Abstract
The use of juvenile conventional pigs as a preclinical animal model to perform pharmacokinetic (PK), pharmacodynamic (PD) and safety studies for the paediatric population is increasing. Repetitive oral administration of drugs to juvenile pigs is however challenging. A representative method which can be used from birth till adulthood is necessary. The current study presents the placement and use of a gastrostomy button in pigs with a weight ranging from 2.4 to 161 kg. The surgical placement was performed via a laparotomic procedure on, each time, 12 pigs (six male, six female) of 1 week, 4 weeks, 8 weeks and 6–7 months old. For every age category, eight pigs were part of a PK study with a non-steroidal anti-inflammatory drug (NSAID) and four pigs served as a control group. No severe complications were observed during surgery. The button remained functional for 10 days in 40 out of 48 pigs. No significant differences in body temperature or white blood cell count were observed during the trial. Three control pigs showed signs of inflammation indicating a NSAID might be warranted. Autopsy revealed minimal signs of major inflammation in the abdominal cavity or the stomach. A limited number of pigs showed mucosal inflammation, ulcers or abscesses in the stomach or around the fistula. These results indicate that the laparotomic placement of a gastrostomy button might be considered safe and easy in growing pigs to perform repetitive oral dosing preclinical studies. However, the method is not advised in pigs weighing more than 100 kg.
Introduction
Juvenile animal studies during paediatric drug development are gaining more awareness and are stimulated by different regulating authorities. Juvenile animal studies have proven to be useful in evaluating developmental differences in pharmacokinetics (PK), pharmacodynamics (PD) and safety aspects of paediatric drugs. 1 Animal species selection should be based on the properties of the drug, comparative ontogeny of the major organs of interest, accurate prediction of the outcome in human and the feasibility of conducting the study. Due to the availability of a large historical dataset, among other practical advantages, rats are usually the species of choice. 2 Nevertheless, pigs are being increasingly used as experimental animals in research on several topics going from nutritional studies to surgery or preclinical pharmacological studies. The use of adult pigs in preclinical PK/PD and safety studies for humans is already well established.3–5 However, the use of juvenile pigs is currently still minimal despite the good anatomical and physiological similarities in developmental windows between the two species, namely neonate, infant, child and adolescent. 6 Recently, several studies demonstrated the successful use of juvenile pigs as human paediatric surrogate for preclinical studies.6–8
Accurate oral administration of drugs to juvenile pigs is challenging. Roth et al. trained pigs to accept and swallow tablets during a 2-week socialization period. 8 However, intensive training might not always be possible, especially when the developmental stage of interest concerns the neonatal phase. The formulation of the drug is also important. Tablets can be more easily administered by placing it on the tongue of the pig and then holding the snout until the pig swallows the tablet. Liquids on the other hand, are more challenging to administer. These can be given via a syringe in the mouth, but exact dosing is not possible via this method as the pig mostly does not swallow everything. Therefore, oral dosing by gavage is frequently used, where the pig is shortly restrained in order to put a tube via the mouth into the stomach or esophagus to provide accurate dosing. The placement of an orogastric or oro-oesophageal tube in a conscious pig can be technically challenging, stressful for the animal and requires at least 3 technicians to perform. Moreover, stress may alter the pharmacokinetics of the drug.9,10 Furthermore, placement of the oro-oesophageal or orogastric tube may cause oro-oesophageal trauma or accidental dosing into the lung or pharyngeal injuries might occur by incorrect placement.11,12 In order to perform multiple oral dosing studies or oral dosing for several days in pigs from different ages, a standardized technique is necessary to evaluate developmental changes. In newborn piglets, orogastric and oro-oesophageal tubes with exits through the cheek are easily placed, but are less suitable for use in weaned and older pigs as they are technically more difficult to place and are often regurgitated or chewed by the molar teeth.13,14 A more suitable technique for these older pigs would be permanent gastrostomy tubes. Several techniques have been successfully performed in pigs such as percutaneous endoscopic gastrostomy (PEG), blind percutaneous gastrostomy (BPG) or a laparoscopic gastrostomy (LG).15–17 Despite the use of several different techniques for repetitive oral administration by gastrostomy tube placement in pigs, to the authors' knowledge, no literature is available where the same method has been described for different age groups of piglets. Although LG seemed to be a suitable method, the authors selected laparotomy since LG requires more trained personnel, special apparatus and has a higher cost. The aim of the current study was to evaluate the use of a gastrostomy tube placement method via laparotomy in growing conventional piglets.
Animals, material and methods
Animals
The present study was approved by the ethical committee of the Faculties of Veterinary Medicine and Bioscience Engineering of Ghent University (EC2016/105). Care and use of the animals was in full compliance with the national (Belgian Royal Decree of 17 February 2017) and European legislation on animal welfare and ethics (Directive 2010/63/EU).18,19 All pigs were part of a PK and safety study with ibuprofen, a frequently used non-steroidal anti-inflammatory drug (NSAID) in humans. Four age categories were included in the study corresponding with the main age groups of the human paediatric population, namely neonate, infant, child and adolescent.6,20 One-week-old piglets (3.0 ± 0.4 kg BW; Landrace ×Large White × Maximus, RA-SE Genetics, Merkem, Belgium), 3.5-week-old piglets (7.0 ± 0.8 kg BW; Maximus, RA-SE Genetics, Bassilly, Belgium), 8-week-old piglets (20.1 ± 3.4 kg BW; Landrace ×Large White, RA-SE Genetics and Convis, Ettelbruck, Luxembourg) and 6–7-month-old pigs (134 ± 4.6 kg BW for males and 142 ± 9.8 kg for females; Landrace × Large White, RA-SE Genetics and Convis, Ettelbruck, Luxembourg) were used representing the respective human age groups. Each age category consisted of 12 pigs (6 male, 6 female), of which eight pigs (four male, four female) received ibuprofen in the PK trial and four pigs (two male, two female) served as control group in the PK trial and were sham-treated. The pigs were randomly allocated to a treatment group and all 12 pigs in each age group underwent the same surgical procedures. When four pigs had surgery per day, at least one pig served as control in the PK trial and if six pigs had surgery per day, two pigs served as control. The control pigs of the PK trial were used to evaluate the functionality and safety of the stomach button. All male pigs were intact. Since females reach puberty later than males, the females were 7 months old, while the males were 6 months old. 21 All pigs arrived at least 24 hours (1–5 days) prior to surgery at the test facility.
