Abstract
Laboratory animal welfare has received increasing attention in recent years as housing protocols move toward favoring environments that allow natural behaviors. Within this study, the effects of housing male and female Sprague Dawley rats in standard cages versus taller cages with an upper shelf were investigated. To determine differences in behavior and physiology based upon cage type, home-cage assessment of ultrasonic vocalizations and analysis of fecal corticosterone metabolites, as well as various behavioral tests, were performed. Rats in shelved cages produced significantly less 50 kHz calls, demonstrated better working memory in the spontaneous alternation task and had higher concentrations of fecal corticosterone metabolites. No differences were observed in the open field, elevated plus maze or light–dark box. While no significant treatment differences were found in the ultrasonic vocalization playback paradigm, results confirmed previous evidence of approach behavior upon 50 kHz call playback. The observed differences in behavior and physiology as a consequence of housing conditions inevitably have implications for experimental reproducibility as comparing studies across laboratories may be difficult if different housing parameters are utilized. The results of this study can also be used in guiding future animal welfare protocols given that certain cage modifications such as increased vertical space and/or the presence of a shelf might improve welfare. Investigation of additional parameters and strains of rodents will enhance our understanding of optimal laboratory animal housing conditions.
Introduction
Understanding the influence of various environmental factors on laboratory animals is important for animal welfare and experimental consistency. While environmental enrichment of laboratory animals exists in many forms, from social enrichment such as housing animals in groups to physical enrichment such as adding a physical item to the cage, 1 variations in cage layout (i.e. larger floor area, increased vertical height, or multi-level shelfing) might also influence animal behavior and physiology. Agencies governing the ethical use of laboratory animals often have strict protocols and standards in place which have been gradually transitioning toward housing arrangements that allow natural behaviors. For example, ‘standard’ cages often prevent rodents from standing upright, a natural behavior that is evidenced to be beneficial for welfare,2–4 thus many agencies, such as the Canadian Council on Animal Care (CCAC), are transitioning toward the implementation of cages with an increased vertical height. 5 In addition to preventing various natural behaviors such as upright standing, burrowing, and climbing, 3 conventional rodent housing has also been demonstrated to cause anxiety-like behaviors 6 and compromise the immune system. 7 Alternative cage designs such as the Double Decker cage (Tecniplast, Montreal, QC, Canada) and the Optirat Plus cage (Animal Care Systems, Centennial, CO, USA), which both feature a shelf and increased vertical height, strive to overcome the welfare concerns associated with standard housing. Additionally, it is important to consider that environmental enrichment might be strain-, 8 age-, 9 and sex-dependent. 10 A frequent concern with modifying the home environment is that it will increase data variability; however, previous studies suggest the effects of home-cage enrichment on data integrity to be minimal.7,11,12 Overall, an understanding of the effects of different housing scenarios is essential in guiding future protocols and standards for the improvement of laboratory animal welfare as well as potentially detecting more subtle experimental differences.
While previous studies have begun to explore the impacts of different housing conditions on laboratory animals, additional tests are necessary given the difficulty with interpreting some of the outcomes. Tests designed to assess the same domain of behavior, such as anxiety, often yield contradictory results. 13 For example, the open field, 14 elevated plus maze, 15 and light–dark box 16 are all referenced as tests that assess anxiety-related behavior through observation of an animal’s response to a novel environment, yet numerous studies have observed discrepancies in the results of such tests. 17 Incorporation of additional tests that assess positive affect may further assist with interpretation of experimental findings. Ultrasonic vocalizations (USVs) are sounds produced by rats to communicate that may be passively recorded within the home-cage with the use of a specialized microphone to monitor affective state.18–21 Furthermore, the playback of USVs has also been used to induce a positive affective state in rodents within the home-cage. 22 Rats typically communicate via two main frequencies: 50 kHz calls (positive valence) and 22 kHz calls (negative valence); 50 kHz calls have been further classified into various categories based upon frequency modulation.18,23 Playback of 50 kHz USVs typically induces approach behavior while 22 kHz USVs induces inhibitory behavior. 24 The USV playback paradigm is a standardized assessment of social behavior that is not confounded by the presence of another stimulus animal. 25 Environmental enrichment is also often associated with increases in cognitive capacity, particularly spatial working memory.26–28 The spontaneous alternation task is an exploratory-driven test which assesses spatial working memory. Given an animal’s natural tendency to explore novelty, the test assesses memory through analysis of the sequence of arm entries. Additionally, concentration of fecal corticosterone metabolites is a relatively non-invasive method which relies on natural defecation as a passive method of sample collection that is more stable than blood corticosterone. 29
To determine behavioral and physiological impacts of housing rats in commercially available cages featuring a shelf and increased vertical height (Double Decker cages), males and females housed in standard versus shelved cages were assessed via traditional behavioral tests (open field, elevated plus maze, and light–dark box), the spontaneous alternation task, and the USV playback paradigm. Fecal corticosterone metabolite concentration and production of home-cage USVs were also assessed. It was expected that animals housed in shelved cages would demonstrate a significantly different behavioral and physiological profile compared with those housed in standard cages. Rats in shelved cages produced fewer 50 kHz calls, demonstrated improved working memory and had higher concentrations of fecal corticosterone metabolites. The results indicated significant differences between animals in standard versus shelved cages, confirming the importance of understanding the effects associated with cage modifications.
