Abstract
Pigs are commonly used in diabetes research due to their many physiological similarities to humans. They are especially useful in imaging procedures because of their large size. However, to achieve imaging procedures the pig must lie completely still, and thus needs to be anaesthetized. Most anaesthetic drugs used in laboratory animals affect carbohydrate metabolism by the inhibition of insulin release. The aim of this pilot study was primarily to develop an anaesthetic protocol for pigs that did not have an effect on blood glucose levels throughout the 3 h of anaesthesia; and secondly, to evaluate the most promising protocol in combination with an oral glucose tolerance test (OGTT). Two anaesthetic protocols were used in four growing pigs. Intravenous propofol infusion caused hyperglycaemia in three out of four pigs within 5–10 min after induction and was therefore excluded. Intravenous infusion with tiletamine, zolazepam and butorphanol (TZB) for 3 h did not affect blood glucose levels. The pigs underwent OGTT twice, once without anaesthesia and once with TZB induction after glucose intake. Anaesthesia during OGTT resulted in a lower area under the curve (AUC) of glucose (P < 0.05), higher AUC of glucagon (P < 0.05) and an insulin response less than 10% of that during OGTT without anaesthesia. In conclusion, long-term infusion anaesthesia with TZB does not affect glucose homeostasis in pigs. However, the protocol is not effective when combined with OGTT, as glucose, insulin and glucagon levels are affected.
The pig is a commonly used large animal model in diabetes research because of its many physiological similarities to humans. 1 The same metabolic alterations occur in pigs with streptozotocin-induced diabetes as well as in human patients with diabetes, and the metabolic changes can be reversed by insulin treatment.2,3 In addition, the oral glucose tolerance test (OGTT), a valuable tool in diabetes research, can be carried out in a similar manner in pigs as in humans. 4 Pigs are especially useful in different imaging procedures such as positron emission tomography (PET) scans, since a larger-sized animal is often required to achieve high resolution images. However, to achieve imaging procedures the pig needs to lie completely still and must therefore be anaesthetized; and these anaesthetic agents might affect various physiological mechanisms and thereby impact research results. Most anaesthetics and sedative agents that are used in laboratory animal medicine are known to affect carbohydrate metabolism, including medetomidine,5–7 xylazine,5,8 volatile agents such as isoflurane and sevoflurane,9–13 and barbiturates.14–16 Individuals anaesthetized with these agents typically show decreased insulin secretion and hyperglycaemia.5–16 An anaesthetic protocol without any effects on carbohydrate metabolism in pigs has not been described.
N-methyl-D-aspartate (NMDA) receptor antagonists such as ketamine and tiletamine have little or no effect on carbohydrate metabolism in rats, 17 humans, 14 non-human primates 18 or cats. 8 A 1:1 mixture of tiletamine and the pyrazolodiazepinone derivative, zolazepam, does not interfere with intravenous glucose tolerance test (IVGTT) in non-human primates 7 or Japanese black bears. 6 The anaesthetic agent, propofol, has been reported to be a good choice in keeping blood glucose at a constant level during surgery in human patients with diabetes. 19 However, anaesthetic protocols in human patients are often complicated by peri- and intraoperative use of several different drugs. Publications describing the effects of propofol alone on carbohydrate metabolism are rare, although propofol has been reported to not affect insulin secretion in rat β-cells in vitro or during IVGTT in rabbits. 13
The aim of the present pilot study was to evaluate the effects of two anaesthetic protocols: (1) propofol and (2) a combination of tiletamine, zolazepam and butorphanol (TZB), on blood glucose concentrations during 3 h of anaesthesia in pigs. A second aim was to test which protocol had the least effect on glucose homeostasis in combination with OGTT.
Animals, materials and methods
All procedures were approved by the Ethics Committee for Animal Experimentation, Uppsala, Sweden. The study ran for a total of six weeks.