Before surgery took place, the animals were group-housed in either rescue decks (0.90 × 1.40 m 2 , Provimi, Rotterdam, The Netherlands) (1-week-old pigs), standard pig stables with partially slatted floors (2.30 × 2.40 m 2 ) (3.5- and 8-week-old pigs) or sow stables (0.65 × 2.20 m 2 ) (6–7-month-old pigs). After surgery, the animals were housed individually to avoid pen mates biting the catheters and stomach buttons. All age groups had ad libitum access to feed (1 week: RescueMilk®, Provimi; 3.5 and 8 weeks: Biggistart Opti®, Aveve, Leuven, Belgium; 6–7 months: Optivo Pro®, Aveve) and water. Natural light was provided by translucent windows and the stable temperature was 24.3 ± 2.14℃ during the whole duration of the trials. Higher temperatures (30–35℃) in the rescue decks were obtained by heating lamps. 22 One day prior to surgery, a cotton towel was given to the piglets (three youngest age categories), which was then passed on after surgery to mimic the smell of the other piglets when they were housed separately. The 1-week-old pigs could also hear, smell and see (Plexiglass®) each other. All stables were enriched with suspension chains, rubber toys and balls, which were rotated on a daily basis.
Prior and after surgery, all pigs were weighed on a daily basis for the whole duration of the trial (10 days). The 6–7-month-old pigs were only weighed the day of surgery. Technicians spent at least 15 minutes every day per pig to make the pigs comfortable with the presence of personnel in the stable and to facilitate the handling with the catheter and button. Both lumens of the jugular catheter were flushed at least once daily with a sterile diluted heparin solution (heparin Leo 5000 UI/ml, Leo Pharma, Lier, Belgium; 1-week-old piglets: 0.04% v/v; 4- and 8-week-old piglets: 1% v/v; 6–7-month-old pigs: 2% v/v). Sealing caps and bandages were changed when needed and wound healing was monitored. The stomach button was flushed daily with tap water and the skin surrounding the stoma was visually inspected and cleaned daily with chlorhexidine digluconate (Hibiscrub® 40 mg/ml, Mölnlycke Health Care, Antwerp, Belgium). After the PK trial, pigs were euthanized by intravenous injection of an overdose of pentobarbital (Sodium pentobarbital 20%®, Kela, Hoogstraten, Belgium). In case the double-lumen catheter was no longer functional at the time of euthanization (six pigs), euthanasia was performed by intramuscular injection of a mixture (1:1) of 4.4 mg/kg BW of xylazine hydrochloride (Xyl-M 2%®, VMD, Arendonk, Belgium) and 4.4 mg/kg BW tiletamine-zolazepam (each 50 mg/ml, Zoletil 100®, Virbac, The Netherlands), followed by intracardiac injection of an overdose of pentobarbital.
Experimental design
The experimental design was identical for all age categories and is graphically shown in Figure 1. Briefly, following a one-day postoperative recovery period, a single dose of 5 mg/kg BW ibuprofen (stock solution of 100 mg/ml in 0.9% NaCl, Ibuprofenum, Fagron Inc., Meer, Belgium) was administered intravenously using the proximal lumen of the jugular catheter. Next, one-day wash-out was respected before starting the multiple intragastric dosing study with a paediatric ibuprofen suspension (5 mg/kg BW, Ibuprofen EG® 40 mg/ml, Eurogenerics, Brussels, Belgium). This dose was administered three times daily for five consecutive days. After intragastric dosing, the gastric tube was flushed with tap water (≥5 ml) to make sure all ibuprofen reached the stomach. The control pigs did not receive any ibuprofen during the trial but were sham treated with the same volume of tap water for the oral dosing. Blood samples were taken from the distal lumen of the catheter every morning from both the control pigs and the pigs in the PK trial to determine white blood cell count (WBC). Furthermore, during the PK trial, blood was taken at different time points from the pigs receiving ibuprofen, to determine plasma concentration-time curves and perform further PK analysis (data not presented). On day 10, all pigs were euthanized for macroscopic and histological examination of possible lesions due to the stomach button.
