Abstract
One of the most commonly used protein sources in rodent diets is soy, which is naturally rich in phytoestrogens. Although phytoestrogens have shown potential health benefits in humans, they may also have the ability to disrupt reproduction. Consequently, there has been a tendency to try to exclude them from rodent diets. In the current study, we investigated whether phytoestrogen content in the mouse diet could affect reproduction in mice used as embryo donors. Donor mice (C57BL/6JOlaHsd) were maintained with three different diets: high phytoestrogen (ca. 400 mg/kg genistein), low phytoestrogen (ca. 10 mg/kg genistein) and standard breeding diet (ca. 120 mg/kg genistein). Mice fed a high phytoestrogen diet had a high yield of plugs, embryos, and injectable embryos, as well as producing good quality embryos. Results from donor mice fed a low phytoestrogen diet were consistently but only slightly inferior, whereas mice fed a standard diet performed the poorest. Interestingly, the largest number of born and weaned offspring were observed when recipient females received embryos from the standard diet group. Sperm yield and quality of stud males did not differ between the groups. We surmize that for experimental endpoints requiring fertilized embryos it may be more beneficial to feed mice a diet containing phytoestrogen, but if the goal is to produce transgenic mice, a diet high in phytoestrogen may be inadvisable. In conclusion, care should be taken when selecting a diet for experimental mouse colonies as phytoestrogen could influence the study outcome.
Introduction
Successful reproduction is critically important when managing the production and breeding of experimental mice. Successful and effective reproduction would lead to fewer mice needed for breeding, thus resulting in an overall reduction in mice needing to be produced for experimental work, which would fall agreeably with the 3 R concept. 1 It can be assumed that good reproduction is at least partially a result of well-being. 2 Efforts to improve mouse well-being in modern animal facilities have become standard, and a lot has been achieved by looking into the optimal physical environment, such as temperature, humidity, light cycle and ventilation.3–7 Furthermore, a high-quality diet is often provided to make sure that not only are the nutritional needs met, but successful reproduction is also supported. 8
Commercial manufacturers offer a large number of different diets for mouse colonies, many of them targeting specific colonies, strains or individuals. The protein, fat and carbohydrate composition is balanced depending on whether it is intended to be used, for example, as a maintenance diet (i.e. for potentially obese strains requiring low fat and low energy) or as a breeding diet (i.e. for in-bred transgenic strains often requiring higher fat, protein and energy content).8, 9 Not only should the relative amounts of protein, fat and carbohydrates be taken into account, but also the quality of the original raw material. 10 One of the most commonly used ingredients in mouse diets is soy, as it is abundantly available and relatively cheap. 11 Soy is naturally rich in phytoestrogens, a group of plant-derived substances that are structurally similar to estradiol, and as such can mimic its effects. 12 Phytoestrogens have received a lot of attention lately due to the potential health benefits in humans. 13 They have been suggested, for instance, to reduce the risk of cancer, osteoporosis and heart disease, and they may have beneficial effects on obesity and diabetes. 13 Despite the potential benefits of phytoestrogens for human health, however, their adverse effects as endocrine disruptors may be underappreciated. 14
Phytoestrogens have been found to disrupt and affect rodent reproduction in different ways. They have been suggested to have deleterious effects on the reproductive system as a whole, disrupting ovarian function and causing reduced fertility and even infertility. 15 They have been associated with decreased body weight, accelerated vaginal opening, reduced litter size, and alteration of the oestrus cycles in young mice. 16 Dietary phytoestrogen consumption has been associated with dampening and partially defeminizing female sexual behaviour. 17 Phytoestrogens are also suggested to cause persistent demasculinization of male mice, 18 and to cause low sexual receptivity. 19 Due to these potential effects of phytoestrogens on mouse reproduction, it is increasingly recommended that they be excluded from rodent diets.17, 20, 21 However, many of the studies performed today have used mice as a model for different human conditions and diseases rather than specifically focusing on mouse reproduction, raising questions about how far the results can be directly applied to mouse husbandry and breeding.
The aim of this study was to determine whether long-term exposure to different levels of phytoestrogens in mouse diets could affect mice that will be used as embryo donors. Specifically, we looked at pregnancy rates, embryo yield and quality, as well as litter size, as indicators of successful reproduction in the context of transgenic mouse production via DNA microinjection.
