Abstract
NSG™ mice are highly immunocompromised thus demonstrate high efficiency engraftment of patient-derived xenografts (PDXs) for pre-clinical oncology research. It has previously been reported that NSG™ mice are hyper-sensitive to doxorubicin due to the impairment of DNA damage repair mechanisms. As such, doxorubicin causes a wide spectrum of toxicities including cardiotoxicity, hepatotoxicity and intestinal toxicity in NSG™ mice. Doxil is an alternative clinical formulation of doxorubicin, where doxorubicin is encapsulated within pegylated liposomes and displays improved toxicity profiles compared to conventional doxorubicin. Doxil was substituted for doxorubicin in our study to determine its toxicity profile in female NSG™ mice. The mice that were treated with Doxil developed dose-dependent histopathological alterations associated with non-glandular gastritis, with non-Helicobacter spp. bacterial infiltrates, as well as oesophagitis. Of note, a study using a dose of 2 mg/kg Doxil was terminated early due to significant weight loss while the use of Doxil at 1 mg/kg allowed for repeated treatment of twice a week for a duration of three weeks. A dose optimised treatment regimen has now been established and can be applied to assess Doxil-related anti-tumour efficacy in a range of PDX-bearing NSG™ mice.
Introduction
Patient-derived xenografts (PDXs) are derived from surgically removed tumour fragments or cancer cells that are transplanted from a cancer patient into immunodeficient mice. 1 In recent years, PDXs have emerged as useful tools for pre-clinical translational research for evaluating anti-cancer drug efficacy and enabling a personalised approach for selection of cancer therapies. 1 The NSG™ mice, also commonly known as NOD scid gamma, are considered as one of the best mouse strains for establishing different PDXs, since NSG™ mice are extremely immunosuppressed and exhibit high levels of successful xenotransplantation.2,3 This is attributed to the two mutations carried by NSG™ mice: the severe combined immune deficiency (Prkdcscid) leading to a deficiency of B cells and T cells, and loss of interleukin-2 receptor gamma chain (IL2rgnull) leading to an absence of functional natural killer cells. 4 The Prkdcscid mutation also results in impaired DNA double-stranded break repair in NSG™ mice and this causes them to be susceptible to agents that induce DNA double-strand breaks such as doxorubicin, 5-fluorouracil and radiation.4–7
Doxorubicin remains as one of the most important chemotherapy drugs used clinically as it has a wide spectrum of activity against different types of cancers including leukaemia, lymphoma, breast cancer and sarcoma. 8 However, the use of doxorubicin is limited by a number of dose-limiting and other well-documented side effects. 9 Studies on NSG™ mice have found that doxorubicin-induced toxicity is dependent on drug dose, route of delivery and selected drug combinations. 5 An alternative formulation of doxorubicin is pegylated liposomal doxorubicin, known as Doxil in the USA or Caelyx in Europe. In comparison to conventional doxorubicin, the pharmacokinetic profile of Doxil is altered by the presence of pegylated liposomal components that encapsulate doxorubicin hydrochloride, 9 maintaining the anti-tumour efficacy of doxorubicin while having favourable toxicity profiles, with reduced cardiotoxicity, myelosuppression and alopecia. 10
To date, the tolerable dose of doxorubicin in NSG™ mice has been established as three daily doses at 1.5 mg/kg in a 4-weeks study, 5 however, combination with cytarabine was required to avoid drug toxicity, and there has been no comparable studies with Doxil. In this study, we investigated Doxil-mediated toxicity and found that it causes dose-dependent non-glandular gastritis and oesophagitis in NSG™ mice. We also determined the optimal dose of Doxil tolerated by NSG™ mice in three cycles of repeated treatments. This provides conditions that can be utilised for assessing the chemotherapeutic efficacy of Doxil in NSG™ mice that have been transplanted with different types of patient-derived cancers.
Material and methods
Animal handling and drug treatments
The use of animals and drug treatment protocols were approved by the Animal Ethics Committee at La Trobe University under ethics number ‘AEC15-88’. All animal experiments complied with all provisions of the Prevention of Cruelty to Animals Act, 1986, and the Australian Code for the Care and Use of Animals for Scientific Purposes, eighth edition, 2013.
