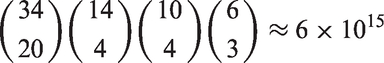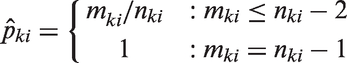Abstract
The hyperactive FVB/N inbred mouse strain is widely used for transgenic research applications, although rarely for behavioral studies. These mice have visual impairments via retinal degeneration, but are considered highly intelligent and rely largely on olfaction. While investigating diet-induced obesity in autotaxin transgenic FVB/N mice, we observed an increase in the necessity for male, but not female, cage separations. Based on the observations, we hypothesized that feeding FVB/N mice a lean diet increases nocturnal bouts of aggression between male littermates. The diets of adult littermates were switched from normal chow to either ad libitum high-fat (45% fat) or lean (10% fat) matched diets for 27 weeks, whereby the mice reached an average of 43 g versus 35 g, respectively. Then, cage separations due to nocturnal bouts of aggression became mandatory, even though littermates peacefully cohabitated for 10–16 weeks previously. Since the data was of an unusual nature, it required uncommon statistical methods to be engendered to evaluate whether and where significance existed. Therefore, utilizing the randomization and population models, we established a methodology and postulated that either testosterone, the autotaxin transgene or diet alteration was the causal factor. Statistical evaluation demonstrated a significant correlation between cage separations and aggressive behavior associated with the lean-diet-fed mice, not autotaxin. Biochemical data did not appear to explain the behavior. In contrast, energy metabolism highlighted differences between the groups of normally hyperactive mice by diet. This characteristic makes FVB/N male mice unsuitable subjects for long-term studies with lean-diet modifications.
The FVB/N inbred mouse strain is widely used in laboratory research applications. It is spontaneously hyperactive, only slightly more anxious when compared to C57BL/6 mice, which are also widely used for research.1,2 FVB/N mice possess unique characteristics that make them suitable to generate transgenic animals for experiments, including their high mating frequency, their consistent ability to produce large, healthy litters and their zygote survivability. 3 Female FVB/N mice exhibit strong maternal behavior whereby moms display exceedingly low rates of infanticide or cannibalism, which allows robust numbers of pups to survive, even with a litter size of 12 and an average of 9.5. 4
Although FVB/N mice are described as ‘unsuitable for behavioral studies’ because they have visual impairments via retinal degeneration, that conclusion is based largely on the reliance of commonly used tests that require acute vision to perform the function, not olfaction.5,6 For example, Morris water navigation tasks to find a visible platform and novel object recognition are significantly easier without impaired vision. However, more recent studies show FVB/N mice are superior in learning and memory performance over C57BL/6 mice using an olfactory tube maze test. 7 This suggests that vision, not intelligence, has led to false impressions about what constitutes normal behavior in mice.
There are somewhat conflicting studies surrounding the degree of aggressive behavior in FVB/N males. One study concluded that FVB/N males have only ‘somewhat higher levels of aggressiveness’ when compared to C57BL/6 males. 1 In contrast, another study concluded that in comparison to C57BL/6 male mice, FVB/N male mice display a higher duration of offensive behavior and significantly longer attack durations against intruder mice, which might result from their inability to maintain regular circadian rhythm patterns. 8 Studies on aggression typically focus on behavior among males, not females.
Initially, we set out to conduct a study about the interaction between diet-induced obesity with autotaxin transgene expression. Transgenic mice overexpressing autotaxin have larger adipocytes, biological differences with obesity and develop spontaneous cancers after 12 months of age.9,10 Herein, we observed an unexpected and striking increase in FVB/N male aggression among certain cages, which necessitated permanent separations. This required incessant cage patrolling and routine separations throughout the course of the experiment.
Due to the unusual nature of the data, we required the creation of a new statistical method to evaluate whether statistical significance existed within these observations. Several aspects of these data combine to prevent it from fitting neatly into any existing data analysis paradigm. The data are both clustered and partially missing. The clustering is due to the treatment assignment being received by cages of animals, rather than individual animals. The resulting data structure is similar to a group randomized clinical trial. The partial missing-ness is due to the way aggressive behavior is objectively measured by cage separation – aggressive animals are isolated in individual cages; however, animal separation is an imperfect surrogate for aggressiveness.
Herein we describe the study and the statistical computations along with the data that inspired the mathematical exploration. Based upon these parameters, we conclude that violent behavior ensues among cohabitating groups of adult male FVB/N mice through diet modification to lean chow with 10% fat. To our knowledge, this is the first description of such a scenario in FVB/N, largely due to the non-reliance on the strain for behavioral research. Therefore, the behavior of this model makes the males unsuitable for long-term studies using lean-diet modifications; however, this model could be ideal to study male mouse aggression that is not reliant on visual stimulation.
Materials and methods
Animals
All protocols for the use of vertebrate animals at the University of Georgia were approved by The Institutional Animal Care and Use Committee. The Guide for the Care and Use of Laboratory Animals is endorsed in this study. Transgenic mice were the kind gift of Drs Gordon B Mills and Susan S Smyth and, as described previously, generated on an FVB/N genetic background. 11 Pups were weaned after 21 days and fed ad libitum water and standard rodent chow containing kcal% of 30% protein, 57% carbohydrate and 13% fat (Lab Diet, #5001, St Louis, MO) prior to experiments employing the diet modifications. There were no restrictions on the quantity of food or water available and cages were kept well stocked with pellets. Polymerase chain reaction (PCR) analysis was used to confirm the genotyping and expression from ear clips with an Agilent 2100 bioanalyzer (Agilent, Santa Clara, CA) and the following primers: 5’-GATCCCAGCCAGTGGACTTA-3′ (forward) and 5′-TCTGACACGACTGGAA CGAG-3′ (reverse). In total, 140 male mice (106 AT-ATX transgenic and 34 wild-type) were fed with standard rodent chow, for which the data are included in this study.