Animals, material and methods
Animals and husbandry
Female donor mice and stud males (both C57BL/6JOlaHsd), as well as recipient mice (Crl:CD1(ICR)), were bred in-house and maintained in specific pathogen-free conditions (SPF; according to FELASA 2014 recommendations 22 ), in an individually ventilated cage housing system (type 1145T; Tecniplast, Buguggiate, Italy) at Max Planck Institute of Molecular Cell Biology and Genetics, Dresden, Germany. Each cage was provided with aspen bedding and nesting material (Tapvei, Paekna, Estonia), which was routinely changed once per week under laminar flow (InterActive Cage Changing Station; Tecniplast). Filtered and acidified (pH 3.0) water was provided ad libitum. Animal rooms were maintained at 22 ± 1℃, and at a relative humidity of 55 ± 10%. Light–dark cycle was 12:12 h with lights on at 05:00 h.
All animal housing, handling and experimental techniques were in accordance with the principles set out in the Declaration of Helsinki, as well as in accordance with the ethical standards of the European and German Animal Welfare legislation. Experiments were planned and conducted to adhere closely to the 3 R (replacement, reduction, refinement) principles of animal welfare. All procedures were licensed (Landesdirektion Sachsen Aktenzeichen 24-9168.11-9/2005-1) and carried out according to the Institutional Animal Care and Use Committee guidelines as regulated by German Federal law governing animal welfare.
Study design
Female mice used as embryo donors, as well as stud males, originated from three different feeding groups. One group received high phytoestrogen diet (High-PE) (Harlan Teklad TD.55133, 20% soy, 18% protein, ca. 400 mg/kg genistein). The second group received a low phytoestrogen diet (Low-PE) (Harlan Teklad TD.55132, 0% soy, 18% protein, ca. 10 mg/kg genistein). The third group received the standard diet (Normal-PE) typically used for most of the genetically modified in-house mouse colonies (Harlan Teklad 2018S, approximately 10% soy, 18% protein, ca. 120 mg/kg genistein). Each feeding regime was followed for a minimum of two generations before mice were used for the experiments. Altogether, 108 donor mice (22 experimental days) were used from each of the High-PE and Low-PE feeding groups, and 162 donor mice (17 experimental days) were used from the Normal-PE group. Mice from different feeding groups were colour coded. Donor mice aged 3–4 weeks were pooled together and distributed randomly to stud males. Futhermore, the order in which the recipient mice received embryos from different feeding groups was changed in each experimental day. Operators performing hormone priming, mating with stud males, organ collection and embryo transfers were not aware of the origin (feeding group) of the mice (blinding).
Hormone priming
Data was collected using previously published methodology. 3 In short, on day zero, each female donor mouse (3–4 weeks of age) received an intraperitoneal injection of five international units (IU) of pregnant mare’s serum gonadotropin (PMSG), followed by an intraperitoneal injection of 5 IU of human chorionic gonadotropin (hCG) on day two (46 h later). Females were mated with fertile males (within their respective feeding groups) immediately after hCG injection. Early in the morning of day three, mice were checked for vaginal plugs and euthanized by cervical dislocation. All oviducts were collected and embryos flushed out.
Embryo collection
Embryos were separated from the cumulus cells and transferred in the first drop of the culture medium (M2). The total number of embryos was counted under the stereomicroscope. During the subsequent wash procedure (transferring embryos from one drop of culture medium to the next 10 times), only healthy-looking embryos were selected in each transfer step. In the last drop, the embryos were counted again and scored as ‘good quality’ (regular round form, intact zona pellucida). Embryos were then placed in a drop of KSOM incubation medium covered with oil for 1–2 h until they were used for pronucleus injection. Injections were performed under a microinjection stage (Zeiss Axiovert 200 M [Carl Zeiss AG, Oberkochen, Germany]; Eppendorf TransferMan NK2 [Eppendorf AG, Hamburg, Germany] and Eppendorf Femtojet [Eppendorf AG]), and only those embryos that had a well-developed (clearly visible) pronucleus were used for DNA injection. These embryos were counted and scored as ‘injectable quality’. After the DNA injection, the embryos were further incubated for 2–3 h, and the embryos that did not survive this period were scored as ‘lysed’. After this, all surviving embryos were transferred to recipient mice.