Animals
In total, five specific pathogen-free pigs (Yorkshire × Swedish Landrace × Hampshire) from the same litter, two males and three females (prepubertal), were obtained from the university herd (Swedish Livestock Research Centre, Lövsta, Sweden). One pig was difficult to ventilate during experiment 1; thus, it did not participate in experiment 2. Instead, a reserve sibling was used in experiment 2, together with the other three pigs. The animals were six weeks old upon arrival; after a two-week acclimatisation period their mean weight was 33.6 ± 1.6 kg. They were housed at the Department of Clinical Sciences in individual pens measuring 3 m2, within sight and sound of one another. Straw and wood shavings were used as bedding. A 14:10 h light/dark schedule (lights on at 06:00 h) was used, and an infrared lamp (24 h) was provided in each pen. The room temperature was 16–18℃. The pigs were fed a commercial finisher diet without growth promoters (SOLO 330, Lantmännen, Sweden) twice daily (07:00 and 15:00), the amount of which was dependent on their body weight (BW) and according to the Swedish University of Agricultural Sciences regimen for growing pigs. 20 Water was provided ad libitum. During the two-week acclimatisation period the animals were handled frequently, trained to be bottle-fed with glucose dissolved in water and to step onto an electronic scale. The pigs were bottle-fed every day during the experimental period.
Surgical implantation of an indwelling venous catheter
After the acclimatisation period, the pigs underwent surgery and an indwelling silicon catheter (SIL-C70 with rounded tip; Instech Solomon, Plymouth Meeting, PA, USA) was inserted into the vena jugularis dextra under general anaesthesia as previously described. 3 The surgical procedure was completed within 45 min. The catheters were flushed twice daily with heparinized saline 100 IE/mL (Heparin LEO, 5000 IE/mL; Leo Pharma, Ballerup, Denmark) and facilitated frequent blood sampling without stress.
In connection with the surgical procedure, venous blood samples were obtained. Blood preserved in EDTA was analysed for total and differential white blood cell (WBC) counts and haemoglobin (Hb) by an electronic cell counter validated for porcine blood (Advia 2120; Siemens, Erlangen, Germany). Serum samples were analysed for creatinine (CREA), and enzyme activities of aspartate amino transferase (ASAT), alanine amino transferase (ALAT), γ-glutamyltransferase (GT) and glutamate dehydrogenase (GLDH), by an automated equipment (Architect C4000; Abbott Diagnostics, North Ryde, Australia).
Experiment 1
Two anaesthetic protocols were evaluated for their effect on blood glucose levels. Four pigs were anesthetized using both protocols, with five days’ washout between the different protocols. Anaesthesia was induced in the pig’s home pen without any pre-anaesthetic treatment. After induction the pigs were placed on a stretcher and intubated while still in the stable, thereafter they were moved to the surgical ward once the endotracheal tube was in place. On the way to the surgical ward (1–2 min) the pigs were manually ventilated with a bag valve, if needed. In the surgical ward the endotracheal tube was connected to a pressure-controlled ventilator (FLOW-i; Maquet Critical Care AB, Solna, Sweden), and the airways were ventilated to maintain an end-tidal carbon dioxide (CO2) of approximately 5 kPa. Ringer acetate (RAc) infusion (5–10 mL/kg/h) was provided as maintenance therapy. During anaesthesia, respiratory rate, respiratory minute volume, positive end expiratory pressure, peak inspiratory pressure, end-tidal CO2, inspired/end-tidal oxygen (O2), O2 saturation, heart rate, non-invasive blood pressure (cuff placed on upper forelimb), RAc infusion rate, rectal temperature (measured repeatedly with a digital thermometer), palpebral reflex, nystagmus, shivering and anaesthetic infusion rate were monitored and noted every 15 min. The above-listed physiological parameters were measured with Datex Ohmeda S/5 monitors (Datex-Ohmeda, Inc, Madison, WI, USA). The depth of anaesthesia was assessed for adequacy by checking for absence of spontaneous movements, absence of palpebral reflex and absence of response to manipulation of the extremities and the endotracheal tube. Blood glucose was measured before anaesthesia, and at 5, 10, 20, 30, 45, 60, 75, 90, 105, 120, 135, 150, 165 and 180 min after the induction of anaesthesia.
