Abstract
For a wide range of biomedical approaches, an accurate estimate of the age of embryos or pups is important. Overnight mating is the method that is mostly used to establish timed pregnancies. The oestrus cycle in mice repeats every four to five days. So, not all females will get pregnant because they are not in oestrus. Therefore, the aim of this study was to analyse whether polygamous mating could increase the rate of timed pregnancies per breeding cage and female. We compared overnight timed mating regimes with up to four females per male, using C57BL/6 and BALB/c mice as well as F1 hybrids of these two strains. The number of vaginal plugs, number of females that gave birth and weaned litter (including size and weaning weight) were recorded. Our results showed that the plug and pregnancy rate decreased, but the productivity per breeding cage increased for polygamous mating regimes. The proportion of females with vaginal plugs and females that gave birth was significantly higher in monogamous mating. The proportion of plugged females that gave birth, as well as litter size and weaning weight, were not influenced by the mating regime. After analysing 513 breeding cages with a total of 1090 females, we found that polygamous mating with up to three females per male can increase the number of timed pregnancies. However, in the mating regime with more than three females, the rate of timed pregnancy as well as number of pups per female declined.
Introduction
Selection of the best mating regime for the induction of timed pregnancies is important for variety of approaches such as cryopreservation, hygienic re-derivation programmes and generation of genetically modified mouse models, as well as in developmental biology research. In all these approaches, embryos or offspring at defined developmental stages are needed.
At first sight, it seems appropriate to use polygamous mating regimes for the production of a large number of progeny if the reproductive performance of the male is able to afford this. In addition, females in permanent polygamous mating tend to raise their offspring together in one or two nests. However, successional studies by Peters and Festing 1 showed that mating ratios of more than two to three females per male resulted in a significant decline in the productivity per female in some strains.
Due to an exponential growing number of mouse models 2 and more stringent legal regulations for husbandry (Directive 2010/63EU, Appendix A), space is a limiting factor in many animal facilities. Quite often, researchers prefer polygamous mating regimes to save breeding cages. However, to our knowledge, whether this is also the most effective approach to generate timed pregnancies has not yet been examined.
Therefore, the aim of our study was to find the best reproductive mating regime for timed pregnancies using the inbred mouse strains C57BL/6 and BALB/c. To reduce inter-individual differences of embryo development, overnight matings were arranged. In line with the findings of Peters and Festing, 1 we mated one male overnight with up to four females.
Methods
Animals and husbandry
C57BL/6JHanZtm (B6), BALB/cJHanZtm (C) and F1 hybrids were bred at the Central Animal Facility of the Hannover Medical School, Hannover, Germany. We used 10 strain combinations (female × male): B6 × B6, C × C, B6C × B6, B6C × C, B6 × B6C, C × B6C, CB6 × B6, CB6 × C, B6 × CB6 and C × CB6.
All mice were bred and maintained in an individually ventilated caging system (IVC, BioZone, Orlando, FL) in type 22 cages (440 cm2) with positive pressure under controlled standardised conditions: temperature 21 ± 2℃, relative humidity 50 ± 5%, artificial light (14 h light, 10 h dark), sterilised commercial softwood granulate bedding (Lignocel; Altromin, Lage, Germany) and additional nesting material (Nestlets; ANT Tierbedarf, Buxtehude, Germany). The mice received autoclaved commercial pelleted diet (Altromin 1314) and autoclaved water ad libitum.
Females were kept in sibling groups until mating, whereas males were separated at weaning. The microbiological status was examined as recommended by the Federation for Laboratory Animal Science Associations. 3 The mice were free of the listed microorganisms, except for Pasteurella pneumotropica and Helicobacter hepaticus.
The experiments were performed in accordance with the German Animal Welfare Legislation, approved by the local Institutional Animal Care and Research Advisory Committee and permitted by the Lower Saxony State Office for Consumer Protection and Food Safety (reference number 42500/1H and 13A404).
Mating regime
Mice were used for mating an age of approximately 8–10 weeks old. Each male was mated overnight to one (1:1), two (2:1), three (3:1) or four (4:1) females. Mating was set up in the afternoon around 4:00 pm by transferring the females into the male’s cage. Depending on the reproductive performance of a strain combination, we set up 5–24 ‘breeding cages’ per mating regime and strain combination. The order of mating was arranged randomly to exclude seasonal effects. The females were checked once for the presence of a vaginal plug the next morning, approximately one hour after the lights were turned on again. In most cases, the vaginal plug is visible by inspection of the vaginal area. Plug-positive females were marked, and all females were re-transferred to their home cage. Males were not reused for this study. Visible pregnant females were separated shortly before parturition. In total, 1090 female mice were mated to 513 males.
