Abstract
Production of genetically-modified mice is strongly dependent on environmental conditions. Mice are commonly housed at 22℃, which is significantly lower than their thermoneutral zone. But, when given a choice, mice often seem to prefer higher ambient temperatures. In the current study we investigated the effect of higher ambient temperature on the production of transgenic mice, with emphasis on embryo and sperm yield and quality. Mice (C57BL/6JOlaHsd) were housed under four different ambient temperatures (22, 25, 28 and 30℃). Female mice were superovulated, and mated with males. As indicators for reproductive fitness, the success of the mating was observed, including embryo yield and quality, as well as sperm count, motility and progressivity. Female mice were found to produce high amounts of high quality embryos from 22 to 28℃. Sperm count dropped continuously from 22 to 30℃, but sperm motility and progressivity remained high from 22 to 28℃. We conclude that mice can be housed at significantly higher temperatures than is commonly recommended without compromising embryo production and quality, or sperm quality. These results could lead to fundamental changes in how mouse facilities are built and operated – especially in warmer climates whereby energy consumption and therefore costs could be significantly reduced.
The mouse has become one of the major animal models in biomedical research, not only because its genome has been fully sequenced, but also because its genome can be precisely engineered.1,2 Producing genetically-engineered mouse strains1–3 require a reliable and high yield of fertilized embryos. The embryos for microinjections must be collected from donor mice that have to be euthanized in the process. 4 To generate one new transgenic strain one generally has to use many tens, and sometimes more than 100, mice as embryo donors and as recipients. With an increasing need to constantly produce more transgenic strains it has become essential to look into the production and breeding efficiency with the aim of producing more strains with fewer mice. This is both a practical and an ethical issue.
One way to improve breeding efficiency could be by ensuring that welfare is optimal. Good welfare is an essential requirement when housing and breeding animals for experimental purposes, and it is actively enforced by regulations and laws.5,6 Better welfare could be achieved by ensuring that animals are content with their immediate environment.7–9 Consequently, it can therefore be assumed that animals would also breed better when content with their environment.10,11 Mice, for example, are typically housed in a relatively narrow temperature range varying from the German standard of 20 to 24℃ to the US American standard of 20 to 26℃.6,12 In practice, most mice are housed in a much narrower range of about 20 to 22℃, conveniently at the temperatures that match the human comfort zone. 13 However, mice are able to alter their microenvironment through their own behaviour by nesting and huddling. 14 This behavioural thermoregulation may no longer be so efficient with modern mouse facilities that utilize effective room ventilation and individually-ventilated cages (IVCs). 15 Thus, the way mice experience their thermal environment could play an important role in improving the welfare of experimental animals.
In contrast to the common practice of housing mice at about 22℃, a growing number of studies seem to suggest that mice could thrive in higher temperatures. It has been known for decades that the thermoneutral zone (TNZ) of a mouse, the so-called ambient temperature range in which the metabolic rate of an animal is minimal, is between 28℃ and 34℃. 16 Higher ambient temperatures of up to 32℃ could even in some cases be advantageous for mouse reproduction. 17 Furthermore, mice housed under routine animal facility room temperatures at around 22℃ can already be under chronic cold stress. 18 Karp 13 concludes in his review that there is already a considerable amount of data suggesting that the cold stress endured by a laboratory mouse under standard room temperatures profoundly affects its physiology, and that we may therefore be better off keeping laboratory mice at higher ambient temperatures. Indeed, when given a choice, mice often seem to prefer higher ambient temperatures.19,20 Therefore, the suggestion that many experiments in the past may have been performed under suboptimal conditions is hardly surprising, 21 and leads to the conclusion that mice could indeed be held in warmer temperatures. 22 To date, however, the effect of warmer ambient temperature for the breeding performance of mice has not been thoroughly studied.
The aim of this study was to determine whether higher ambient temperatures influence the reproductive fitness of mice housed in IVC systems. We looked into pregnancy rates as well as embryo and sperm yield and quality as indicators of good welfare in connection with the production of transgenic mice via DNA microinjection.
Materials and methods
Animals and husbandry
Mice (C57BL/6JOlaHsd), maintained in pathogen-free conditions (according to FELASA 2002 recommendations), were housed in an IVC system (type 1145T; Tecniplast, Buguggiate, Italy). Each cage was provided with aspen bedding and nesting material (Tapvei, Paekna, Estonia) and was routinely changed once per week under laminar flow (InterActive Cage Changing Station; Tecniplast). Pelleted autoclaved mouse diet (Harlan Teklad 2018S; Harlan, Indianapolis, IN, USA), as well as filtered and acidified (pH 3.0) water, was provided ad libitum. The standard room climate, unless experimentally changed, was maintained at 22 ± 1℃, and at a relative humidity of 55 ± 10%. Light–dark cycle was 12:12 h with lights on at 05:00 h.
Female mice were ordered weekly from the vendor (Harlan, Horst, The Netherlands) at an age of three weeks, placed directly either in the control or the experimental room, and subsequently used for collecting embryos. Male mice were ordered before the beginning of the experiments at an age of eight weeks, placed singly in cages in both the control and experimental rooms, and test-bred once with fertile females. A pool of 30 males was set up for both conditions, and males were used for breeding on average three times per week.
All animal housing, handling and the experimental techniques were in accordance with the principles set out in the Declaration of Helsinki, as well as in accordance with the ethical standards of the European and German Animal Welfare legislation. Experiments were planned and conducted to adhere closely to the 3R (replacement, reduction, refinement) principles of animal welfare. All procedures were licensed and carried out according to the Institutional Animal Care and Use Committee guidelines as regulated by the German Federal law governing animal welfare.