Animals, materials and methods
Animal husbandry
Male (n = 60) and female (n = 60) Sprague Dawley (CD®) rats (total = 120) were obtained from Charles River Laboratories (Saint-Constant, QC, Canada) at ~7–8 weeks old. Sprague Dawley rats were used as they are a general-use model. Four groups of 30 animals were utilized; each group contained animals of the same sex with 15 animals in five standard cages (control) and 15 animals in five shelved cages (treatment) with 3 animals/cage. Upon arrival, rats were allowed a one-week acclimatization period within standard cages (Figure 1). Following acclimatization, rats were habituated to the testing suite for 5 min, 1× per day for five days, and handled via picking up and placing them back in the cage 1× per day for five days as previously described. 13 Rats were then randomly allocated via random number generator to standard cages (508 mm × 406 mm × 216 mm) (Ancare Corporation, Bellmore, NY, USA) or GR1800 Double Decker cages (381 mm × 305 mm × 394 mm) (Tecniplast, Montreal, QC, Canada), termed ‘shelved cages’ throughout the study. Animals were maintained on an ad libitum diet of Laboratory Rodent Diet 5001 (LabDiet, Saint Louis, MO, USA) and water. All cages were bedded with Hardwood Beta Chips (North-Eastern Products, Warrensburg, NY, USA), contained a PVC tube as shelter, and changed twice a week. Daily animal welfare checks were performed throughout the duration of the study. No animals were excluded. Conditions within the colony room were as follows: room temperature at 19–22°C, humidity at 30–52%, overhead fluorescent lighting at 300–400 lux, ambient room sound ~60 dB. A 12-h reverse light–dark cycle (lights off 06:00h–18:00h) was utilized; a red lamp was used to provide necessary light during the dark cycle. All behavioral testing was conducted between 08:00h and 15:00h. At the end of the study, animals were anesthetized using isoflurane and decapitated. All procedures performed were conducted in accordance with the guidelines of the CCAC and approved by the University of Prince Edward Island Animal Care Committee protocol #21-032.
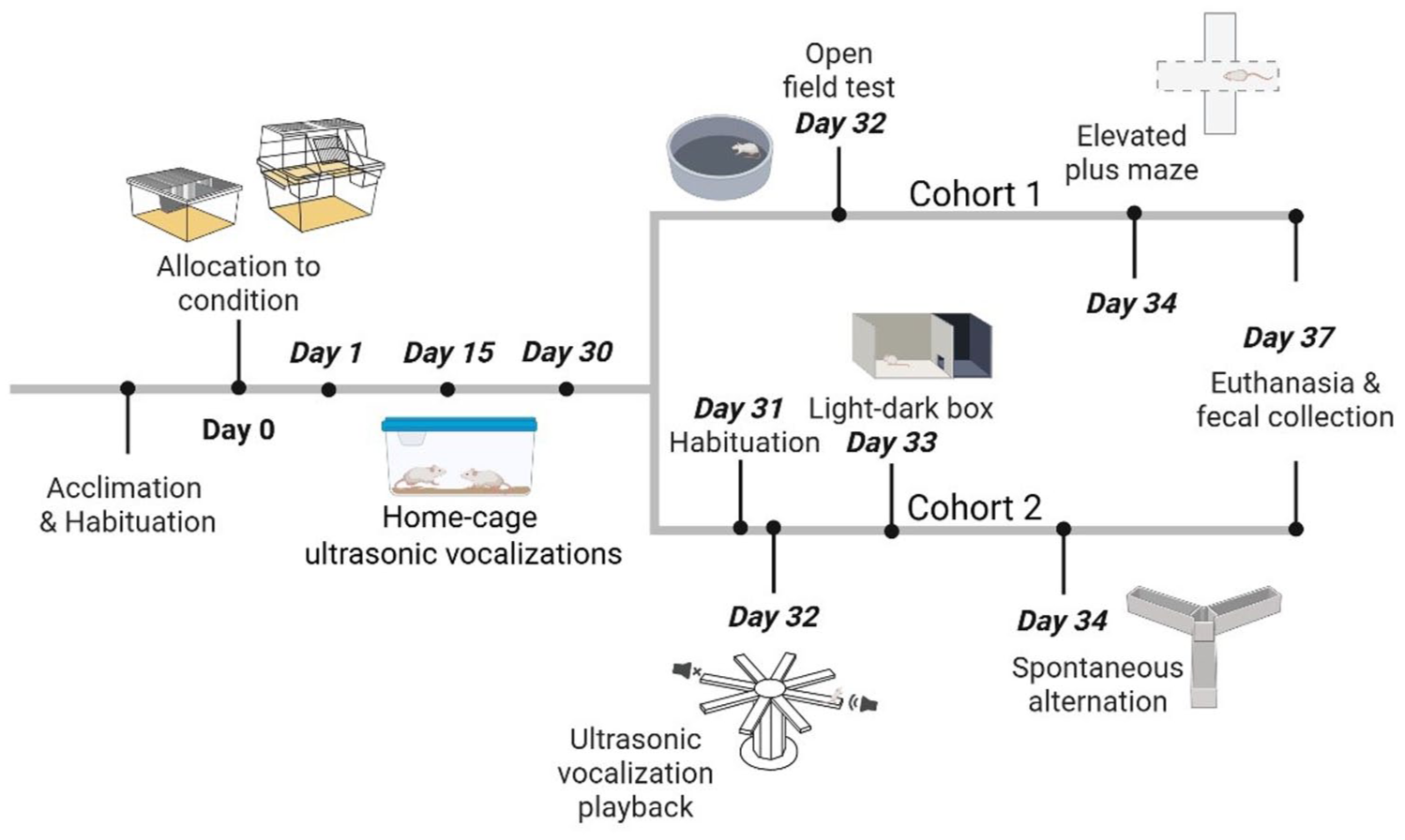
Home-cage USVs