Mating regimes were assessed by recording following characteristics: vaginal plug rate (VPR) – proportion of females with a visible vaginal plug; plug pregnancy rate (PPR) – proportion of females with a plug that gave birth; pregnancy rate (PR) – proportion of all females that gave birth; productive cages (PC) – proportion of cages that produced at least one litter; litter size – the number of live pups per female counted at weaning (females without litter were not included); productivity per female – the number of live pups referred to all females (females with no litter were also included); and productivity per breeding cage: the number of weaned pups per cage (i.e. number of weaned pups sired by one male). All breeding cages were included.
Statistics
We used Biomath 4 to calculate the group size and Shapiro–Wilk test to show if the results were normally distributed. 5
Results were analysed using GraphPad Prism v5 (GraphPad Software, San Diego, CA). The experimental unit was the respective mating regime (i.e. 1:1, 2:1, 3:1 and 4:1). To test whether the VPR, PPR and PR of each female correlated with the mating regime, we used chi-square statistics (Fisher’s exact test). A p-value of < 0.05 indicated a correlation. Kruskal–Wallis tests with Dunn’s multiple comparison tests were used to analyse litter size, weaning weight, as well as productivity per female and breeding cage, because these results were not normally distributed. p-Values of < 0.05 were considered significant. Because the reproductive performance of some strain combinations was lower than expected, we increased the number of breeding cages for some mating combinations to meet the assumptions of the statistical approach.
Results
Reproductive outcome without considering the pairing combination
Correlation of VPR, PPR, PR and PC for all offspring combinations.
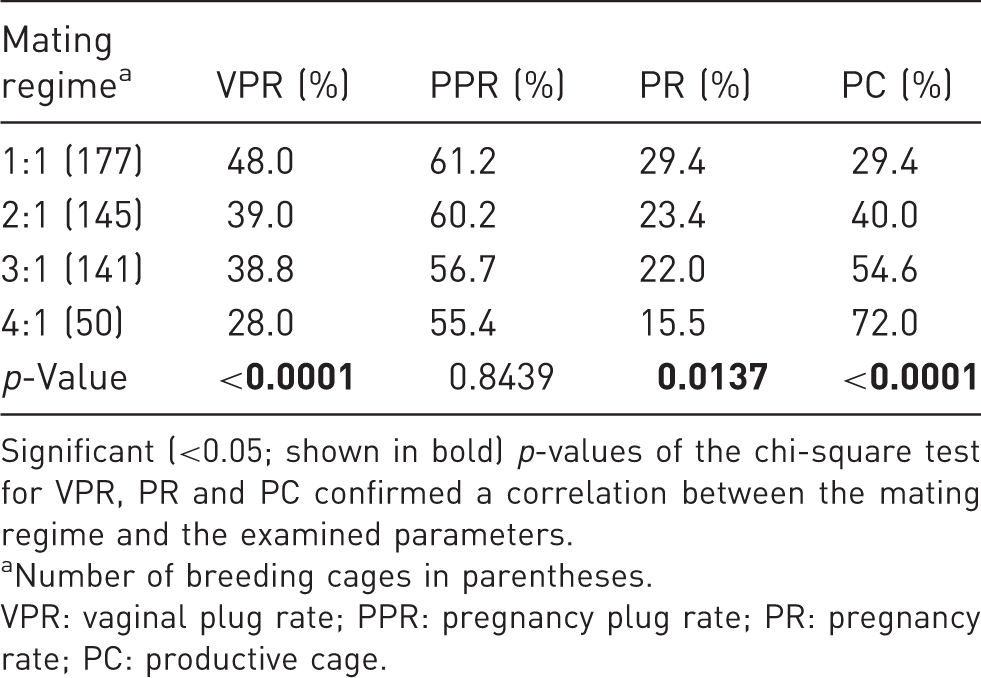
Significant (<0.05; shown in bold) p-values of the chi-square test for VPR, PR and PC confirmed a correlation between the mating regime and the examined parameters.
Number of breeding cages in parentheses.
VPR: vaginal plug rate; PPR: pregnancy plug rate; PR: pregnancy rate; PC: productive cage.