Embryo transfer to pseudopregnant recipients
Embryo transfer was performed by oviduct transfer as described by Nagy. 23 Nulliparous 0.5 d pseudopregnant Crl:CD1(ICR) recipients (8–15 weeks of age, weighing 28–36 g) that had been bred in-house under SPF conditions were used. Embryo transfers were aseptically performed in a sterile lab within the barrier unit. Recipients were anesthetized intraperitoneally with ketamine, acepromazine and xylazine. 24 Prior to embryo transfer, the quality of the recipient mice was evaluated by observing the swelling of the ampulla together with a good blood circulation of the ovary–oviduct–uterine tract. Post-operatively recipients were placed in their home cage under the heating lamp until they were fully recovered, and subsequently transferred back to the animal room. The recovery period was intensively observed by the operator, and during the subsequent 48 h animals were observed several times per day. All mice recovered successfully without a single incident. On average, 24 embryos were transferred to each recipient mouse. Altogether, 46 recipients received embryos from High-PE donors, 48 recipients from Low-PE donors, and 68 recipients from Normal-PE donors. Recipient mice were all fed with the standard diet (Normal-PE).
Sperm collection
Sperm was collected from both High-PE (n = 4, 41–48 weeks of age) and Low-PE (n = 6, 24–45 weeks of age) mice using previously published methodology. 3 In short, to isolate the sperm, the epididymis and the first few millimetres of the vas deferens were dissected, and the surrounding fat tissue was carefully removed. The epididymis were then transferred to cryoprotective agent (CPA; 18% raffinose and 3% skim milk, prepared by the Transgenic Core Facility, and stored at –20℃ for up to 6 months), opened with forceps, and incubated for 5 min at room temperature to allow the sperm to be released. An aliquot (4 µL) of the sperm suspension was transferred to 196 µL of human tubal fluid medium and incubated for 5 min at 37℃. A 30 µL aliquot (dilution of 1:50) was transferred to a Standard Count 2 Chamber Slide from Leja (Nieuw-Vennep, The Netherlands) and measurements were made with a Hamilton Thorne Automated Sperm Analyser (Hamilton Thorne, Beverly, MA, USA) with default settings for mouse sperm.
Experimental setup
Experiments with High-PE and Low-PE groups were performed on the same days with the same DNA (exact pairwise comparison). Experiments with Normal-PE group were performed during the same time frame, but not necessarily on the same days as High-PE and Low-PE groups. Typically, five females were used per experimental batch (within 1 day) for High-PE and Low-PE groups; however, eight to ten were typically used per experimental batch for the Normal-PE group. This was to safeguard enough material for transgenic work. Each experiment was part of a standard production of transgenic mice, and each successful experiment resulted in mice that were the foundation for several transgenic strains. No additional mice outside of the standard production of generating transgenic strains were used for these experiments.
Statistics
Data were processed and analysed using PASW Statistics 23.0 software (SPSS Inc, Chicago, IL, USA). All data were tested for normal distribution (Shapiro–Wilk) and homogeneity of variance (Levene’s test). One-way analysis of variance (ANOVA) was performed for all data that showed a normal distribution, followed by post hoc analysis (Tukey’s honest significant difference (HSD)). A non-parametric Kruskal–Wallis test, followed by the post hoc Dunn–Bonferroni analysis for pairwise comparison was used for the data when normality could not be assumed. Pearson’s chi-square was used to demonstrate the association between the pregnancy output and different diets. The statistical significance level was set at p < 0.05. All results are shown as means (± standard deviation) unless otherwise indicated.
Results
High dietary phytoestrogen content led to good mating success and high embryo yield
To produce the maximum number of embryos, all female mice were superovulated with hormones (PMSG and hCG). The hormone treatment not only increases the production of oocytes, it also times the oestrus cycle so that the maximum number of copulations can be anticipated in any given day. To confirm the potential pregnancy of a mated female mouse, we investigated the success of copulation by observing vaginal plugs the morning after mating. The yield of vaginal plugs was highest with the High-PE group at 85%. The Low-PE group had a slightly, but not significantly, lower yield at 79%. The Normal-PE group showed the poorest success rate at 76%, which was significantly different from that of the High-PE group (Kruskal–Wallis global X2(2) = 7.147, p = 0.028; pairwise comparison Normal-PE with High-PE p = 0.023) (Figure 1). These results indicate that a high level of dietary phytoestrogen has a positive effect on mating success, although this effect cannot be based on the phytoestrogen only, as the comparison with the low phytoestrogen group did not yield a significant difference.
