Abstract
Metabolism cages are designed to conduct absorption, distribution, metabolism and excretion (ADME) studies, enabling an ‘excretion balance’ scientific objective to be met. Historically, the design of dog metabolism cages has involved single housing. This type of housing has limitations for normal social behaviours and has been largely unchanged for 25–30 years. Improving animal welfare is a focus area for the authorities as well as the industry throughout the European Union. A collaboration was developed between Novo Nordisk and Covance to enhance the design of metabolism cages, allowing dogs to be pair housed. The purpose of the study was to compare excretion balance data from pair-housed and singly housed dogs in order to demonstrate that conducting excretion balance studies with a pair-housing design improves animal welfare without compromising the scientific integrity of the study. A radiolabelled test compound, [14C]-Quetiapine, was selected for this investigation based on its excretion profile. The assessment of the dogs’ stress levels was investigated by measuring the levels of serum cortisol as an indicative biomarker. Results were inconclusive due to large variations in cortisol levels. However, dogs appeared calmer in the pair-housing setting. The overall mean recovery (±standard deviation) for pair-housed animals (94.0 ± 0.66% of the dose) was equivalent to that from singly housed dogs (93.0 ± 2.29%). Based on these data, we conclude that pair housing of dogs for future metabolism ADME studies does not compromise the scientific integrity, and therefore is a major progression in the design of these studies, enhancing welfare.
Introduction
The absorption, distribution, metabolism and excretion (ADME) properties of small- and large-molecule pharmaceuticals are primarily assessed using in vivo studies. These studies are conducted to provide information on test substance and metabolite kinetics, including the rates and routes of elimination, 1 and are required in regulatory submissions of new pharmaceuticals. 2 The animals used in ADME studies are based on the choice of species used in the long-term safety evaluation of a new pharmaceutical, and those most commonly used are the rat, as the model for rodent, and the dog, the minipig, 3 and the non-human primate, 4 as models for non-rodents, although the non-human primate is only used when there is no suitable alternative. Currently available metabolism cages for the purposes of conducting non-rodent ADME studies involve single housing of dogs and have been largely unchanged for 25–30 years.
Dogs are known to form social groups and strong relations to conspecifics and humans easily, and are therefore highly social animals.5,6 Studies have shown that single housing of beagles has a negative influence on their behaviour, leading to bizarre movement, decreased sleep duration and increased vocalisation rates. 7 Similar behaviours have also been observed in singly housed kennel dogs. 8 Preventing dogs from having social contact causes distress in most dogs and can lead to separation anxiety.9,10 EU Directive 2010/63/EU recognises that single housing of dogs is stressful and stipulates that dogs should not be single housed for more than four hours 11 unless for valid scientific reasons and with the permission of the local competent authority. EU Directive 2010/63/EU also include a classification of the severity of procedures involving animals. It suggests that a procedure where animals are single housed in a metabolism cage for more than five days with severe restriction of movement over a prolonged period should be classified as a severe procedure. 11
Metabolism cages are typically constructed from brushed stainless steel with a slatted floor to enable surfaces to be easily wiped down in order to ensure the complete collection of excreta voided during the confinement of the animal. Cages normally contain limited environmental enrichment due to the concerns over absorption of test-related material to this enrichment during the experiment. The length of the housing period in a metabolism cage is usually five to seven days. 12 However, it depends on the ADME of the compound under investigation. Although tactile interactions between animals in an ADME study are removed, auditory, visual and olfactory stimuli between animals are maintained, with a cohort of animals on test simultaneously and within the same room.
Today, there is an enhanced awareness of the importance of animal welfare within the life-sciences industry and initiatives to explore and introduce new measures which lead to benefits for animals, 13 without compromising the scientific outcome and, at best, even improving the quality of scientific data.14–16 This enhanced awareness of animal welfare and the benefit to scientific output helps the ethical review process not only to challenge new procedures but also to improve existing procedures in the spirit of the application of the 3Rs (replacement, refinement and reduction) principles. Therefore, we set out to refine and challenge the single-house paradigm in ADME studies by redesigning current metabolism cages, allowing dogs to be pair housed.
The objective of this study was to compare the excretion balance data from pair-housed and singly housed dogs, following intravenous single-dose administration of [ 14 C]-Quetiapine. Data from pair-housed dogs were evaluated to determine whether they were sufficiently robust to meet the scientific – and therefore meet expected regulatory – standards for data from a study of this type. An additional objective was to determine and compare the pharmacokinetics of radioactivity following intravenous administration of [ 14 C]-Quetiapine in either singly housed or pair-housed animals, demonstrating that the PK from singly and dual-housed animals were equivalent. To support the hypothesis that pair housing reduced the stress of the dogs, the levels of cortisol in sera were measured during the study in both singly house and pair-housed dogs.