After approximately 10–16 weeks of age, transgenic (39 male) and wild-type (17 male) mice were then re-assigned to a different diet, from initially eating standard chow. Mice were fed either a high-fat diet with kcal% of 20% protein, 35% carbohydrate and 45% fat (Research Diet D12451, New Brunswick, NJ) or the matched control, lean diet that included 17% sucrose, lean diet of 20% protein, 70% carbohydrate and 10% fat (Research Diet D12450H). Male cages required significant management and cage separations for fighting and wounding, as governed by the Animal Use Protocol and recommendations of the veterinarian attendant. The diet study in male mice lasted 27 weeks.
The mouse facility automates the lighting in the rooms on a 12-hour cycle, whereby the lights come on at 7:00 am EST and then go off at 7:00 pm EST. The temperature is maintained at 72°F ± 4°F. The mice are housed in Individually Ventilated Cages (IVC) mouse DGM (digital ready green line mouse) racks and Sealsafe Plus GM500 cages measuring 15.40 × 7.83 × 6.30 in (Tecniplast, West Chester, PA). The area of the cage floor is 77.66 in 2 . The mouse cages include Bed-o’Cobs® bedding and one Bed-r’Nest® portion-controlled nesting material (The Andersons, Inc, Maumee, OH). For enrichment, each cage includes one plastic red mouse house that allows only red wavelength light to pass through.
Aggressive behavior was flagged by our veterinary staff. The animal cages are checked daily for health and hygiene monitoring. Tail handling is the main type of handling. The facility also performs full changes of the cages every other week. During the daily monitoring, they observed heightened and unexpected fighting without cessation, wounding, chasing and biting within the cages. Most often this was concurrent with bloody bedding, visual wounds and open cuts on victims, whereas perpetrators perched on top of mouse houses and lacked extensive bleeding wounds. Cages were cleaned on Monday mornings, every other week, whereas separations and deaths from aggressive bouts occurred randomly throughout the week. All of these observations required intervention and cage separations per guidelines for care of the animals. As these results were unexpected, there was no blinding.
Humane endpoints were based on an overlapping study. 10 Prior to this study, we did not foresee aggressive bouts occurring to this degree. The humane endpoints governing our animal use protocol included: excessive tumor burden, inability to move, seizures, abdominal distension that interferes with mobility, inability to feed, severe lethargy with little/no spontaneous movement, difficult or labored breathing, blue mucus membranes, bleeding from deep wounding or severe ulceration where there is an absence of an epidermal layer overlying the skin that is >50% of the wound size. At the conclusion of the study, animals were euthanized with a method consistent with the guidelines of the American Veterinary Medical Association.
Statistical methodology
The treatments and control in this experiment were randomly assigned to the cages. The effects of the treatments on aggressiveness can, therefore, be evaluated using two different statistical modeling approaches. We examine both approaches to analyze these data. The first approach is based on a randomization model and a sharp null hypothesis. 12 The second approach is based on a population model and a null hypothesis about population parameters. 13 For our purposes, the randomization model has the advantage that the limited sample size will not invalidate a randomization-based permutation test (unlike a central limit theorem-based test), so that reliable p-values can be generated with this approach. On the other hand, the population model has the advantage of permitting a more detailed description of the effect size and uncertainty estimates for comparisons of treatment groups and control.
The unusual data structure
Unlike all the humane endpoints governing this protocol, the endpoint of aggression has an unusual statistical structure. First, aggressiveness cannot be observed in isolation, but is a behavior that requires the presence of other mice in the same cage. When no other mice are present, then it is no longer possible to observe aggressiveness for the remaining mouse. This gives rise to a specific type of right censoring that must be taken into consideration during the analysis. Second, the data are also clustered by cages, so that proper analysis requires taking into account the clustering structure and possible correlations of observations in the same cage. Third, the number of cage separations can be modeled as a discrete random variable taking on values between 0 and the total number of animals in a cage minus one. The induced distribution due to the right censoring will be non-standard and must be mathematically calculated (see Supplemental Figure 2 for calculations).
Randomization model methodology
The sharp null hypothesis used for the randomization model states that there is no effect on the response (aggressiveness) for any of the treatments. Any observed differences are, therefore, attributable to the randomization itself – that is, to the proverbial ‘luck of the draw.’ Therefore, every other possible randomization would have resulted in the exact same set of observations. The ‘unusualness’ of the observed data can, thus, be quantified by a permutation test. For this experiment, there is one control condition and four different treatment conditions. The number of cages per condition were 20 for the control, and 4, 4, 3, 3 for the different treatments, for a total of 34 cages. Therefore, the total number of distinct permutations of the cages is as follows:
This number suggests that we can obtain a very small p-value for the test.
Population model analysis
For the population model approach, the natural population parameter of interest is the proportion of animals in the target population of mice that turn aggressive when exposed to the control or a specific treatment. Let
Biochemical analysis
Testosterone content in serum was determined using a colorimetric competitive enzyme immunoassay ELISA kit (Enzo Life Sciences, Farmingdale, NY) following the protocol according to the manufacturer's instructions. A calibration curve was created using the testosterone standard included in the kit, which has a range from 2000 pg/mL to 7.81 pg/mL. The absorbance was read at 405 Nm using a SpectraMax M2 plate reader (Molecular Devices, Sunnyvale, CA).
The adenosine diphosphate/adenosine triphosphate (ADP/ATP) ratio was determined using single-cell suspensions with a luciferin–luciferase bioluminescent assay kit (Abcam, Cambridge, MA) according to manufacturer's instructions. The ratio was calculated based on a calibration plot made with a standard ATP solution with a range from 1 nM to 10 nM (ATP Disodium Trihydrate, Amresco, Solon, OH). Single-cell suspensions were created using 200 mg of flash-frozen tissues and dissociated with collagenase type I (Stemcell Technologies, Vancouver, Canada) and suspended in phosphate buffered saline (PBS). Luminescence was determined using a Molecular Devices SpectraMax M5 plate reader (Sunnyvale, CA). The data are plotted as a scatterbox using GraphPad Prism to display the average and range of the data points.