Graphical overview of the experimental setup. On day 1, a double lumen jugular catheter and a gastrostomy button were placed in 12 pigs (six male, six female) respectively 1 week, 4 weeks, 8 weeks and 6–7 months old. A pharmacokinetic (PK) study in eight pigs per age group after both intravenous (IV) and oral (PO) administration was performed. The four control pigs per age group were sham-treated with water. The red tubes mean blood was sampled on those days. On day 10, euthanasia and post-mortem examination was performed in all pigs to evaluate possible lesions due to the stomach button.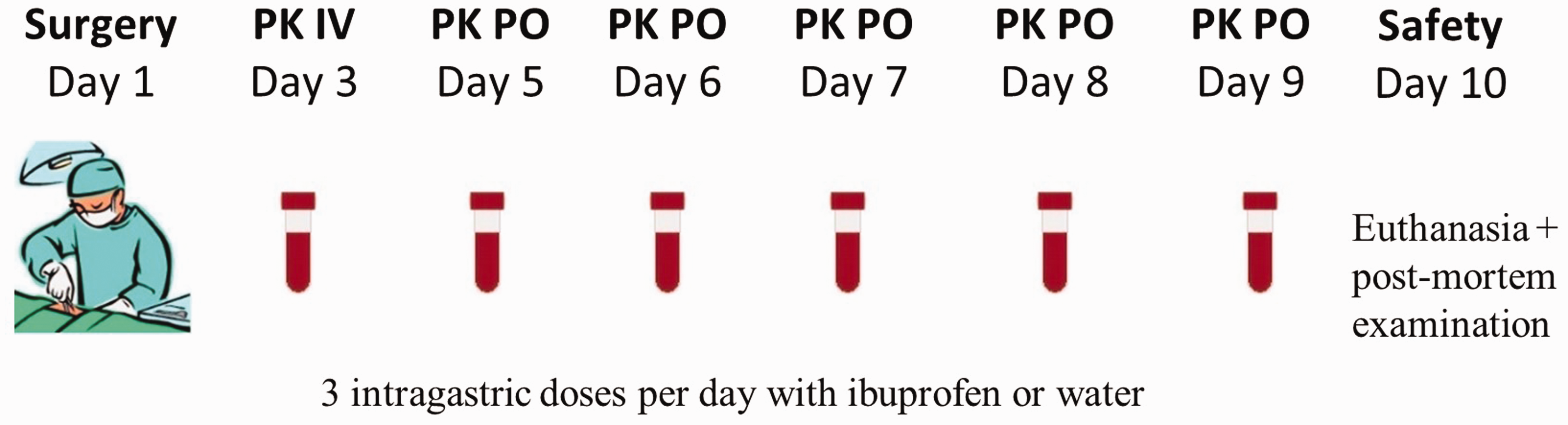
Anaesthesia and surgical procedures
All pigs were fasted 12 hours prior to surgery, except the 1-week-old piglets to reduce the risk of hypoglycemia. Anaesthesia was performed as described by Gasthuys et al. (2017). 23 After intramuscular premedication in the lumbar muscles with a mixture of morphine (0.1 mg/kg BW; Morphine.HCl® 10 mg/ml, Sterop, Anderlecht, Belgium), midazolam (0.5 mg/kg BW; Midazolam Mylan 5 mg/ml, Mylan, Hoeilaart, Belgium) and ketamine (10 mg/kg BW; Ketamidor 100 mg/ml, Richter Pharma AG, Wels, Austria), a catheter was placed in the marginal ear vein (24 standard wire gauge (SWG) in the 1-week-old piglets, 22 SWG in the other age groups). General anaesthesia was then induced using propofol (Propovet™ 10 mg/ml, Abbott Animal Health, Maidenhead, UK) which was administered slowly intravenously until endotracheal intubation was possible. Anaesthesia was maintained with sevoflurane in an oxygen/air mixture (inspired oxygen fraction 60%) administered in a semi-closed circle system (AV-1, Dräger, Lübeck, Germany). Monitoring included pulse oximetry, inspired and expired gas analysis, electrocardiography and measurement of body temperature and blood pressure (oscillometry) using a multiparameter monitor (S/5, D-LCC15-03, Datex Ohmeda, OR, USA). During surgery, the body temperature was stabilized using a resistive polymer warming device (Hot Dog®, Augustine Biomedical, Eden Prairie, MN, USA). Recovery of the 1-week-old piglets was facilitated with 0.02–0.04 mg/kg flumazenil IV (Anexate® 0.5 mg/5 ml, Roche, Bazel, Switzerland) and a nursery-hospital incubator (Bird Pavilion, Autoelex Co. Ltd, Gyeongsangnam-do, Korea).
Surgery was performed in right lateral recumbency. First, a double-lumen catheter (two-lumen central venous catheterization set, Arrow International, Cleveland, USA – 1-week-old piglets: 4 Fr., 30 cm, other age groups: 7 Fr., 60 cm) was surgically placed in the left jugular vein according to Gasthuys et al. (2009 and 2017).23,24 The external jugular vein was surgically exposed and the heparinized catheter was introduced using the Seldinger technique. The jugular vein was punctured with a trocar, and a guide wire was advanced through the trocar towards the vena cava. Hereafter, the catheter was advanced over the guide wire, and the guide wire was then removed. The catheter was tunnelled using the trocard tunnelling device from the neck region, immediately cranial to the withers (small stab incision), towards the exposed surgical area, and attached to the skin with sutures. After closure of the incisions with a three-layer continuous pattern, the laparotomic procedure was started.
The gastrostomy button (Entristar™ skin level gastrostomy kit, Kangaroo™, Covidien, Dublin, Ireland – 1-week-old piglets: 12 Fr., 1.2 cm, 4-week-old piglets: 16 Fr., 2.0 cm, 8-week-old piglets: 16 Fr. 2.0 cm and 12 Fr. 3.0 cm, 6–7-month-old pigs: 12 Fr., 5.0 cm) was placed by means of a laparotomic procedure during the same general anaesthesia. The abdomen was opened by a paracostal incision. The incision was made 2–4 cm caudal to the costal arch. To facilitate the procedure, the spleen was exteriorized and covered with gauze swabs soaked in physiological saline. Subsequently, the gastrosplenic ligament was used as a guide towards the major curvature of the stomach. The major curvature of the stomach was exteriorized and secured with two atraumatic clamps. The gastrostomy button was introduced into the abdomen with its introducer stylet in place through a separate stab incision 1 cm craniodorsal from the paracostal incision. A monofilament USP 2/0 purse string suture (Poliglecaprone, Monocryl, Ethicon Inc., Somerville, USA) was placed in the serosa muscularis layer just cranial from the major curvature on the ventral surface of the fundus. The gastrostomy button was introduced into the stomach through a stab incision in the middle of the purse string suture and keeping the internal retention bolster in extention via the obturator. The tube was secured into the stomach by releasing the internal retention bolster and leakage of gastric content into the abdominal cavity was avoided by tightening and knotting the purse string suture. After removal of the introducer stylet and obturator, the surface of the stomach was rinsed with 0.9% sterile NaCl solution before repositioning of the stomach in the abdominal cavity. The paracostal incision was routinely closed in a three-layer continuous pattern using vicryl 2/0 (antibacterial polyglactin 910, Vicryl Plus, Ethicon Inc., Somerville, USA). The stomach was pulled against the abdominal wall by application of the provided clip under the external retention bolster of the stomach button. Figure 2 gives an overview of the placement of the button and the catheter.