Testing occurred on days 1, 15, and 30 following allocations to condition. Order of testing was randomized. On testing days, each cage was transported to the testing suite and allowed to acclimate for 5 min prior to a 5-min USV recording performed while the animals remained in the home-cage. USV recordings were performed using Avisoft Ultrasound Gate with a CM16 microphone and analyzed using SAS LabPro (Avisoft Bioacoustics, Glienicke/Nordbahn, Germany). Analysis of spectrograms occurred blind to experimental treatment. The microphone was placed approximately 125 mm away from the front of the wire lid of the shelved cage and suspended 125 mm above the wire lid in the center of the standard cage. The total number of calls emitted throughout the 5-min trial was determined and each 50 kHz call was classified based upon characteristic using three categories: flat, frequency-modulated, or trill. 21 The number of 22 kHz calls produced within the 5-min interval was also recorded.
Behavioral and cognitive tests
Two cohorts of animals of each sex were utilized. One cohort was tested in the open field and elevated plus maze while the second cohort was tested in the light–dark box, spontaneous alternation task and USV playback paradigm (Figure 1). Following the last day of home-cage USV recordings, behavioral testing was initiated. For all tests, cages were randomly selected via random number generator and each rat was transported from the colony room in a transfer cage. Once in the testing suite, each rat was given a 5-min habituation period prior to being placed in the maze. Upon placement in the maze, a video recording was initiated using a HiSeeU infrared camera at a wavelength of 590nm (London, United Kingdom) and later analyzed using Noldus Ethovision XT 14 (Wageningen, Netherlands). Video recordings were analyzed blind to experimental treatment. Some files were excluded from analysis owing to technical difficulties (see Supplemental material online).
The open field consisted of a circular arena (inner diameter: 91.5 cm; side height: 50.8 cm) where each rat was placed in the center of the maze and allowed to explore for 10 min. The elevated plus maze consisted of a plus configuration elevated off the floor by 83.8 cm with two closed arms (side height: 30.5 cm) and two open arms (119 cm × 10.2 cm) where each rat was placed in the center of the maze facing an open arm and permitted to explore for 5 min. The light–dark box contained a light and a dark chamber (each chamber with dimensions of 40.6 cm × 40.6 cm × 39.4 cm) with an opening (9.5 cm × 8.3 cm) separating the two chambers, where each rat was placed in the dark chamber and permitted to explore for 5 min. The primary measures of interest were time spent in the more aversive area (center of open field, open arms of elevated plus maze, light chamber of light–dark box).