The percentage yield of vaginal plugs in mated females shown as a boxplot. The values were counted from pooled females (typically between 5 and 10 females per day) from each experimental day. High-PE n = 22 (108 mice), Low-PE n = 22 (108 mice), Normal-PE n = 17 (192 mice). Mean values are shown as white diamonds, second and third quartiles as rectangles with the median value indicated by a vertical line, and the minimum and maximum values at the end of the whiskers. Significance levels are shown as *p < 0.05 (Kruskall–Wallis followed by pairwise comparison (Dunn–Bonferroni test)).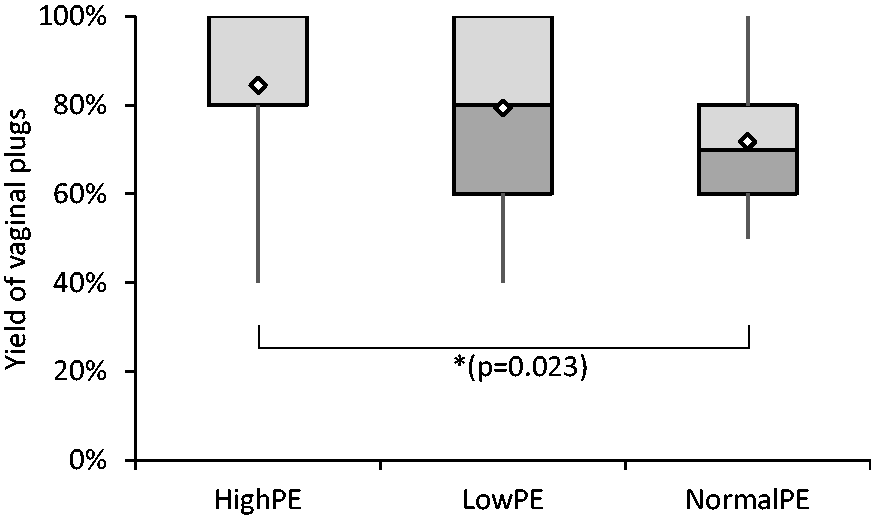
Although mating success can be estimated by observing the yield of vaginal plugs, this is no guarantee. Therefore, it is necessary to determine the total yield of embryos. The best yield of embryos per mated female was obtained with mice fed a high phytoestrogen diet (32.0 ± 9.7 embryos), whereas the embryo yield dropped slightly with mice fed a low phytoestrogen diet (26.3 ± 8.2 embryos) and with mice fed by standard diet (23.5 ± 6.7 embryos). The embryo yield in the Normal-PE group was significantly lower than that of the High-PE group (ANOVA F(2, 58) =5.069, p = 0.009, Tukey HSD Normal-PE vs. High-PE p = 0.009) (Figure 2). These results show that dietary phytoestrogen led to a high embryo yield.
The yield of embryos per mated female shown as a boxplot. High-PE n = 22, Low-PE n = 22, Normal-PE n = 17 (n = experimental days). Mean values are shown as white diamonds, second and third quartiles as rectangles with the median value indicated by the horizontal line, and the minimum and maximum values at the end of the whiskers. Significance levels are shown as **p < 0.01. (ANOVA followed by Tukey’s HSD).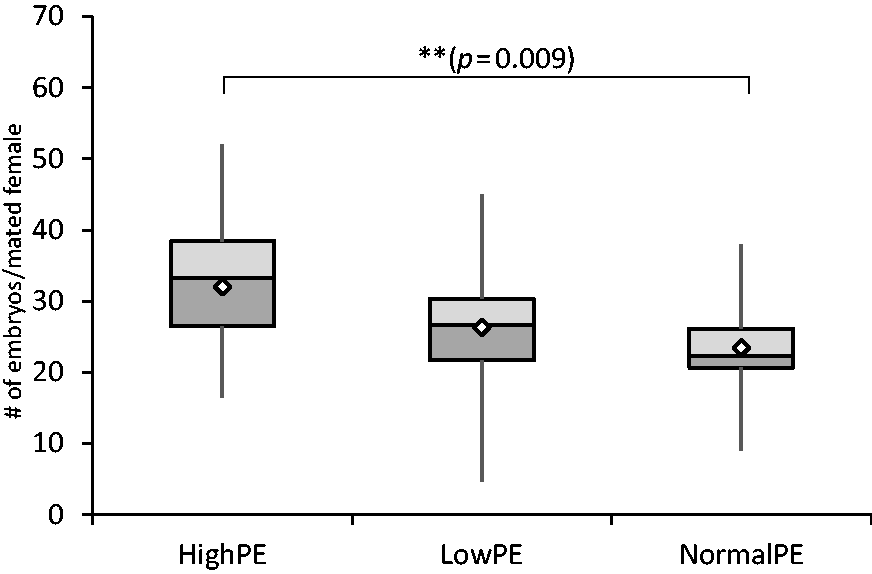
Embryo quality remained high with both high- and low-dietary phytoestrogen content
The proper development of embryos in vitro is essential for successful pronucleus microinjection. To determine whether the collected embryos could be successfully cultivated for further use, embryos were selected for a healthy round form and intact zona pellucida. The yield of good quality embryos per mated female at the healthy round form stage was highest in the High-PE group (29.6 ± 10.1 embryos), somewhat reduced in the Low-PE group (24.8 ± 8.4 embryos) and clearly reduced with Normal-PE group (21.5 ± 6.2 embryos). The Normal-PE group had significantly fewer embryos compared with the High-PE group (ANOVA F(2, 58) =4.129, p = 0.021, Tukey HSD Normal-PE vs. High-PE p = 0.018) (Figure 3).
