Abstract
Swine models had been popular in paediatric oesophageal surgery in the past. Although being largely replaced by rodent models, swine experienced a revival with the establishment of minipig models. However, none of them has ever been investigated for similarity to humans. We conducted a pilot study to determine whether three-week old Pietrain piglets and three-month old Aachen Minipigs are suitable for experimental paediatric oesophageal atresia surgery. We tested the operation's feasibility, performed a necropsy, weighed organs, measured organ length and calculated relative weights and lengths, and measured laboratory parameters. We used multidimensional scaling to assess the similarity of the swine breeds with previously published human data. Pietrain piglets had a higher a priori bodyweight than Aachen Minipigs (Δ = 1.31 kg, 95% confidence interval (CI): 0.37–2.23, p = 0.015), while snout-to-tail length was similar. Pietrain piglets had higher absolute and relative oesophageal lengths (Δ = 5.43 cm, 95% CI: 2.2–8.6; p = 0.0062,
Introduction
The core aspects for choosing a surgical animal model in studies are to achieve meaningful results by humanlike anatomy and physiology.1,2 Only an equivalently usable animal model for the interpretation of human data pursues the purpose of animal welfare of the 3Rs due to the minimization of aspects hampering transferability to humans. The suitability of swine for paediatric research has been pointed out earlier: on the first days of life, humans and swine have similar physiologic characteristics, which other species do not come close to. 1 The choice for a particular model organism is not exclusively based on similarity of anatomy and physiological parameters, but may also be influenced by aspects requiring genetically modified organisms, which would favour mice due to the short generation time and the well-established techniques of genetic modification. However, for translation of experimental results the researcher should be aware of the strengths and limitations of the respective experimental model. 2 Consequently, swine had been the favourite model of experimental paediatric oesophageal surgery. 3 It was used to pioneer 4 one of the first broadly used surgical procedures to treat long-gap forms of oesophageal atresia without oesophageal replacement: the Livaditis myotomy to reduce anastomotic tension. 5 Beyond physiologic similarities between new-born piglets and infants, it has even been shown that their oesophagus exhibits a similar reaction to traction compared to humans and thus seem to have similar tissue properties. 6 Therefore, the animal model ‘swine’ is particularly suitable for paediatric oesophageal surgery.
Despite these encouraging facts, swine have subsequently been neglected in paediatric oesophageal surgery, with the exception of robotic surgery, and largely been replaced by rodent models. 7 Rodents are cheaper than swine, easier to house and handle, antibodies, assays and genetically modified models are available. 8 In many studies in which swine models were used, the authors failed to report the exact subspecies9,10 or age and bodyweights. 11 The translation of experimental animal results to human studies and pathologies is thereby limited and may thus contribute towards irreproducible results. 12
Previous studies in experimental paediatric oesophageal surgery favoured piglets, 3 whereas minipig models were gaining ground recently.13,14 However, their anatomy and physiologic parameters have not been evaluated in respect for their similarity to humans. Since new minipig breeds like the Aachen Minipig 15 become available, we conducted a pilot study to determine the suitability of three-week old Pietrain piglets in comparison to three-month old Aachen Minipigs as a model for long-gap oesophageal atresia. We performed the planned surgery, conducted a necropsy, determined the weight of all suitable internal organs and analysed blood as well as urine samples for basic laboratory parameters. Using available organ weight data from humans in different developmental stages and postnatal ages, we conducted a similarity analysis to determine which breed comes closest to humans based on the respective anatomical characteristics, which are considered important factors in a surgical model.
Animals, materials and methods
Human disease to model: long-gap oesophageal atresia
Nationwide data from the French oesophageal atresia register found a mean birthweight of 2519 g (SD 699) in 275 affected children.
16
The most common operative approach for long-gap oesophageal atresia is a delayed primary anastomosis around eight weeks of age, because children will double their birthweight in that time paralleled by an overproportionate growth of oesophageal remnants.
