Abstract
The parenteral administration of hydrophobic substances in vivo requires the use of organic solvents to ensure sufficient solubility and avoid precipitation. Dimethyl sulfoxide is commonly used for this purpose. Based on the common assumption that polyethylene glycol (PEG) is non-toxic, our local regulatory authorities recently recommended the use of PEG instead. However, mice injected intraperitoneally (i.p.) with PEG 200 at a dose of 8 mL/kg (i.e. 9 g/kg) did not tolerate PEG 200 well, and half of the animals had to be euthanized. Our results demonstrate that although PEG 200 is generally considered to be harmless, it can be toxic when injected i.p. and is painful for the recipient mice. Nevertheless, it can be used as a solvent for repeated i.p. injections in mice at a dose of 2 mL/kg (i.e. 2.25 g/kg) without obvious signs of systemic toxicity.
Introduction
To minimise the suffering of experimental animals, solvents for injections should ideally be isotonic, used at body temperature and have a neutral pH of 7.0–7.3. 1 However, alternative solvents have to be employed if substances are poorly soluble in aqueous solution.
Dimethyl sulfoxide (DMSO; CAS number 67-68-5) is commonly used as a vehicle for hydrophobic substances to improve their solubility and avoid precipitation (Figure 1(a)). DMSO has a relatively low toxicity when applied systemically, and is rapidly cleared from the circulation and tissues.2,3 However, the injection of DMSO causes transient pain. Moreover, DMSO influences a variety of biological processes, and induces several side effects, limiting its range of applicability.
4
Chemical formulae of (a) dimethyl sulfoxide (DMSO), (b) polyethylene glycol (PEG) and (c) tetraethylene glycol. (b) In the case of PEG 200 that was used in our study, n is 4.2.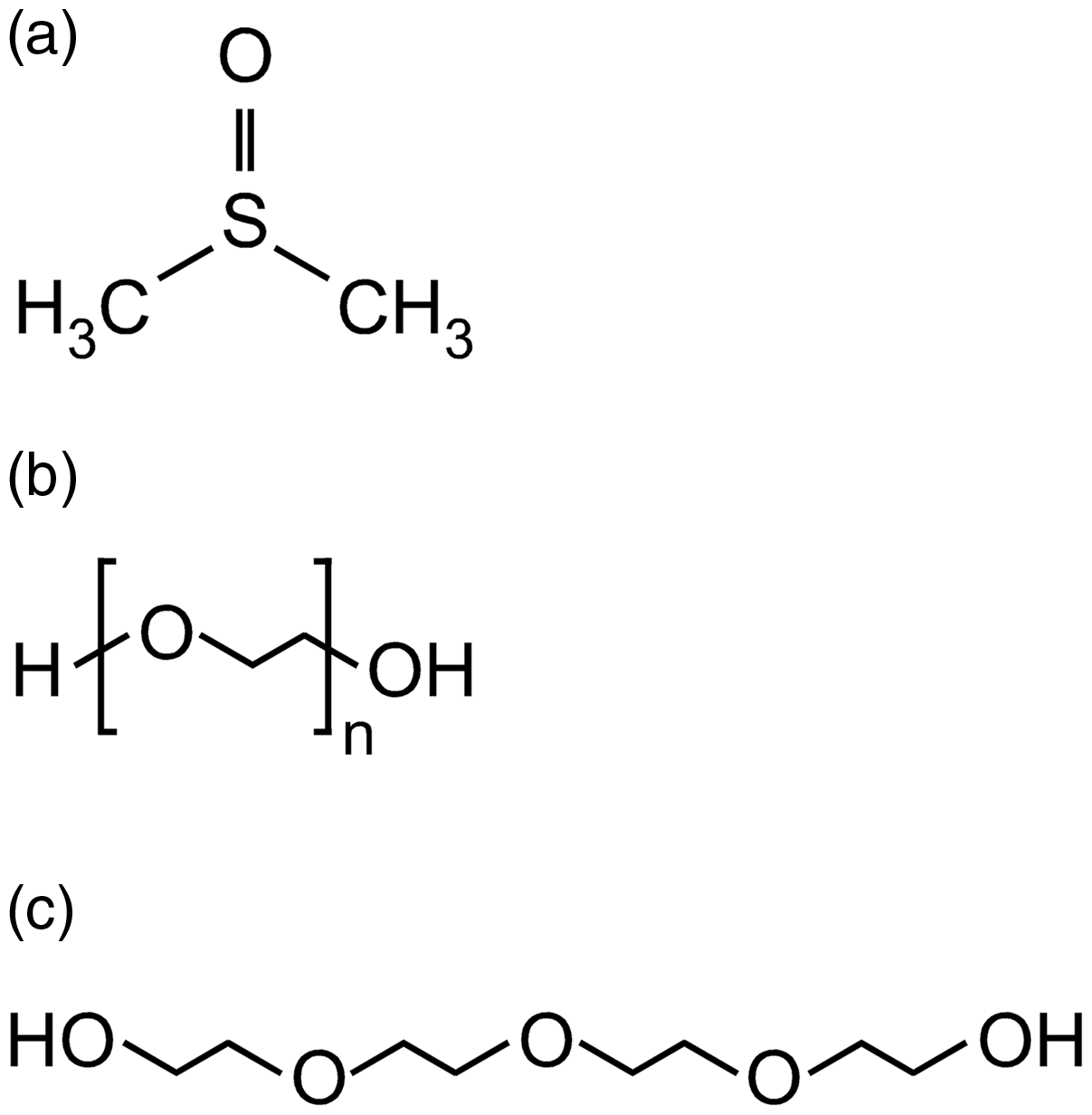