The spontaneous alternation apparatus consisted of three closed arms (length: 57 cm; side height: 30 cm) at 120° where the animal was placed into an arm facing away from the center and allowed to explore for 5 min. The primary measure of interest was percentage alternations, calculated as the number of alternations divided by the possible number of alternations. The number of alternations was defined as the number of times all three arms were visited before revisitation of an arm and the total number of possible alternations was determined based on the number of arm entries. 30
The playback apparatus was a radial arm maze, previously described.24,31 Two speakers (one control and one emitting noise) were positioned at opposite ends of the maze (position randomized), about 30 cm from the end of each arm. Prior to testing, rats were habituated by allowing exploration of the radial arm maze for 10 min without sound. The following day, each rat was placed in the maze and allowed to explore for a 10-min trial. No noise was played for the first minute and white noise (70 dB) played for the second minute. A 2-min interval during which no noise was played subsequently occurred followed by either 50 kHz or 22 kHz USVs (order randomized) at ~70 dB, which were played for a period of 1 min, each followed by a 2-min interval of no noise. The white noise was previously recorded from the same room where testing occurred. The calls used for the 50 kHz and 22 kHz playback originated from a female rat at the same age exploring an open field arena. For the 50 kHz playback, a sequence approximately 10 s long was repeated six times which consisted of a total of 270 calls, 45 different calls repeated (13 flats, 16 frequency-modulated and 16 trills). For the 22 kHz playback, a 60 s sequence consisting of all unique calls was used. All unique calls were utilized for the 22 kHz playback since a repeated sequence of these calls would risk losing information owing to the temporal pattern inherent to this call type. 24 The primary measure of interest was time exploring the proximal arms (the arm with the sound emitting speaker and two adjacent arms).
Fecal corticosterone metabolites
Three days after behavioral testing, a fecal sample was collected. Rats were placed singly into standard cages until defecation occurred. If a rat did not defecate during the allotted 6-h time, it was returned to the colony room. No further attempts at fecal collection were made owing to concerns regarding temporal sampling consistency and welfare. Once defecation occurred, fecal matter was immediately retrieved using sterile forceps, placed in a sterile collection tube and frozen at −80°C until further use. In preparation for enzyme-linked immunosorbent assay (ELISA), samples were thawed, flash frozen in liquid nitrogen, and homogenized using mortar and pestle. Samples were combined with 80% methanol at 0.1 g:1 mL, vortexed for 30 min and centrifuged for 15 min at 2500 × g. The supernatant was collected and stored at −80°C. Sample supernatants were heat inactivated at 95°C for 15 min immediately before use and diluted 1/20 in assay buffer. Fecal corticosterone metabolites were determined via commercial ELISA kits (Arbor Assay kit K014-H1, Ann Arbor, MI, USA). Experimenters were blinded to treatment during analysis.
Statistical analysis
All data were analyzed using Jamovi version 2.3 for Windows (Jamovi Software) or GraphPad Prism 9 version 6.01 for Windows, with all graphs generated using GraphPad Prism 9 (GraphPad Software, San Diego, CA, USA). Model assumptions were first assessed for all data sets; Levene’s test was utilized to test homogeneity of variances, and the Shapiro–Wilk test was performed to assess normality. For data sets that did not violate assumptions, a generalized linear mixed model was utilized to account for cage as a random effect within tests where each rat was considered the statistical unit (behavioral tests and the USV playback paradigm). Factors of sex and treatment were used for the behavioral tests and factors of sex, treatment and type of playback were used for the USV playback paradigm. The elevated plus maze did not meet the assumption for normality as assessed via the Shapiro–Wilk test (W = 0.922, p < 0.001). It should be noted that there is a significant debate surrounding the violation of the normality assumption for generalized linear mixed models given that these tests are fairly robust in terms of fitting non-normal data, 32 thus this parameter was still analyzed based on a generalized linear mixed model. For home-cage USV analysis, a mixed effects model was utilized (owing to some missing values) using treatment and sex as factors and time as a repeated measure; the statistical unit for this test was cage. The Geisser–Greenhouse correction was utilized where the assumption of sphericity was not met according to Mauchly’s test. Fecal corticosterone metabolite analysis was performed using the non-parametric Kruskal–Wallis test given that the assumption of normality was violated (W = 0.930, p = 0.040) according to the Shapiro–Wilk test. Significance in all tests was determined by p < 0.05 and significant findings were followed up with appropriate post-hoc analysis.