(A) The different parts of the gastrostomy button just before insertion into the stomach of an 8-week-old pig. (B) The position of the jugular vein double lumen catheter and the stomach button after euthanasia in the same pig.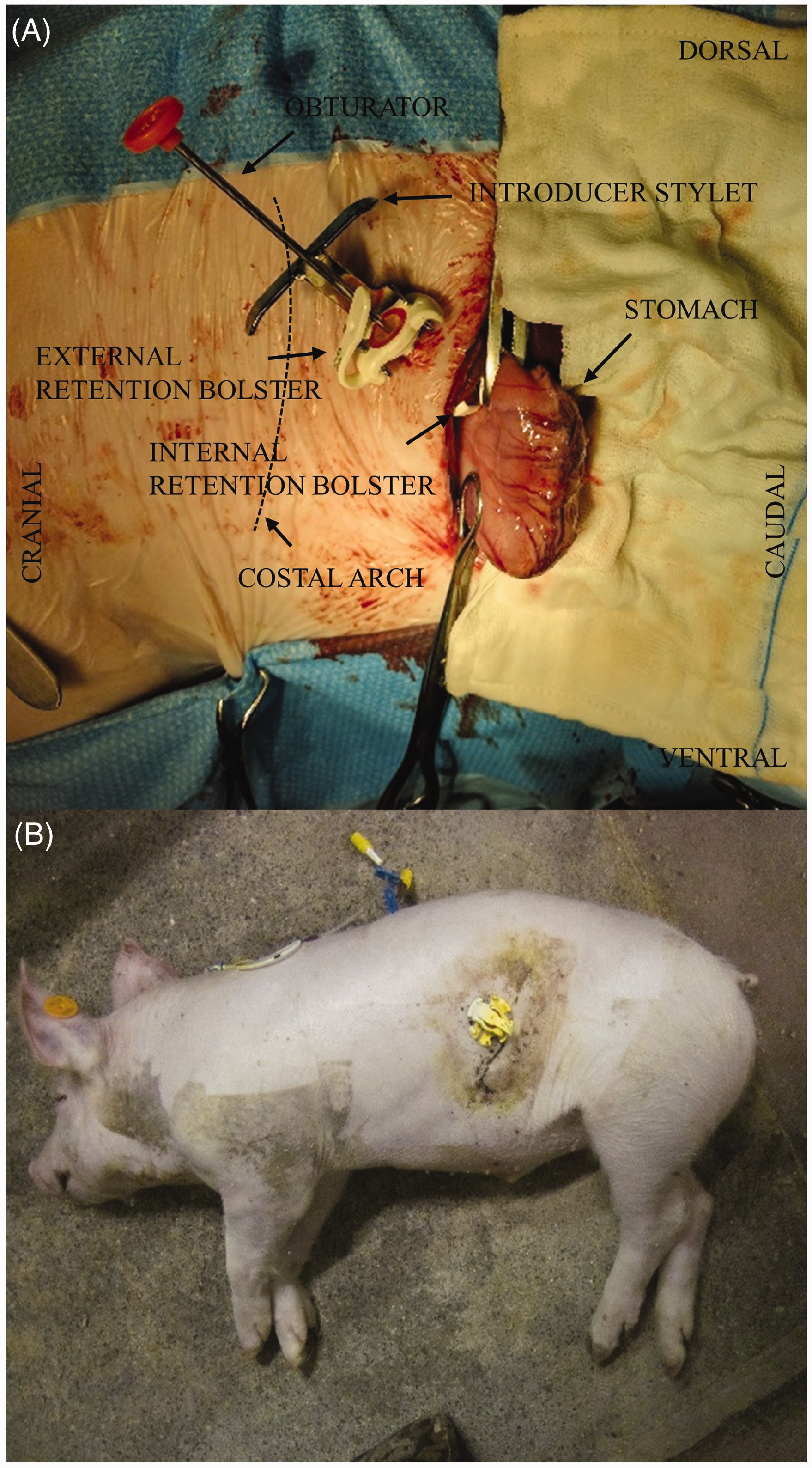
Clinical and post-mortem examination of the pigs
After surgery of the pigs, their water and feed intake, as well as their body temperature and interaction with animal caretakers were monitored twice daily. Temperature was measured via a LifeChip® (Allflex Europe SA, Vitré, France) placed in the left semitendinosus muscle during anaesthesia. Total WBC count was performed daily, from the day after surgery till the end of the trial by taking 1 ml blood via the double lumen catheter in a K3 EDTA coated tube (Vacutest® Kima, Arzegrande, Italy, analysis performed by Medvet BVBA, Antwerp, Belgium).