Protocol 1
Anaesthesia was induced with an IV infusion of propofol (Diprivan®, 20 mg/mL; AstraZeneca, Södertälje, Sweden) at a dose of 4–10 mg/kg over a period of one minute. For maintenance, propofol was given as a constant rate infusion (CRI) of 8–20 mg/kg/h, and was continuously adjusted to achieve an adequate depth of anaesthesia.
Protocol 2
Tiletamine and zolazepam (Zoletil®, 50 mg + 50 mg/mL; Virbac, Carros, France) was mixed with butorphanol (Butomidor® vet, 10 mg/mL; Salfarm Scandinavia, Helsingborg, Sweden) and sodium chloride (Natriumklorid Fresenius Kabi, 9 mg/mL; Fresenius Kabi, Bad Homburg, Germany) to produce a solution of 25 mg/mL of tiletamine/zolazepam and 1 mg/mL of butorphanol. Anaesthesia was induced intravenously at a dose of 5–6.25 mg/kg tiletamine/zolazepam and 0.2–0.25 mg/kg butorphanol over a one-minute period. For maintenance, doses of 5–6.25 mg/kg/h tiletamine/zolazepam and 0.2–0.25 mg/kg/h butorphanol were given as CRIs, and continuously adjusted to achieve an adequate depth of anaesthesia.
Experiment 2
Four pigs underwent OGTT without anaesthesia, and subsequently OGTT with anaesthesia (protocol 2), which was induced after glucose intake. Oral glucose tolerance was tested after an 18 h overnight fast. Water was withheld one hour before the experiment. The pigs were bottle-fed with an oral glucose load (orally soluble glucose, Glukos; APL, Stockholm, Sweden) of 1.75 g/kg BW, maximum 75 g, mixed with 2 mL of water per gram of glucose. The glucose solution had to be consumed within 5 min.
Blood samples were collected before and at 5, 10, 20, 30, 45, 60, 90, 120 and 180 min after glucose intake. Blood samples were collected in BD P800 tubes (BD Diagnostics, Franklin Lakes, NJ, USA) containing protease-, esterase- and dipeptidyl peptidase-4 inhibitors, and EDTA for measurements of insulin and glucagon. Blood glucose was measured immediately; the blood samples were then centrifuged, and plasma was stored at –80℃ until analysis.
Glucose, insulin and glucagon analyses
Blood glucose concentrations were measured with a blood glucose meter (Accu-Chek, Roche Diagnostics, Basel, Switzerland; validated for porcine blood at the Department of Clinical Chemistry, SLU, Sweden). Plasma concentrations of insulin were determined by Porcine Insulin ELISA (Mercodia, Uppsala, Sweden). All samples were run in duplicate with a coefficient of variance (CV) of < 10%. Results were converted from ug/L to pmol/L using a conversion factor of 174 as recommended by the manufacturer. Plasma concentrations of glucagon were analysed by Glucagon ELISA (Mercodia, Uppsala, Sweden). All samples were run in duplicate with a CV of < 10%.
Euthanasia and post-mortem examination
The pigs were euthanized with an IV overdose of pentobarbital sodium (Euthasol vet, 400 mg/mL; Virbac) at the end of the experiment. The pigs underwent gross examination post-mortem.
Statistical analyses
All values are presented as mean ± standard error of the mean (SEM). Area under the curve (AUC) values are presented as the net area, and were calculated using the trapezoid rule with the 0 min value as baseline. Comparisons between groups were determined by Kruskal–Wallis one-way analysis of variance on ranks with Dunn’s post hoc test. P values of < 0.05 were considered to be statistically significant.
Results
During social training, all pigs became accustomed to the staff. As a result the experimental procedures and all experiments could be carried out without the animals being distressed. Blood concentrations of Hb, total and differential WBC counts and serum levels of CREA, ALAT, ASAT, GLDH, and GT were all within the reference ranges for pigs. The animals did not show any signs of disease either during the habituation and experimental periods or during post-mortem examinations.
