demonstrated using the standard method.
With these results in mind, it is conceivable that the Foley catheter will be an adequate replacement for the standard method and allow for longitudinal studies. Furthermore, it serves as a simple replacement for researchers wishing to simplify their techniques and avoid time-consuming and/or risky surgery for endpoint or longitudinal studies. This technique need not be limited to HNG MRI imaging studies. Micro-CT 19 and positron emission tomography (PET) imaging with dedicated small animal PET imaging systems 20 as well as physiology-based investigations 21 could benefit from the use of this minimally invasive intubation technique by allowing for longitudinal imaging or by simplification or elimination of surgical methods in endpoint studies, where bronchial alveolar lavage or compliance curves are also required. Any animal experiments requiring accurate and precise control of pressure and volume in a longitudinal study such as quantitative measurements of ADCs, absolute ventilated lung volume and the diffusing capacity of xenon could benefit from this technique.
Although the feasibility of the Foley catheter is demonstrated here, it is not without its shortcomings. The catheter is designed for single sterile use; however, it is used multiple times in this work after being cleaned but not autoclaved. The cuff was initially piloted by inflating it using room air; however, it was observed that for times greater than 20 min, deflation occurred leading to sudden losses of breath-hold quality. Inflation using water maintained the volume and pressure of the cuff indefinitely. Also observed was that after repeated use, the cuff volume became asymmetric and inflation did not occur uniformly. The catheter was replaced with a new one when this occurred. Due to the long length and smaller airway diameter of the Foley catheter its flow resistance was high (theoretically 9× higher) relative to the IV catheter, though not measured. The ventilation parameters necessary to support life required adjustment with breathing rates of 20 breaths/min and I/Es less than 1.0 to allow adequate time for passive exhalation. It may be difficult to achieve large enough tidal volumes in a short time period suitable for more exotic imaging schemes with HNG MRI, where signal is proportional to the amount of gas supplied. To our knowledge, this model of Foley catheter was the most suitable at the time of the study. A significant shortening of the catheter from 30 to 6 cm may allow for more adequate gas delivery though this requires further study. This would have the effect of reducing airway resistance in the dead space by a factor of 5 due to airway resistance's linearity with length. Though further improvements could be achieved by increasing the airway's diameter, it may reduce the application of this method due to the fact that the outer diameter is already very close to the inner diameter of the trachea for the size of animals used (2.6–2.7 mm). Furthermore, the limiting dimension of the catheter occurs 11 mm from the tip where the balloon resides. The cuff segment must be able to clear the vocal folds (during intubation and extubation) without tearing them in either direction. This may be overcome by restricting its use to mature animals only. Although animals as small as 250 g have been successfully intubated with this method it is suggested to use rats 400 g or larger to avoid damaging the vocal folds.
A modified Foley catheter can serve as a suitable replacement for invasive surgery for breath-hold ventilation and 3He MRI of the rat lung. The gas leakage rates observed using the Foley technique (−0.20 (0.03) cmH2O/s) are equivalent to the leakage rates of using the standard technique in rats (−0.16 (0.05) cmH2O/s) and are well below the maximum allowable leak rates of −0.5 and −0.7 cmH2O demonstrated for fast 3D 3He imaging of rat lungs. Rats are fully recoverable after application of the Foley catheter when mechanical ventilation is not involved and largely healed after one week as confirmed by histology. Though some denuding of the respiratory epithelium results from the procedure, it is resolved by day 6 (i.e. grade of 0). This approach may allow longitudinal studies using HNG breath-hold MRI and other modalities in individual rodents in future, but most importantly, it may help reduce the total required number of animals for a given study.
Footnotes
ACKNOWLEDGEMENTS
The authors would like to thank Heather Cadieux and Lynda McCaig for invaluable assistance and advice related to animal care. The authors thank Darcy O'Neil and Kelly Galloway for processing H&E-stained slides and finally Alexei Ouriadov for assistance in performing 3He imaging.
The authors would like to acknowledge contributions from the following sources of funding: Natural Sciences and Engineering Research Council (NSERC), Canadian Institute for Health Research (CIHR) and Cancer Imaging Network of Ontario (CINO).