Immunoblotting
For protein resolution, liver tissue from mice was homogenized in 500 µL of lysis buffer (RIPA buffer with a protease inhibitor in a 1:100 ratio). Buffered tissue was sonicated, centrifuged and the supernatant extract was obtained. Protein concentrations were determined by a bovine serum albumin (BSA) protein assay (Pierce BSA protein assay kit, Thermo Fisher Scientific, Rockford, IL). Approximately 30 µg per sample was resolved using sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) for 120 min at 110V. Proteins were then transferred to nitrocellulose membranes on ice for 90 min at 100 V. Membranes were blocked in milk buffer at room temperature for 1 h and incubated on a shaker plate with Androgen receptor AN4441 (MA5-13426, Thermo Fisher Scientific, Rockford, IL) overnight at 4 ℃. Membranes were washed with Tris-buffered saline with Tween 20 (TBST) three times, 10 min each, then incubated with secondary for 120 min at room temperature and washed again, as stated previously. Chemiluminescent substrate Dura (SuperSignal West Dura extended duration substrate, Thermo Fisher Scientific, Rockford, IL) was applied to the membrane for 5 min and then imaged for 6 min. A glyceraldehyde 3-phosphate dehydrogenase (GAPDH) image was obtained the same way with a regular chemiluminescent substrate (SuperSignal West Pico, Thermo Fisher Scientific, Rockford, IL) applied for 3 min and imaged for 2 min. Imaging was obtained with a Li-cor Odyssey Fc (Li-cor Biosciences, Lincoln, NE).
Software and calculations
Calculations were carried out in RStudio with R version 3.4.1. GraphPad Prism was used to create graphical presentations and assess the statistical significance among some of the data, where indicated.
Results
Diet-induced aggression in male FVB/N mice
During a diet-induced obesity study using FVB/N inbred mice, we observed an unexpected manifestation of aggression among male, but not female, cages that encumbered the investigation. In the colony containing several hundred FVB/N mice, cages were provided with a diet of standard rodent chow ad libitum. The female cages required no separations for aggressive behavior (fighting, wounding, chasing, biting, blood discovered in the cage, etc.). During the same period, the male cages required only a few separations (Figure 1(a)).
FVB/N males, but not females, are more likely to require cage separations. (a) Bar graphs showing the number of total cages required overall with a standard, chow diet. This is reflective of typical cage separations over a four-month time period with FVB/N mice and standard conditions of sterile bedding, supplemental nesting material, mouse houses, ad libitum food and water. Males began with ∼5 littermates in each of the 20 cages (n = 100) and ended with 26 cages. Females began with ∼5 littermates in each of the 28 cages (n = 140) and ended without any changes. (b) Four male littermates fed a high-fat diet peacefully cohabitating in their cage. The fifth mouse is just outside of the image and slightly away from the group.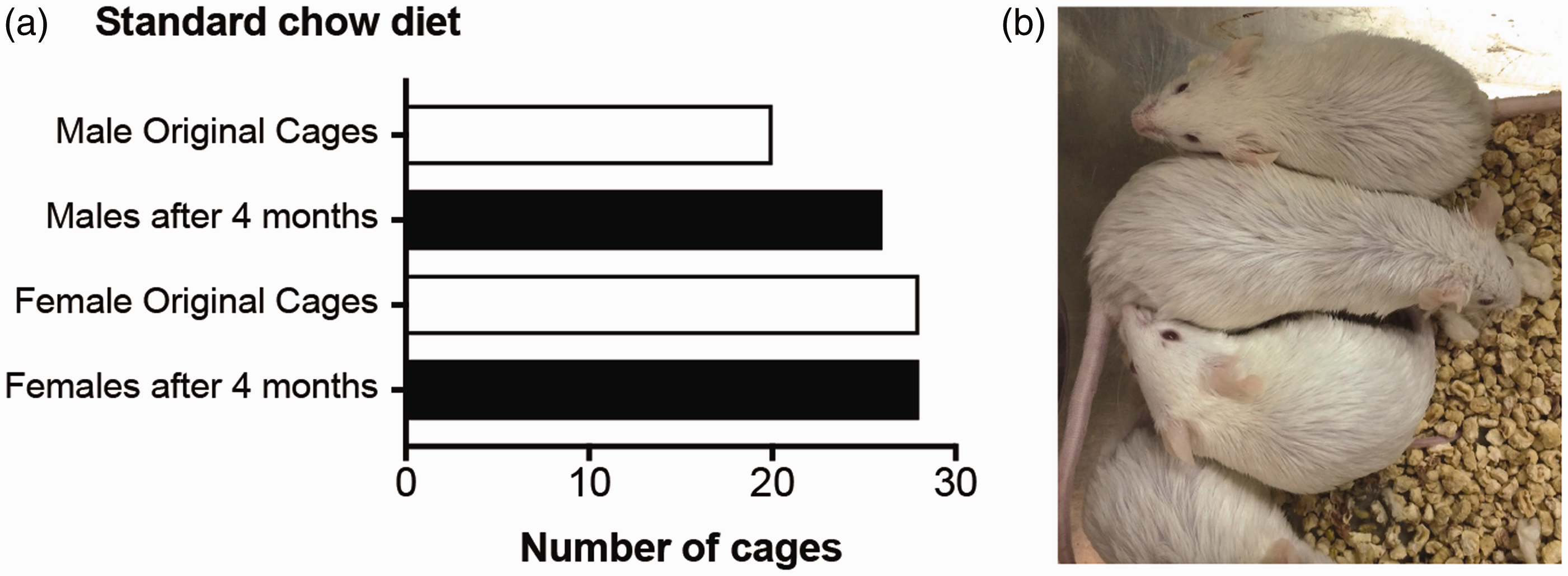
Diets were altered in a small group of the animals after 10–16 weeks to measure weight changes leading to adult-onset, diet-induced obesity. In this regard, some male cages were provided ad libitum high-fat (45%) or lean (10% fat) diets over a period of 27 weeks. Males were assigned a cage based on their genotype (Supplementary Figure 1) and littermate status before the food was altered, with a stringent prerequisite to keep siblings together and encourage peaceful cohabitation (Figure 1(b)).