Experiment 1
Anaesthesia induction was smooth with both the anaesthetic protocols; the pigs lay down calmly and could be lifted onto the stretcher for prompt endotracheal intubation after drug administration. The time from induction until the endotracheal tube was connected to the ventilator in the surgical ward (approximately 5–10 min) was similar for both protocols. After TZB induction, the pigs continued to breathe spontaneously without any need for manual ventilation; while all pigs had to be manually ventilated after propofol induction until the endotracheal tube was connected to the ventilator. Initial tachycardia (Figure 1) and low blood pressure (data not shown) were observed with propofol induction. Two of the pigs shivered during both anaesthetic protocols while the other two pigs did not. Apart from the above-mentioned changes, no abnormal physiological parameters were noted. For the TZB protocol, doses of 5–6.25/0.2–0.25 mg/kg/h were sufficient to produce general anaesthesia, whereas doses of 13–20 mg/kg/h of propofol were needed to produce the same effect.
Heart rate and blood glucose concentrations during 3 h of anaesthesia with propofol (open circles) and tiletamine/zolazepam/butorphanol (filled circles) in growing pigs (Yorkshire × Swedish Landrace × Hampshire; n = 4). Values are presented as mean ± SEM.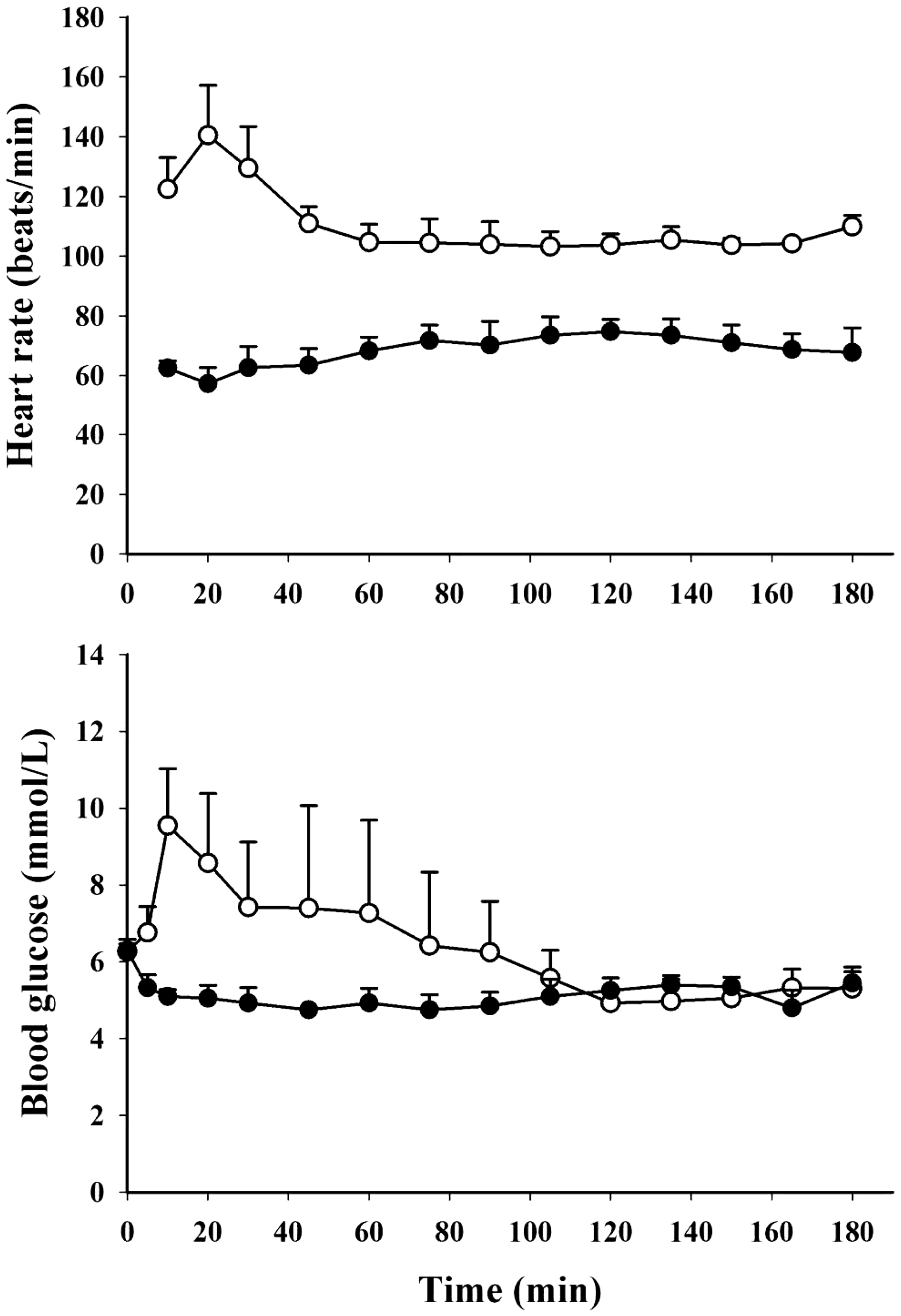