Results
Home-cage USVs
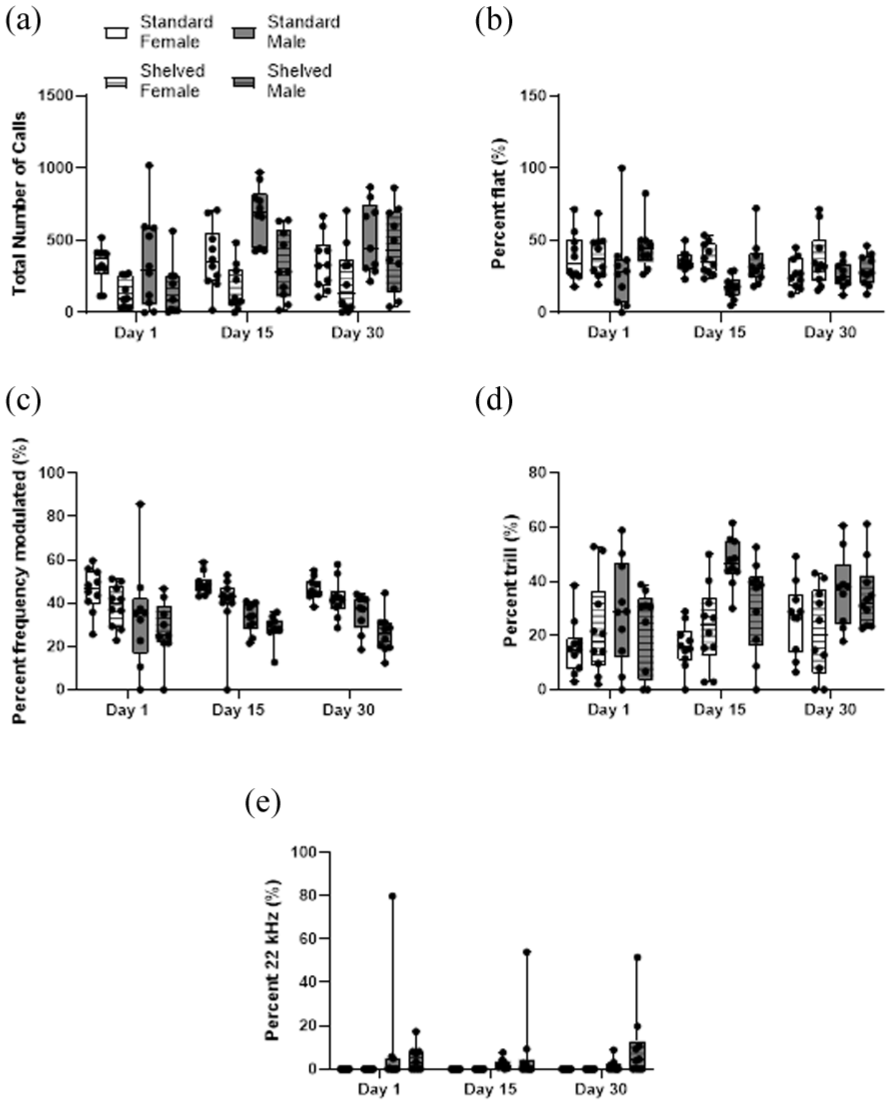
Assessment of total number of calls produced over a 5-min interval throughout the study (days 1, 15, and 30), indicated a significant treatment effect (F (1, 18) = 11.49, p = 0.003), sex effect (F (1, 13) = 7.16, p = 0.026), and time effect (F (2, 36) = 9.17, p < 0.001) (Figure 2(a)). Female rats in standard cages produced significantly more USVs compared with those in shelved cages (t (4) = 6.25, p = 0.003). No significant differences in male rats were observed (t (4) = 1.68, p = 0.168). Rats produced significantly more calls on day 30 compared with day 1 (t (17) = 4.77, p < 0.001. Analysis of percentage of flat calls showed a significant treatment (F (1, 18) = 5.29, p = 0.034) and time effect (F (2, 36) = 3.97, p = 0.028), but no sex effect (F (1, 15) = 2.89, p = 0.114) (Figure 2(b)). Animals in shelved cages produced significantly more flat calls compared with those in standard cages (t (116) = 2.74, p = 0.007). Analysis of percentage of frequency-modulated calls showed a significant treatment (F (1, 18) = 9.42, p = 0.007) and sex effect (F (1, 14) = 39.42, p < 0.001), but no time effect (F (2, 36) = 0.10, p = 0.901) (Figure 2(c)). Animals in standard cages produced significantly more frequency-modulated calls compared with those in shelved cages (t (115) = 3.10, p = 0.002). Analysis of percentage of trill calls showed no significant treatment effect (F (1, 18) = 0.53, p = 0.476), but there was a significant sex (F (1, 16) = 13.33, p = 0.003) and time effect (F (2, 36) = 5.38, p = 0.009) (Figure 2(d)). Males produced significantly more trills than females (t (116) = 4.58, p < 0.0001). No female rats produced 22 kHz calls, therefore only a sex effect was observed (F (1, 87) = 9.64, p = 0.004).