Study design
The temperature of the experimental animal room was modified using an electric heater installed inside the supply air duct, and when insufficient, two additional stand-alone electric heaters with thermostats were used within the room. The air humidity was maintained constant by using an additional air humidifier (B300; Brune, Aglasterhausen, Germany). The ambient temperature and humidity were recorded using the building’s in-built monitoring system with gauges installed in the supply and exhaust air ducts (IBS8, Messner, Schkeuditz, Germany), and verified by placing temperature data loggers (SK100; Dickson, Addison, IL, USA) in different parts of the room, and inside the cages.
Experiments were performed within a time frame of 12 weeks. During this period the control room was kept at a constant temperature of 22 ± 1℃ and a relative humidity of 55 ± 10%. The data from the control group were obtained over 47 experimental days involving 290 mice. During the same period the experimental room was used with different ambient temperatures. The data were obtained over 22 experimental days at 25 ± 1℃ (125 mice), 15 experimental days at 28 ± 1℃ (90 mice) and 10 experimental days at 30 ± 1℃ (60 mice). The relative humidity was maintained constant throughout the experiments at 55 ± 10%. There was no statistical time-related bias between the different weeks, so the control group was used as a whole when compared with different experimental conditions. Each experimental day included an average of six mice per experimental group. For all conditions mice were acclimatized for three days before any experiments were performed.
On day zero each female mouse received an intraperitoneal injection of five international units (IU) of pregnant mare’s serum gonadotropin (PMSG), followed by an intraperitoneal injection of 5 IU of human chorionic gonadotropin (hCG) 46 h later. Females were mated with fertile males immediately after hCG injection. Early in the morning of day 1, mice were checked for vaginal plugs and euthanized by cervical dislocation. Embryos were collected and counted from all mated mice. During embryo collection the swelling of the ampulla of each female was observed to verify the success of the hormone treatment. The embryos from mice in both experimental and control rooms were scored and used for routine production of transgenic mice via pronuclear injection of DNA.
Scoring embryos
The embryos, collected with a cumulus complex isolated from the ampulla, were separated from the cumulus cells by treating them for 5 min with 300 µg/mL hyaluronidase (Sigma 3884, Sigma-Aldrich, St Louis, MO, USA). Embryos were then transferred in the first drop (100 µL) of the culture medium (M2) and the number of embryos per female was counted under the stereomicroscope. During the subsequent wash procedure (transferring embryos from one drop of culture medium to the next for 10 times), only healthy looking embryos were selected in each transfer step (regular round form, intact zona pellucida). In the last drop, the embryos were counted again and scored as ‘Good Quality’. Embryos were then placed in a drop of KSOM incubation medium covered with oil for 1–2 h until they were used for injection. Injections were performed under a microinjection stage (Zeiss Axiovert 200M [Carl Zeiss AG, Oberkochen, Germany]; Eppendorf TransferMan NK2 [Eppendorf AG, Hamburg, Germany] and Eppendorf Femtojet [Eppendorf AG]), and only those embryos that had well-developed pronucleus were used for DNA injection. These embryos were counted and scored as ‘Injectable Quality’. After the DNA injection, the embryos were further incubated for 2–3 h, and the numbers of embryo that did not survive this period were scored as ‘Lysed’. All surviving embryos after this stage were transferred to pseudopregnant recipient females.
Scoring sperm
In the second experimental week a few males were selected from each ambient temperature for sperm analysis. The males used were all proven breeders from the general pool of males used for weekly mating. To isolate the sperm, the epididymis and the first few millimetres of the vas deferens were dissected, and the surrounding fat tissue was carefully removed in 0.9% NaCl. Both epididymis were transferred to 170 µL of cryoprotective agent (CPA; 18% raffinose and 3% skim milk, prepared by the Transgenic Core Facility, and stored at –20 °C for up to six months), opened with forceps, and incubated for 5 min at room temperature to allow the sperm to be released. An aliquot (4 µL) of the sperm suspension was transferred to 196 µL of human tubal fluid (HTF) medium and incubated for 5 min at 37℃. An aliquot of 30 µL was transferred to a Standard Count 2 Chamber Slide from Leja (Nieuw-Vennep, The Netherlands) and measurements were made with a Hamilton Thorne Automated Sperm Analyser (Hamilton Thorne, Beverly, MA, USA) with default settings for mouse sperm.
Statistics
Data were processed and analysed using PASW Statistics 18.0 software (SPSS Inc, Chicago, IL, USA). All data were tested for normal distribution (Shapiro–Wilk) and homogeneity of variance (Levene’s test). One-way analysis of variance (ANOVA) was performed for all data, followed by the post hoc analysis (Tukey’s HSD). Additionally, the linear regression analysis was performed for the sperm data. The statistical significance level was set at P < 0.05. All results are shown as means unless otherwise indicated.
Results
Embryo yield is not affected by increased ambient temperatures of up to 28°C
The yield of vaginal plug positives from mated females (VP+), the yield of swollen ampullas, indicating the success of the hormone treatment, from mated females (SA), the yield of embryos from mated females (EM), and the yield of embryos per hormone positive response (EH).
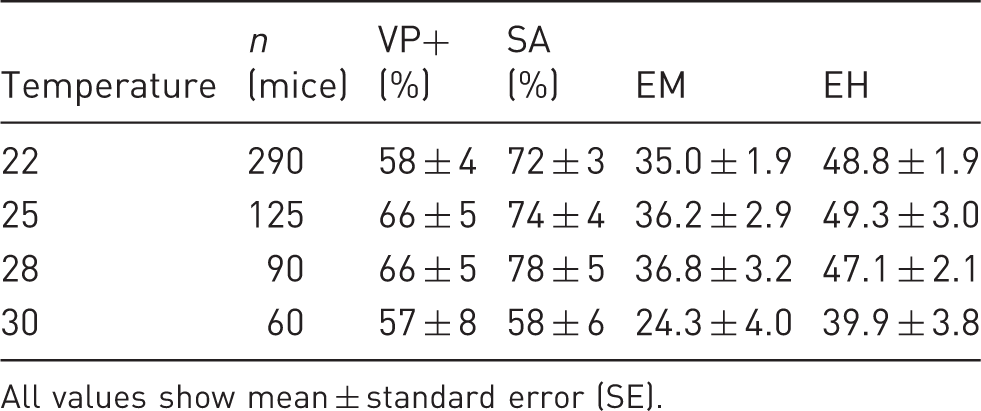
All values show mean ± standard error (SE).