During TZB anaesthesia blood glucose was within the reference range throughout the 180 min (range 3.6–6.8 mmol/L) while hyperglycaemia was observed in three out of four pigs during propofol anaesthesia within 5–10 minutes (peak levels range 8.3–15.4 mmol/L), see Figure 1. Blood glucose levels were within the reference range within 20–105 min after induction of anaesthesia. Due to the hyperglycaemia observed after propofol induction, TZB was chosen for further evaluation with OGTT.
Experiment 2
Blood glucose levels, plasma insulin and glucagon concentrations, and AUC values during OGTT are presented in Figure 2. During OGTT without anaesthesia, blood glucose levels increased, with peak levels at 30 min (7.1 ± 0.3 mmol/L). When anaesthesia was induced immediately after glucose intake, blood glucose increased by 0.6 mmol/L in one pig and none at all in a second pig. Consequently, a 15 min delay was introduced between glucose intake and anaesthesia in the remaining two pigs. In these two pigs, the initial increase in blood glucose was comparable to the values from OGTT without anaesthesia, with peak levels of 7.4 and 6.5 mmol/L at 20 min. However after 20 min, blood glucose concentrations fell more rapidly, and the AUC of glucose was lower than during OGTT without anaesthesia.
Blood glucose levels and plasma insulin and glucagon concentrations, and corresponding net AUC values during OGTT in pigs (Yorkshire × Swedish Landrace × Hampshire; n = 4). Awake (filled circles, filled bars), anaesthetized with a combination of tiletamine, zolazepam and butorphanol