Only a few cages produced the maximum possible number of litters. In the mating regime with two females 33.79% (49/145) of the breeding cages produced one litter. Only nine (6.20%) had two litters. In cages with three females, 46.10% (65/141) produced one litter, 8.51% (12/141) produced two litters and none had three litters. In cages with four females, 60% (30/50) produced one litter, 10% (5/50) had two litters (10.00%) and 2% (1/50) had three litters. In 3:1 and 4:1 pairings, the maximal possible number of litters per cage (i.e. three and four litters, respectively) was never achieved.
The number of pups produced per breeding cage increased with the number of breeding females, but only up to three females. However, this number declined when four females were mated to one male (Table 3). This increase or decrease in the number of pups was significant for the 1:1 mating regime compared to the 3:1 and 4:1 regimes, as well as 2:1 compared to 3:1. Furthermore, the number of pups per female (all females included) decreased in 4:1 mating. The difference was significant for the 1:1 mating regime compared to the 4:1 regime.
Reproductive outcome per breeding cage and female (without considering the strain combination).

The increase in number of pups/cage was significant from one to three and four females,* as well as from two to three females.# The decrease of pups/female was significant for mating regime 1:1 compared to 4:1.§ Differences in litter size were not significant. Results are given as the mean ± standard error of the mean (SEM).
Reproductive outcome for the different strain combinations
The results for the analysed parameters differed in the single strain combinations (Table 3). The reproductive performance of the intra-strain pairings (B6 × B6 and C × C) declined dramatically when four females were mated to one male. In addition, a mating regime of up to three females also seemed to be superior to the mating to four females in other strain combinations analysed in this study.
Reproductive outcomes for the strain combinations.
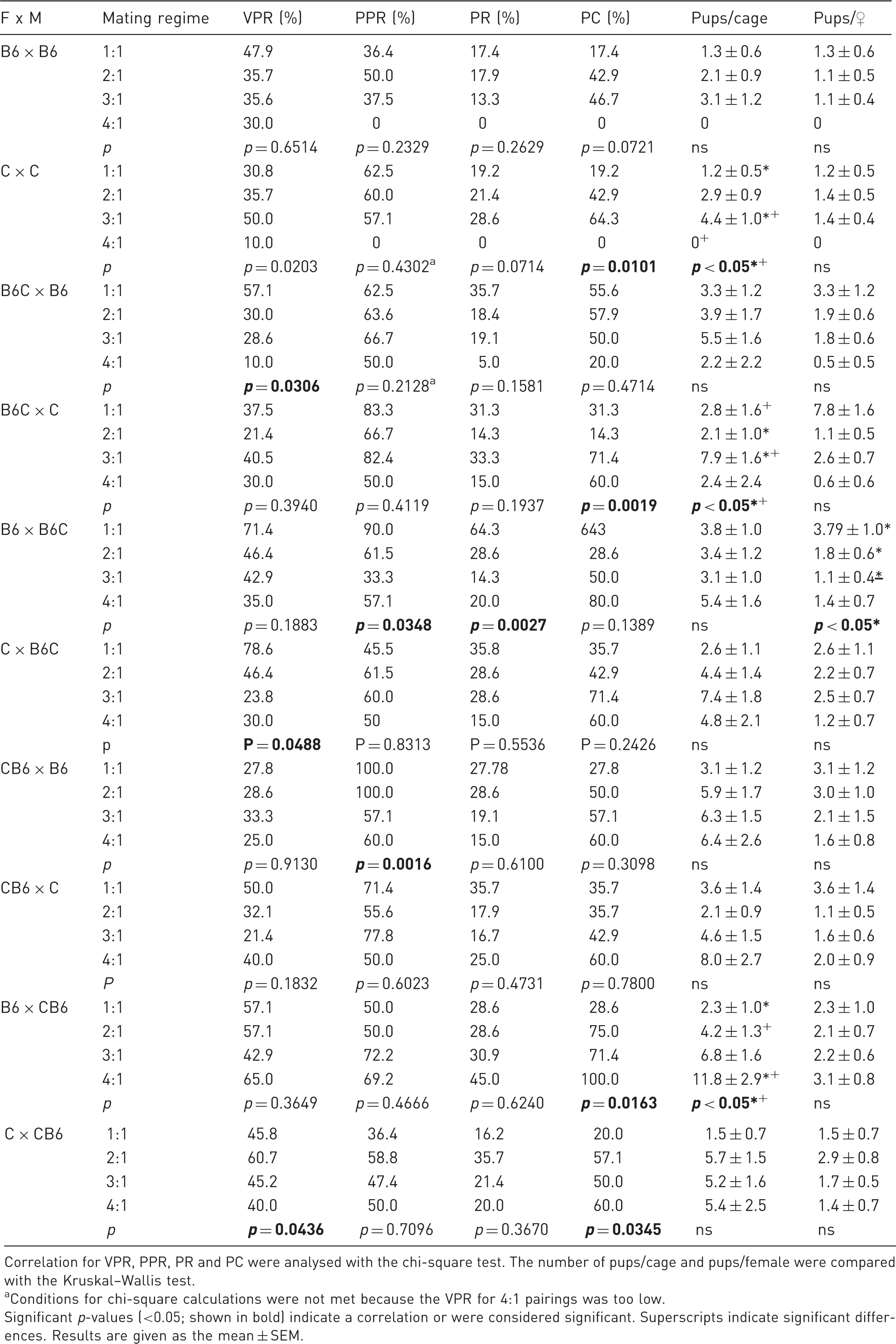
Correlation for VPR, PPR, PR and PC were analysed with the chi-square test. The number of pups/cage and pups/female were compared with the Kruskal–Wallis test.