The yield of good quality embryos per mated female shown as a boxplot. High-PE n = 22, Low-PE n = 22, Normal-PE n = 17 (n = experimental days). Mean values are shown as white diamonds, second and third quartiles as rectangles with the median value indicated by the horizontal line, and the minimum and maximum values at the end of the whiskers. Significance levels are shown as *p < 0.05 (ANOVA followed by Tukey’s HSD).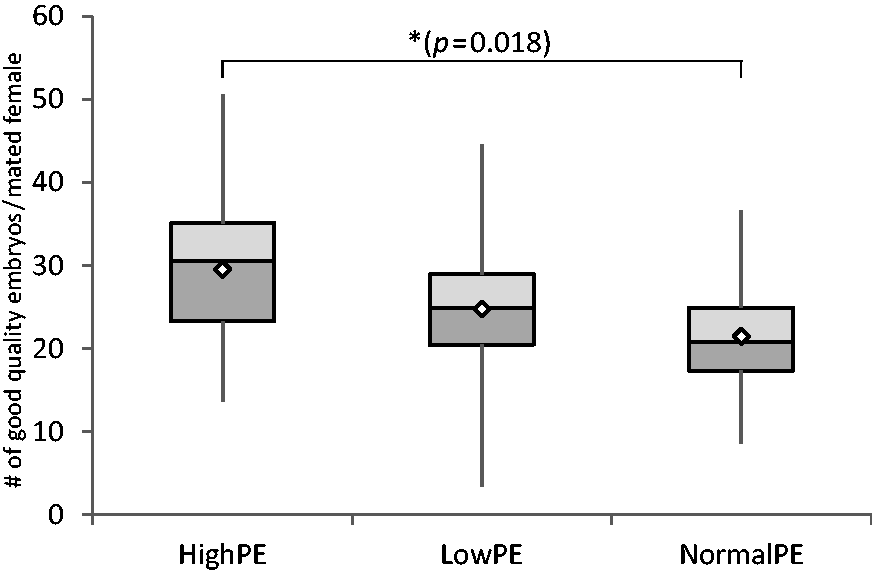
The injectability of embryos was examined next, as only embryos with a properly developed and visible pronucleus can be successfully used for microinjection. The overall numbers of embryos selected for injections was highest in the High-PE group (16.3 ± 5.4 embryos), but not significantly lower in the Low-PE group (12.3 ± 5.9 embryos). Only 11.5 ± 3.7 embryos were observed on average at this stage in the Normal-PE group, which is significantly lower than in the High-PE group (ANOVA F(2, 58) = 4.780, p = 0.012, Tukey HSD Normal-PE vs. High-PE p = 0.020, High-PE vs. Low-PE p = 0.040) (Figure 4).