Methods
Animals and ethical statement
Animal procedures were conducted in an Association for Assessment and Accreditation of Laboratory Animal Care International accredited facility in accordance with the UK Home Office Animals (Scientific Procedures) Act 1986 and EU Directive 2010/63/EU, with ethical approval from Covance Local Ethical Review Body and Novo Nordisk ethical review council.
Six pure-bred male beagle dogs were obtained from a UK Home Office approved source (Envigo). The six dogs ranged in weight from 6.77 kg to 8.17 kg and were approximately six months of age at the start of dosing. Dogs were acclimatised for a minimum period of three weeks during which time they were incrementally habituated to metabolism cages for periods of up to a maximum of four hours per day. Each dog was allocated to a pen with a floor area of approximately 2.25 m2. Routinely, dogs of the same sex and group were housed in groups of two or three by removal of the barrier(s) dividing the pens. To enrich the environment and welfare of the animals, provision of ‘toys’ (balls, inert nylon chews etc.), raised platforms, bedding and exercise/socialisation periods were included throughout the study. Dogs were allowed free access to water ad libitum and approximately 300 g of a high-density canine diet (5L66 Diet, LabDiet) daily; during feeding periods, dogs were separated. A veterinary inspection was performed before the start of dosing to ensure their suitability for the study. The animals were kept in rooms thermostatically maintained at a temperature of 15–21℃ and exposed to fluorescent light (nominal 12 hours) each day.
Group details.
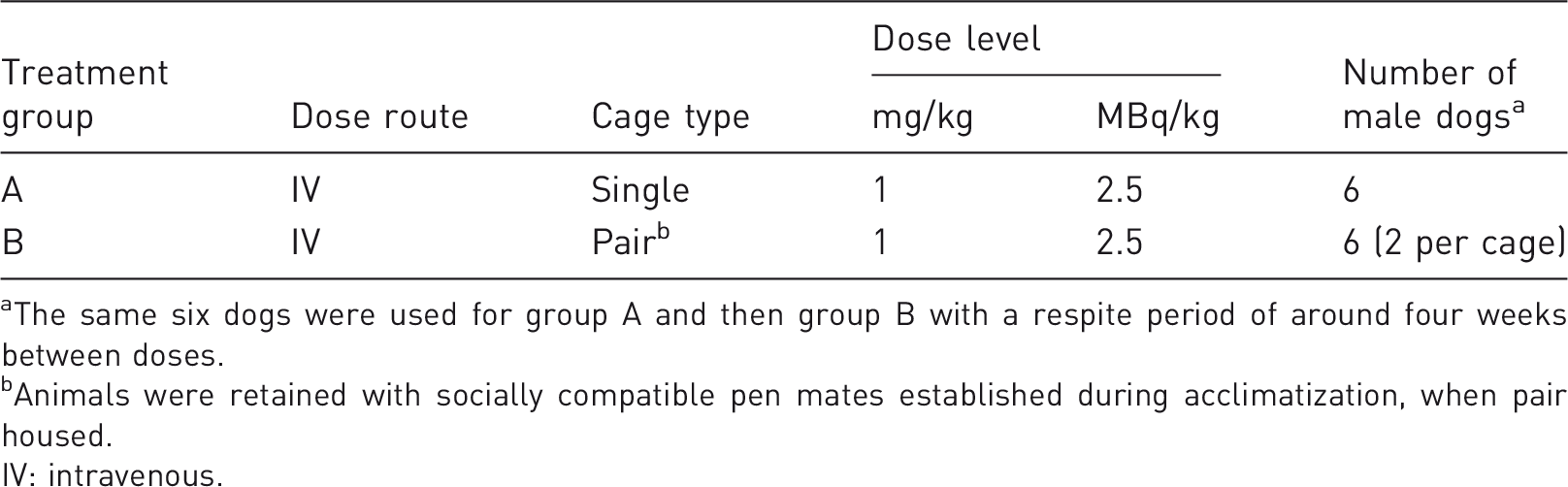
The same six dogs were used for group A and then group B with a respite period of around four weeks between doses.
Animals were retained with socially compatible pen mates established during acclimatization, when pair housed.
IV: intravenous.
Justification for number of animals and single sex
Direct comparisons of recovery data from both singly housed and dual-housed dogs are required to investigate the scientific integrity of data generated from the dual-housed animals. The study was designed to compare the recovered radioactivity in excreta from singly housed dogs (n = 6) and pair-housed dogs (n = 3 × 2) following administration of [ 14 C]-Quetiapine. Only males were chosen, as the vast majority of ADME investigations involve the use of a single sex (sex differences are not expected in the second species). The number of selected animals in each group was considered sufficient for demonstrating equivalence within ±10% recovery at the statistical power of 96%, based on a statistical evaluation of historical data (t-test with 95% confidence interval). Animals were randomly allocated a study number.