Behavioral and cognitive tests
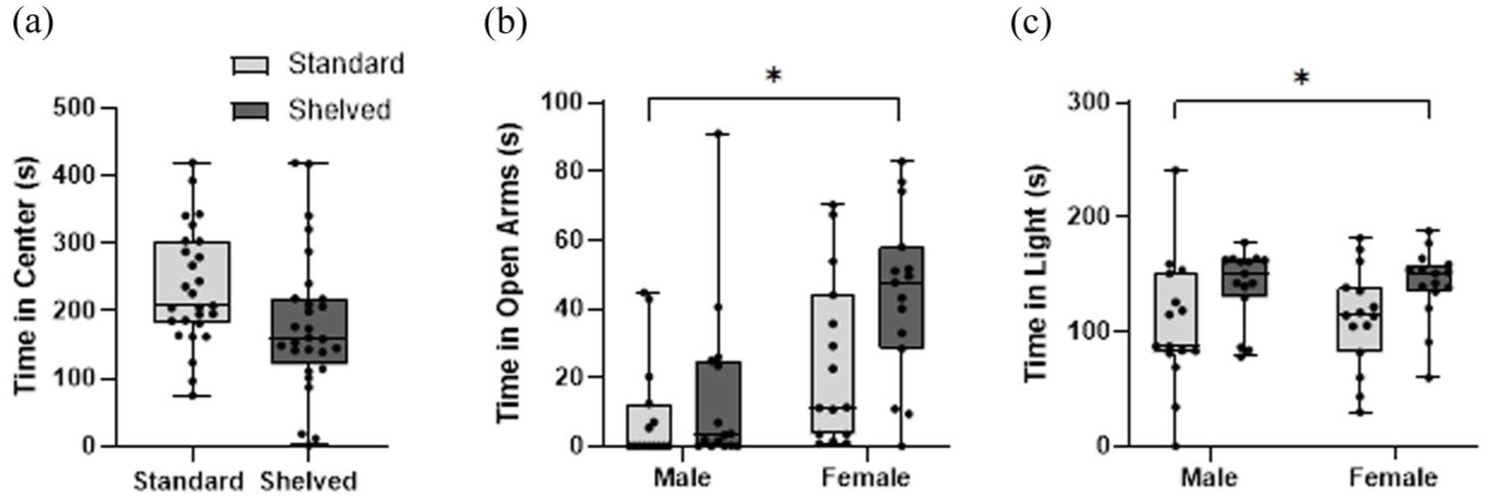
Assessment of time in the center of the open field showed no treatment (F (1, 17) = 2.97, p = 0.103) or sex effects (F (1, 17) = 0.0047, p = 0.946) (Figure 3(a)). Assessment of time in the open arms of the elevated plus maze showed no treatment effects (F (1, 16) = 4.33, p = 0.054), although there was a sex effect (F (1,16) = 13.29, p = 0.002) (Figure 3(b)). Assessment of time spent on the light side of the light–dark box indicated no treatment effect (F (1, 16) = 4.30, p = 0.055) (Figure 3(c)), although there was a sex effect (F (1, 16) = 12.16, p = 0.003). There were no effects for cage as a random variable (open field: p = 0.219, elevated plus maze: p = 0.770, light–dark box: p = 0.800).
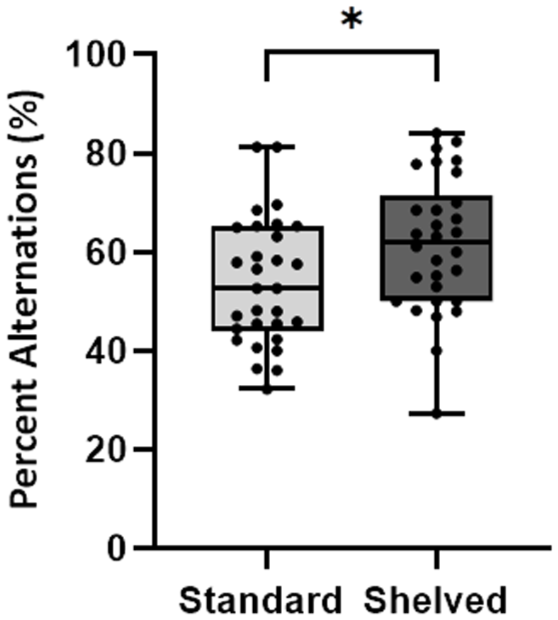
When assessing percentage of alternations, a significant difference between animals in standard versus shelved cages was observed (F (1, 56) = 4.99, p = 0.029) (Figure 4); no sex effects were observed (F (1, 56) = 0.02, p = 0.886) with no effect for cage as a random variable (p = 1.00).