Polyethylene glycols (PEGs; α-Hydro-ω-hydroxypoly(oxy-1,2-ethanediyl); CAS number 25322-68-3) are synthetic hydrophilic polyethers that consist of repeating ethylene oxide units. The general chemical formula of PEGs is H[OCH2CH2] n OH, where n represents the average number of oxyethylene groups (Figure 1(b)). 2 Pharmaceutical grade PEG preparations are mixtures of polyethers with different molecular weights and chain lengths. The pharmaceutical term PEG 200 denotes a PEG mixture with an average molecular weight of 200 g/mol and an average number of 4.2 oxyethylene groups. 2 PEGs increase the solubility of poorly soluble compounds, and represent a potential alternative to other organic vehicles. 2 They are widely used in pharmaceutical and cosmetic products, and are generally considered harmless, as they are non-toxic when applied orally or dermally.2,5–7 Furthermore, PEGs are used as food additives, and a daily intake of 10 mg/kg bodyweight is considered acceptable. 8 However, reports about the toxicity of parenterally applied PEG in mice are scarce, and it is not widely accepted that the injection of PEG can be harmful.9–11
We have used DMSO as a solvent for preclinical parenteral in vivo studies for more than a decade.12–15 In 2017, we applied for permission to inject a test substance dissolved in DMSO intraperitoneally (i.p.) into tumour-bearing mice. However, the local regulatory authorities recommended that DMSO should be replaced with PEG, which they considered less harmful than DMSO. Having had no previous experience with the use of PEG, we followed this recommendation. Consequently, we used PEG 200 as a vehicle, and injected it at the approved dose. Unexpectedly, PEG 200 was not tolerated well by the mice, and half of them had to be euthanized. A subsequent literature search revealed that the toxicity of parenterally applied PEG had been reported more than five decades ago. 10 As this literature is not widely read, here we highlight the often overlooked fact that PEG 200 is toxic with a LD50 of 7.5 mL/kg in i.p. injections. 10 Moreover, we observed that PEG 200 causes pain when injected i.p., even at a moderate dose.