17
This would result in a weight at the time of operation of 5038g for the nationwide French cohort. This estimated weight at surgery was chosen as the fixpoint for our animal models whose age was determined by this bodyweight based on available growth curves
18
and recommendations from their breeders from their experience when this bodyweight would typically be reached. We simulated a normally distributed population using the weights described above and bootstrapped a mean and its 95% confidence interval (CI) (
Animals
We conducted our experiments in accordance with the German Animal Welfare Act and its subsequent statutory acts, which is in accordance with the Council of Europe Convention ETS 123. The competent state agency approved the study to determine the adequate animal model for a larger study (permit number: G-17-1-033). We obtained five freshly weaned, two-week old Pietrain piglets of both sexes (Figure 1(a)) in conventional microbiological status from a local organic farmer, licenced by authority to supply swine for animal experiments to our institution. We bought five three-month old Aachen Minipigs of both sexes (Figure 1(b)) from an official breeder (Gerd Heinrichs, Heinsberg, Germany), whose microbiological status has been described in detail.
15
Based on the Monte-Carlo-simulations by Allgöwer and Mayer,
19
which demonstrated five animals per group to be sufficient for a pilot study to achieve a statistical power of at least 80%, we used five animals per group.
The investigated swine breeds. (a) Three-month old Aachen Minipigs. (b) Pietrain piglets, aged three weeks.
At our closed facility, the animals were kept with five conspecifics in two separate swine box of 3.2 m2 equipped with appropriate enrichments, as approved by the local authorities. European spruce ground granulate of 2.5–3.5 mm (JRS Lignocel FS-14, J. Rettenmeier & Söhne, Rosenberg, Germany) was used as bedding material. Two infrared-heating lamps ensured an active warming at the resting period. The temperature was adjusted between 20.3 and 21.7 ℃ with a relative humidity between 21.4 and 49.0%. Air within the stall was exchanged 13 times per hour. A dark–light cycle of 12 hours with artificial lighting between 6 and 18 o’clock was set. Drinking water was provided ad libitum, as was 180 g of feeding slurry (120 ml of hot water compounded with 60 g of piglet fodder (Protec pre, Denkavit, Warendorf, Germany)) three times per day with an additional amount of 30 g of dry fodder per animal between the feeding times. The swine had one week of acclimatization to husbandry conditions at our facility before the experiments were started.
Anaesthesia and operation
All animals used were collected from a pilot study. For this reason, the euthanasia of the animals took place after surgery. Following a right-sided thoracotomy an oesophagotomy and re-anastomosis was performed: The oesophagus was cut at the carinal level and re-anastomosed with simple interrupted sutures to test the feasibility of the planned procedure. At the end of the surgical procedure, the swine were euthanized without awakening by an overdose of sodium pentobarbital (100 mg/kg bodyweight).
Necropsy, blood and urine sampling
The animal's length was determined from snout to the beginning of the tail using a tape measure. The whole animal and subsequently all organs were weighed on a laboratory balance (PCB6000-1, Kern & Sohn, Balingen, Germany). Necropsy was conducted as described before. 20 Blood were drawn via the central venous catheter before application of the pentobarbital overdose and urine was obtained by transcutaneous bladder puncture. All analyses were conducted at the Institute for laboratory medicine of our institution.
We calculated relative organ weights by division with the animal's bodyweight and multiplied it with 100 to obtain percentages. Relative organ length was determined similarly yet calculated relative to snout-to-tail length. All raw data are included in Supplemental data 1.
Statistical analysis
All values are presented as mean and 95% confidence. Due to the small sample size, we calculated them using bias corrected and accelerated bootstrapping as described by Efron,
21
which has been demonstrated to be applicable to analyses hampered by their small sample size, like our present study.
22
As recommended, we used 10,000 resamplings to achieve an adequate accuracy of the confidence interval.
23
We tested our results for a Gaussian distribution using the Shapiro–Wilk test and for homoscedasticity with Levene's test. If data was non-normally distributed, we compared the two groups by the Wilcoxon–Mann–Whitney U-test and if data had missing homoscedasticity, but were normally distributed, we used Welch's test. To account for the problem of alpha-inflation by the high number of tests, we corrected for multiple comparisons employing the Benjamini–Hochberg procedure to obtain the corrected level of significance q*.