The hormone treatment does not only increase the production of oocytes, it also times the oestrus cycle so that the maximum number of copulations can be anticipated in any given day. In order to confirm the potential pregnancy of a mated female mouse, the authors investigated the success of the copulation by observing vaginal plugs the morning after mating. 4 The yield of vaginal plugs was highest at 25℃ and 28℃ (both 66%) and lowest at 22℃ and 30℃ (58% and 57%, respectively) (F(3, 90) = 0.85, P = 0.471) (Table 1). These results show that the hormone treatment produced a good response from 22℃ to 28℃, and that the yield of vaginal plugs was not affected by the higher temperatures of 25 or 28℃, but was slightly reduced at both 22℃ and 30℃.
Although mating success can be estimated by observing the yield of vaginal plugs and swollen ampullas, it is not guaranteed by these. Therefore we need to determine the total yield of embryos. The best yield of embryos per mated female was obtained at ambient temperatures from 22 to 28℃ (35.0 to 36.8 embryos), whereas the embryo yield dropped notably (about 35%, 24.3 embryos) at 30℃ (F(3, 90) = 2.27, P = 0.085) (Table 1). The yield of embryos from females that had swollen ampullas, and therefore were likely to have been successfully mated with males, followed the same pattern as the general embryo yield, with most embryos produced by females housed in temperatures from 22℃ to 28℃ (F(3, 90) = 1.43, P = 0.239) (Table 1). These results show that embryo yield is not affected by increased ambient temperatures of up to 28℃, but is potentially reduced at 30℃.