Materials and methods
The animal experiment was approved by the local regulatory authorities, and was performed according to German legal requirements.
In control experiments, 16 (eight male) in-house-bred C57BL/6 J mice aged 12–14 weeks (mean ± SD weight males: 29.9 ± 2.2 g; mean weight females: 22.9 ± 1.6 g) were randomly allocated to two groups with eight animals (four male) each. These groups were injected subcutaneously with either 5000 or 20,000 syngeneic B16-F10 cells in 100 µL phosphate-buffered saline per animal. On day 4 post tumour cell inoculation, the animals were injected i.p. with 8 mL/kg (9 g/kg; approximately 200 µL/animal) undiluted BioUltra PEG 200 (Sigma–Aldrich, Steinheim, Germany; lot number: BCBS0153V). The PEG 200 dose used was based on the solubility in PEG 200 of the chemical substance we intended to test in subsequent experiments with other groups of tumour-bearing mice. Treatment was suspended when we observed that the mice did not tolerate the injected PEG 200, and was resumed with the surviving mice on day 6 post tumour cell inoculation, with a reduced PEG 200 dose of 2 mL/kg (2.25 g/kg; approximately 50 µL/animal). During the experiment, the animals were monitored at least four times per day. Tumour growth was assessed with a calliper twice per week. Animals were killed by cervical dislocation. The experimental scheme is depicted in Figure 2.
Schematic overview of how the experiment was performed. Two groups of mice, each with four male and four female animals, were used for the experiment. Group 1 was subcutaneously injected with 5000 syngeneic B16-F10 melanoma cells; group 2 received 20,000 B16-F10 cells. On day 4 post tumour cell inoculation, the animals were intraperitoneally injected with