Welfare considerations of cage design
In general, the metabolism cages used in academic research and pharmaceutical development are designed with dimensions and materials suitable for optimal recovery of urine and faeces. The cage dimensions for single housing of animals used in these investigations were 1.0 m × 0.6 m × 1.0 m (w × h × d) and contained a roll bar style open flooring to allow the recovery of urine and faeces below the cage unit, and an internal shelf to provide a different viewing height for the animals. The viewing was typically out of the front of the cage, through the meshed enclosure surrounding the water and food hoppers.
Traditional dog metabolism cage for single housing without any tactile contact between dogs.
The metabolism cage was redesigned to improve the dogs’ welfare by combining two metabolism cages, including an adjustable removable shelf, replacement of the stainless-steel front side panel with a clear Perspex panel and the introduction of a sliding hatch incorporated into the side of each cage to allow pair housing.
Dosing and sample collection
[ 14 C]-Quetiapine was administered at a nominal dose level of 1 mg/kg as a bolus intravenous injection in physiological saline at a dose volume of 1 mL/kg. Each animal received two intravenous doses: one in group A and one in group B. Blood samples (nominally 2 mL) in K2EDTA tubes were collected from a suitable vein at the following intervals: pre dose (–24 hours) and then at 5 minutes, 15 minutes, 30 minutes and 1, 2, 4, 8, 12, 24, 48, 72 and 96 hours post dose. An additional 1 mL blood was also collected into serum tubes at pre-dose and at 0.5, 24, 48, 72 and 96 hours post dose.
Concentrations of radioactivity in blood and plasma were determined by liquid scintillation counting at all time points. Cortisol concentrations were also determined in serum samples on a daily basis for groups A and B. Urine was collected pre dose and then at 0–12 hours, 12–24 hours and daily for up to 168 hours. Faeces were collected at 24-hour intervals up to 168 hours post dose, and the radioactivity was quantified.
For singly housed animals, excreta were collected and analysed separately for each animal, whilst for pair-housed animals (Figure 2), excreta from both halves of the cage were pooled at each collection (faeces) or after analysis for urine and cage wash due to the large volumes of these samples. Animals were separated during blood collection, sample collection during cage cleaning and during feeding. This was a period of approximately one to two hours in total for every 24-hour period.
Schematic/photo representation of the dual cage system with a hatch between the two connected cages with pair-housed dogs.
Sample preparation
Volumes and/or weights of biological samples were measured. Faeces and cage debris were homogenised in an appropriate volume of deionised water. Swabs taken from the cage interior following the removal of urine and faeces were soaked in a suitable solvent. Aliquots of the solvent were added to liquid scintillant prior to liquid scintillation counting (LSC). Portions of test substance solutions and formulations (suitably diluted), plasma, urine and cage washings were added directly to liquid scintillant prior to LSC. All remaining samples were analysed after solubilisation or combustion analysis.
Combustion analysis
Faeces samples submitted to combustion analysis were combusted using a Packard Sample Oxidiser. The combusted products were absorbed in Carbosorb® (PerkinElmer LAS (UK) Ltd) and mixed with Permafluor E+ scintillation fluid (PerkinElmer LAS (UK) Ltd) before being analysed by LSC.
Solubilisation
A suitable volume of solubilising agent (e.g. Solvable™; PerkinElmer LAS (UK) Ltd) was added to the blood and cage debris samples. After an appropriate period of incubation, liquid scintillant was added, and the samples allowed to adapt to the dark prior to LSC.
Liquid scintillation counting
A suitable scintillation counter was used. Radioassays were performed at least in duplicate. Efficiency correlation curves were prepared and routinely checked by the use of [ 14 C]-toluene and Ultima Gold™ quenched standards (PerkinElmer LAS (UK) Ltd). The limit of quantification for each batch of samples analysed by direct counting was taken as twice the mean background disintegration rate obtained from vials containing an equivalent volume of an appropriate solvent, if applicable, in liquid scintillant. The limit of quantification of each batch of samples analysed by combustion was taken as twice the mean background disintegration rate obtained when Combusto-Cones™ containing Combusto-Pads were combusted.
Data collection and analysis
Liquid scintillation counts and weighing data were either entered manually or captured online using a validated data acquisition system (Debra v6.1.1.87, supplied by LabLogic Systems Ltd). The pharmacokinetic analysis of the total radioactivity in blood and plasma for [ 14 C]-Quetiapine hemifumarate were performed using Phoenix WinNonlin v6.4.
Statistical methods
Means and standard deviations (SD) were calculated. Mean recovery data from groups A and B and cortisol data were evaluated using unpaired t-tests.
Results
There were no adverse clinical signs observed in any animals following any of the procedures conducted in these investigations. A comparison of the recoveries of radioactivity in the singly housed and pair-housed animals following intravenous administration of [
14
C]-Quetiapine are presented in Table 2 and Figure 3.