Embryo quality is not affected by increased ambient temperature of up to 28°C
The proper development of embryos in vitro is essential for successful microinjection. To determine whether the collected embryos can be successfully cultivated for further use, embryo quality (indicated by a healthy round form and intact zona pellucida) was analysed. The yield of good quality embryos per mated female remained at the same range (from 32.1 to 33.3 embryos) from 22 to 28℃, but was reduced (21.7 embryos) at 30℃ (F(3, 90) = 2.09, P = 0.107) (Figure 1).
The yield of good quality embryos per mated female. Mean values are shown as white diamonds.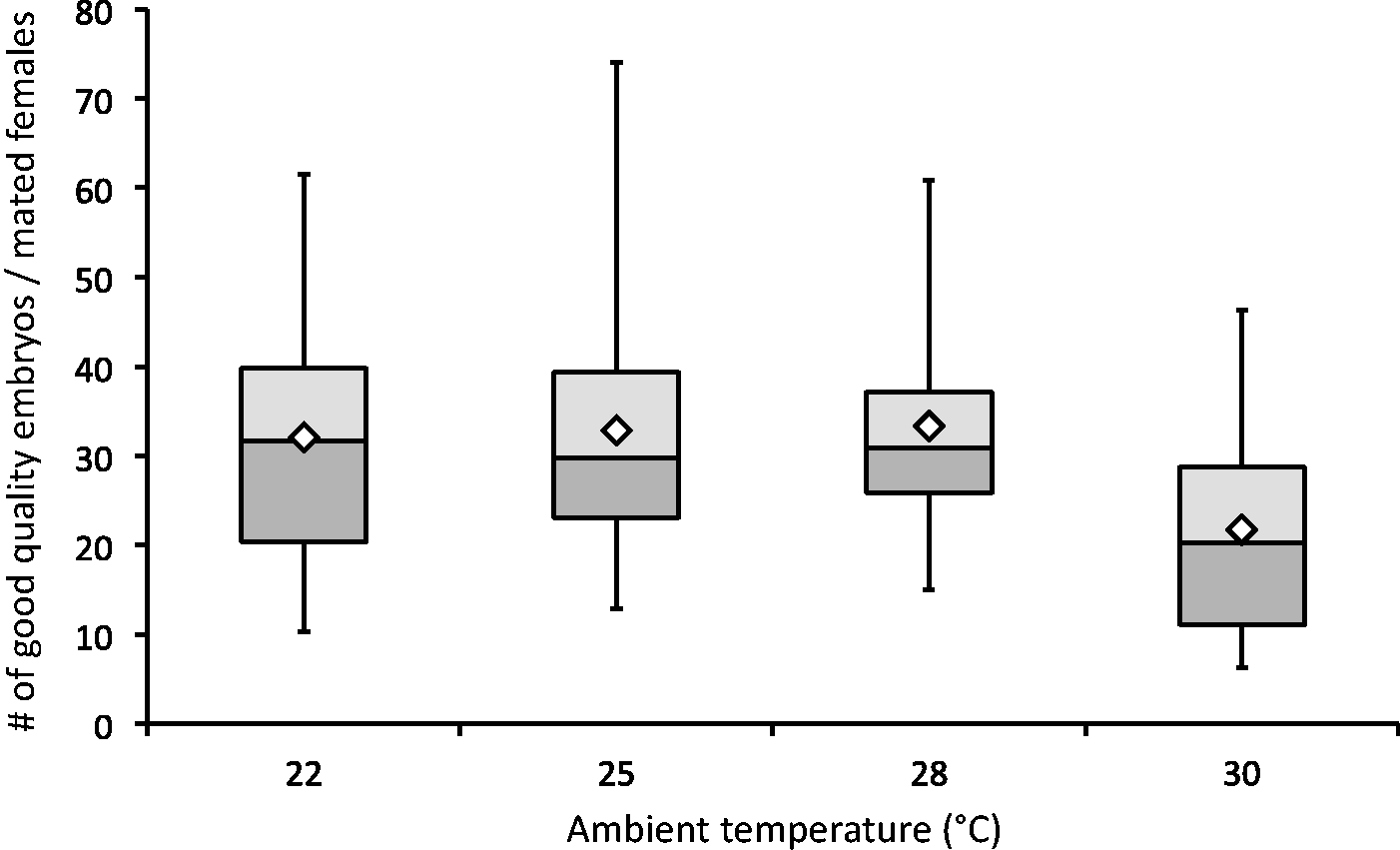
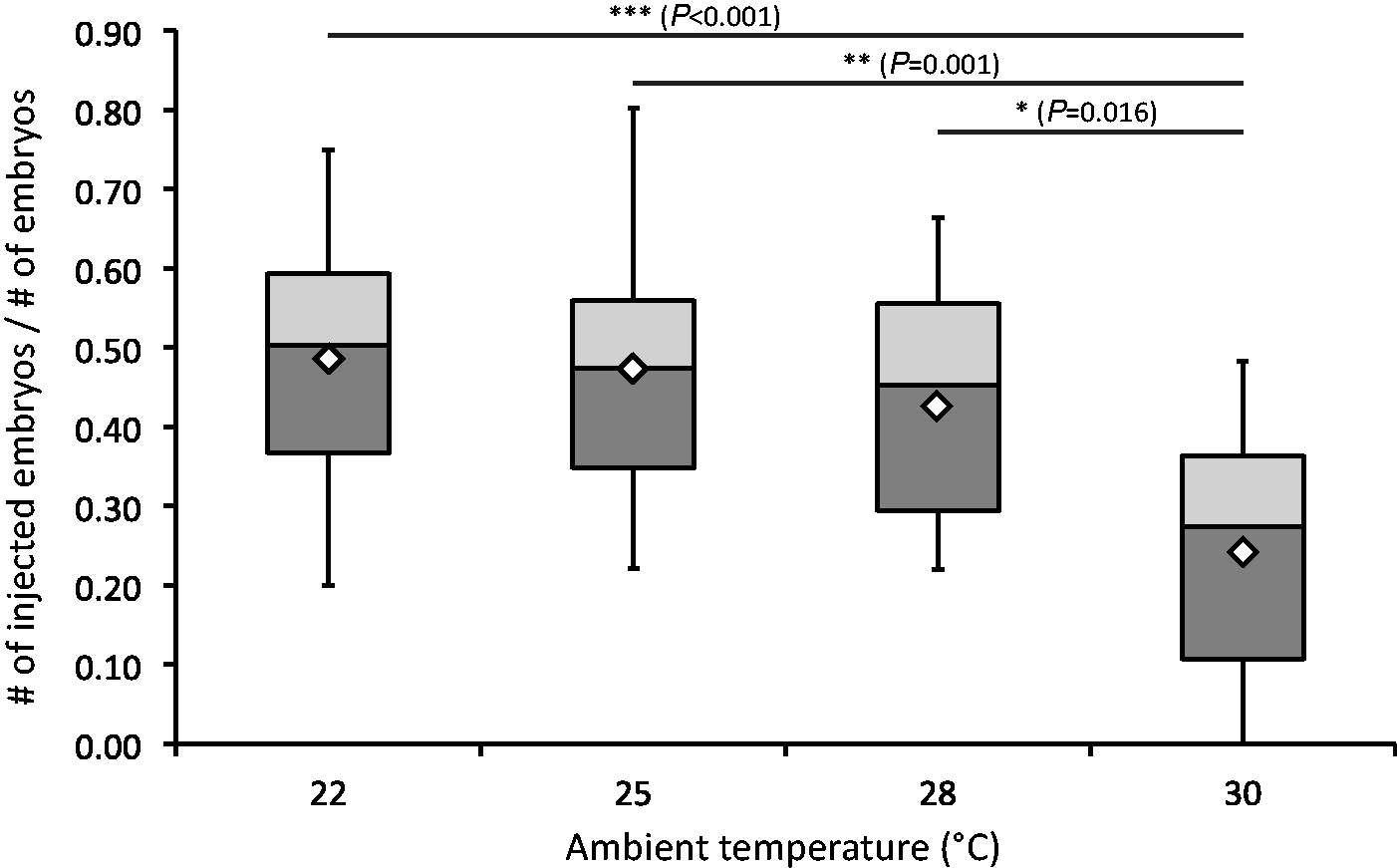
The injectable quality of embryos was examined next, as only embryos with properly developed and visible pronucleus could be successfully used for microinjection. The overall numbers of embryos with good injectable quality per mated female was highest at 22℃ (17.3), but not significantly lower at 25℃ (16.3) or at 28℃ (15.8). At 30℃ a significant drop was observed with only 6.0 injectable embryos per mated female (F(3, 90) = 5.69, P = 0.001) (Figure 2). Analogous results were obtained when the injectable quality was plotted against the overall number of embryos produced. The largest numbers of injectable embryos were found in the 22℃ (48.6%), 25℃ (47.4%), and 28℃ (42.6%) groups, but numbers were significantly reduced at 30℃ (24.1%) (F(3, 90) = 7.74, P < 0.001) (Figure 3). These results show that higher ambient temperatures of up to 28℃ do not affect the injectable quality of embryos. However, the quality is noticeably reduced at 30℃.