Conditions for chi-square calculations were not met because the VPR for 4:1 pairings was too low.
Significant p-values (<0.05; shown in bold) indicate a correlation or were considered significant. Superscripts indicate significant differences. Results are given as the mean ± SEM.
Litter sizes and weaning weights for the different strain combinations (females without litter were excluded).

Litter size and weaning weight were not influenced by the mating regime. Therefore, the mating regime was not considered here.
No litter for mating regimes 4:1 for B6 × B6 and C × C. The most pups/litter were born in combination CB6 × B6. The difference was significant compared to combinations B6 × B6, C × C, B6 × B6C and B6 × CB6. The smallest litters were found in combination C × C. The difference was significant when compared to combinations B6C × B6, B6C × C, C × B6C, CB6 × B6, CB6 × C and C × CB6.
The lowest weaning weight was found in strain combination B6 × B6. The difference was significant compared to B6 × B6, B6C × B6, B6C × C, B6 × B6C, C × B6C, CB6 × B6, CB6 × C, B6 × CB6 and C × CB6. The highest weaning weight was found for strain combination C × CB6. The difference was significant compared to combinations B6 × B6, B6 × B6C and B6 × CB6.
Results are given as the mean ± SEM.
Figure 1 summarises the results for all analysed parameters. Except for the proportion of PC, the most strain combinations showed the best results after applying the 1:1 mating regime.
Success of mating regime for the analysed parameters. VPR: vaginal plug rate; PPR: plug pregnancy rate; PR: pregnancy rate; PC: productive cages (cages with a litter). The height of the bars corresponds to the number of strain combinations (y-axis), with the best results of the parameter given in the x-axis.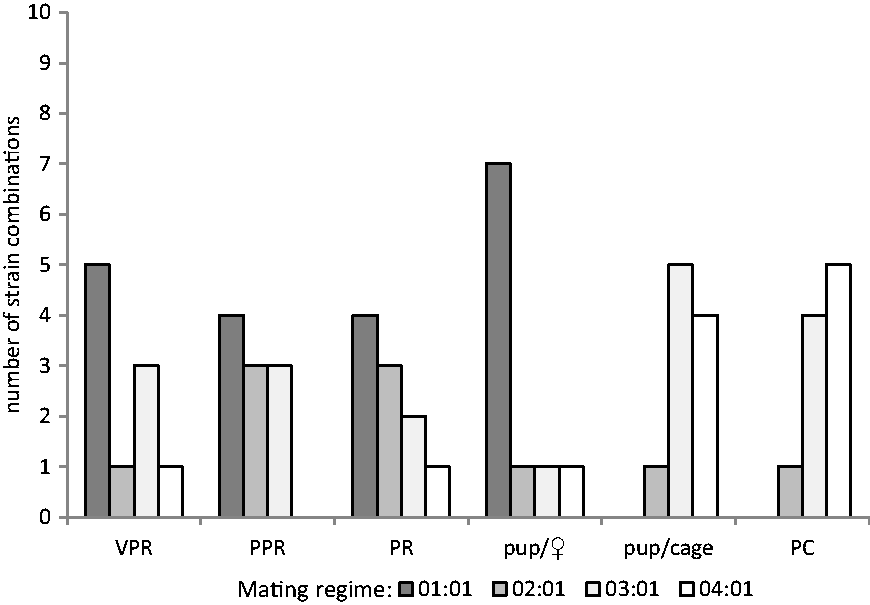
Discussion
The aim of our study was to find the most appropriate mating regime in mice for timed pregnancies when defined embryonal stages or offspring are needed. This mating regime should be economic (i.e. saving time and money) and effective at the same time. In contrast to Byers et al., 6 we decided not to determine the stage of the oestrus cycle. We also did not synchronize the females with hormones or by using the Whitten effect with co-housing as suggested by Stiles et al. 7 Surprisingly, even without using such approaches, we observed more plug-positive females after overnight mating than expected. Whitten 8 showed that only 13–14% of young virgin females had a vaginal plug on the first two days after mating. Only on the third day after mating did the VPR increase to >45%. This effect was also observed when females were only exposed to male pheromones that are present in bedding or carried by the air from a male’s cage over a distance up to 2 m. 9 This phenomenon was termed the Whitten effect. We observed almost the same VPR after overnight mating in the monogamous mating group (48.02%; Table 1). In addition, VPRs were also above the expected values for the pairings with more than one female. The inbred strains used in this study (C57BL/6JHanZtm and BALB/cJHanZtm) have been separated from the respective strains of the Jackson Laboratory since the early 1970s. Analyses confirmed the genetic difference. The reproductive performance, however, is not altered (Wedekind, unpublished data). We also never observed that males copulated a female not in oestrus – this would have been visible next morning. So, the separation from the original strains seems not to be an explanation for the increased VPRs. One explanation for increased VPRs might be that in our IVC system, pheromones were distributed from one cage to another, causing the Whitten effect. This, however, has to be evaluated in more detail because we excluded a direct airflow from cage to cage.