Comparison of recovery of radioactivity in singly housed (group A) and pair-housed (group B) animals. Excretion balance investigation. Unpaired t-test confirmed no statistical significance between mean values (p > 0.1). SD: standard deviation.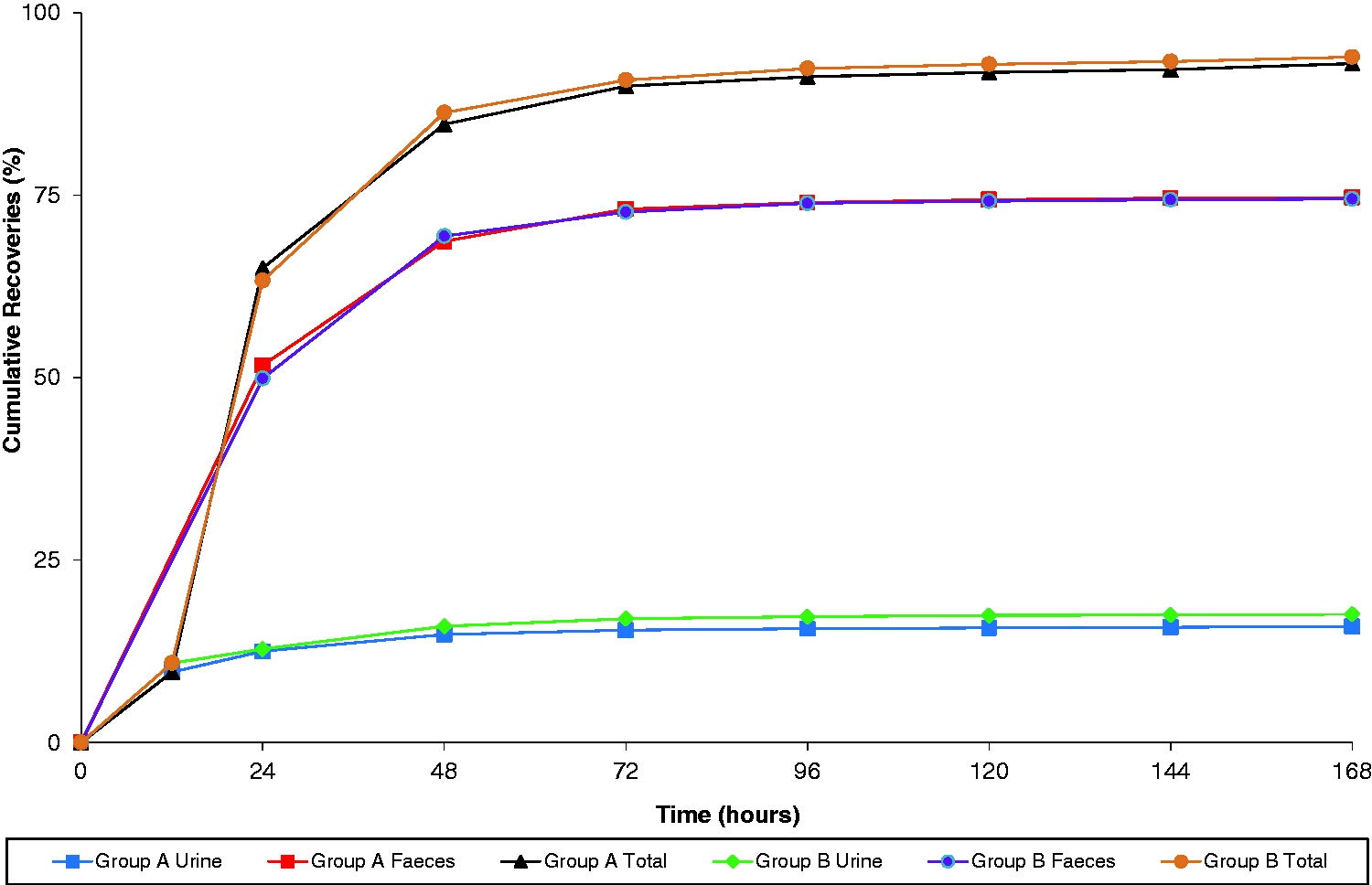
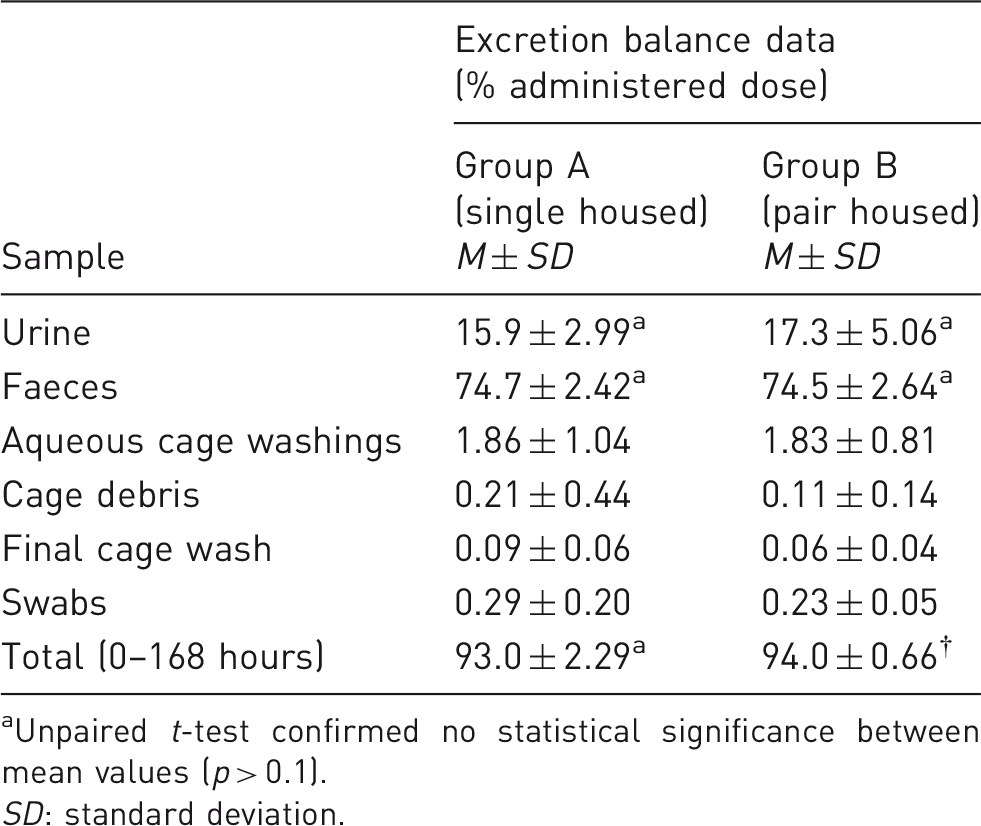
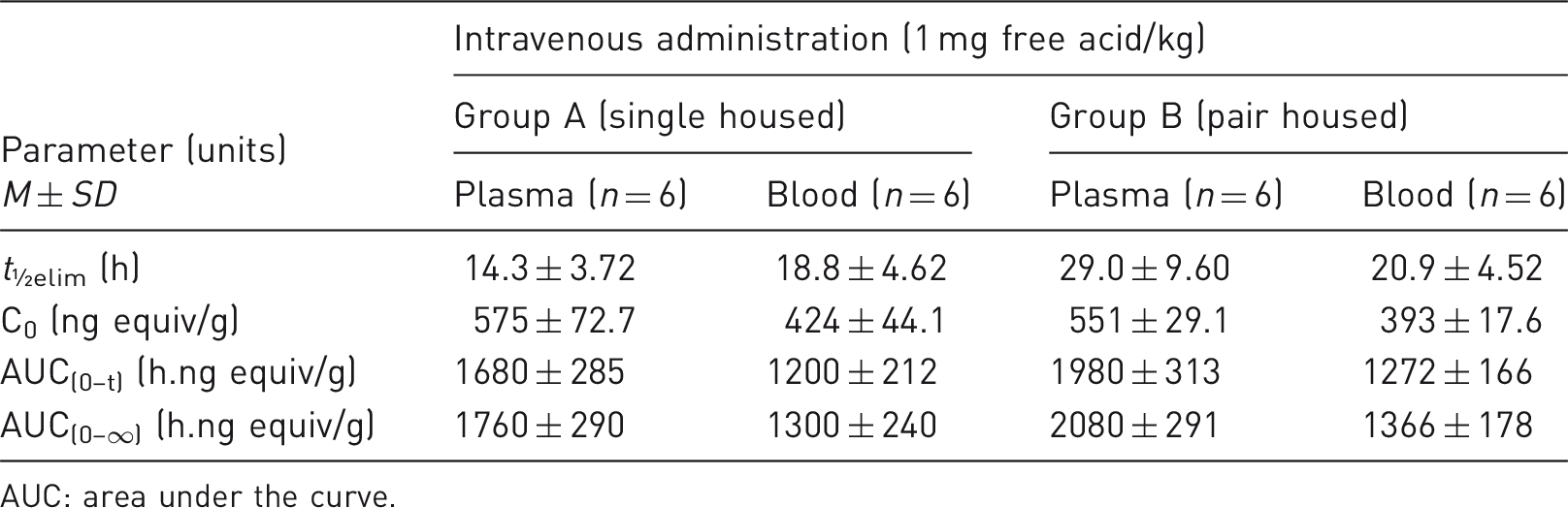
The overall mean recovery for pair-housed animals (94.0 ± 0.66% of the dose) was within the variability range of data from singly housed dogs (93.0 ± 2.29%), and therefore equivalent to the reference group. Faecal excretion represented the principal route of elimination (around 72–77% dose for both groups), whilst around 13–20% was eliminated by renal routes. The majority of radioactivity was eliminated within the first 48 hours. Low levels of radioactivity were still detected in urine and faeces at the final sampling time point (≤0.1%).