PEG 200 at 8 mL/kg. The animals did not tolerate the treatment well, and eight animals had to be euthanized by cervical dislocation. The remaining mice that had recovered from the first PEG injection were then injected again intraperitoneally on days 6–8, 11–15, 18–22 and 25–29 post tumour cell inoculation with PEG 200 at a dose of 2 mL/kg. The remaining animals were killed on day 29 post tumour cell injection by cervical dislocation.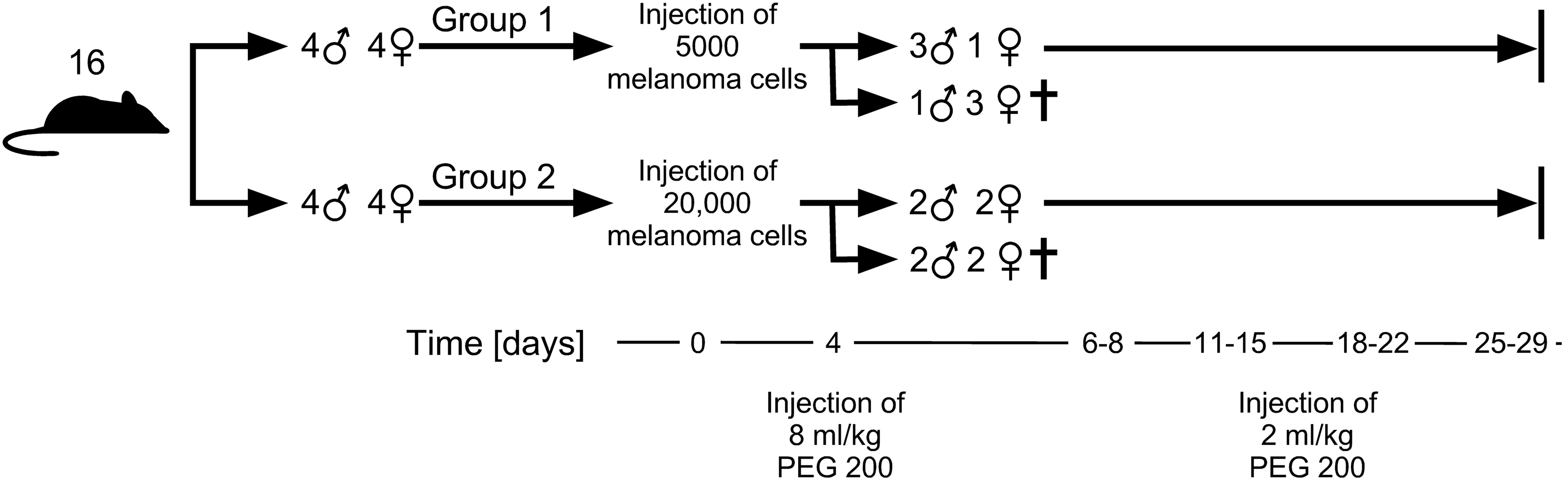
Mice were kept in groups of four in type III macrolon filtertop cages (Tecniplast, Hohenpeißenberg, Germany) containing SAFE fs14 bedding (Rettenmaier & Söhne, Rosenberg, Germany). Rat/mouse extruded food (SSNIFF, Soest, Germany) and sterilised water acidified with HCl (pH 2.8–3.1) were provided ad libitum. The specific pathogen-free area was kept at 20℃ and 30–60% humidity on a 07:00–20:00 hours light cycle. The health status of the animals in the facility is routinely assessed by a commercial veterinarian laboratory (mfd Diagnostics, Wendelsheim, Germany) in serological examinations every three months (epizootic diarrhoea of infant mice, mouse hepatitis virus, murine norovirus, minute virus of mice, Theiler’s encephalomyelitis virus, Pasteurella pneumotropica) or annually (Clostridium piliforme, Mousepox, lymphocytic choriomeningitis virus, mouse adenovirus type 1 and type 2, Mycoplasma pulmonis, pneumonia virus of mice, Reovirus type 3, Sendai virus). The serology was found to be negative for the parameters tested.
Results
PEG 200 is not tolerated well by mice upon i.p. injection
Overview moribund mice.
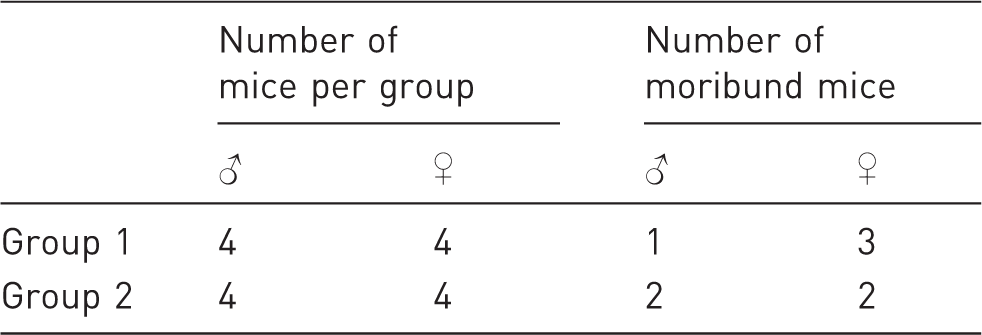
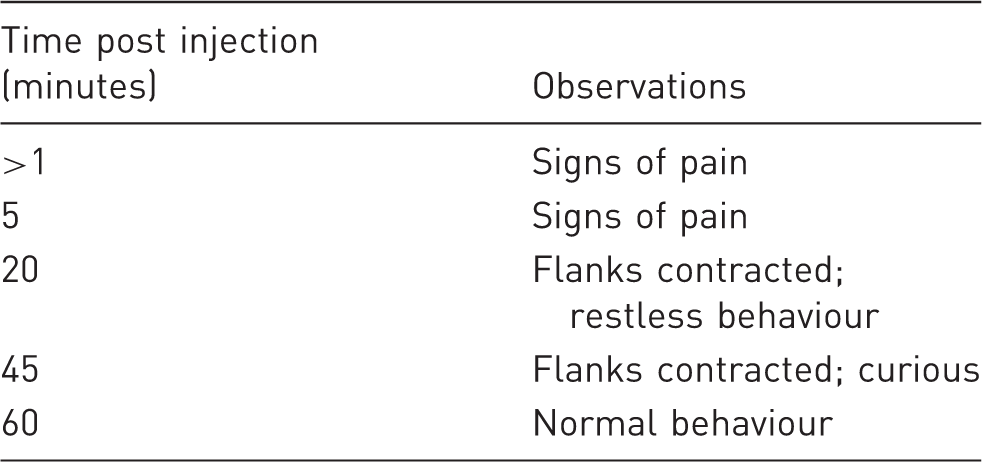
Effects of intraperitoneal injection of PEG 200 at 2 mL/kg in mice.