Over the 27 weeks of the diet experiment, the weight of the males in the respected cages began to increase or stabilize, based on the food provided (Figure 2(a)). The final average weights of the lean-diet-fed cages were approximately 35 g compared with 43 g for high-fat diet cages (Figure 2(b); comparing lean with high-fat diets with the same genotype). The changes in weights ranged from 5 g to 18 g (Figure 2(c)). This was reflected in the fur of high-fat-diet-fed mice as it changed in appearance due to the oily red food and the propensity of the males to eat and nest in it, creating a greasy coat (Figure 2(d)). The study was terminated when one of the largest males, who exceeded 60 g, suddenly died for unknown reasons, although obesity and/or fatty liver disease were suspected by the veterinarian upon necropsy (data not shown).
Male FVB/N mice show significant increases in weight with high-fat diets. (a) Males were fed either a lean or high-fat diet for 27 weeks and then weighed. Box plots shows the range of (b) the final average weight or (c) the change in weight, in grams, per group at the end of the study. The association herein is related to diet, not the transgene, which has no affect. (d) Image comparing a mouse fed a lean diet (bottom) with a mouse fed a high-fat diet (top). Note the difference in coat appearances between the mice due to the fat content of the diets.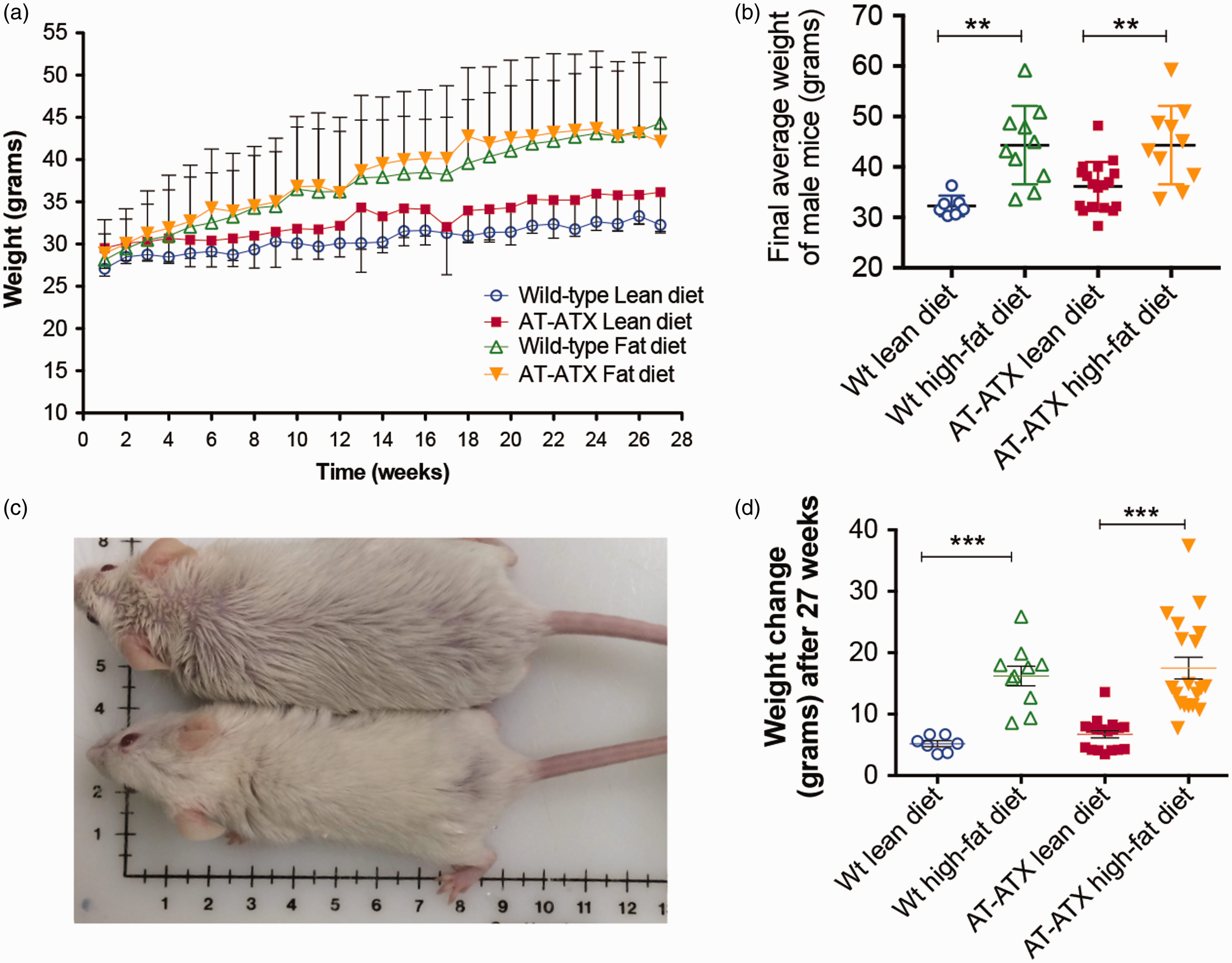
Initially the experiment commenced with peaceful cohabitation among the groups, and this continued in the male high-fat diet cages (Figure 3(a)). Unexpectedly, males in the lean diet cages began to show wounds during morning observations (Figure 3(b)). Cage separations were necessary to stop fighting, with some unfortunate victims of nocturnal bouts of aggression, during the time of day when rodents are active. Although there were more victims of aggression in the lean diet-fed cages, over the course of the experimental period, there was not a significant difference in overall survival between groups (Figure 3(c)).