On day 10, all pigs were euthanized, and a post-mortem examination of the pigs was performed to evaluate possible lesions and complications due to the stomach button. During necropsy, the position of the stomach button and tissue reaction was macroscopically evaluated in the abdominal cavity as well as in the stomach. Major complications were defined as removal of the button and buried bumper syndrome (BBS). Minor complications were abscesses and inflammation in the stomach. Samples of the stomach wall adjacent to the button were collected for histopathological evaluation. After fixation in 4% formaldehyde and paraffin embedding, 5 µm thick slices were cut and stained with haematoxylin-eosin.
Statistical analysis
Changes in body temperature and WBC were evaluated using a univariate type III repeated-measures ANOVA in R (RStudio version 1.1.456). Mauchly's sphericity test was significant (p < 0.05) which means the Greenhouse-Geisser correction was applied.
Results
Surgery
Overview of the number of pigs per age category and treatment group which had a successful surgery for both double lumen catheter and gastrostomy button and lost their button during the trial due to obstruction or removal. The average daily weight gain ( ± SD) is also given for the three youngest groups.
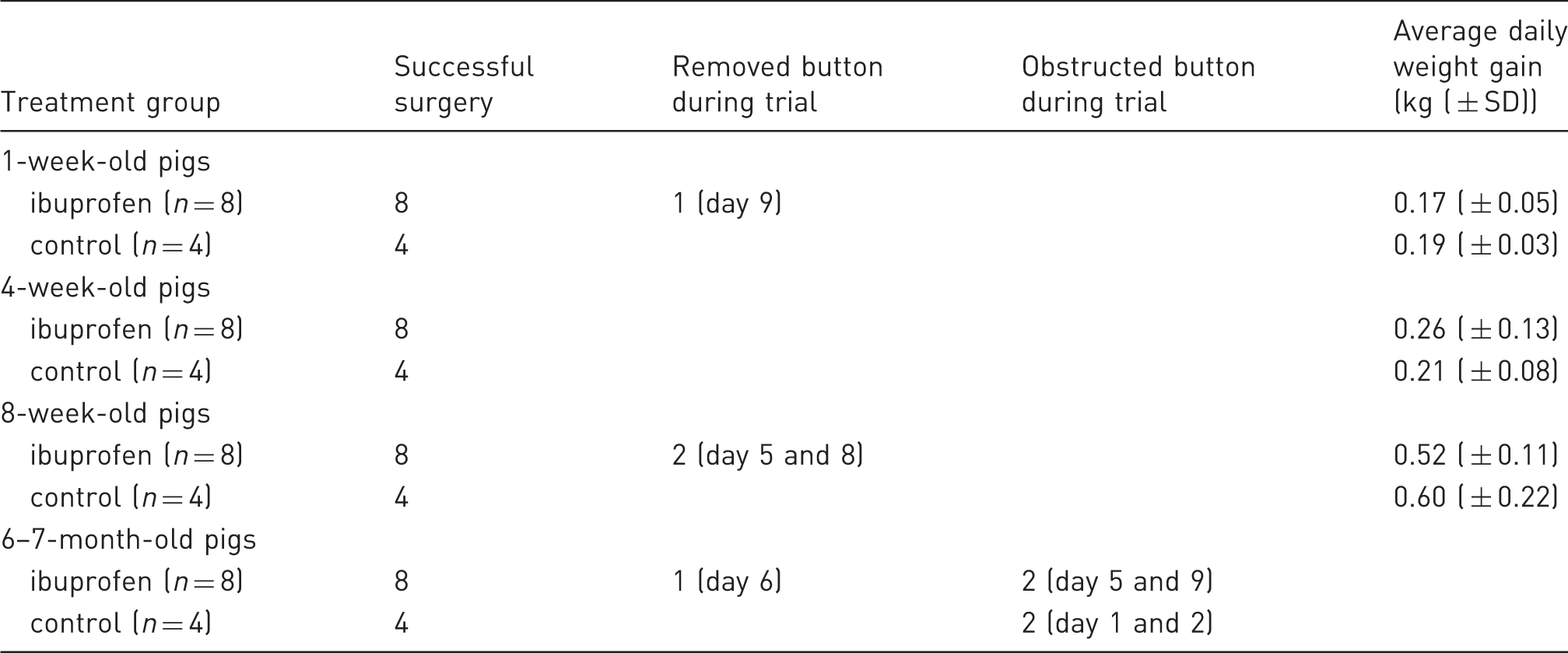
Initially, the stomach button of all pigs was functional except for one 6-month-old control pig. During the trial, the functionality of several buttons was impaired due to removal by the pig (four pigs) or obstruction of the button (four pigs) (Table 1). When the button was removed, the wound was cleaned, disinfected and properly bandaged. Two days after removal, the wound was almost closed. If the button was obstructed, it was left in place until autopsy as the pigs did not experience any nuisance.
Clinical examination
Control pigs
A limited number of control pigs showed clinical abnormalities. Two 1-week-old control piglets had fever ( > 40℃) on day 7 and were apathic. One 4-week-old control pig showed the same signs on day eight. They were treated with an intramuscular injection of 0.4 mg/kg BW of meloxicam (Metacam® 5 mg/ml, Boehringer Ingelheim Vetmedica GmbH, Ingelheim/Rhein, Germany). In one 1-week-old piglet, the meloxicam administration was repeated after 24 hours. These three pigs with fever had no complications during surgery.
No significant changes (p > 0.05) in body temperature within each age group were observed during the trial nor between the different age groups. Similarly, no significant differences were observed in WBC over time for the control group (Figure 3).