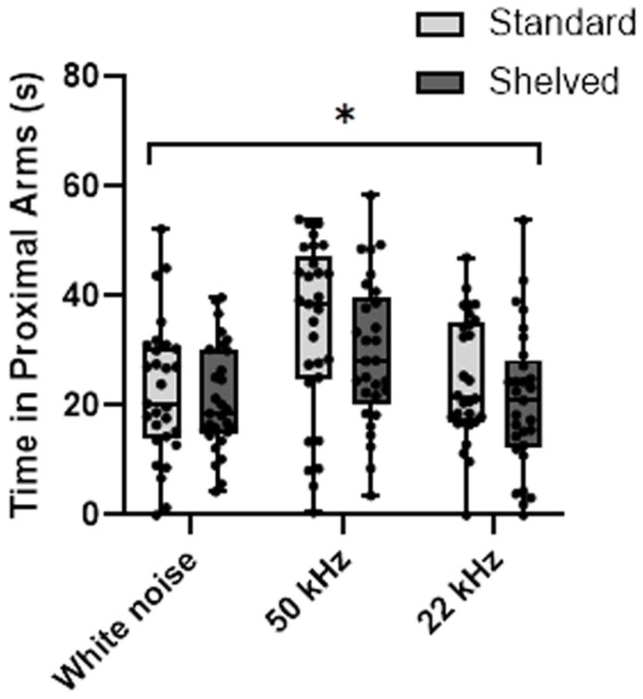
When assessing the playback of USVs, there was no treatment effect (F (1, 17) = 0.54, p = 0.471) or sex effect (F (1, 17) = 0.22, p = 0.646), but an effect for type of sound emitted (F (3, 201) = 19.43, p < 0.001) (Figure 5). Rats spent significantly more time in proximal arms during playback of 50 kHz calls compared with both 22 kHz calls (t (114) = 3.52, p = 0.0006) and white noise (t (114) = 4.06, p < 0.001). No significant difference in time spent in proximal arms between 22 kHz calls and white noise was observed (t (114) = 0.46, p = 0.6456).
Fecal corticosterone metabolites
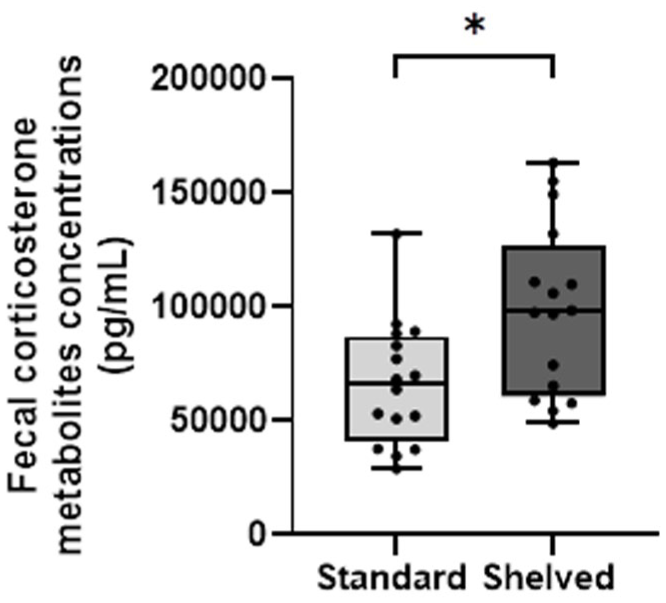
There was a significant treatment effect for corticosterone concentration, with higher concentrations observed in animals in shelved cages (H (1) = 6.76, p = 0.009) (Figure 6).
Discussion
The results of this study indicated some significant differences between male and female rats housed in standard versus shelved cages. Rats in shelved cages exhibited a decrease in 50 kHz home-cage USVs (Figure 2), exhibited improved working memory (Figure 4), and had higher fecal corticosterone metabolite concentrations (Figure 6). While all rats had a greater response to 50 kHz calls compared with white noise and 22 kHz calls, no treatment differences were observed in the ultrasonic vocalization playback paradigm (Figure 5). No significant differences within the open field, elevated plus maze, and light–dark box were observed (Figure 3).
Analysis of USVs demonstrated an overall increase in the number of 50 kHz calls in standard cages, with an increase in the proportion of frequency-modulated calls and decrease in the proportion of flat calls (Figure 2). Given the association of 50 kHz calls with positive affect in rodents, these results suggest a moderate increase in affective state of standard housed rats. Alternatively, the differences observed in USVs could represent a more affiliative relationship between cage mates within standard cages. Furthermore, frequency-modulated calls have been shown to be reflective of a rewarding situation while flat calls are more typical in everyday social interactions, 21 providing evidence that, within this study, animals in standard cages relayed a more positive affective state compared with those in shelved cages. A possible explanation for such results might be explained through some of the sex differences noted for USV production.
Only male rats within this study emitted 22 kHz calls, which are commonly associated with a negative affective state. Social arrangements and/or hierarchies might modify the impact of enrichment. While it is clear that social hierarchies exist in male rats, less work has been conducted using females. 33 Access to the upper shelf within the shelved cages may be a resource worth defending, subsequently influencing stress and welfare. It is possible that male rats may emit 22 kHz calls when fighting and/or attempting to defend their territory. Male rats in shelved cages were previously found to spend more time fighting than those in standard cages, leading to the possible conclusion that cage mates might fight in an attempt to defend this resource. 23 Future studies examining the relationship between 22 kHz USVs produced and fighting may assist in elucidating the cause for the 22 kHz calls. Additionally, fighting behavior in females might be useful in understanding whether this behavior is exclusive to males or evident in both sexes. Assessment of a shelved cage featuring only increased vertical height in the absence of an upper shelf might also provide further insight into the individualized effects of each type of enrichment.