The numbers of injectable embryos per mated female shown as a boxplot. High-PE n = 22, Low-PE n = 22, Normal-PE n = 17 (n = experimental days). Mean values are shown as white diamonds, second and third quartiles as rectangles with the median value indicated by the horizontal line, and the minimum and maximum values at the end of the whiskers. Significance levels are shown as *p < 0.05 (ANOVA followed by Tukey’s HSD).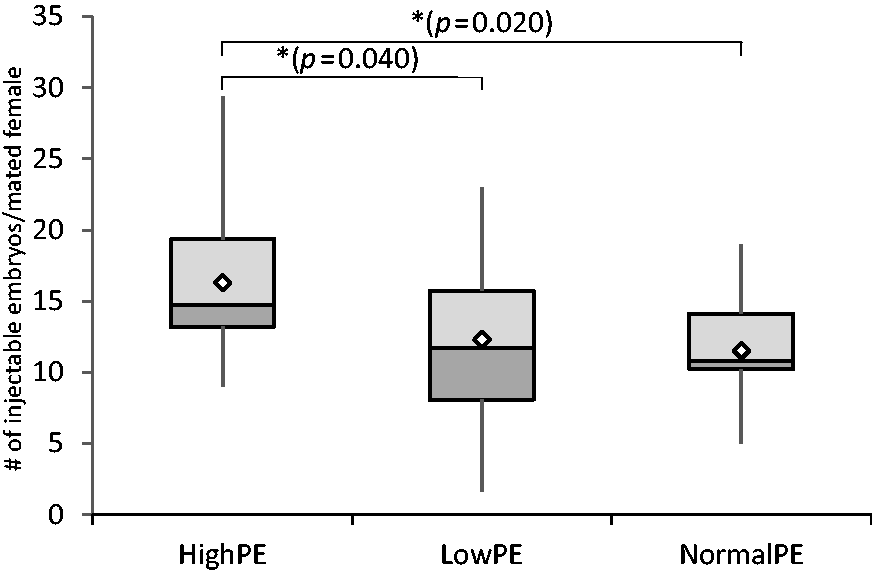
To test the robustness of the embryos that had been microinjected with DNA, the final embryo scoring was performed by removing the lysed (broken) embryos. The relative number of transferrable embryos per mated female was not statistically different between the treatments (ANOVA F(2,56) = 2.250, p = 0.115), whereas on average 14.3 ± 5.0 transferable embryos were obtained with High-PE group, but only 12.0 ± 5.5 with Low-PE and 11.1 ± 3.5 with Normal-PE groups.
These results show that embryo quality remained high with high phytoestrogen content, but was somewhat, but not significantly so, reduced with low phytoestrogen content. However, the quality of embryos was reduced in mice fed with a standard diet.
Fewer offspring were produced with embryos derived from mice fed a high phytoestrogen diet
Although high embryo yield and quality are essential to safeguard successful pronucleus injection experiments in vitro, success can be ultimately analysed by studying the numbers of pups born and weaned. Although embryo yield and quality indicated that mice fed a standard diet produce fewer embryos and that the quality of the embryos was inferior, we observed a significant association between birth events and different diets (X2(2) = 15.37, p < 0.001, Pearson’s chi-square) indicating that significantly more pregnancies and birth events were observed with recipients that received embryos from the Normal-PE group. Recipients from the Normal-PE group had a birth event in 91% of the cases, whereas 77% of recipients gave birth from the Low-PE group. Significantly fewer birth events (57%) were observed with recipients from the High-PE group.
Average litter size indicated a significant difference: High-PE recipients produced on average only 3.69 ± 1.8 pups, whereas Low-PE and Normal-PE recipients produced more (5.90 ± 3.4 and 7.38 ± 3.3 pups per litter, respectively). The litter size produced by High-PE recipients was significantly smaller than that of Normal-PE recipients (Kruskal–Wallis global X2(2) = 14.21, p = 0.001; pairwise comparison Normal-PE with High-PE p = 0.001) (Figure 5).