Discussion
Organic vehicles such as DMSO have higher toxicity and unwanted side effects compared to isotonic aqueous vehicles, and the injection of DMSO is more painful for the recipient animals. During evaluation of our recent application to perform animal experiments, the local authorities strongly recommended the use of PEG, which they considered harmless, with the intention of replacing DMSO with a vehicle that is better tolerated. Unaware of its toxicity, we complied with this recommendation, and injected PEG 200 i.p. into tumour-bearing mice. Unexpectedly, we observed that PEG 200 was more harmful than DMSO, that the injection of PEG 200 caused pain even at a dose of 2 mL/kg and that a dose of 8 mL/kg was not tolerated well by the animals.
While generally considered non-toxic when applied orally or dermally or when used in cosmetic products, adverse reactions to PEG have been reported, especially for PEGs with low molecular weight or when PEG has been applied to mucous membranes or damaged skin.2,5–7 For example, three burn patients died due to renal failure caused by a burn cream containing PEG. 16
The toxicity of PEGs injected intravenously as a 5% solution was found to be very low in rabbits (LD50: 10,000 mg/kg). 17 However, few studies have examined the toxicity of parenterally applied PEG in mice. Shideman and Procita compared the toxicity of polypropylene glycols and PEGs. 9 They report a LD50 of 9.2 g/kg for PEG 400 upon i.p. injection into mice – a value for this size of PEG that is also available in Scifinder.9,18 In 1965, Worthley and Schott found that the LD50 of PEG 200 was 7.5 mL/kg when administered i.p. in Swiss albino mice. 10 As PEG can reduce plasma volume without altering osmolality, it has been suggested that the lethal effect of parenterally applied PEG is caused by hypovolaemic shock. 11 We report here that a single i.p. injection of PEG 200 at a dose of 8 mL/kg (9 g/kg; densityPEG 200 = 1.125 g/mL at 20℃) into mice caused pain and was toxic. The lethality within our population was 50% (8/16 animals), suggesting an LD50 of 8 mL/kg, which is slightly above the LD50 reported by Worthley and Schott for PEG 200, and slightly below the LD50 determined by Shideman and Procita for PEG 400.9,10 However, the pain that we observed directly after the injection was not reported in previous studies.9,10 Clearly, this pain should be considered when the suffering of the animals is assessed.
Our observations suggest that animals injected with PEG i.p. should receive analgesics, although these may distort experimental outcomes. Similarly, use of alternative vehicles requires careful consideration. For example, vegetable oils that contain linolic acid and vitamin E influence metastasis in vivo.15,19,20 Nanocarriers can also be toxic and can bias results. 21 For example, pegylated phospholipid micelles differentially induce apoptosis in cancerous compared to non-cancerous cell lines. 22
We used a high grade (‘BioUltra’) PEG solution that is tested to ensure minimal trace impurities below 10 mg/kg, except for sodium (200) and potassium (500). 23 Assuming a maximum of 500 mg/kg in the PEG 200 preparation, and given a PEG dose of 9 g PEG 200/kg and an average weight of 25 g per mouse, 0.225 g PEG 200 and approximately 0.1125 mg potassium were applied per animal. The LD50 of KCl is 15.925 mg/mouse, and no lethality was observed at 12.5 mg/mouse. 24 Thus, the estimated potassium dose that we applied is 1/72th below the LD50, making it highly unlikely that potassium is responsible for the systemic toxicity that we observed upon injection of PEG 200.