24
The first set of comparisons was the anatomical parameters, which we expected to be similar with a
In order to compare the investigated swine breeds to previously published human data on organ weights,25–28 we used metric multidimensional scaling, which reduces high-dimensional data to a two-dimensional representation based on the inter-object proximities expressed in Euclidian distances. 29 If only a cumulative weight of pairs or organs was provided,25,26 we used the mean weight for both sides. Mean weights were also the input parameters for all groups including our experimentally assessed swine breeds. From the study by Maroun and Graem, 26 we used the organ weights from non-macerated specimens.
All statistical analyses were conducted using R (version 3.4.3) with the stats4-package (version 3.4.3). 30 Levene's test was from the car-package (version 3.0.0) 31 and bootstrap confidence intervals were obtained by the groupwiseMean-function from the rcompanion-package (version 1.13.2). 32
Results
We first tested whether there was an a priori difference of bodyweight between the two breeds using the planned five animals per group. Pietrain piglets were heavier than Aachen Minipigs by 1.31 kg (95% CI: 0.37–2.23, p = 0.0073) (Figure 2(a)), while the snout to tail length of both breeds was similar (Figure 2(b)).
Differences in anatomical parameters. (a) The Pietrains were 1.31 kg heavier than the Aachen Minipigs (95% CI: 0.37–2.23, p = 0.0073). (b) The snout–rump length of both swine breeds was similar (95% CI: –5.8–1, p = 0.1626). (c) The oesophagus was 5.43 cm shorter in Aachen Minipigs compared to Pietrain piglets (95% CI: 2.2–8.6, p = 0.0022, 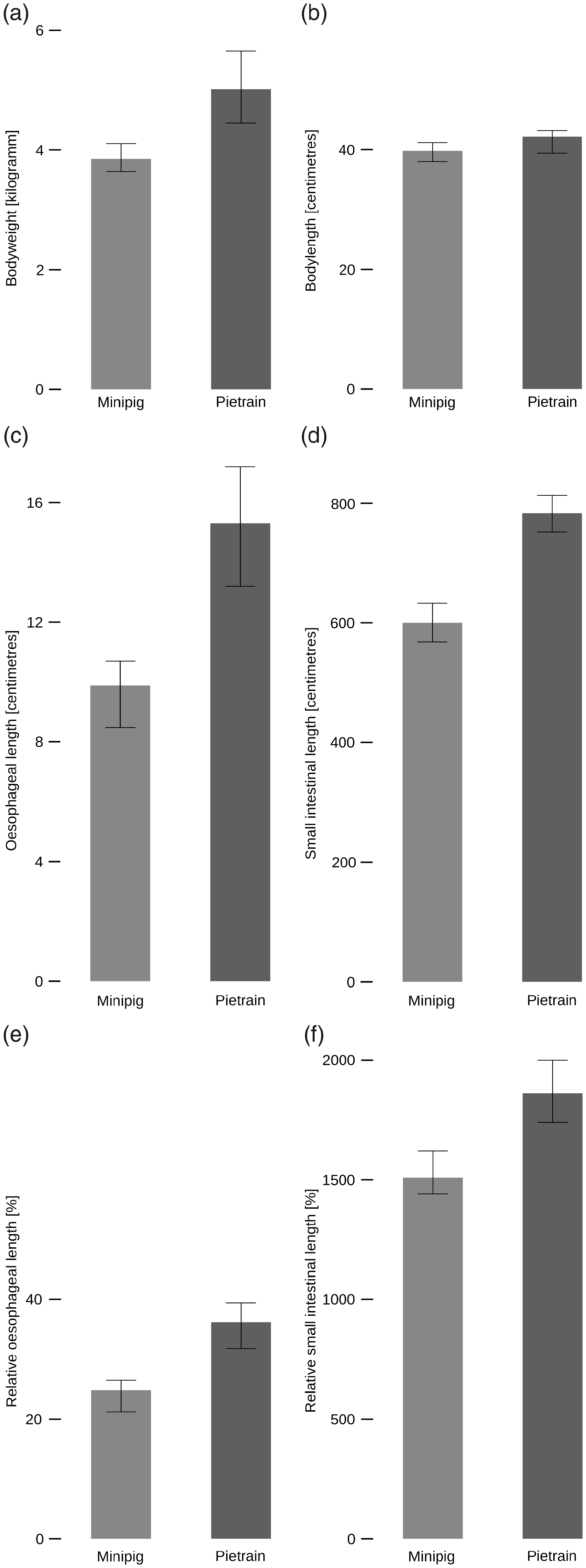
Anatomical differences between Pietrains and Aachen Minipigs
Aachen Minipigs (
Likewise, the small intestine was shorter (Δ = 182 cm, 95% CI: 122–243, p = 0.0001,
Anatomical parameters of the investigated swine breeds.