The amount of injectable embryos per mated female. Significance levels are shown as *P < 0.05 and **P < 0.01, (analysis of variance [ANOVA] followed by Tukey’s honest significant difference [HSD]). Mean values are shown as white diamonds. The relative amount of injectable embryos compared with total embryo yield. Significance levels are shown as *P < 0.05, **P < 0.01, ***P < 0.001 (analysis of variance [ANOVA] followed by Tukey’s honest significant difference [HSD]). Mean values are shown as white diamonds.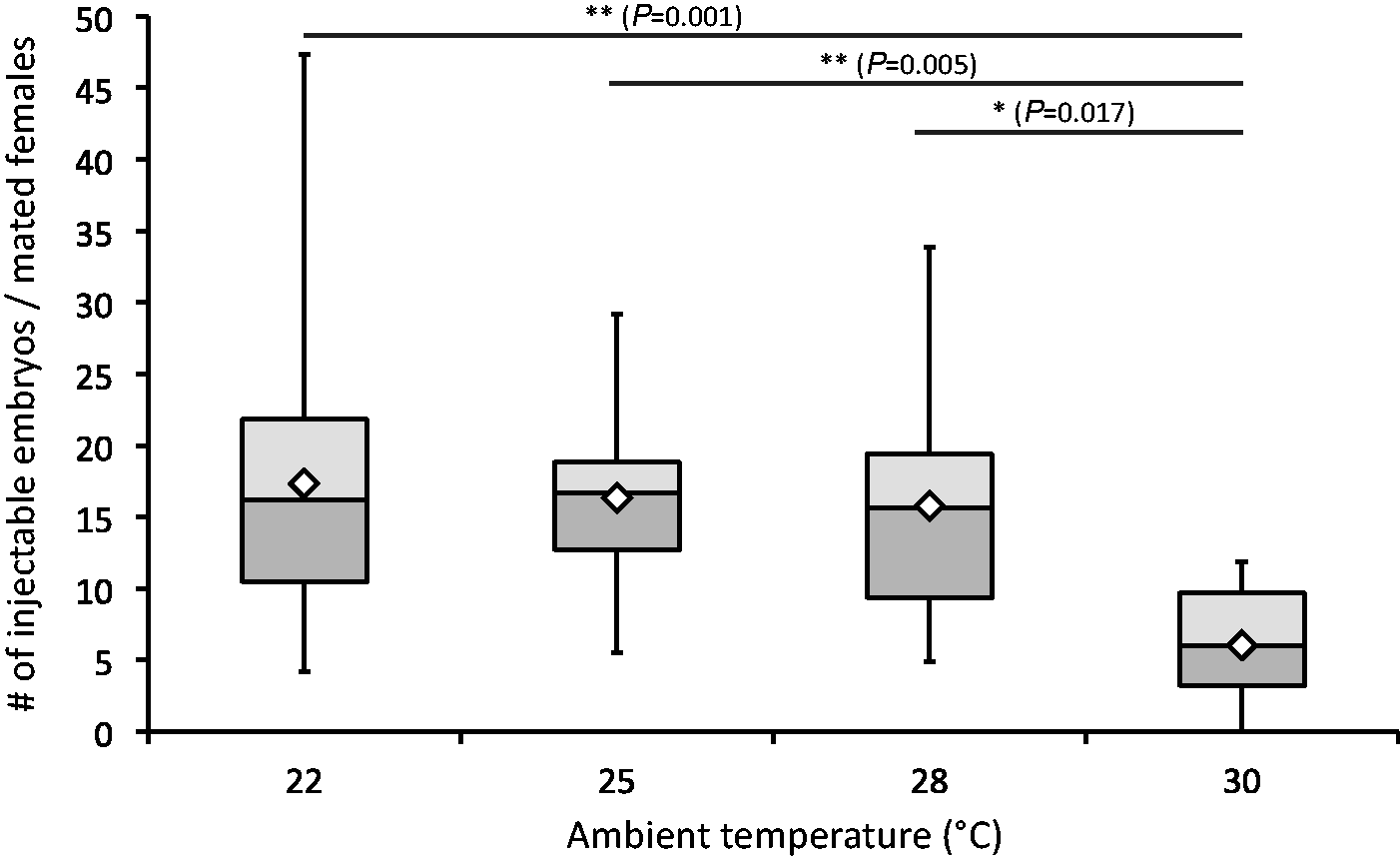
In order to test the robustness of the embryos that had been microinjected with DNA, the final embryo scoring was performed by counting the lysed (broken) embryos. The relative number of lysed embryos after the injection was at its highest at the ambient temperature of 30℃ (26%). By contrast, only 17%, 16% and 16% of the injected embryos were lysed at the three lower ambient temperatures of 22℃, 25℃ and 28℃, respectively (F(3, 88) = 1.88, P = 0.139). These results show that the higher ambient temperatures of up to 28℃ do not affect the robustness of the injected embryos.
Reproductive fitness remains excellent up to an ambient temperature of 28°C
Our observations that the female mouse reproductive system performs extremely well at higher ambient temperatures prompted us to ask whether this is only due to a preference of the female mice or if male performance also plays a role. In order to understand the role the stud males play in the mating success, the sperm count and quality were determined at each of the ambient temperatures. The average sperm count reduced continuously from 2048 (the actual number of cells counted in all fields analysed with 1:50 dilution) at 22℃ to only half that (1009) at 30℃ (F(3, 11) = 3.16, P = 0.068) (Figure 4, panel a), demonstrating a continuous reduction (significant linear regression (F(1, 13) = 10.75, P = 0.006) with an R2 of 0.453) of sperm count with increasing ambient temperatures. The authors next examined whether the quality of the sperm can influence the mating success by analysing sperm motility and progressivity (progressive motility). Sperm motility remained at a high level from 22℃ through to 28℃ (between 70% and 75%), but dropped significantly at 30℃ (45%) (F(3, 11) = 7.41, P = 0.005) (Figure 4, panel b). Sperm progressivity was highest at 25℃ (26% progressive), slightly reduced at 22℃ and 28℃ (23% and 21%, respectively), and significantly reduced at 30℃ (12%) (F(3, 11) = 12.94, P = 0.001) (Figure 4, panel c). Surprisingly, these results show that, unlike sperm count, neither motility nor progressivity is affected by higher ambient temperatures of up to 28℃. This indicates that sperm quality is a more significant factor than sperm count for mating success.