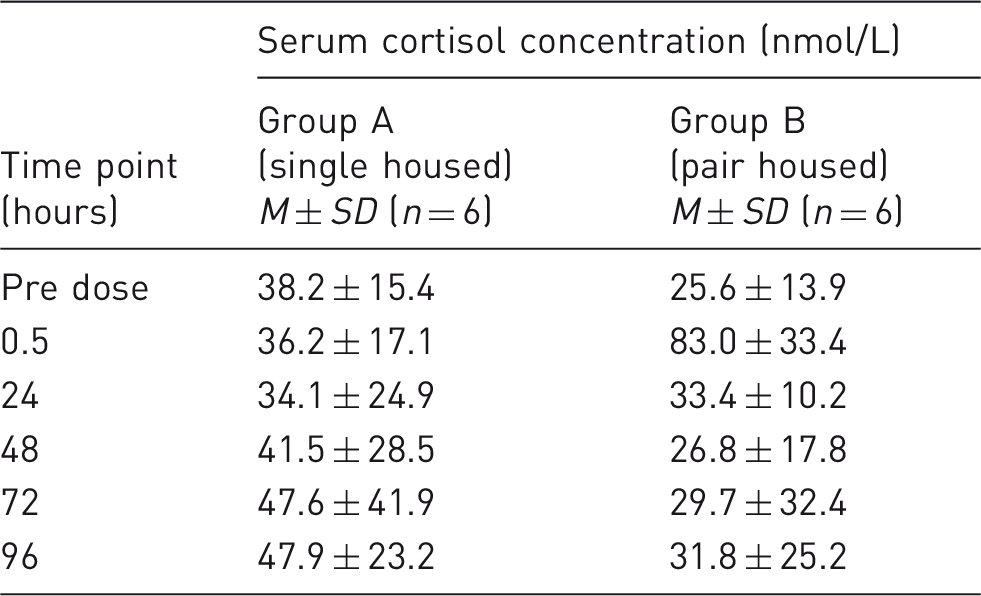
Serum cortisol concentrations.
Plasma and blood pharmacokinetic parameters
A summary of the pharmacokinetic parameters is shown in Table 4 and Figure 4. There were no notable differences in the blood radioactivity exposure, evaluated by area under the curve (AUC) assessments, and the concentration versus time profiles were comparable between treatment groups over the first 48 hours. The plasma concentrations at the last time point (96 hours) were below the limit in one group, which resulted in slightly different terminal elimination half-lives. Mean AUC values were similar, irrespective of housing.
Mean plasma concentrations of radioactivity in singly housed male dogs (group A) and pair-housed dogs (group B). Plasma and blood pharmacokinetic parameters. AUC: area under the curve.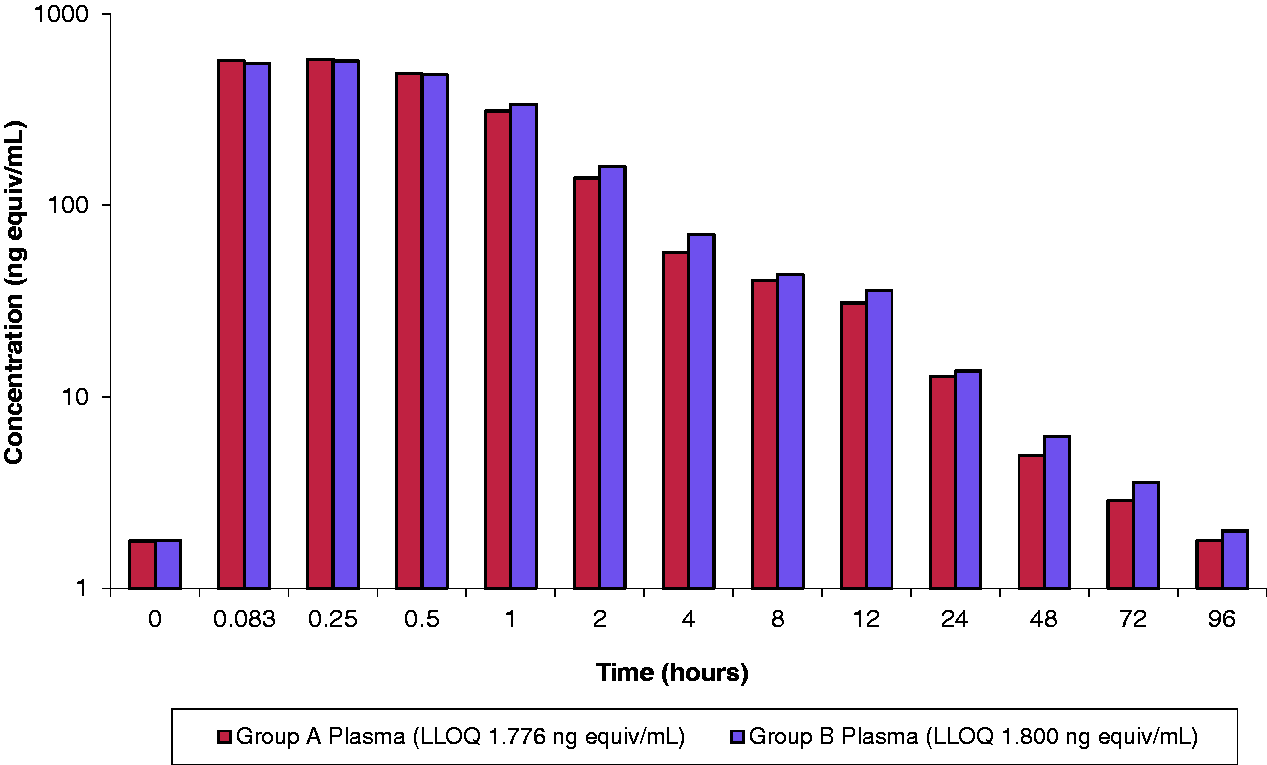
Discussion
The improvement of animal welfare is of paramount importance at both Novo Nordisk A/S and Covance Laboratories Ltd, and therefore in a joint collaboration, a metabolism cage for pair housing of dogs was designed to generate data for the assessment of an excretion balance, while refining the procedure to improve the welfare impact on the dogs.
Dimensions
One of the major challenges for co-location of multiple animals was the need to increase the cage dimensions substantially. One option was to redesign and manufacture a new cage with dimensions that were compliant with current EU Directive 2010/63/EU. However, this would require the production of a completely new stainless-steel cage by a professional cage manufacturer. The overriding consideration was how the licensed animal technicians and associated staff would be able to gain access to the animal effectively for timed blood sampling, or how they would be able to access the entire cage physically sufficiently well to collect samples and clean the cage at each timed interval, with a significantly larger surface area. This led to a more cost-effective solution of proposing the joining of two cages of similar dimensions together and allowing access for two animals to each ‘half’ of the new double-sized unit (Figure 2).
Animal number
In terms of number of animals that are selected for use in an excretion balance study, this should always be commensurate with the fewest number required to achieve the scientific objectives of the study. In general, for ADME studies, no formal statistical analyses of the data are performed, since the objectives are to investigate and understand the pharmacokinetics, explore tissue distribution, investigate the rates and routes of elimination and provide samples for further investigation of the metabolite identities. Historically, three animals (per sex as appropriate) have been selected for a study of this nature in order to generate sufficient data for the initial evaluation of the test compound absorption and pharmacokinetics, and how it is metabolised and excreted. These data are usually reported as mean values in regulatory submission documents, with limited statistical interpretation due to the small sample size.