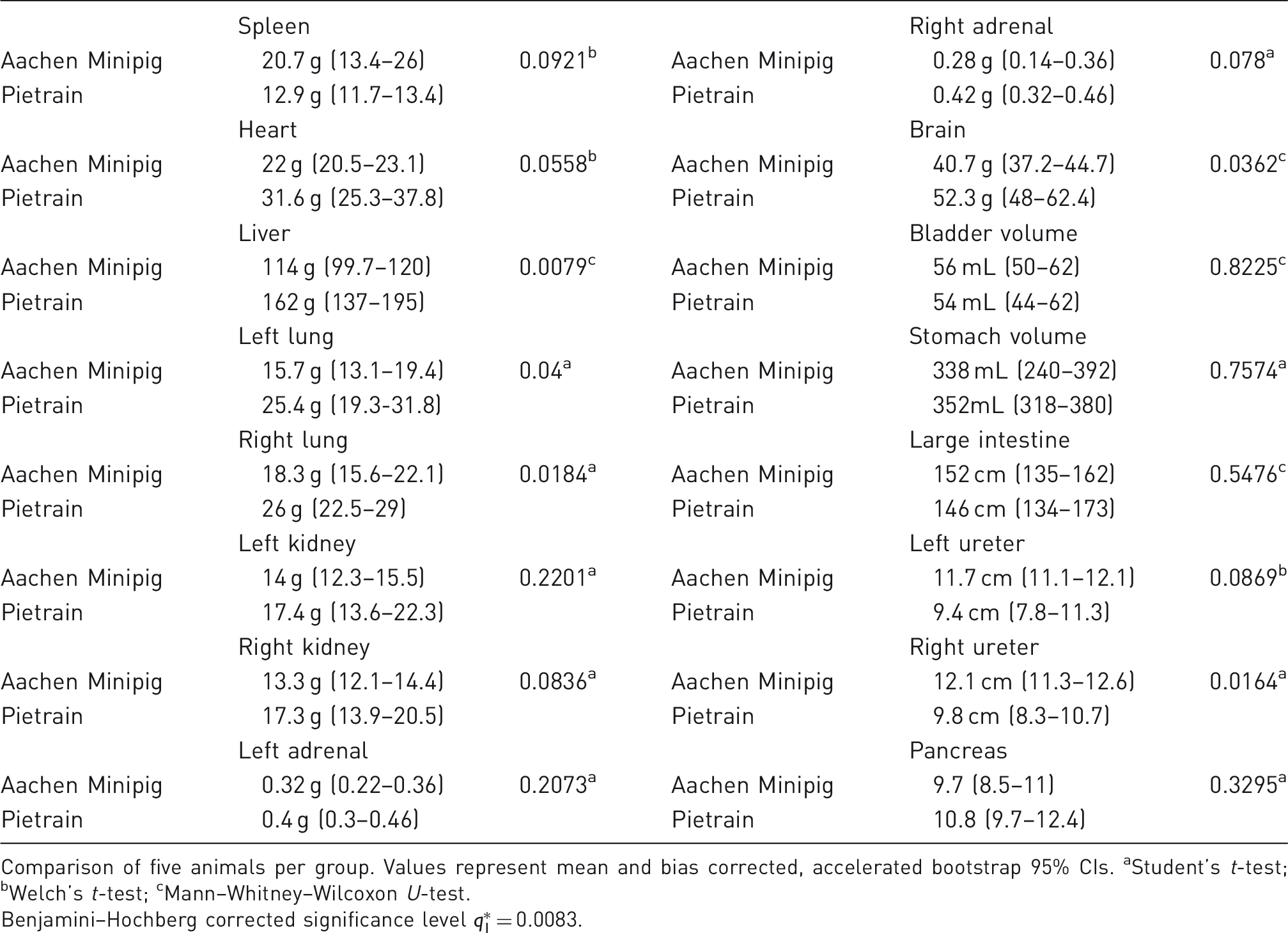
Comparison of five animals per group. Values represent mean and bias corrected, accelerated bootstrap 95% CIs. aStudent's t-test; bWelch's t-test; cMann–Whitney–Wilcoxon U-test.