Female NSG™ (NOD.Cg-Prkdc scid Il2rg tm1Wjl /SzJ/Arc) (JAX stock #005557) at 7–8 weeks old (20.3–22.4 g) were sourced from the Animal Resource Centre, WA, Australia and allowed to acclimatise for a week.4,11 They were housed in a specific pathogen free area in La Trobe University Central Animal House (12:12 h light:dark cycle, 21°C ± 2°C, 45% ± 10% humidity), in GM500 individually ventilated cages (Tecniplast, NSW, Australia) (three mice/cage) containing appropriate bedding, ad libitum access to irradiated Barastoc rat and mouse pellets (Ridley, VIC, Australia) and autoclaved water. Routine monitoring of the health status in the facility was performed and excluded these microorganisms: adenovirus type 1 and 2, ectromelia virus, lymphocytic choriomeningitis virus, minute virus of mice, mouse hepatitis virus, mouse parvovirus, pneumonia virus of mice, reovirus type 3, Sendai virus, Theiler’s encephalomyelitis virus, Citrobacter rodentium, Clostridium piliforme, Corynebacterium kutscheri, Mycoplasma pulmonis, Salmonella spp., Streptobacillus moniliformis, Streptococcus pneumonia (α-haemolytic), Streptococcus spp. (β-haemolytic), Demodex musculi, Giardia muris, Mycoptes musculinus, Myobia musculi, Aspiculuris spp., Syphacia spp., Polyplax serrata, Radfordia spp., Spironucleus muris and Tritrichomonas muris.
Mice were handled in a UV-sterilised BS48 biosafety cabinet (Tecniplast, NSW, Australia), weighed and monitored daily for any signs of distress including reduced activity, depressed behaviour and abnormalities in gait, coat, breathing rate, eyes or faeces. A total of nine mice were randomly allocated into three mice/group by an independent animal technician prior to drug treatment. Mice were treated with intravenous injection of saline (B. Braun, NSW, Australia), 1 mg/kg Doxil (Caelyx, Janssen-Cilag, NSW, Australia) or 2 mg/kg Doxil twice per week for a total duration of 3 weeks (six doses in total). The procedure was performed by a trained animal technician. Mice were humanely euthanised by carbon dioxide asphyxiation at either the experimental endpoint or ethical endpoint. The ethical endpoint as approved by the Animal Ethics Committee represents body weight loss of more than 20%, or weight loss between 10–20% coupled with any signs of distress listed above.
Pathology assessment
After euthanasia, dissection was performed along the peritoneal wall and chest, followed by fixing whole mice in 10% neutral buffered formalin prior to pathology examination at Cerberus Sciences (VIC, Australia). The tissues (2–3 mm) were placed in histology cassettes, embedded in paraffin and sectioned at 4 µm. Gram staining as well as haematoxylin and eosin staining were performed. These sections were imaged by an Olympus BX40 light microscope (Olympus Lifesciences, Tokyo, Japan) and pathological changes were described and interpreted independently by a board-certified pathologist who was blinded from the treatment groups (LR).
Polymerase chain reaction (PCR) analysis
Fresh mouse faecal pellets were collected in a sterile microfuge tube and transported to Cerberus Sciences (VIC, Australia). DNA extraction was performed using the Complex200_v6_DSP protocol and QIASymphony® DSP Virus/Pathogen mini kit (Qiagen, VIC, Australia) as described by the manufacturer. PCR was performed in a Rotor-Gene (Qiagen, VIC, Australia) using the extracted DNA and a QuantiNova Multiplex PCR kit (Qiagen, VIC, Australia). The primers and probes for Helicobacter spp. were designed on the basis of published information. 12 An internal control primer, targeting mouse genetic material was run simultaneously during PCR amplification for Helicobacter spp.
Statistical analysis
Data analysis was carried out using Prism® 7 software (GraphPad, CA, USA). Statistical evaluation between groups was performed by two-way ANOVA, followed by multiple comparison test. Values of p < 0.05 were considered statistically significant.