The average litter size shown as a boxplot. Values are calculated as number of offspring born per pregnant recipient (successful births). High-PE n = 16 litters, Low-PE n = 20 litters, and Normal-PE n = 32 litters. Mean values are shown as white diamonds, second and third quartiles as rectangles with the median value indicated by the horizontal line, and the minimum and maximum values at the end of the whiskers. Significance levels are shown as **p < 0.01 (Kruskall–Wallis followed by pairwise comparison (Dunn–Bonferroni test)).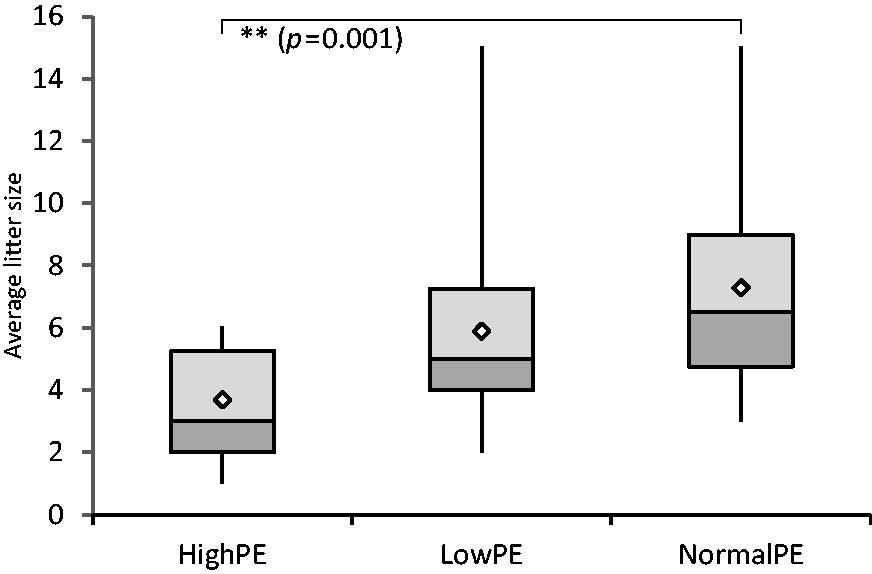
Finally, the overall effectiveness regarding productivity was calculated. Whereas on average only 0.46 donors fed with the standard diet and 0.92 donors fed with low phytoestrogen diet were required to produce one born offspring, noticeably more were needed (1.83) if donor mice were fed the high phytoestrogen diet. Likewise, on average 19.25 transferred embryos were required to produce one pup in the high phytoestrogen group, whereas only 9.66 transferred embryos were needed in the low phytoestrogen group, and only 4.86 transferred embryos with the standard diet.
Sperm count and quality were not affected by dietary phytoestrogen
To analyse the role played by the stud males in female mating success, sperm count and quality were determined. The average sperm count (the actual number of cells counted in all fields analysed with 1:50 dilution) was not significantly different between males fed with the high and low phytoestrogen diets (High-PE 2317 ± 686, Low-PE 2261 ± 641). We also examined whether the quality of the sperm could influence mating success by analysing sperm motility and progressivity (progressive motility). Sperm motility (percentage of motile sperm from the total sperm count) remained the same between high and low phytoestrogen groups (High-PE 59 ± 16%, Low-PE 67 ± 12%). The same tendency was observed with sperm progressivity (percentage of sperm with progressive motility from the total sperm count), where 18 ± 5% of the High-PE group sperm, and 16 ± 3% of the Low-PE group sperm, was found to be progressive. The sperm data from mice fed with the standard diet was not collected during this study, however the observed sperm count and quality was very similar to that of our earlier study with mice fed with the same standard diet. 3 This indicates that dietary phytoestrogen was not a significant factor for sperm count and quality.
Discussion
This study demonstrated that high dietary phytoestrogen content did not negatively affect the early stages of reproduction in mice. Mating success, embryo and sperm yield, and embryo and sperm quality all remained very high regardless of whether the mice were fed diets with high or low phytoestrogen content. This disagrees with a previously published report by Ramin et al., 25 who found that the embryo yield was reduced when mice were fed a diet containing approximately 110 mg/kg genistein. Their phytoestrogen-rich diet was similar to the standard phytoestrogen diet (ca. 120 mg/kg genistein) used in this study with no apparent reduction in embryo yield. The high phytoestrogen diet used in our study contained significantly more phytoestrogen (400 mg/kg genistein) and produced significantly more embryos when compared with mice fed a normal diet. It is to be noted that the mice in the study by Ramin et al. were fed only for a very short time (2 weeks), in contrast to our study where not only the mice used in the experiments, but also their parents, were fed for more than two generations with the respective diets. It could be speculated that the change in diet in the Ramin et al. study was responsible for the lower embryo yield rather than the phytoestrogen itself. Furthermore, one could consider whether long-term exposure to a phytoestrogen-rich diet eventually induces epigenetic changes 26 that cannot be observed in short-term studies.
Overall, the embryo yield and quality were very similar between the low and high phytoestrogen diet groups. Unexpectedly, we observed a reduction in embryo yield and quality when mice were fed the standard diet containing approximately 120 mg/kg genistein. This effect cannot be explained by the dietary phytoestrogen content alone, therefore other factors must play a role here. Although, based on the experimental setup, it is not possible to conclude definitively what might have caused this reduction, one could speculate that the standard diet was not exactly the same quality as the low and high phytoestrogen diets. The standard diet was produced in large batches, whereas the experimental diets were specially prepared in much smaller batches. Although all diets were produced by the same company and the experimental diets were adjusted to resemble as closely as possible the standard diet, there may have been differences in their production or the selection of ingredients (such as amino acid composition or fatty acid quality) that increased the overall nutritional value of the experimental diets. As a consequence, we could argue that the source of the mouse diet, and how it is made, could in itself have an impact on mouse reproduction, regardless of the label values presented by the manufacturer. This could be an important aspect to be analysed in a more specific experimental animal research setting. Furthermore, it has been shown that genistein can induce epigenetic changes, 27 but as epigenetic effect was not within the scope of our study this was not further investigated.