Males fed a lean diet significantly increase aggressive behaviors. (a) Bar graphs displaying the overall number of cage separations over the entire diet experiment. The study was required to be terminated due to spontaneous death among an obese male mouse. (b) Image of mice from a lean-diet-fed cage showing a victim (left) that suffered a serious wound as a result of the perpetrator (right) from nocturnal activities that were discovered the next morning. The perpetrator is seemingly sniffing the opponent, even after inflicting horrific wounds overnight. The veterinarians requested that both animals be euthanized using a method consistent with the guidelines of the American Veterinary Medical Association. (c) A Kaplan–Meier statistic was used to estimate the overall survival of the mice in the study, based on diet. The solid red line indicates those on the high-fat diet versus the dashed line for the lean diet. Results were not statistically significant. (d) Group cage separations that were required during the diet study.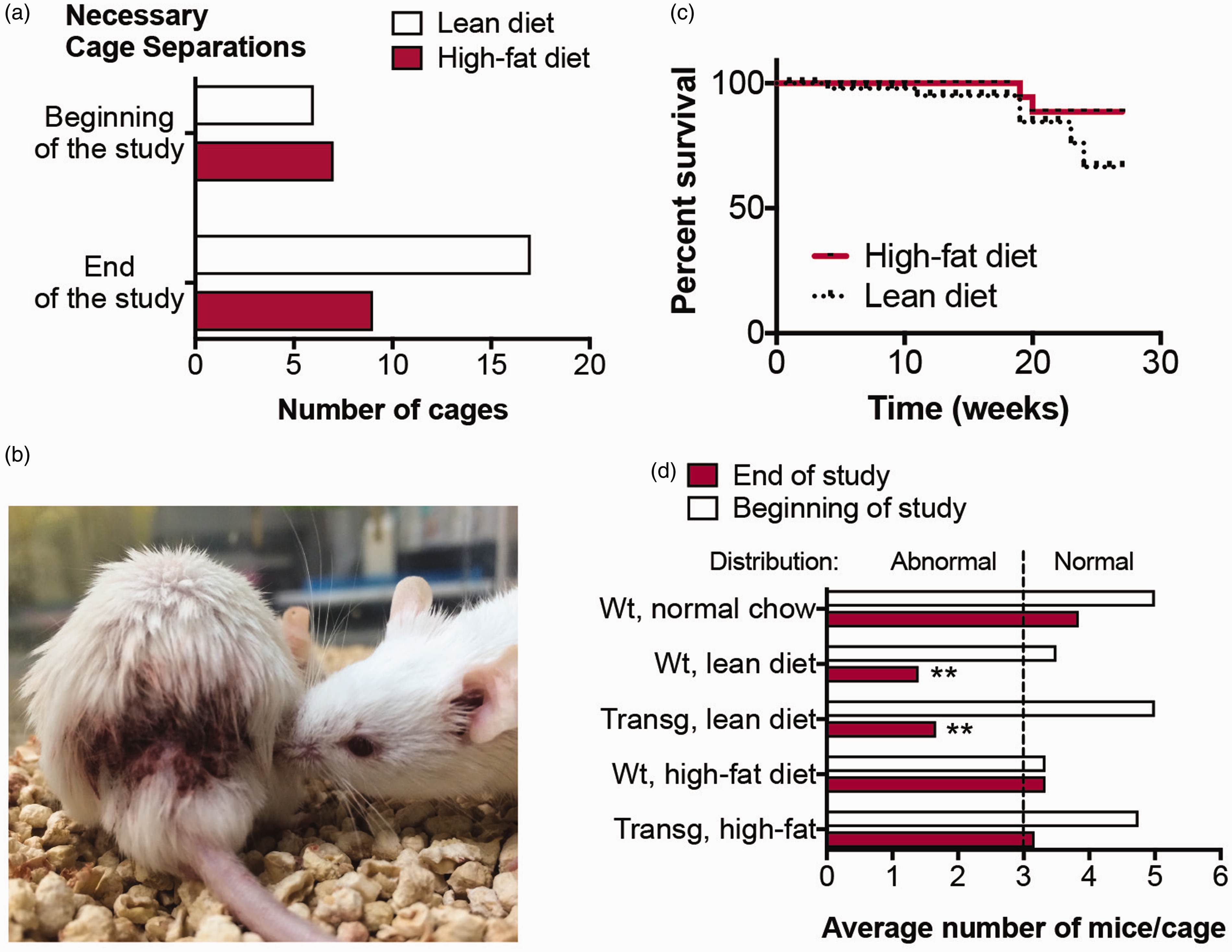
At first we attributed this to male FVB/N behavior and hypothesized that either the transgene, colony size or diet was the source. Initially, each male cage had an average of 3–5 littermates in it when the diet study commenced, dependent on the litter size and sex at birth. However, the number per cage dropped to an average of 1.4 (among wild-type, lean diet cages) to an unchanged 3.3 (among wild-type, high-fat diet cages) by the study's end (Figure 3(d)). Once fighting had occurred exclusively in nearly every lean cage, it became apparent that the behavior was abnormal.
The most striking reduction in the number of mice per cage occurred in the transgenic lean diet cages, where the average number started at 5.0 and ended with 1.6 (p < 0.01). Although we hypothesized that the transgene, autotaxin, may be related to the aggressive behaviors observed, the following statistical data did not support that. Among the high-fat diet transgenic cages, the average number of mice per cage started at 4.75 mice and finished at 3.2 mice. Similarly, the wild-type lean diet cages started with an average of 3.4 mice and finished with 1.3 mice. This suggested that diet was the causal factor.