Benjamini–Hochberg corrected significance level
Differences in standard laboratory parameters between the two breeds
Laboratory parameters of the investigated swine breeds.
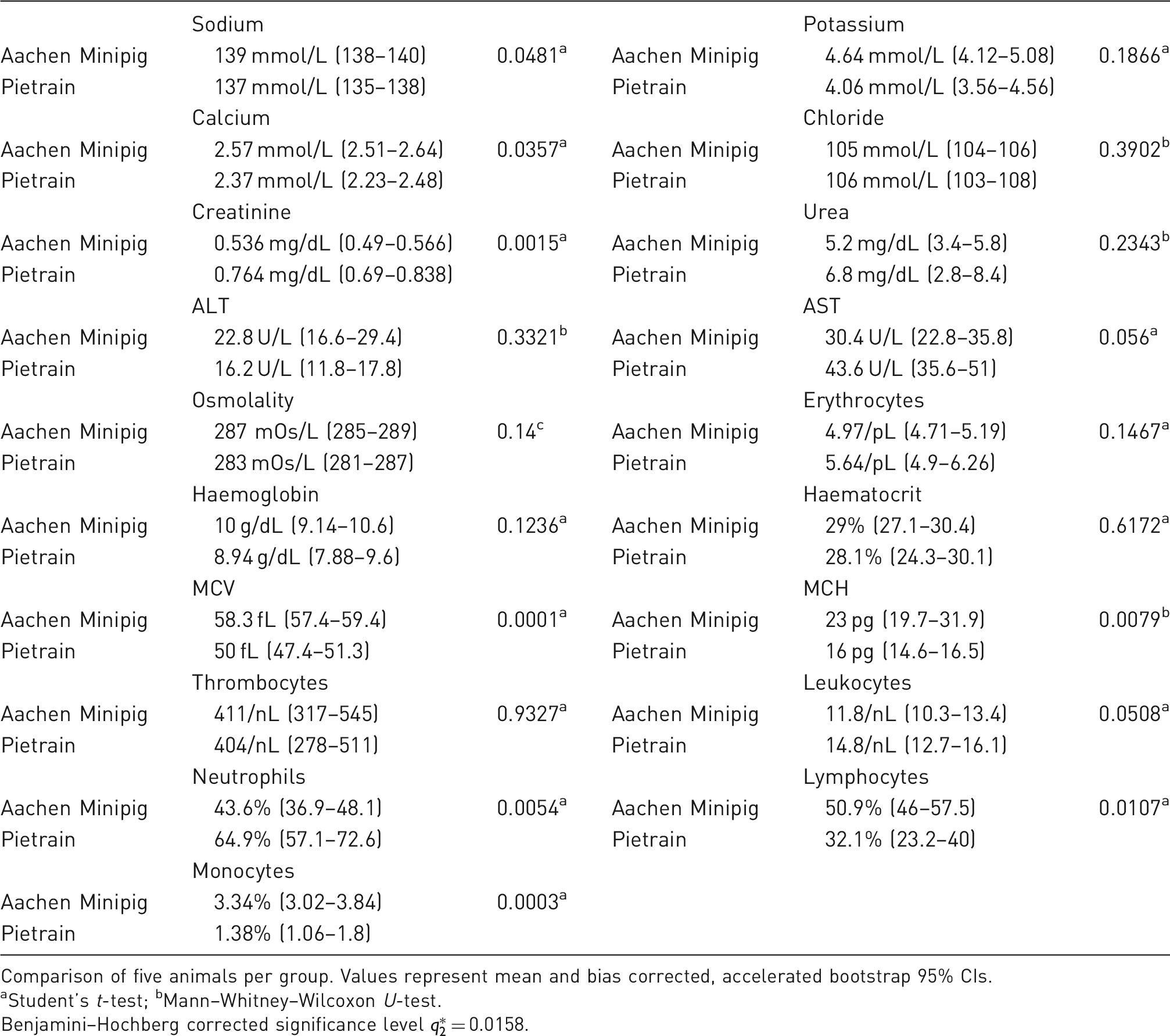
Comparison of five animals per group. Values represent mean and bias corrected, accelerated bootstrap 95% CIs.
aStudent's t-test; bMann–Whitney–Wilcoxon U-test.