All six animals were subjected to group A procedures (single housing) before being subjected to group B procedures (pair housing) following the four-week wash-out period. In terms of the aims of the study in comparing ADME properties following single or pair housing, these would not be impacted by the dosing sequence. However, the effect and timing of procedures on biomarkers of stress could potentially have an impact, since acclimatisation or sensitisation to procedures could occur.
The revised study design, from single to pair housing, does reduce the number of samples available for analysis at each time point for urine and faeces (from six to three) due to pooling. However, pharmacokinetic profiles remain available from all six animals, irrespective of single or pair housing. This allows a holistic review of the data from the study, enabling the investigator to explore any atypical absorption of elimination profiles of radioactivity from the systemic circulation and to apply any observations to the scientific assessment of the excretion balance recoveries and subsequent metabolite profiling assessments conducted as part of ADME studies.
Food sharing
During the initial evaluation, it became readily apparent that even when housed in pairs, a social hierarchy could affect the food consumption of the animals in a pair. The inclusion of a period of up to four hours per day in the study design when animals could be separated to allow for relaxed feeding was considered an appropriate compromise to ensure each animal obtained a fair share of the food on offer. However, provided animals were fed together from arrival, it was possible to ensure that compatible animals were housed together and thereby enable animals to be fed together, thus preventing separation for feeding.
Blood sampling
During experimental procedures, such as blood sampling, the flexibility of the dual caging enabled animals to be isolated during this time if required in order to allow for a calm procedure for both dog and animal technician, restricting the animals from running freely between cages.
Social housing and sample collection
The use of a dual-caging system has the added benefit that animals can be moved into one side while the other side is being cleaned and urine and faeces collected. This removed the possibility that animals could come into contact with any cleaning liquids or interfere with the technician collecting the samples.