(a) Average sperm count (2048 [n = 3], 1575 [n = 5], 1306 [n = 4] and 1009 [n = 3] at ambient temperatures of 22 ℃, 25 ℃, 28 ℃ and 30 ℃, respectively). (b) Sperm progressivity. (c) Sperm motility. (d) Sperm motility (black triangle, solid line) and progressivity (black circle, dashed line) versus mating success shown as plugging frequency (left bar, light grey) and swollen ampulla frequency (right bar, dark grey). Mean values in panels (a), (b) and (c) are shown as white diamonds. Significance levels are shown as *P < 0.05, **P < 0.01, ***P < 0.001 (analysis of variance [ANOVA] followed by Tukey’s honest significant difference [HSD]).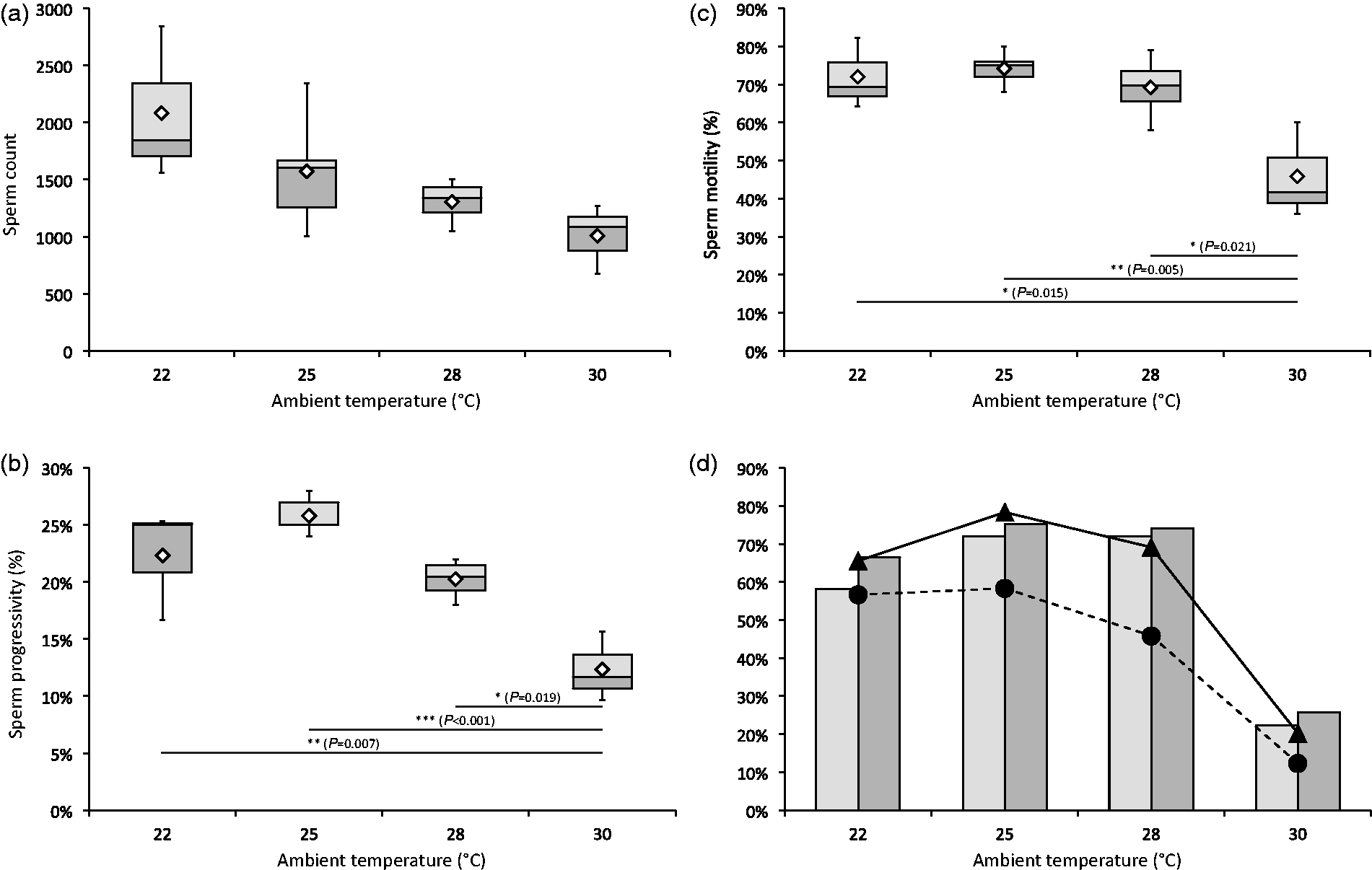
Finally, to determine the combined breeding performance of male and female mice, their reproductive fitness as a whole was looked into. When analysing the correlation of sperm motility and progressivity together with the mating success and swollen ampulla frequency, a clear tendency towards higher motility/progressivity resulting in better mating success was observed (Figure 4, panel d). When female reproductive fitness, defined as good embryo yield and embryo quality, was analysed in comparison with male sperm count, sperm count did not seem to influence female reproductive fitness until a threshold of roughly a 50% reduction in sperm count was reached (Figure 5). These results indicate that the reproductive fitness of a mating pair is more predictable by sperm quality and not by sperm count. In other words, sperm count does not play an important role for mating success as long as the quality of the sperm is maintained at a high level.