Benjamini–Hochberg corrected significance level
Similarity analysis of the swine breeds' organ weights in comparison with human autopsy data
Proximity analysis showed both swine breeds to be far from being an adequate model of humans in developmental stages close to birth (Figure 3(a)). However, brain weights differed highly between swine and humans – who had a six-fold higher brain weight at minimum than the investigated swine breeds. This prompted us to exclude brain weights from the multidimensional scaling as we felt brain weight to be of lesser importance in a non-neurosurgical model. The resulting proximity matrix demonstrated the three-week old Pietrain piglets (–1685/–26) to be in striking distance to two-month old humans (–1767/3) indicating the high degree of similarity between the organ weights, whereas Aachen Minipigs were still distant from all investigated human developmental stages (Figure 3(b)).
Model similarity of swine breeds and human autopsy results from the literature.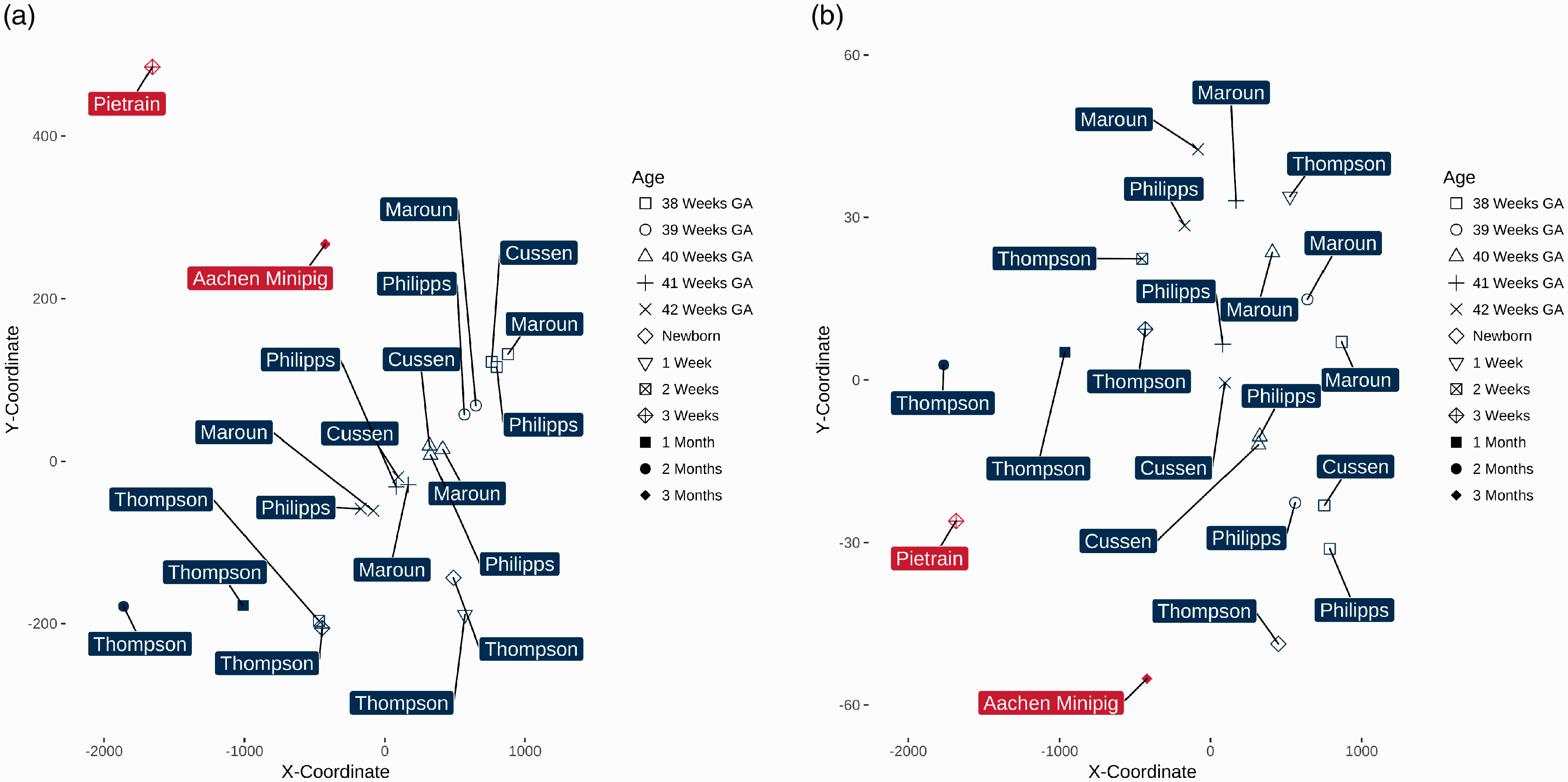
Discussion
For experimental animal research, the choice of the right model organism is crucial. To gain meaningful, translatable results in experimental surgical research, the model needs to have similar anatomical and functional characteristics which mimic the investigated human condition as close as possible. 33 Swine had been the favourite experimental animal in paediatric surgical research, 3 but have largely been replaced by rodent models. 7 Piglets were often chosen in earlier studies of oesophageal diseases,4,34 but older swine were used in more recent studies.9–11 A trend boosted with the promulgation of minipigs outside their initially intended scientific habitat – xenotransplantation surgery. 15 Subsequently, minipigs have also been used in paediatric surgery.13,14 This raises an important question: what aspects and parameters should influence model choice?