immediately (open circles, bars with horizontal stripes) and 15 min (open triangles, bars with vertical stripes) after glucose intake. Values are presented as mean ± SEM. *Indicates a significant difference of P < 0.05. AUC: area under curve; OGTT: oral glucose tolerance test.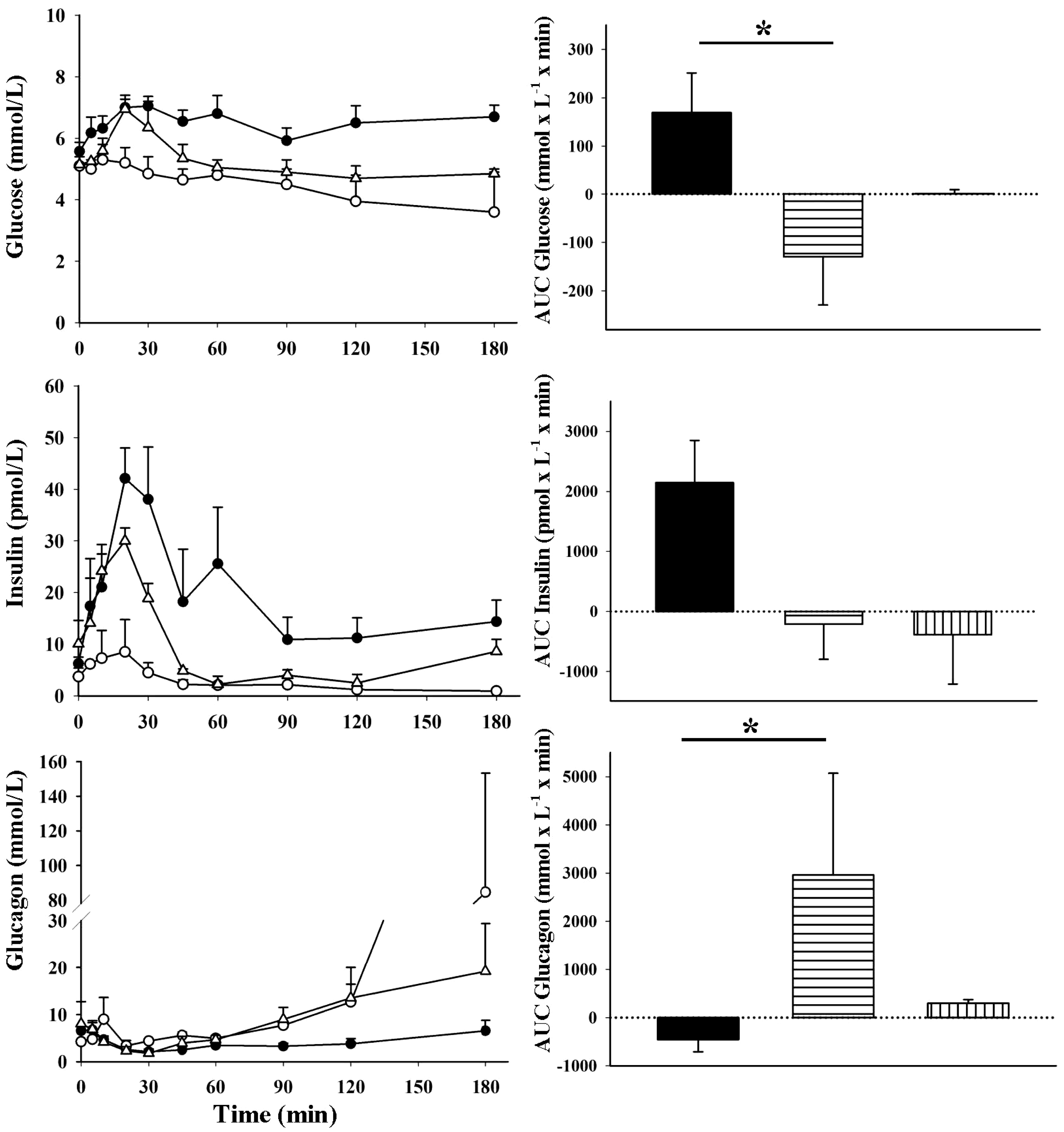
During OGTT without anaesthesia, peak insulin levels (42.1 ± 5.9 pmol/L) and glucagon nadir (2.1 ± 0.3 mmol/L) were observed at the 20 and 30 min sampling periods, respectively. During OGTT with anaesthesia, peak insulin levels were also observed at 20 min after glucose intake; however, peak values were lower, both when anaesthesia was induced immediately (80% lower) and 15 min after glucose intake (25% lower). The AUC of insulin was lower with anaesthesia than without; this difference did not reach statistical significance, but the insulin AUC was more than 10 times higher during OGTT without anaesthesia. When anaesthesia was induced immediately after glucose intake, glucagon levels were suppressed by 0.9 mmol/L, while the glucagon suppression was 6.3 mmol/L during the first 30 min when anaesthesia was delayed, which is comparable to the suppression of OGTT without anaesthesia. With anaesthesia, glucagon levels increased in all pigs during the second half of the OGTT, especially between 120 and 180 min. In one pig, glucagon levels increased to 153.4 mmol/L at 180 min; in this pig the corresponding blood glucose concentration was 2.2 mmol/L.