Female reproductive fitness, shown as embryo yield per mated female (grey triangle, grey dashed line), good quality embryo yield per mated female (grey circle, grey dashed line), injectable quality embryo yield per mated female (star, grey dashed line), plotted against male sperm count (black square, black solid line).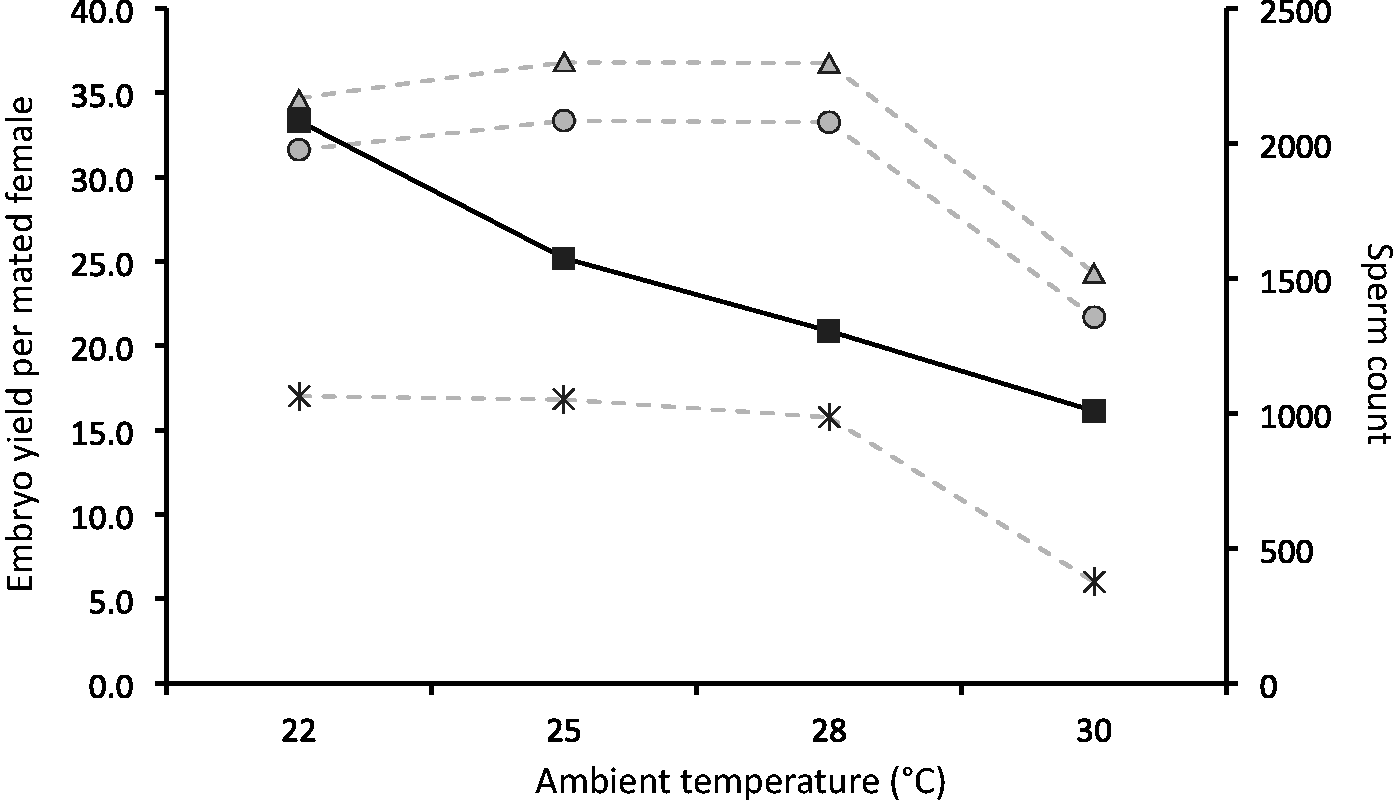
Discussion
This study demonstrates that the reproductive fitness of mice is maintained at a very high level up to an ambient temperature of 28℃. This was confirmed throughout the study – vaginal plug rate and response to hormone treatment, embryo yield and quality, and sperm quality all displayed an excellent reproductive outcome up to an ambient temperature of 28℃. This contrasts with current recommendations, which do not allow mouse facilities to function at temperatures above 24–26℃.6,12 The NIH Guide for the Care and Use of Laboratory Animals endorses a temperature range of 20℃ to 26℃, further recommending using the middle range as a standard. 6 Although the guide recognizes the preference mice may have for higher temperatures during resting times, it also acknowledges that mice often seek lower temperatures during high activity.14,23,24 Therefore, to avoid potential heat stress, the guide generally recommends mice are housed below the lower critical temperature (26℃) of the TNZ. 16 German standards, as well as European standards in general, set their recommendation for mice even stricter at 20℃ to 24℃, implying that temperatures above 24℃ should not be used. 12 Regulatory bodies (IACUC, Overseeing Governmental Officials) most often do not allow deviations from these temperatures for normal husbandry or breeding. Nevertheless, our results show not only that higher ambient temperatures do not have adverse effects on mouse reproduction, but that certain aspects of mouse reproduction, such as the presence of vaginal plugs, embryo yield and quality, or sperm quality, may even be improved from 25℃ up to 28℃. Although the commonly used mouse room temperatures of 20℃ to 24℃ are still suitable for normal husbandry, we must conclude that significantly higher temperatures may be preferable for optimal mouse reproduction. Based on our findings with mice housed in an IVC system, the current recommendations may be too rigid, and ought to be revised to allow more flexibility for mouse facilities to choose the best temperature.