Interestingly, the positive effect of high phytoestrogen on embryos observed in our study did not translate into an ability to produce live offspring. Only about one out of 22 embryos from the high phytoestrogen group developed into live offspring, whereas less than half that number was needed in the normal and low phytoestrogen groups. Although Li et al. 28 observed that dietary phytoestrogens had no significant effects on embryo implantation and early pregnancy, we could argue that in our experimental setup either the implantation or the early pregnancy were compromised. Jefferson et al. 15 found that, although phytoestrogens affected embryo development, once the embryos were removed from the maternal environment at the two-cell stage, cultivated until blastocyst stage, and subsequently transferred for pseudopregnant recipients, both genistein-treated and untreated embryos were equally competent to produce live pups. However, they also showed that cumulative effects of the phytoestrogen genistein (given subcutaneously) eventually results in infertility by disrupting embryo development and implantation. The different results in many of these studies could be explained by the method of phytoestrogen delivery, such that dietary phytoestrogen could result in lower serum levels than that achieved with injected phytoestrogen, leading to a greater (negative) effect. 29 Surprisingly, in our study the dietary phytoestrogen had a clear effect and disrupted the embryos’ ability to develop live offspring. This could potentially have been caused by the cumulative effects of phytoestrogen (multigenerational feeding). Consistent with this, it has been suggested that long-term exposure to phytoestrogens may have the potential to generate epigenetic changes that could explain some of these results.26, 30 More significantly, all recipient females were raised using the standard diet and embryos most competently developing to live offspring came from the same diet group. Therefore, it could be argued that high phytoestrogen embryos struggled to survive and implant in a uterus environment derived from a different (standard) diet. This cannot be concluded with this study and would require further investigation involving embryo donors and recipients raised on the same or different diets.
Finally, as the purpose of embryo production typically is aimed at producing offspring to establish new transgenic strains, one should take great care selecting the optimal diet for both producing donors and recipients. Our study suggests that high phytoestrogen content in the diet of the donor colony may have a significant negative effect on the amount of offspring being produced, but it could also be a result of the different diets used for the donor and recipient colonies. Therefore, we would suggest facilities generating transgenic mice look into the diet compositions of both donor and recipient production with the aim of harmonizing them as much as possible – this could be especially important to those facilities that order donor mice directly from the vendor, but produce recipients themselves. In practical terms this could eventually lead to a paradigm change in how diets are used for generating transgenic animals, but to give a more concrete recommendation further research must to be performed.
Furthermore, it is to be noted that recipients in this study were anesthetized as published by Arras et al. 24 Although we have experienced excellent recovery of recipients during this study (and beyond), the anaesthetic procedure has recently been changed to include post-surgical analgesia (Metamizol) as lately recommended by GV-SOLAS. 31
Conclusions
Our data showed that phytoestrogen content in mouse diet could influence embryo yield or quality. Furthermore, our data showed that embryos produced by feeding donor mice with a high phytoestrogen diet had a significantly lower yield developing to live offspring (after DNA injection and subsequent transfer to recipient female). We conclude that for experimental endpoints requiring fertilized embryos it may be more beneficial to feed mice with a diet containing phytoestrogen instead of phytoestrogen-free diets, but if the goal is to produce transgenic mice via DNA microinjection, a high phytoestrogen content in the diet seems to be inadvisable. In conclusion, care should be taken when selecting a diet for experimental mouse colonies as phytoestrogen in diet may in certain cases influence the study outcome.
Footnotes
Acknowledgements
We thank the following services and facilities of the Max Planck Institute of Molecular Cell Biology and Genetics for their support: Biomedical Services, Transgenic Core Facility and Photolab. We would like to thank especially Tina Mittasch, Anke Muench-Wuttke and Wolfgang John for technical assistance. We gratefully acknowlegde Iain Patten and Raine Kortet for their constructive criticism of the manuscript.
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge support from the Max Planck Institute of Molecular Cell Biology and Genetics, Dresden, Germany.