Results
To confirm doxorubicin toxicity in NSG™,5,13 the mice were treated with 2 mg/kg doxorubicin twice in a week, but the mice showed intolerance to doxorubicin treatments as the mice were severely ill with significant weight loss of more than 20% within 9 days after receiving three doses of doxorubicin (Supplementary Figure 1). Therefore, this study focuses on the response to Doxil as an alternative formulation of doxorubicin, which is known to have a lower toxicity profile compared to conventional doxorubicin, 10 was used in NSG™ mice. The mice were treated with saline, 1 mg/kg or 2 mg/kg Doxil twice a week, for 3 weeks. The exposure of Doxil at 2 mg/kg (three doses in total) in mice also resulted in a substantial weight loss of 14% within 10 days, and this was coupled with signs of distress including hunched back and ruffled fur (Figure 1). As a result, this treatment group was terminated earlier than the anticipated 3-week study even though the body weight loss was improved compared to doxorubicin treatment. In contrast, 1 mg/kg Doxil-treated mice displayed minimal weight loss of around 5% with no signs of distress compared to approximately 4% of weight gain in saline-treated mice, thus indicating NSG™ mice experienced minimal acute toxicity with 1 mg/kg Doxil treatments, and a lowered Doxil dose must be used in NSG™ mice to allow three repeated cycles of Doxil treatments.
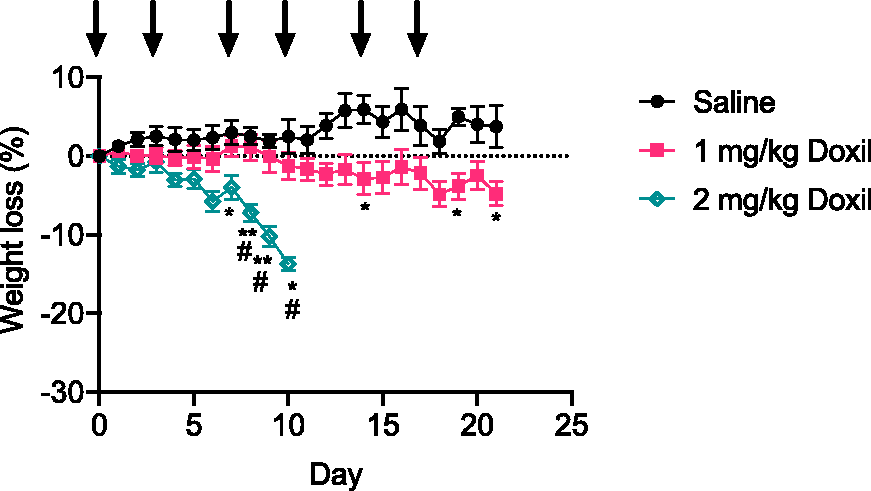
Body weight loss of NSG™ mice in response to saline and 1 mg/kg or 2 mg/kg Doxil treatments. Arrows represent the day in which the mice received drug treatments (n = three mice/treatment). Data are presented as mean and error bars represent standard error of mean. Significance compared to saline: *p < 0.05; **p < 0.01. Significance compared to 1 mg/kg Doxil: #p < 0.05).
To examine the potential cause of toxicity observed in 2 mg/kg Doxil treated NSG™ mice, gross examination showed that all three mice in this treatment group had developed dilated stomachs, filled with air (Supplementary Figure 2). Pathological assessment showed that these mice developed moderate to severe non-glandular gastritis as indicated by mucosal hyperplasia, hyperkeratosis, cellular atypia with karyomegaly and cellular pleomorphism (Figure 2; Table 1). The non-glandular stomach showed severe, diffuse, submucosal oedema with multifocal dense infiltrations of neutrophils. The non-glandular stomach epithelial lining was attenuated in areas, eroded in others and there was marked cellular atypia in the remnant mucosa. The epithelium was also severely hyperkeratotic, and neutrophils were present in the layers of keratin (Supplementary Figure 3). Doxil treatment at 2 mg/kg also caused moderate to severe cellular atypia of the oesophagus, indicating oesophagitis in two of the three mice (Figure 2; Table 1).