There is no perfect animal model for experimental paediatric surgery. However, comparable body weights may have been a landmark, but its limited usefulness as a sole parameter might be exemplified by oesophageal length: it has traditionally been determined using the patient's height, 35 whereas others favour crown–rump length, 36 height and bodyweight 37 or simply dismiss all anatomical parameters. 38 The gold standard would be to compare experimentally determined oesophageal lengths to those obtained from autopsy results, but to our knowledge there are no such data available, as bemoaned before. 39 Estimation of oesophageal length using one of several formulae is impossible too. One has only been validated for prematurely born 37 and another only for children older than three years. 38 The remaining two at least require a height measurement,35,36 which is not only missing in the French oesophageal atresia cohort, 16 but also in the only autopsy dataset reporting measurements from non-stillborn. 28 Transferral of weight to height using growth charts would introduce too much uncertainty as their correlation is far from being perfect with R ≈ 0.7, 40 thus precluding usage of the formulae as a marker of orientation for model choice based on oesophageal lengths. Hence, evaluation of model suitability has to be based on more than one parameter.
Comparisons between different minipigs or standard pig breeds had been undertaken to assess differences in certain aspects between them, for example hypercholesterolaemia and atherosclerosis, 41 and gut microbiota influencing obesity. 42 A more general comparison of different swine breeds to get insights into baseline values to assess similarities and, even more importantly, dissimilarities remained an exception, but has recently been conducted for Göttingen and Aachen Minipigs. 18 The influence of age on a study's outcome was investigated in isolated, perfused lungs, 43 tissue-engineered bladder wall replacements, 44 surgical hepaticojejunostomy, 45 and found to be often of relevance. Studies comparing porcine data from different breeds directly with human data are rare: acetylcholinesterase enzymatic activity and kinetics 46 and the properties of one of its inhibitors 47 were determined and compared between swine and humans. These analyses were limited to one specific point of interest, which eases the methodological complexity as it increases with the number of parameters. However, pair-to-pair comparisons of a certain aspect cannot provide an overall picture of the included parameters in a comprehensive fashion.