Randomization model results
Thus, it became necessary to design a strategy to quantify the effect of each group. In the beginning of the study, six lean and seven high-fat diet cages were created, and at the end of the study, 17 lean and nine high-fat diet cages were recorded. To obtain the p-value, a statistic must be found that gives some measure of the differences between the treatment groups and the control. A natural cage-level response variable is the number of separations in a cage divided by the total number possible. A corresponding statistic typically used is an F-test statistic. Note that in this case the data need not be normal, and the F-statistic need not have an F distribution, because our results will not rely on any asymptotic, large-sample theory. There is a total of 156 mice, with an average of 4.5 per cage. The maximum number of possible separations for a cage is the number of animals in that cage minus one. The observed F-statistic based on the proportion of separations was 3.72 and the p-value from 100,000 permutations was 0.025 (estimated standard error 0.0005). The histogram of the permutations is shown in Figure 4 and can also be found at: https://github.com/dobbinke/Rmarkdown4cagesep. Since the p-value is less than 0.05, we reject the null hypothesis at the 0.05 significance level and conclude that the separations differ by treatment assignment for at least some of the treatment groups and control.
F-statistic histogram of the data permutations. The F-statistic shows the ratio of the variances. Because the p-value is less than 0.05, this resolves that it is appropriate to reject the null hypothesis at the 0.05 significance level. Further, the statistics conclude that the separations differ by treatment, for at least some of the treated groups and control. The vertical line is observed.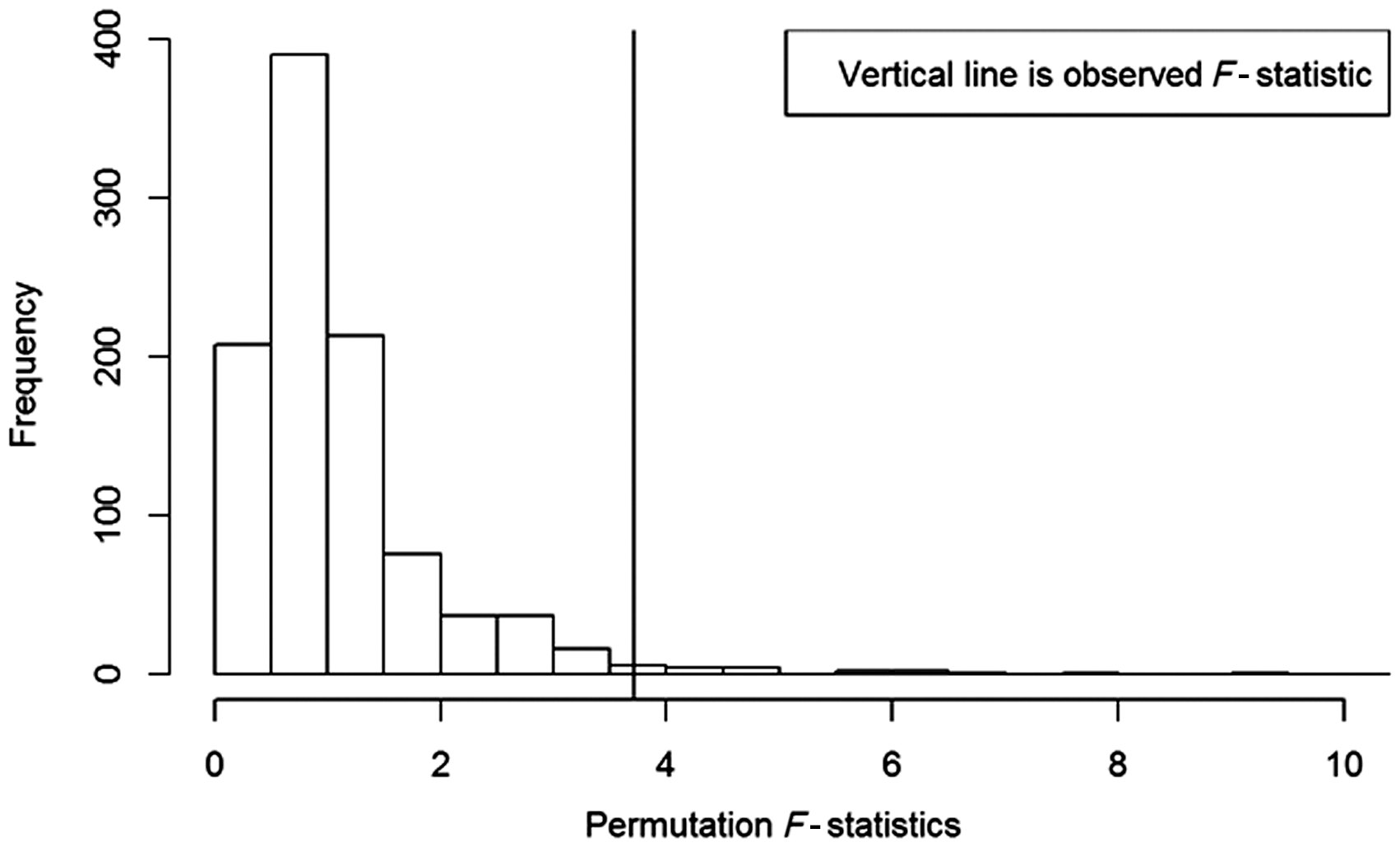
Population model results
Therefore, for cage i assigned to condition
Then
Multiple comparison of means with Dunnett contrasts.
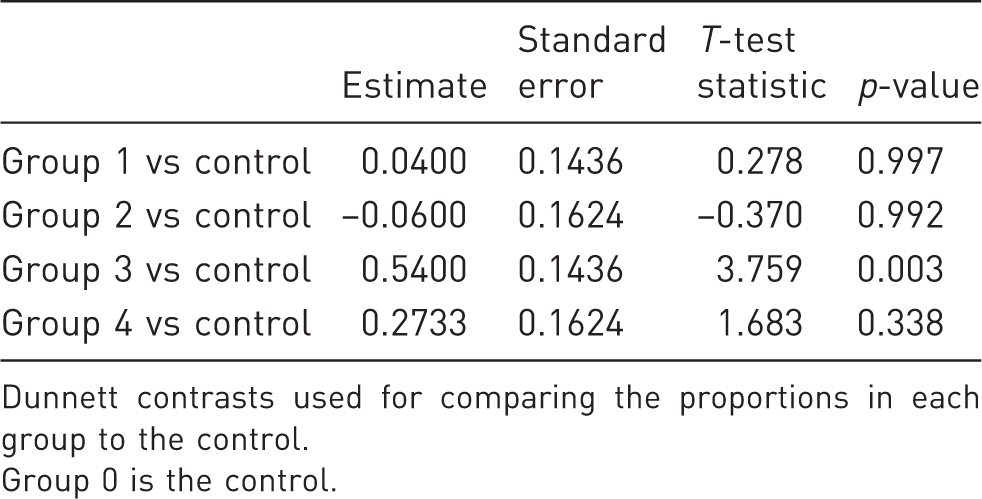
Dunnett contrasts used for comparing the proportions in each group to the control.