(a) The mean body temperature and (b) the mean number of white blood cells for the control pigs (four pigs per age groups, C, dotted lines) and pigs receiving ibuprofen (eight pigs per age groups, IBU, full lines) at 1 week (blue square), 4 weeks (red diamond), 8 weeks (yellow dot) and 6–7 months (green triangle) of age. Measurements started the day after surgical placement of a double lumen jugular catheter and a gastrostomy button.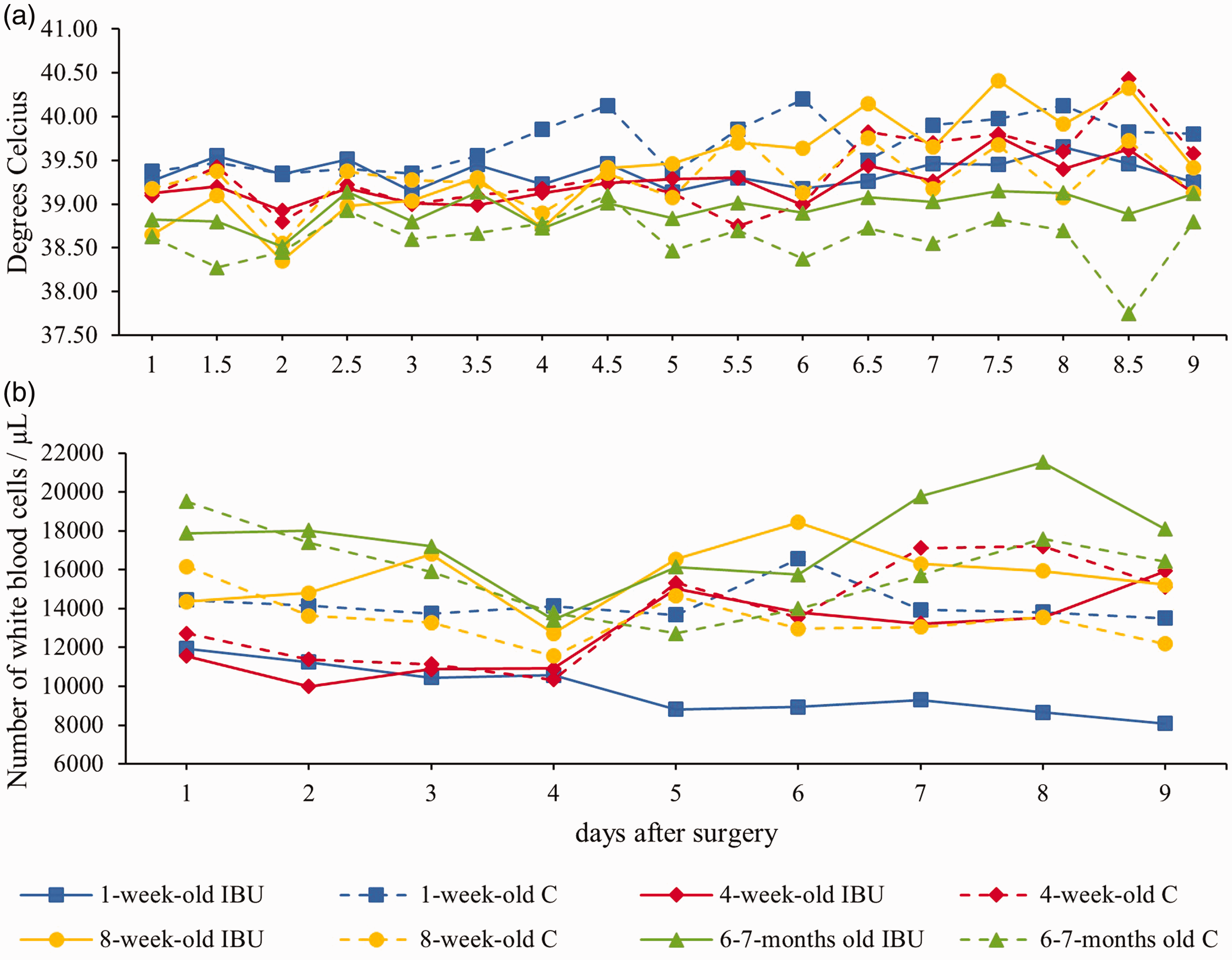
Pigs receiving ibuprofen
None of the pigs who received ibuprofen showed clinical abnormalities during the trial. Similarly to the control pigs, no significant changes in body temperature or WBC were observed during the trial within the age groups, nor between the age groups. Moreover, no significant changes in temperature or WBC were observed between the ibuprofen group and the control group except for the 1-week-old piglets in the ibuprofen group having a significant lower amount of WBC compared to the 1-week-old control piglets (Figure 3).
Post-mortem examination
Control pigs
During post-mortem examination, the presence of macroscopic lesions was evaluated. Gross examination of the surgical wound of the abdominal wall revealed a good wound apposition with adhesive healing (Figure 4(a)). The peritoneum had a normal appearance and there were omental adhesions with stomach, spleen and stoma site in all control pigs (Figure 4(b)). In one 4-week-old and one 8-week-old control pig, there were also omental adhesions with the liver. In three 1-week-old control pigs a small area ( ± 1 cm) of the small intestine was covered with omentum and was retained against the abdominal wall. No signs of major inflammation were observed inside the abdominal wall along the peritoneum. Overall, no excessive damage was observed inside the stomach (Figure 4(c)). Three 8-week-old control piglets showed local redness in the mucosa next and opposite to the stomach button. Three 6–7-month-old control pigs had abscesses at the location of the incision or the internal retention bolster. In two 6–7-month-old control pigs, the button was not present inside the stomach, but in the gastric wall. In the other two 6–7-month-old pigs, the internal retention bolster was buried and surrounded with mucosa because of the high traction forces on this internal retention bolster.