PEG 200 is a mixture of PEGs with an average molecular weight of 190–210 g/mol and an average oxyethylene group number of 4.2. 2 The predominant species in PEG 200 is tetraethylene glycol (CAS number 112-60-7; Figure 1(c)). Due to the manufacturing process, PEG species with a lower molecular weight such as monoethylene glycol (CAS number 107-21-1; 62.07 g/mol) and diethylene glycol (CAS number 111-46-6; 106.12 g/mol) can be found in tetraethylene glycol and PEG 200 preparations. 25 Monoethylene glycol and diethylene glycol are toxic. 26 When injected i.p. into mice, the LD50 for monoethylene glycol is 5.62 g/kg, and the LD50 for diethylene glycol 9.73 g/kg. 27 Although information about the content of monoethylene glycol and diethylene glycol in the BioUltra PEG solution we used is not available, other similar products are reported to have a combined monoethylene and diethylene glycol content of 0.4–7%.28,29 Depending on the presence and actual concentration of monoethylene diethylene glycol and diethylene glycol in the BioUltra PEG solution, these two compounds could conceivably contribute to the toxic effects of PEG 200 that we observed. However, even assuming a monoethylene content of 1% and a diethylene content of 6%, doses of only 0.89 and 0.534 g/kg, respectively, would have been reached at the beginning of the experiment. These doses represent 1/60 and 1/20 of the LD50 of monoethylene glycol and diethylene glycol, respectively, and would not have been sufficient to cause the toxic effects alone. However, we cannot rule out a contribution of monoethylene glycol and diethylene glycol to the pain we observed upon injection of PEG 200.
A general recommendation for injections in experimental animals is that the solutions should have a neutral pH of 7.0–7.3. 1 The poor solubility of our intended test substance meant that undiluted PEG 200 had to be used. When we measured the pH of the PEG 200 at room temperature, we found that the pH was between 8.6 and 9.1. However, the impact of pH varies depending on the route of administration. 1 Therefore, although a contribution of pH to the observed pain caused by the PEG 200 injections cannot be ruled out, it is not trivial to predict the actual impact of the pH on the peritoneum.
Mice injected i.p. with a single dose of 4 mL/kg PEG 400 tolerated the treatment, and were killed 1, 5, 8 or 24 h post injection. 11 Alanine transaminase (ALT) levels were temporarily increased, ascites accumulated and necrosis occurred in the subcapsular regions of the liver. 11 In our experiment, a lower dose of 2 mL/kg PEG 200 (2.25 g/kg) was well tolerated, and no accumulation of fluid in the peritoneal cavity was observed post-mortem. As we did not measure ALT levels in the blood or investigate possible hepatic necrosis, we cannot draw conclusions about possible effects on the liver.
In summary, PEG 200 is not tolerated well when injected i.p. into mice at 8 mL/kg. At 2 mL/kg, PEG 200 still induces local pain but can be administered repeatedly without obvious signs of systemic toxicity. Due to the serendipitous nature of our observations, we did not use injection with control substances to assess the toxicity of PEG 200, and therefore our study can formally only be considered observational rather than conclusive. Nevertheless, our observations highlight the fact that although PEGs are considered harmless and non-toxic when applied orally or dermally to the undamaged skin, i.p. injections of pure, undiluted PEG are not safe at a dose of 8 mL/kg, and cause pain even at a dose of 2 mL/kg.
Footnotes
Acknowledgements
We gratefully acknowledge the expert technical assistance of Selma Huber and Sabine Müller.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