Conventional wisdom would dictate that providing social interactions by pair housing of animals that are naturally social, rather than singly housing, would improve the welfare of the animals utilised. Hormonal biomarkers of stress in animals range from markers in serum, saliva, hair or faeces, such as cortisol, to physical signs that are symptomatic, such as facial expression, atypical behaviour and vocalisation.17–19
In this study, serum cortisol concentrations were assessed from samples taken for the pharmacokinetic investigations as a useful biomarker for stress, 20 although it is clear from these data that additional measurements and observations would have added to the value of these direct evaluations. It is thought that the highest mean serum concentration of cortisol, observed in the pair-housed animals at 0.5 hours, could have been attributed to a number of factors, for example the presence of extra numbers of people observing the dosing procedure in pair cages or memory of the first dosing occasions. A lower mean serum cortisol was observed for pair-housed dogs from 48 hours onwards. However, due to a large variation in serum cortisol, this difference should be treated with caution. From an observational perspective, animals appeared calmer, with less barking when pair housed.
Many of the aspects of cage design discussed in terms of the scientific, regulatory and logistical aspects also overlap with those that have a welfare-related component. For instance, the cage dimensions or the methods for conducting feeding or sampling all affect the welfare of the animals, and a balance needs to be struck between meeting the scientific objectives of the study, the logistical ease and reproducibility of conducting the investigation and the experiences of the animals as the test system.
The cage roll-bar flooring was one area that invoked a considerable amount of discussion. As noted above, the use of roll-bar flooring allows for the separate collection of urine and faeces voided from the animals. However, there is a possibility that animals would find this uncomfortable and unpleasant, or even that they could potentially injure themselves if slipping off the bars. To address this concern, the use of a tenderfoot grid floor (Figure 5) with holes of around an inch in diameter could be introduced to explore whether animals that were pair housed favoured one flooring surface over the other. Data from preliminary investigations (data not reported) demonstrated that the overall recovery of radioactivity (90.9 ± 2.46%) was slightly lower but generally comparable to the singly housed or dual-housed results. From a technical perspective, the tenderfoot flooring led to increased collection times and was practically difficult for technicians to clean and ensure that all of the faecal sample had been collected.
Tenderfoot floor in the dual dog metabolism cage.
Pertinent to this discussion is the single housing of dogs and, in addition to this, for a period of more than five days, which is classified as a severe scientific procedure in the Directive. 11 However, within the UK, this is conventionally classified as a moderate procedure where no tethering or social isolation are involved. Nonetheless, the ideal would be to reduce the experience of any animals used in scientific procedures to one which was mild or below the threshold of classification.
Concluding remarks
The total mean recoveries of radioactivity in excreta, following dosing of a representative radiolabelled test substance, were equivalent and within the variability range, irrespective of housing of the dogs, supporting that pair housing of dogs for future metabolism ADME studies can generate data for regulatory submission.
It is imperative that ADME studies are carried out in metabolism cages only when there is not an alternative and as a necessary means of answering the scientific questions posed. Where triggered, efforts can be made to decrease the numbers of animals that must be used through consideration of study design in respect to sex and meeting the scientific objectives. Through review of the considerations described here, there is supporting evidence and precedent for the conduct of excretion balance studies in dogs utilising dual-housed rather than singly housed animals.
The cortisol concentrations (used as a stress indicator) were inconclusive. However, it is thought that the actual process of blood sampling from the animals (irrespective of housing) is the predominant factor in cortisol concentration elevation during this study.
The additional investigations using tenderfoot flooring (Figure 5) led to some possible faecal contamination of urine samples and lower although still acceptable recoveries of radioactivity. These factors together with the practical difficulties of collection of faeces should be taking into consideration before choosing the tenderfoot floor in mass balance studies.
Future
Discussions with regulatory authorities on the acceptability of the data from pair-housed animals compared to those of singly housed animals are ongoing. However, there is acknowledgement that welfare has a major impact on the scientific validity of study data, and therefore this improvement is widely viewed as an extremely positive development to obtaining the most robust scientific data from an excretion balance ADME study.
Studies utilising alternative isotopes, such as 3H, and those involving large molecules with longer half-lives and a wider range of new chemical entities may be conducted to provide further support for studies with group-housed animals. As the use of the dual housing in ADME studies gains popularity, it is expected that this database will grow to demonstrate further that the robustness of the data generated in dual housing is equivalent to that generated during single housing studies.
With reference to the scientific procedures carried out being refined causing less distress, there is a possibility that animals could be reused on similar studies, reducing the numbers of animals involved in this study type.
The continued welfare benefits should be investigated in further detail, and should include further observations on social interactions, animal behaviour and physiological biomarkers such as heart rate, electrocardiographs and blood pressure.
The possibility of triple housing for future studies is also a consideration, as this will enable the current n = 3 set of pharmacokinetic data to be obtained on a single-dosing occasion, using the minimum number of animals.
Footnotes
Acknowledgements
We would like to express our thanks to the cage manufacturer, Arrowmight, for the implementation of the cage modifications.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