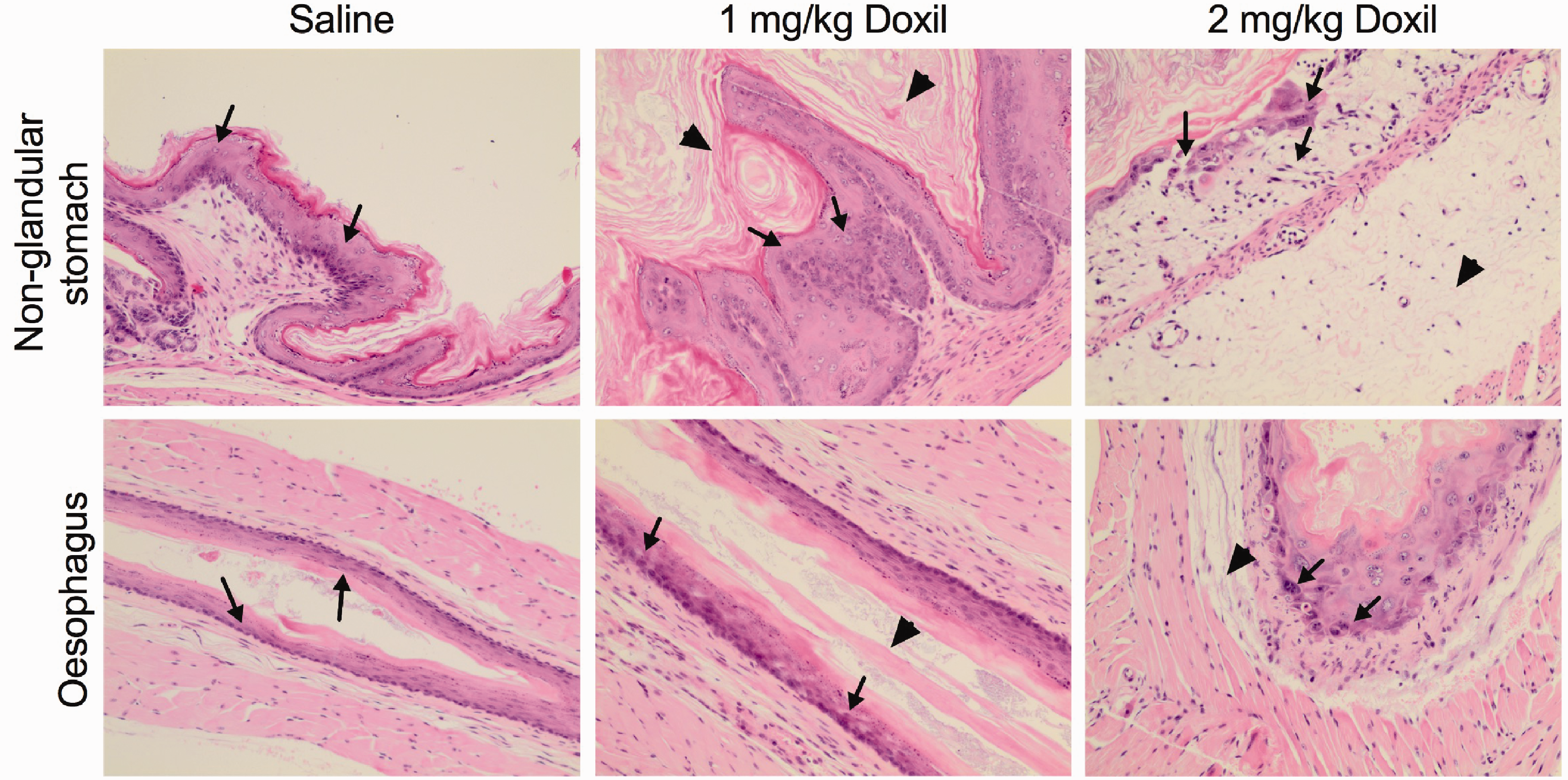
Histopathology changes in non-glandular stomach and oesophagus of NSG™ mice after receiving Doxil chemotherapy treatments (three mice/group). The tissue sections were stained with haematoxylin and eosin, original objective 10×. Non-glandular stomach, saline: normal epithelial cells, mucosa (arrow); 1 mg/kg Doxil: hyperplasia and epithelial atypia, mucosa (arrow), hyperkeratosis (arrowhead); 2 mg/kg Doxil: epithelial atypia, erosion and oedema, mucosa (arrow), oedema, submucosa (arrowhead). Oesophagus, saline: normal epithelial cells, mucosa (arrow); 1 mg/kg Doxil: epithelial atypia, mucosa (arrow) and hyperkeratosis (arrowhead); 2 mg/kg Doxil: epithelial atypia, mucosa (arrow) and oedema, submucosa (arrowhead).