(a) The gastrostomy button (external retention bolster) nine days after surgery of an 8-week-old control piglet. The abdominal incision is located right ventral to the button. (b) Adhesions between omentum and abdominal wall of the same pig. The stomach is nicely retained against the abdominal wall. (c) The internal retention bolster in the stomach of a 4-week-old control piglet. The stomach was cut open along the curvature major. (d) Histological picture of the stomach wall surrounding the button. The dotted line represents the connective tube between internal and external retention bolster of the gastrostomy button.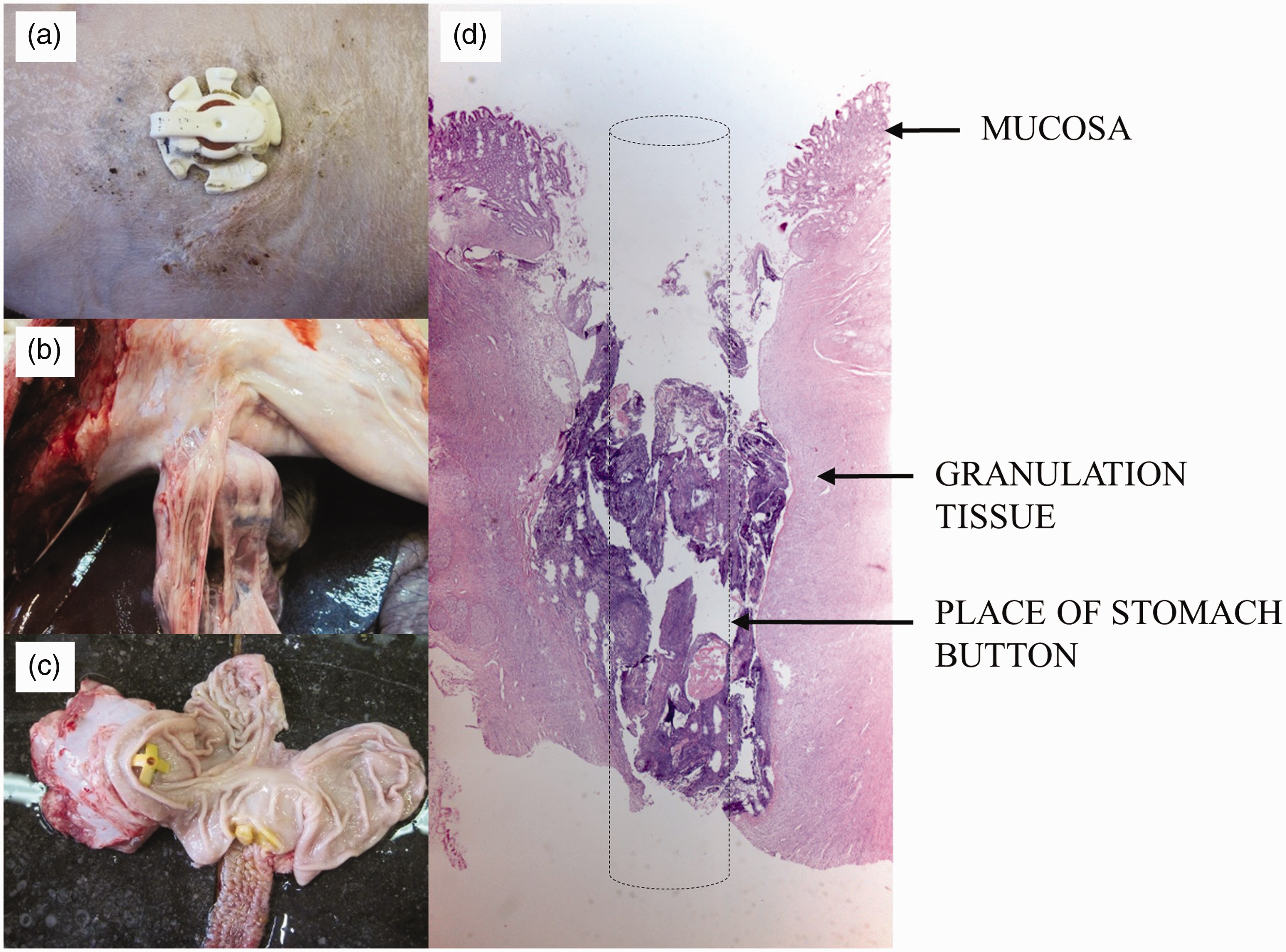
The histological findings at the insertion site of the stomach button confirmed the macroscopic observations. All pigs showed granulation tissue (fibroblastic and vascular proliferation) from mucosa until muscularis propria at the surgical borders of the fistula (Figure 4(d)). The granulation tissue was often covered with fibrin deposit and cellular debris with micro colonies of bacteria. A mild to severe influx with neutrophils was observed which was the most intense in the (sub)mucosal areas. Incomplete reepithelization and focal ulceration of mucosa was observed at the site where the internal retention bolster made contact with the mucosa. The 8-week-old pigs showed in general a more severe inflammation going deeper towards the serosa. The 6–7-month-old pigs showed more tissue necrosis around the internal retention bolster.
Pigs receiving ibuprofen
Generally, the findings during necropsy of the pigs which received ibuprofen were similar to those of the control pigs. Omental adhesions with the liver were observed in four 4-week-old pigs and three 8-week-old pigs. Omental adhesions with colon were observed in six 1-week-old piglets and three 6–7-month-old pigs. And in one 1-week-old piglet, the small intestine was covered with omentum. Local redness in stomach mucosa surrounding or opposite the button was observed in one 1-week-old and three 8-week-old pigs. One 4-week-old pig had an ulcer in the pars oesophagea of the stomach and three 6–7-month-old pigs had an ulcer next to or opposite the button. Abscesses at the abdominal incision site or surrounding the internal retention bolster were observed in four 6–7-month-old pigs. The button was no longer present inside the stomach of the two 6–7-month-old pigs whose button was obstructed during the trial. In the 1-week-old and two 8-week-old pigs who removed their button, the stomach was still attached to the abdominal wall and the fistula was filled with granulation tissue. The histological findings were no different from those of the control pigs.