Group 0 is the control.
Biochemical analysis
To biochemically explain the reason why lean-diet-fed mice exhibit more aggression towards littermates, we initially hypothesized that the concentration of testosterone was variable between the groups. However, a comparison of lean-diet-fed mice versus high-fat-diet-fed mice demonstrated no significant difference in testosterone (Figure 5(a)). In contrast, when the ratio of ADP/ATP was assessed in tissue, the lean-diet-fed mice demonstrated a significant decrease in comparison to high-fat-diet-fed mice (Figure 5(b)). This suggests that energy status and metabolism are the crux of the enhanced nocturnal activity,16–18 which is consistent with this strain. Immunoblotting of the tissues from the animals showed no apparent differences in the expression of the androgen receptor between groups (Figure 5(c)).
Biochemical analysis of male mouse tissue. (a) Serum was collected at necropsy and tested for the concentration of testosterone (ng/mL). (b) The ratio of ADP/ATP was measured in the male tissues to assess the energy status of the groups. (c) Immunoblotting of androgen receptors did not reveal any striking trends between the groups and quantification of the bands revealed no differences.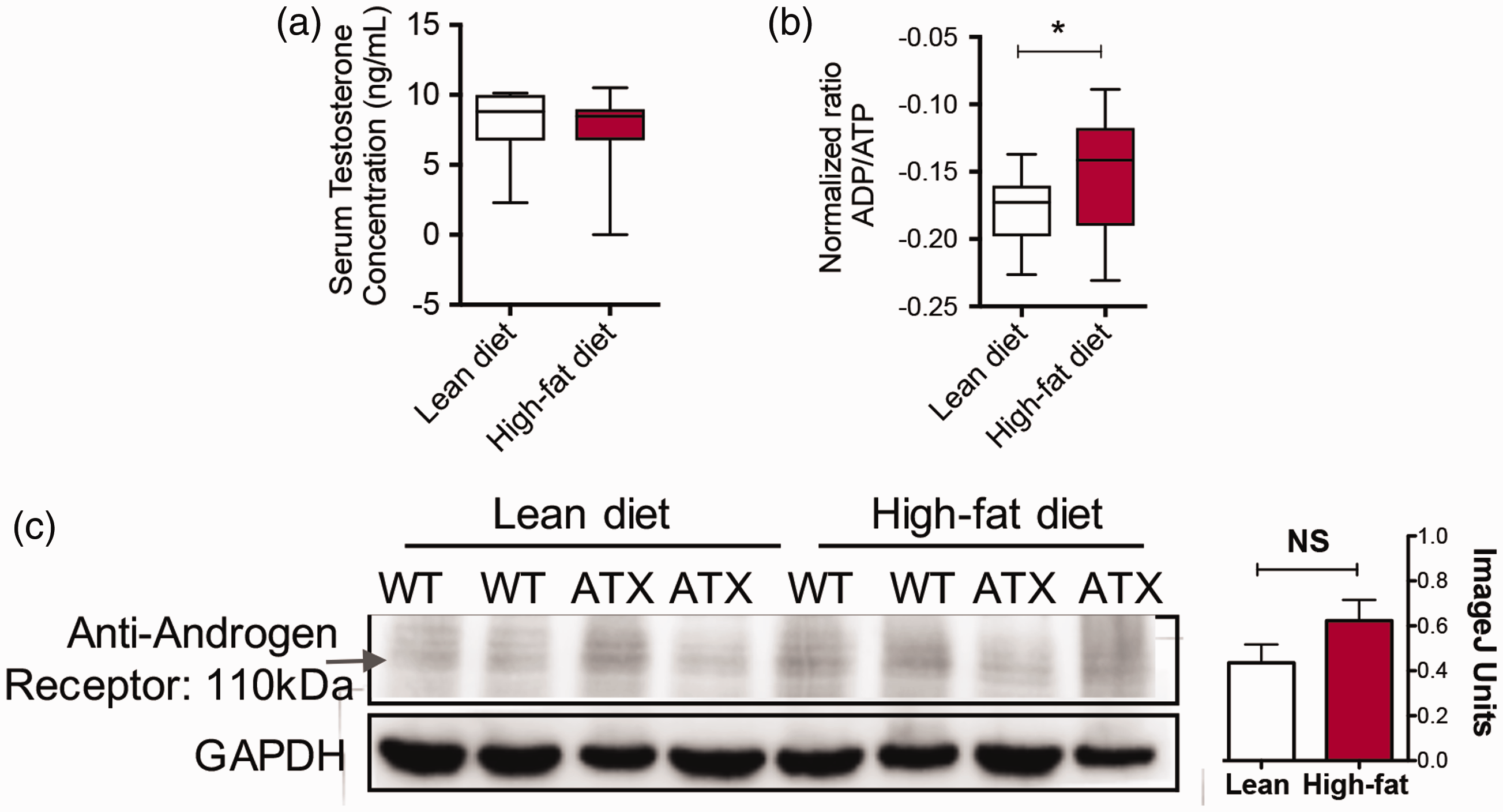
Discussion
In this study, we observed unexpected, heightened manifestations of aggressive nocturnal bouts among FVB/N male cages fed a lean diet. To evaluate this phenomenon, we developed statistical modeling and computation methods to analyze the data using randomization and population models with a censored likelihood. We conclude that male cage separations differ significantly due to a lean diet (i.e. treatment assignments) and are unrelated to transgene, autotaxin, status and the testosterone level of the mice.