Severity of histopathological changes in non-glandular stomach and oesophagus. The presence of Helicobacter spp was examined in non-glandular stomach.
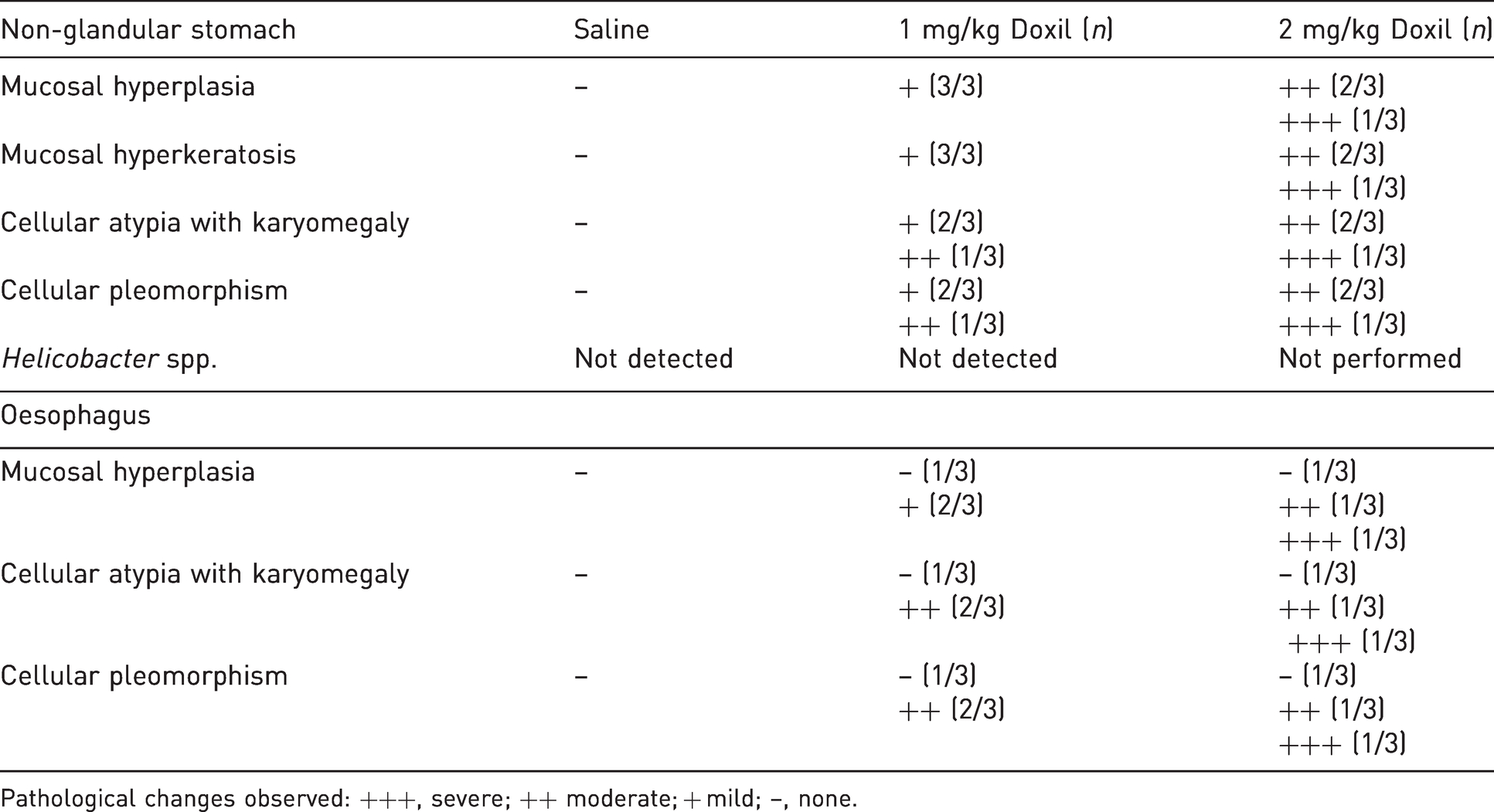
Pathological changes observed: +++, severe; ++ moderate; + mild; –, none.