Discussion
The results from the present experiment confirm that a combination of TZB does not affect blood glucose levels during 3 h of anaesthesia in healthy pigs. With TZB there were no adverse physiological events and anaesthesia induction was smooth. This protocol is useful if anaesthesia is needed during metabolic research in which glucose-related physiology is under study. However, two out of the four pigs shivered during anaesthesia which can be a problem under some conditions, for example during imaging procedures such as computed tomography, magnetic resonance imaging or PET scans, in which the animals need to lie completely still. The cause of shivering in pigs during anaesthesia is unknown, and in the present experiment the same two pigs shivered during both anaesthetic protocols, while the other two pigs did not. Neuromuscular blocking agents (NMBs) may be used in addition to anaesthetics to reduce shivering, 21 but it takes skill to adequately manage administration of these agents. They should only be administered if one is absolutely certain that an adequate depth of unconsciousness has been reached, since the animals are unable to move in response to painful or stressful stimuli. Since the pigs in the present experiment did not receive NMBs, depth of anaesthesia was easy to assess, and the infusion dose could be changed accordingly.
The propofol protocol was selected for evaluation in the present experiment because it has previously been described as not affecting carbohydrate metabolism in rat β-cells in vitro or during IVGTT in rabbits. 13 It has also been reported to be a good choice for keeping blood glucose at a constant level during surgery in human patients with diabetes. However, how well the protocol would work in pigs could not be predicted with certainty due to species differences in carbohydrate metabolism and the use of different drugs in combinations with propofol in the human study. In the present study propofol was excluded after the first experiment due to severe respiratory depression and the observed hyperglycaemia and tachycardia. Anaesthesia was induced very slowly with IV propofol and proceeded smoothly. Nevertheless, bradypnea followed, and the pigs needed manual ventilation until the endotracheal tube was connected to the ventilator in the surgical ward. Bradypnea causes a large catecholamine release, 22 and consequently, tachycardia and hyperglycaemia. In the present experiment, the effects of propofol per se on carbohydrate metabolism could not be evaluated with certainty, since the bradypnea may have affected the results. However, regardless of biological mechanism, this protocol was excluded from further testing due to the presence of hyperglycaemia.
After TZB was confirmed not to affect glucose homeostasis during a 3 h period of anaesthesia, the pigs were anaesthetized a second time with the same protocol, but only after they were bottle-fed an oral glucose load. Unexpectedly, anaesthesia severely affected the OGTT results, producing lower glucose and insulin levels and higher glucagon levels than during OGTT without anaesthesia. The difference in AUC of insulin secretion between unanaesthetized and anaesthetized pigs did not reach statistical significance. We did not think it would be ethical to add more pigs to this pilot study, since all results clearly pointed in the same direction. In two of the pigs that were anaesthetized 15 min after glucose intake, the first 15 min of OGTT was similar to OGTT in unanaesthetized animals. However, after anaesthesia induction, glucose and insulin levels were lower and glucagon levels higher than during OGTT in the unanaesthetized state. During OGTT under anaesthesia, plasma levels of glucagon were increased, especially during the last hour. In healthy individuals, glucagon is the most important hormone that increases blood glucose levels in response to hypoglycaemia, 23 by inducing glycogenolysis and gluconeogenesis.24,25 In the present experiment, blood glucose did not increase much during OGTT under anaesthesia, and a trend towards lower blood glucose levels was observed in anaesthetized pigs, especially at the end of OGTT. Increased glucagon levels in anaesthetized pigs were probably due to a physiological response to maintain blood glucose at normal levels, rather than a direct effect of the anaesthetic agents on glucagon secretion, since the levels did not increase immediately after anaesthesia induction. The large increase in glucagon and the hypoglycaemia present in one pig were most likely due to low glucose uptake during OGTT, glycogen depletion and an insufficient rate of gluconeogenesis, causing the alpha cells to secrete large amounts of glucagon. Glucagon levels during OGTT with a normal glucose curve under anaesthesia need to be investigated further.