We also demonstrate that at 30℃ all reproductive indicators used in this study dramatically deteriorate. The drastic drop in both number of embryos produced and their quality, as well as in sperm quality, happened very close to the lower threshold of the TNZ. This is physiologically interesting as it has been shown that mice housed individually actually prefer temperatures of between 26.5 to 29.5℃, and group-housed mice prefer temperatures of between 26.2 to 28.4℃, both the higher temperatures exceeding the lower threshold of TNZ.14,16 This raises the question of why this drop in reproduction took place at the lower threshold of the TNZ, instead of at the higher threshold? Unlike most long-lived mammals, laboratory mice are not seasonal breeders, and their ability or willingness to breed is probably not evolutionarily connected, at least under laboratory conditions, with seasonal factors such as photoperiod or foraging conditions. 25 The adaptation to a cooler environment could be promoted by offering a sufficient amount of nesting material. Gaskill et al. 26 have found that nesting material can affect the thermoregulatory processes of a mouse and can allow mice to be housed at lower ambient temperatures without added cold stress. This allows mice to shift their preferred temperature zone through their own behaviour. How much our results could have been influenced by the nesting material introduced in every cage during the routine cage change cannot be concluded from these data and would warrant further analysis in future studies. Moreover, earlier studies have shown a relative insensitivity to extreme temperatures among house and laboratory mice after longer adaptation, so it is possible that mice could adapt to temperatures of even 30℃, with high reproduction if given a chance. 27 However, since this study demonstrates that the reproductive fitness of mice housed in an IVC system is affected by an ambient temperature of 30℃, temperatures of 30℃ or higher therefore cannot be recommended for mouse breeding.
We also assessed how male reproductive factors could influence reproductive fitness of a mating pair. We observed a continuous drop in mouse sperm count at higher temperatures, effectively halving the sperm yield when the ambient temperature was raised from 22℃ to 30℃. Interestingly, whereas we observed the sperm count decreasing with increasing temperatures, the sperm motility remained constant up to 28℃ before dropping significantly at 30℃. Furthermore, the most progressive sperm was found from mice housed at 25℃, although the progressivity was still high at both 22℃ and 28℃. Low sperm quality at high ambient temperatures has been observed for many mammalian species – this has also been demonstrated with mice in a study by Yaeram et al. 28 where mice housed at the higher ambient temperature were shown to have lower fertility. It is also known that short exposures of male mice to elevated temperatures can lead to decreased sperm concentrations, motility and viability. 29 Our results suggest that male sperm count is not an independently significant factor when impregnating female mice – sperm count had to drop by about 50% before resulting in any effect on reproductive fitness of a mating pair, and this was probably due more to the drop in the motility and progressivity than to a lower amount of sperm. It appears that, until a certain threshold at least, sperm motility and progressivity have a significantly greater influence on reproductive fitness than sperm count. This concurs well with what is know about human sperm quality, where the concentration rather than absolute quantity of the motile spermatozoa is considered to be the most significant parameter predicting the chance of natural conception. 30
Allowing mouse facilities more flexibility to select the most optimal temperature for mouse holding, breeding and experiments could be of great economic and environmental importance. First, the climate in mouse facilities may not need to be regulated so rigorously as is currently recommended or demanded by legislation (commonly 22 ± 1℃). Wider permissible temperature range (i.e. 25 ± 3℃) would allow more efficient and economical technology to be installed. Second, mouse facilities located in warmer climates may opt to permanently run their mouse rooms at a higher temperature, leading to significant cost savings as higher animal room temperatures would result in lower energy consumption (and potentially smaller climate control units) needed for cooling. To conclude, a wider temperature range and higher room temperatures would not only lead to potentially significant cost savings but might also have positive environmental effects via a reduced CO2 footprint as well.
The facilities which regularly produce transgenic mice should also consider the effect of temperature to be able to improve their production of embryos and at the same time fulfil the principles of the 3Rs. 31 Using higher ambient temperatures in animal rooms may result in fewer animals being used for transgenic mouse production. Many indicators in our study, such as plug rates, hormone response, sperm motility and sperm progressivity, suggest that ambient temperatures from 25℃ up to 28℃ may even be preferable. On the other hand, informal discussions with animal care personnel at our facility raised the concern that they felt the animal rooms to be uncomfortably hot at 25℃ and above – this could be of a practical concern to animal care staff if higher temperatures were being used. Different working clothes and use of closed cabinets separating a warmer climate of the mice from that of the animal care staff could be considered.
Conclusions
Our data show that ambient temperatures of up to 28℃ can be tolerated by mice without an adverse effect in their early reproductive fitness (pregnancy rate, embryo yield and quality, sperm yield and quality). Furthermore, our data show that the shift from 28℃ to 30℃ results in a significant drop in both male and female reproductive fitness. We conclude that higher ambient temperatures than currently recommended could be used in the context of good performance in early reproduction of mice, and therefore current recommendations for mouse holding temperatures ought to be revised. Higher mouse room temperatures could also lead to significant cost savings regarding ventilation and cooling.
Future perspectives
As this study concentrated only on the effects of higher ambient temperatures on the reproductive success of mice, it would be interesting to determine whether colder temperatures below 22℃ would be more or less suitable, and at what (lower) temperature would reproductive fitness decline. Furthermore, as in this study the relative humidity was kept constant at 55 ± 10%, it would also be interesting to look at the effect of different animal room humidity on embryo quality and quantity. There is some evidence that higher humidity could lead to lower embryo yield 32 – recent observations from our facility (not published) indicate a similar relationship. Finally, in this study the standard superovulation protocol was used in order to minimize the number of female mice needed for embryo collection. It would be interesting to see whether the same effect with higher temperatures on reproductive fitness would be observable with naturally-mated mice as well.
Footnotes
Acknowledgements
We thank the following services and facilities of the Max Planck Institute of Molecular Cell Biology and Genetics for their support: Biomedical Services, Transgenic Core Facility and Photolab. We would like to thank especially Tina Mittasch, Anke Muench-Wuttke and Wolfgang John for technical assistance. We gratefully acknowledge Iain Patten and Ivan C Baines for their critical reading of the manuscript. We thank P Zerial for her statistical support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study acknowledges support from the Max Planck Institute of Molecular Cell Biology and Genetics, Dresden, Germany. This research project received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