Since histopathologic changes induced by 2 mg/kg Doxil were selectively observed in the non-glandular stomach and oesophagus, these two organs were selected for examination in the mice treated with Doxil at 1 mg/kg. In this group of mice, the scoring for pathologic changes in the oesophagus was milder compared to that observed in mice treated with the higher dose of Doxil at 2 mg/kg (Figure 2; Table 1). Exposure to the low dose Doxil at 1 mg/kg resulted in mild non-glandular gastritis with the presence of mild hyperkeratosis, hyperplasia and epithelial atypia when assessed histologically (Figure 2; Table 1). The histology examination revealed that the oesophagus and non-glandular stomach of saline-treated mice contained normal mucosal epithelial cells indicating that the pathologic changes were observed only in Doxil-treated mice.
Gram staining was performed to examine the cause of non-glandular stomach gastritis observed in response to Doxil treatment. There was a mixture of both large gram-positive and gram-negative bacilli present in the non-glandular stomach of the mice treated with 2 mg/kg Doxil, while the mice from the 1 mg/kg Doxil group predominantly contained gram-positive coccobacilli and bacilli (Figure 3). PCR was also performed to test for the presence of Helicobacter spp. in the mouse faeces collected from saline- and 1 mg/kg Doxil-treated groups, and the PCR results showed that Helicobacter spp. was not detected in both groups.
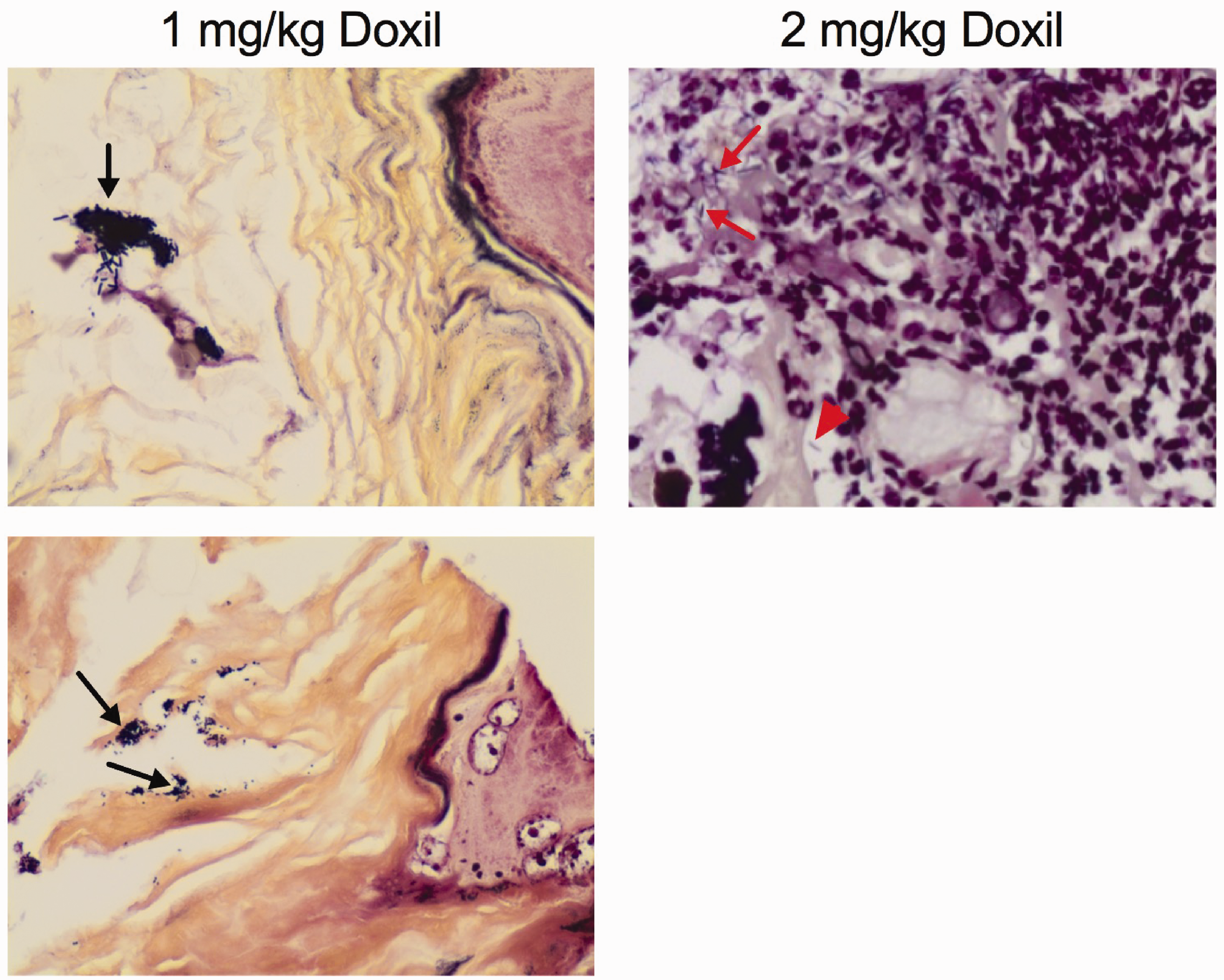
Bacterial gram staining of the non-glandular stomach of NSG™ mice after receiving Doxil treatments, original objective 40× (three mice/group). 1 mg/kg Doxil: (top) gram-positive bacilli (arrow), gastric lumen. (bottom) gram-positive coccobacilli (arrow), keratin and gastric lumen; 2 mg/kg Doxil (right): gram-positive bacilli (arrow) and gram-negative bacilli (arrowhead).