The logical conclusion of our study is that energy among FVB/N males is intensified by feeding males a lean diet because this is a normally hyperactive strain,1,2 which then has amplified activity for nocturnal bouts of aggression. The FVB/N strain is defined in some circumstances by their aggressive nature, although this is almost exclusively assigned to males, not females. 8 Previous studies with crosses of inbred CBA and outbred CFLP mouse strains determined that lean mice exhibit more nocturnal wheel running and higher food intake, compared to fat mice, with lean mice expending substantially more energy towards activity. 18
The opposite is also true – feeding hyperactive FVB/N males a high-fat diet mitigates active behavior because they become sedentary and are less likely to engage in vigorous nocturnal movements. Fat inbred CBA and outbred CFLP mice are also less likely to participate in activities and will steadily decrease participation in nocturnal behaviors, such as wheel running and vertical rearing. 19 Taken together, a lean diet with four-fold less fat enhances active behaviors and keeps the mice agile so that they are more likely to engage in nocturnal bouts of aggression.
Therefore, the contributing factor that causes the hyperactive, aggressive FVB/N mice to increase nocturnal activity is a lean diet with 17% sucrose and twice the amount of carbohydrates. This diet heightens nocturnal behaviors, whereby FVB/N male energy is expended inappropriately on aggressive behavior. To this point, FVB/N mice have a significantly higher total glucose production compared with C57BL/6J, 129S1/SvImJ and ICR mouse strains. 20 Since FVB/N mice exhibit reduced anxiety levels in comparison to other strains, 21 it is unlikely that anxiety contributes to this phenomenon.
We also tested testosterone levels and androgen receptor expression between groups, but the results were not significant (Figure 5(a) and (c)), suggesting these were not contributing factors for this study. However, this is in contrast with a study showing that the highly aggressive CD1 strain ceases fighting upon castration. 22 Nevertheless, castration has major ethical considerations and is an unnatural scenario; and, yet, an adult male living among a group of other adult male mice is an unnatural scenario, too. Most wild mice territories have one adult male, several females and their pups, which is not practical in the laboratory. 23
When planning the study, we sought to mitigate FVB/N aggressive tendencies by housing males with littermates, which facilitates stable social groups and hierarchy from an early age, and by maintaining group housing since periods of single housing can exacerbate aggression. 24 Other studies show that stress can be induced in male mice with olfaction exposure to human male experimenters 25 ; however, all of the human experimenters in this study were female. The only male in the research group was the statistician who did not handle the mice.
On the other hand, cages did have structural enrichment, in the form of a mouse house, that was notably used as a male marking post. Sometimes these types of structures provide opportunities for dominant perpetrators to ambush a victim or attack them as they enter and exit the mouse house. 24 Although nesting material was provided and is supposed to decrease aggression, 26 this did not prevent aggression among the FVB/N males fed a lean diet, but we did not test what occurs in the absence of nesting material.
Prior to this study, we did not anticipate an elevation in overnight wounding to arise in male cages fed lean diets. To control the fighting, cages with wounded mice required separation to avoid future occurrences. By the end of the study, we had an entire housing rack filled with singly housed males in the facility, which requires written justification for each circumstance and communication with the veterinary staff. Studies have shown male mice prefer conspecific housing with another male versus an empty cage, even in the presence of aggression. 27 Therefore, group housing is our standard practice and deviations must be explicitly justified. We avoid solitary housing unless it is absolutely necessary, a position which coincides with the general consensus in favor of group housing for male mice. 23 Recently, Weber et al. strongly recommended limiting necessary male group housing to three animals in a standard cage and not providing shelter enrichments under scenarios that are known to increase aggression. 28
In summary, we developed a novel statistical method for analyzing an abnormal set of biological data, stemming from a study about diet-induced obesity. Although unexpected, we observed a rise in male nocturnal bouts of aggression among cages with ad libitum lean food. Using the randomization and population models, we concluded significance was present in some of the data. Cage separations were not random; rather, they were resultant from a lean diet consumed by FVB/N adult males that enhanced energy status and resulted in nocturnal bouts of aggression. In conclusion, the hyperactive and subsequent aggressive behavior of the FVB/N adult males them an unsuitable model for long-term studies requiring lean-diet modifications; however, they could be utilized to study male mouse aggression that only requires a modification in diet.
Supplemental Material
Supplemental Material1 - Supplemental material for Diet-regulated behavior: FVB/N mice fed a lean diet exhibit increased nocturnal bouts of aggression between littermates
Supplemental material, Supplemental Material1 for Diet-regulated behavior: FVB/N mice fed a lean diet exhibit increased nocturnal bouts of aggression between littermates by Mandi M Murph, Shuying Liu, Wei Jia, Ha Nguyen, Megan A MacFarlane, Susan S Smyth, Sudeepti S Kuppa and Kevin K Dobbin in Laboratory Animals
Supplemental Material
Supplemental Material2 - Supplemental material for Diet-regulated behavior: FVB/N mice fed a lean diet exhibit increased nocturnal bouts of aggression between littermates
Supplemental material, Supplemental Material2 for Diet-regulated behavior: FVB/N mice fed a lean diet exhibit increased nocturnal bouts of aggression between littermates by Mandi M Murph, Shuying Liu, Wei Jia, Ha Nguyen, Megan A MacFarlane, Susan S Smyth, Sudeepti S Kuppa and Kevin K Dobbin in Laboratory Animals
Footnotes
Acknowledgement
Our thanks to Charnel Byrnes for proofreading this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The research was supported by a grant (to MMM) from the National Institutes of Health (grant number 1R15CA176653).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.