Independent from the fact, that the overall VPR was higher than expected in our IVC system, it decreased when more females were mated to one male. This is contrary to the assumption that the chance of there being a female in oestrus should be higher in polygamous mating regimes. One explanation could be that different inbred strains have different times for recovery of their libido (defined as time between attempted matings). For example, libido recovery time for DBA/2 males has been described as being one hour, whereas in B6 males, it can be up to four days. 10
Furthermore, not all females with a vaginal plug got pregnant. A vaginal plug only confirms that copulation and ejaculation occurred. The PPR is influenced by the genetic background 11 but also varies within a strain. 12 The PPRs in our study were in the range of published data, and a correlation with the mating regime could not be verified. However, we saw a tendency that more plugged females gave birth after monogamous mating. Although a negative impact on well-being in polygamous mating regimes has not been shown, the mating regime can influence the behaviour of female mice. 13 Furthermore, recent studies have shown that the number of offspring and/or number of two-cell embryos was reduced after polygamous mating in some strains.13,14 A study by Smarr et al. 15 showed that the success of pregnancy might also be influenced by the physiological state of the female at the time of pairing. Therefore, a decreased PPR could be explained by a higher physiological stress level of the females in polygamous pairings. Also, a decrease in sperm quality and/or sperm count after repeated ejaculations might be an explanation.
As expected, the proportion of PC increased with the increasing number of females per cage. This indicates that at least one female in the cage was in oestrus. However, very seldom did all females become pregnant when more than one female was in the cage. This in line with observed reduction in VPR and PR mentioned above. Furthermore, the decrease in VPR and PR was also responsible for a decline in the number of pups born per female (all females included). The litter size and weaning weight (including only females that gave birth) were not influenced by the mating regime.
The litter sizes of the intra-strain combinations were in the expected range. Moreover, mating of the inbred strains with F1 hybrids produced larger litters.
Mating of F1 females to either one of the inbred males produced bigger litters than mating inbred females to F1 males. Aside from the possibility that F1 females might produce more oocytes, these differences might also be caused by longer recovery time or lower sperm production in the F1 males.
For most strain combinations, the monogamous mating regime achieved the best results in all analysed parameters, except for the portion of PC (Figure 1).
Conclusion
The decision of what mating regime would be the best option to induce timed pregnancies depends on the aim of breeding. When space is the limiting factor, polygamous mating with three females per male (3:1) is recommended, as reproductive outcome per breeding cage was the best for this mating regime. However, if a high number of timed embryos or pups per female are needed, monogamous mating is preferred because reproduction per female was best for this regime.
Footnotes
Acknowledgements
The authors would like to thank Marijana Basic (Institute for Laboratory Animal Science, Hannover Medical School, Hannover, Germany) for critically reading the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