Even though it cannot be ascertained from the results in the present experiment that the anaesthetic agents per se did not directly affect carbohydrate metabolism, it seems unlikely that they would have, based on results from previous studies. NMDA receptor agonists such as ketamine and tiletamine have minimal or no effect on carbohydrate metabolism in rats, 17 humans, 14 non-human primates,7,18 cats 8 or bears. 6 In the present experiment, tiletamine was given together with zolazepam, in a prefabricated mixture. The effects of zolazepam alone on carbohydrate metabolism have not been investigated, but when given in a 1:1 mixture with tiletamine, zolazepam did not affect IV glucose tolerance,6,7 and it therefore seems unlikely that it would affect carbohydrate metabolism to any extent during OGTT.
In the present experiment butorphanol was used because of its anaesthetic sparing effects, and this combination has previously been described in Japanese black bears during IVGTT. 6 In addition, opioids inhibit catecholamine and corticosteroid secretion and thereby limit hyperglycaemia and catabolic hormonal responses due to the activation of the hypothalamic–pituitary axis.26–29 However, one possible explanation for the attenuated glucose and insulin levels observed during OGTT under anaesthesia is a delay in gastric emptying caused by butorphanol administration. Opioids have long been known to affect gastrointestinal motility, 30 although the exact mechanisms are not fully understood. Morphine and other µ-agonists inhibit gastric emptying and intestinal transit,31–34 even at low doses. 35 Butorphanol, used in the present experiment, is mainly a k-agonist 36 and is known to decelerate gastric emptying in horses.34,37 Three other k-agonists, dynorphin-(1–13), U50,488 H and KETO, strongly inhibit gastric emptying in rhesus monkeys; the inhibition of the early (0–10 min) gastric emptying response by these three k-agonists is nearly maximal. 38 Although gastric emptying rate was not monitored in the present experiment, it seems plausible that butorphanol decelerated gastric emptying and thereby affected the OGTT results. With a slower gastric emptying the rise in glucose will be proportionally smaller, 39 and with a strong inhibition gastric content can be retained in the stomach for several hours. 40 It is possible to exclude butorphanol from the anaesthetic protocol as tiletamine and zolazepam alone can produce general anaesthesia in pigs. However, during surgery opioids are important in contributing towards a balanced anaesthesia, and to inhibit catecholamine release due to surgical stress.26–28 Further studies are needed to elucidate whether a combination of tiletamine and zolazepam alone can be a potential anaesthetic protocol for OGTT in pigs. The mixture has been used during OGTT in cats that had received glucose by gavage, after which blood glucose concentrations increased to expected levels. 41 Additionally, a mouse model for measuring gastric emptying was recently described, in which tiletamine/zolazepam anaesthesia was used. 42 In this model, the anaesthesia was considered not to have any effect on gastric emptying. In addition, ketamine, a structurally related NMDA receptor antagonist, has no effect on gastric emptying.43,44 Thus, the almost complete lack of increase in blood glucose during OGTT in anesthetized pigs in the present experiment is not likely to be attributed to tiletamine/zolazepam.
In conclusion, results from this pilot study indicate that IV infusion of a combination of TZB can be used for 3 h of anaesthesia in pigs without affecting glucose homeostasis. The anaesthetic protocol described is especially useful for studies involving glucose-related physiology. The protocol should not, however, be used in combination with OGTT as glucose, insulin and glucagon levels are affected by these agents.
Footnotes
Acknowledgements
Financial support by the Torvald and Birgitta Gahlin’s Foundation is gratefully acknowledged. C Jansson, M Wallbring and L Waern are acknowledged for their excellent technical assistance and animal handling.
Declaration of Conflicting Interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Torvald and Birgitta Gahlin’s Foundation.