Assessment of response to USV playback showed that all animals responded similarly, 24 with no differences in response displayed between treatment groups (Figure 5). Previous findings have shown differences in social approach behavior as a consequence of various enrichment strategies with social approach behavior reduced owing to changes in physical enrichment and social approach behavior increased by social enrichment scenarios. 34 The findings here did not indicate any differences as a result of the change in cage layout.
Shelved cages appeared to improve spatial working memory as evidenced by the spontaneous alternation task (Figure 4). Similarly, various studies have previously provided evidence for improved spatial working memory in animals with an enriched environment,26–28 suggestive of the benefits of environmental enrichment. While greater fecal corticosterone metabolite concentrations were observed in animals housed in shelved cages (Figure 6), indicative of activation of the hypothalamic–pituitary–adrenal axis, such increases are not necessarily indicative of a negative affective state. 35 Increased corticosterone indicates increased arousal, which might be the result of the novelty of the shelved environment rather than attributed to negative stress or poor welfare. It is also possible that these results reflect slightly higher activity levels in rats housed within the shelved environment, which could be beneficial for translation and reproducibility as it is better to work with less sedentary animals. To accurately determine the cause of increased corticosterone metabolites, additional parameters such as behavioral tests must be interpreted in conjunction to provide a broader sense of welfare and emotion. Additionally, the assessment of baseline corticosterone levels for each individual rat as a comparison might give further insight into the interpretation of such results. Given that there were no significant differences between treatments in the open field, elevated plus maze, and light–dark box (Figure 3), it may be suggested that the increase in corticosterone metabolite concentration observed in shelved cages might be due to arousal or positive valence stress rather than anxiety. It is important to note that recent findings have suggested that many tests previously thought to assess anxiety, including the open field, do not reliably show behavioral changes when validated with certain anxiolytic compounds; however, the elevated plus maze and light–dark box appeared most suitable for assessing anxiety-like behavior in mice. 36 No similar re-validation of such tests has yet been conducted in rats. Consequently, while caution must be taken when interpreting the results of fecal corticosterone metabolite concentration owing to the various confounding factors and variability in extraction methods, 37 the outcome of the behavioral metrics supports the conclusion that animals in shelved cages are not predicted to have elevated corticosterone concentrations due to increased anxiety.
In summary, this study investigated the behavioral and physiological effects of housing rats in modified cages featuring increased vertical height and a shelf. Most importantly, this study brings awareness to the impacts of varied cage designs, especially given recent changes in cage requirements within some countries. 5 Given that there are consequences associated with placing rats in different types of cages, this study helps to emphasize the idea that variations in husbandry practices such as cage layout must be controlled for and reported within the literature for all animal-based studies. It is important to clarify that the results presented here are limited to a specific age, strain, and social scenario and that environmental enrichment introduced in adulthood might have effects different from those of enrichment introduced at other developmental time points. 9 Future studies should focus on the assessment of additional behavioral and molecular markers as well as examining the impacts of modified cages on different ages and strains of rats.
Supplemental Material
sj-docx-1-lan-10.1177_00236772251384749 – Supplemental material for Shelved cages impact affective behavior in Sprague Dawley rats – implications for animal welfare
Supplemental material, sj-docx-1-lan-10.1177_00236772251384749 for Shelved cages impact affective behavior in Sprague Dawley rats – implications for animal welfare by Logan J Bigelow, Emily K Pope, Clare PL Lee, Veronica K Sohasky, Sarah K MacLeod and Paul B Bernard in Laboratory Animals
Footnotes
Acknowledgements
Thank you to Tecniplast, Montreal, QC, Canada for providing the Double Decker Cages utilized in this study. These contributors had no role in the preparation of data or the manuscript.
Author contributions
Logan J Bigelow: conceptualization, methodology, investigation, formal analysis, data curation, writing – original draft, writing – review and editing. Emily K Pope: conceptualization, methodology, investigation, formal analysis, data curation, writing – original draft, writing – review and editing. Clare PL Lee: investigation, data curation, writing – review and editing. Veronica K Sohasky: investigation, data curation, writing – review and editing. Sarah K MacLeod: investigation, data curation, writing – review and editing. Paul B Bernard: conceptualization, methodology, supervision, project administration, resources, funding acquisition, writing – review and editing.
Data availability
Data available upon request.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Tecniplast donated five cages utilized in this study, but had no role in the preparation of data or the manuscript.
Ethical considerations
All procedures performed during this study were conducted in accordance with the guidelines of the Canadian Council on Animal Care and approved by the University of Prince Edward Island Animal Care Committee protocol #21-032.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Sir James Dunn Animal Welfare Centre, the Natural Sciences and Engineering Research Council of Canada (RGPIN-2024-05367) and the Atlantic Veterinary College, University of Prince Edward Island.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