Despite the toxicity observed in the non-glandular stomach and oesophagus, the glandular stomach and the remaining intestinal tracts appeared normal for all Doxil-treated groups at different doses. In contrast, the saline-treated mice were healthy and displayed a normal epithelium in both the oesophagus and non-glandular stomach. These results indicate that Doxil treatment causes non-glandular gastritis and oesophagitis in NSG™ mice.
Discussion
NSG™ mice are highly immunocompromised and as such are widely used to perform high efficiency xenotransplantation of patient tumours for oncology studies. However, the usefulness of these models to assess the efficacies of anticancer drugs is limited by hypersensitivities of NSG™ to DNA damaging drugs such as doxorubicin. Female NSG™ mice were utilised in our study as they are the required host for xenotransplantation of human breast tumours, where doxorubicin treatment could be found to be beneficial for individual patients. 14 We have now established an optimal chemotherapy dosing strategy that is clinically relevant for the use of Doxil in NSG™ mice. Utilising Doxil at a reduced dose (1 mg/kg) as an alternative to doxorubicin showed reduced chemotherapy-related toxicities and enabled repeated cycles of chemotherapy. Doxil is the first approved liposomal nanoformulation for cancer treatment. 15 The added advantage of using Doxil is that drug accumulates passively in tumours by means of the enhanced permeability and retention effect. 15 Thus, Doxil treatment of NSG™ mice bearing PDX tumours would be expected to result in a higher tumour drug accumulation compared to doxorubicin, enabling drug efficacy to be evaluated in these models.