Discussion
Oral dosing in juvenile pigs can be challenging. Pigs can be trained to accept and swallow tablets or pills hidden in, for example, a meatball.8,25 However, paediatric formulations are mostly a liquid or suspension for which, to the authors' knowledge, no stress-free administration route has been described which can be applied to pigs of different age. Moreover, Gonzales et al. (2014) have demonstrated that stress can alter the pharmacokinetics of a drug in mice dosed via traditional oral gavage versus mice dosed with the formulation in a peanut butter pellet. 9 Therefore, the aim of the current study was to evaluate whether a laparotomic gastrostomy method could be used in growing piglets to perform multiple dose PK studies with a suspension. Although different mix-breeds of pigs have been used in the current study, this is believed not to have an effect on the surgical placement of the stomach button or the evaluation of this technique. The presented surgical approach is characterized by a shallow learning curve which is reflected in a decrease of anaesthesia time depending on the experience of the surgeon regarding the described technique and training level. The longer anaesthesia time of the veterinarian with the basic surgery training compared to the ECVS diplomates was mostly due to the big size of the 6–7-month-old pigs. The technique can be performed without laparoscopic equipment, and animals have a fast and full postsurgical recovery. Thanks to intensive socializing, bolus administration of drugs and water through the button was easy to perform. If the study design would require aspiration of gastric juice, for example to measure pH, this might also be possible. Easy intra-gastric administration in combination with blood sampling via the double lumen jugular catheter reduced stress for the animals as the pigs did not need to be restrained.
The complications observed during the trial can be divided into major and minor ones. Major complications occurred in 25% of the control pigs and 18.8% of the pigs receiving ibuprofen. Removal of the button was in all cases due to repeatedly scratching against the wall of the pen, probably because of itching. Birck et al. (2015) also observed repeated scratching, but no tubes were displaced. 15 BBS is a complication in which the internal retention bolster migrates alongside the tract of the stoma outside the stomach. This is due to excessive compression of tissue between the internal and external retention bolster of the stomach button. 26 BBS occurred in 41.7% of the 6–7-month-old pigs, despite stoma site measurement before surgery. Longer buttons would provide a solution but were not commercially available. In humans, the general incidence of BBS is estimated to be 1% after placement of a PEG and in the paediatric population this incidence increased up to 22%. 26 Minor complications were observed in 37.5% of control pigs and 31.3% of pigs receiving ibuprofen. Again, the highest incidence was observed in the 6–7-month-old pigs. Abscesses in the oldest age group were probably caused by BBS or the less hygienic wound care, since these pigs removed their bandages daily. The observed complications of the stomach button in all pigs were higher in comparison with the studies of adults and children for PEG and LG. A retrospective study revealed that 13.5% of the paediatric patients in the PEG group suffered from major complications, while minor complications occurred in 10.8 and 25% of the cases for PEG and LG respectively. 27 In adults, major complications occurred in 8.4% of patients undergoing PEG, while minor complications occurred in 24% of cases. 28 However, when only considering the three youngest age groups, the observed complications were similar and even lower compared to the human studies. Major complications were observed in 12.5% of pigs receiving ibuprofen and none were observed in the control pigs. Minor complications were present in 20.8 and 12.5% of pigs receiving ibuprofen and control pigs, respectively. Whether this higher incidence of complications in the ibuprofen group compared to controls is due to the possible gastro-intestinal effects of this drug needs be further investigated.
Overall, the stomach button was functional and well endured by the pigs in the youngest three age groups for the entire study period of 10 days. Nevertheless, three control piglets had fever symptoms although no complications occurred during surgery. Two of the three sick piglets were only 1-week-old. This might be due to the young age and the immaturity of the immune system. Gasthuys et al. (2017) observed inflammation in 25% of 1-week-old piglets after placement of a jugular catheter only. 23 Nevertheless, since the pigs who received ibuprofen did not show any signs of inflammation, a non-steroidal anti-inflammatory drug might be warranted when a gastrostomy button is placed. The histological findings of both control and ibuprofen group were acceptable as could be expected with the insertion of a foreign device.
In conclusion, the current laparotomic placement of a gastrostomy button is proven to be safer than the BPG described by Gades et al. (2001) as less complications were experienced and the pigs could already eat one hour after the procedure. 16 Moreover, no laparoscopic equipment was necessary which made the method easier than the one described by Birck et al. (2015) and the method could be performed by people without specialized surgical training. 15 Laparotomic placement of a gastrostomy button could be successfully used in studies which require multiple oral dosing in pigs aged 1 week, 4 weeks and 8 weeks. However, this method is not advised in pigs weighing over 100 kg.
Supplemental Material
Supplemental material for Gastrostomy tube placement via a laparotomic procedure in growing conventional piglets to perform multi-dose preclinical paediatric drug studies
Supplemental Material for Gastrostomy tube placement via a laparotomic procedure in growing conventional piglets to perform multi-dose preclinical paediatric drug studies by Joske Millecam, Thomas van Bergen, Mathias Devreese, Stijn Schauvliege, Ann Martens, Koen Chiers, Siska Croubels and Gunther Antonissen in Laboratory Animals
Footnotes
Acknowledgements
The help of the colleagues during the animal experiments was gratefully appreciated.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Agency for Innovation by Science and Technology in Flanders, currently known as the Agency for Innovation and Entrepreneurship in Flanders (IWT, SB141427), as well as the SafePedrug consortium, ![]() .
.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