For a surgical model, it is seldom a single parameter that determines model suitability: the interplay of several anatomic factors influence not only the surgical approach – size differences may preclude the usual access to the target organ – but also the technical feasibility of the procedure itself. Inevitably a certain amount of space is necessary to perform all steps of the surgery, in our case oesophageal resection and anastomosis under increasing tension. Another issue to consider in surgical model choice is the interplay between organs: the gastrointestinal tract is a continuum along which traction forces will be distributed, not just present at the anastomosis, but also at neighbouring organs such as the diaphragm that is also loosely connected to the oesophagus. All these organs will absorb and distribute some of the traction forces occurring due to oesophageal resection. This may be exemplified by traction forces measured at the anastomosis of 1.6 N after a 33% oesophagectomy, 6 whereas more than 60 N were measured at explanted oesophagi. 48 Therefore, investigation of a single parameter is likely to be insufficient and represents one of the reasons why we used multidimensional scaling as a form of principle component analysis to simultaneously take more than one parameter into account. We thus aimed to establish a close proximity to humans based on autopsy studies providing weights of several organs.
The higher number of parameters introduces a problem named the ‘curse of dimensionality’, 49 which require different analytical methods to overcome the phenomenon. Previous studies demonstrated the applicability of multidimensional scaling for several problems including laboratory analyses to separate patient groups, 50 discrimination of patients with Alzheimer's disease and controls based on magnetic resonance imaging, 51 and similarities between immunoglobulin repertoires. 52 Therefore, we employed multidimensional scaling for dimensionality reduction to graphically assess the overall similarity between our experimental groups. Multidimensional scaling achieves this by the reduction of dimensions based on an iterative process that keeps the distances between the investigated subjects constant for each dimension and displays this high-dimensional data in a two-dimensional representation of distances based on all distances calculated for the original dimensions, 29 the organ weights in our study. A classic example to illustrate the effect of multidimensional scaling in assessing similarity is the distance between cities. The distances between these cities may be presented in a table of pairwise distances between them, just like the comparison of organ weights in our case. A different representation of these distances would be their depiction on a map, which allows graphical assessment not only of absolute, but also of relative distances and may thus answer at a glance how close or far different cities are from another. Transferred to our study, distances within the graphical representation represent similarity: the closer two points are on the arbitrary axes, the higher is their similarity. Consequently, multidimensional scaling visualizes hidden structure within the examined data, in our case the similarity of organ weights between eight-week old humans and three-week old Pietrain piglets.
As long as brain weights were included in the analysis, the investigated swine breeds were highly different from humans in all included developmental stages. Due to the human brains being at least six fold heavier than the swine brains, we excluded brain weights from the proximity calculation based on the assumption that brain weight may not be representative for oesophageal length, the parameter we were interested in to define a model of oesophageal surgery. The subsequent multidimensional scaling plot revealed a high degree of similarity between the freshly weaned Pietrain piglets and two-month old humans.
Our study is limited by the small number of animals per group, substantially smaller than the group size in similar comparative studies,18,44 one of which also differentiated between sexes. 18 In favour of the 3Rs, we limited our analysis to five animals per group as it has been demonstrated by simulation studies that this conventional group size achieves a power of 80% in pilot studies. 19 To preserve this power, we used Benjamini–Hochberg's correction of the false discovery rate, which is considered superior to a correction of the family-wise error rate if a high number of comparisons will be conducted. 53 Besides these methodological issues, it is unclear whether the oesophagus underlies the same principles as the other internal organs included in our comparative analysis between swine and humans that is the similarity between the porcine and human organs. In addition, it needs to be emphasized that the extraordinary degree of similarity is only valid for the presented organ weights and may be totally different if other parameters are included or targeted as exemplified by the high degree of dissimilarity between human and porcine comparators if brain weights were included. Consequently, similarity analysis might also result in a different relationship between humans and pigs if older and heavier animals were to be compared to older and heavier humans, because growth rates and target weights differ highly between both investigated pig breeds. This may favour the Aachen Minipig as a model for diseases in older humans, because Pietrain swine might just be too large and heavy to be comparable to human anatomy.
However, based on the available data, we can conclude that three-week old Pietrain piglets can model two-month old infants based on their internal organ weights whereas the three-month old Aachen Minipig is more dissimilar to humans. This makes the Pietrain piglets a suitable model of paediatric oesophageal atresia surgery, because delayed primary anastomoses to correct long-gap forms are typically conducted at this age. 54
Footnotes
Acknowledgements
We thank Ann-Kristin Riedesel for her invaluable assistance in piglet husbandry.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