The main toxicities reported in NSG™ mice after receiving doxorubicin treatment (such as cardiotoxicity, hepatotoxicity and intestine inflammation) 5 were not observed in Doxil treatments, even though NSG™ mice are highly susceptible to doxorubicin-related damage. 13 There are a few factors that may contribute to this observation. Firstly, Doxil is mainly delivered through intravenous injection while doxorubicin is commonly administered through intraperitoneal injection in mice. This mode of delivery may reduce the localised impact of doxorubicin-induced necrosis towards organs close to peritoneal areas, such as the intestinal abnormalities and retention of peritoneal fluid. 5 Secondly, encapsulating doxorubicin in pegylated liposomes generates a more favourable pharmacokinetic profile with reduced toxicities.16,17 The liposomal formulation essentially sequesters free doxorubicin and prevents doxorubicin escape into organs that have vasculature with tightly joined capillary junctions (such as hearts). 16
The main Doxil-related toxicities exhibited by NSG™ mice are dose-dependent non-glandular gastritis and oesophagitis as increasing the dose of Doxil increases the severity of these clinical signs. Although NSG™ mice are highly immunocompromised and susceptible to bacterial infections, the signs of non-glandular gastritis and oesophagitis are specific to Doxil treatments as these are not observed in the saline control mice that are housed and handled under the same conditions. Macro-pathology examination of NSG™ mice treated with Doxil showed that the stomach was empty. This may be due to a loss of appetite as a result of oesophagitis and gastritis, thus contributing to the significant weight loss. This can be further validated by measuring food intake in future studies. Clinically, the major adverse reactions of Doxil in humans are predominantly mucocutaneous eruptions, while adverse reactions showing gastritis and oesophagitis are not common. 18 This is likely due to the anatomical differences between humans and rodents where humans lack a non-glandular forestomach. 19 However, there is a rare case study on a multiple myeloma patient who received a single course of Doxil and immediately developed serious oesophagitis and chronic stomach inflammation. 18 This is consistent with our observations in NSG™ mice, which are likely due to the damage induced by Doxil to the mucous membranes in the upper gastrointestinal tract. The pathogenesis of non-glandular gastritis induced by Doxil is unknown and requires further investigation. It is possible that it is a combined effect of the chemotherapy drug, Doxil as well as the hypersensitivity and immunosuppressive background of this particular mouse strain, therefore promoting the inflammation and colonisation of bacteria in the non-glandular stomach.
In summary, we have determined an optimum treatment regime of Doxil that can now be employed in future studies utilising NSG™ mice to assess the efficacy of doxorubicin-based treatments in PDX models. We have also demonstrated that Doxil induces a dose-dependent non-glandular gastritis and oesophagitis in NSG™ mice. NSG™ mice have an enormous potential for high efficiency engraftment with multiple different types of cancer derived from patients to enable pre-clinical translational research and to ultimately assist the development of personalised cancer therapy.
Supplemental Material
sj-pdf-1-lan-10.1177_00236772211009338 - Supplemental material for Observation of non-glandular gastritis associated with Doxil chemotherapy treatment in NSG™ mice
Supplemental material, sj-pdf-1-lan-10.1177_00236772211009338 for Observation of non-glandular gastritis associated with Doxil chemotherapy treatment in NSG™ mice by Alison Cheong, Lorna Rasmussen, Tina Robinson, Ruqaya Maliki and Suzanne M Cutts in Laboratory Animals
Supplemental Material
sj-zip-2-lan-10.1177_00236772211009338 - Supplemental material for Observation of non-glandular gastritis associated with Doxil chemotherapy treatment in NSG™ mice
Supplemental material, sj-zip-2-lan-10.1177_00236772211009338 for Observation of non-glandular gastritis associated with Doxil chemotherapy treatment in NSG™ mice by Alison Cheong, Lorna Rasmussen, Tina Robinson, Ruqaya Maliki and Suzanne M Cutts in Laboratory Animals
Footnotes
Acknowledgement
The authors acknowledge the contribution from La Trobe Animal Research and Teaching Facilities.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Breast Cancer Foundation [IIRS-18-026].
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
