Abstract
Directive 2010/63/EU on the protection of animals used for scientific purposes requires that the killing of mammal foetuses during the last third of their gestational period should be accomplished through effective and humane methods. The fact that murine foetuses are resistant to hypoxia-mediated euthanasia renders the current euthanasia methods ineffective or humane for the foetuses when these methods are applied to pregnant female mice. We have assessed the time to death of foetuses after performing either indirect (dam euthanasia) or direct (via intraplacental injection – a new approach to euthanasia) euthanasia methods in order to determine a euthanasia method that is appropriate, ethical and efficient for the killing of mouse foetuses. The respective times to death of foetuses after performing the three most commonly used euthanasia methods (namely cervical dislocation, CO2 inhalation and intraperitoneal sodium pentobarbital administration) were recorded. Absence of foetal heartbeat was monitored via ultrasound. We consider that the most effective and humane method of foetal euthanasia was the one able to achieve foetal death within the shortest possible period of time. Among the indirect euthanasia methods assessed, the administration of a sodium pentobarbital overdose to pregnant female mice was found to be the fastest for foetuses, with an average post-treatment foetal death of approximately 29.8 min. As for the direct euthanasia method assessed, foetal time to death after intraplacental injection of sodium pentobarbital was approximately 14 min. Significant differences among the different mouse strains employed were found. Based on the results obtained in our study, we consider that the administration of a sodium pentobarbital overdose by intraplacental injection to be an effective euthanasia method for murine foetuses.
The humane euthanasia of animals is a compulsory practice in laboratory mouse facilities for diverse reasons (for example to comply with established humane endpoints, protocols, among others). Attending veterinarians are frequently asked what would be the euthanasia method of choice for a specific situation, in which factors such as an appropriate balance between technical and equipment requirements, effectiveness, and ethical considerations of the euthanasia method have all to be weighed in. A tough situation arises when pregnant females in the last third of their gestation period need to be euthanized. In this scenario, the welfare of foetuses has also to be taken into account. A lack of published literature on foetal euthanasia makes it harder to identify a method that is suitable, effective and humane to both the pregnant females and their foetuses.
Foetal euthanasia as an ethical topic is included in the current EU Directive (2010/63/EU). The regulations outlined in this directive apply to mammal foetuses that are in the last third of their gestation period. Therefore, all the provisions in this directive must be complied with while handling pregnant females. As stated in this EU Directive 2010/63/EU, there is scientific evidence showing that foetuses in the last third of their development period are at an increased risk of experiencing pain, suffering and distress, which may in turn negatively affect their subsequent development.
Previous consensus has supported the belief that neonates and foetuses are not likely to perceive pain. Thus, Mellor et al. have established that rodent foetuses, as well as those of other mammals, are unconscious in the uterus and that hypoxia does not elicit a response. They argue that sustenance of awareness in adult animals requires relatively high levels of oxygen, and, therefore, the low oxygen levels found in foetal circulation together with the actions of other suppressors make it unlikely that awareness occurs in foetuses. 1 Other studies have shown, however, that the former may not be the case. The neural tube of mouse foetuses is already developed into a functional brain by the last third of the gestation period, and the likelihood that they actually may perceive pain should thus be considered.2,3 The reflexive responses to painful stimuli observed in late-term foetuses (i.e. E19–E20 in the rat) correlates with the responses to painful stimuli observed in adult animals.4,5 This is also true for 26-week-old human foetuses. 4 Although the electroencephalographic responses to tail clamping recorded in lightly anaesthetized 5–7-day-old rat pups did not provide conclusive evidence as to whether or not rat pups experience pain they nevertheless did suggest that differences might exist regarding the intensity and the nature of pain perceived by pups at different ages. 6 In the latter study, one of the most relevant ethical and technical issues, when dealing with foetuses, was the application of an appropriate euthanasia method. Although most guidelines2,7–10 recommend that CO2 should not be used to terminate foetal forms, no effective alternatives or other specific methods have so far been proposed.
An appropriate euthanasia agent or method ought to fulfil the following requirements: it should act quickly, be free of signs of pain or distress, be reliable, and it should also be of minimal distress to the individual performing the procedure. Additionally, the extent of handling required when performing the entire procedure should be minimized in order to avoid possible interferences with experimental data.
In searching for new alternatives we came across the latest euthanasia guidelines from the American Veterinary Medicine Association (AVMA 2013) 7 which indicated that non-inhaled agents such as injectable barbiturates alone are acceptable for use in foetuses or neonates. The rodent euthanasia report issued by the American College of Laboratory Animal Medicine (ACLAM), based on the AVMA guidelines, proposes the skilful injection of chemical anaesthetics in quantities sufficient to ensure the death of foetuses over 15 days post coitus (dpc) as a method of euthanasia. 9 Unfortunately, there is a lack of detailed information on the actual implementation of the former method of euthanasia in the literature cited in that study, thus effectively hindering the reproducibility of the proposed technique. This deficiency led us to the formulation of a new method. Additionally, decapitation or other physical methods deemed as acceptable approaches 7 to euthanasia are not valid methods in experimental situations where the preservation of anatomical structures matters, as well as those in which pain and distress play an important role.
To settle on a euthanasia method that is appropriate, ethical and efficient, foetal times to death after application of either indirect euthanasia procedures (dam euthanasia) or direct methods via intraplacental injection were evaluated in this study.
Materials and methods
Animals
Pregnant female mice of the following three widely used mouse strains: Crl:CD-1 (ICR), C57BL/6J and 129S2/SvPasCrl were obtained from the Spanish National Cancer Research Centre (CNIO) mouse breeding colonies (origin: Charles River Laboratories, Lyon, France). All animal procedures were performed in accordance with current Spanish regulations for animal protection (RD53/2013), and in compliance with requirements of the European Union Directive 2010/63/EU regarding the protection of animals used for experimentation. Prior to commencement of the study, approval of all the experimental procedures used had been obtained from the Institutional Laboratory Animal Care and Use Committee (IACUC) of CNIO.
The mice were housed in a controlled environment (12:12 light/dark cycle with dawn and dusk transitions, with an average room temperature of 22℃, and relative humidity of 55%) and were given ad libitum access to a commercially available pelleted diet (Teklad 2014; Harlan Teklad Laboratory, Madison, USA) and drinking water. The mice were kept in individually ventilated cages (IVCs; Tecniplast, Buguggiate, Italy). The cage bottoms and drinking bottles were changed weekly. During the whole study, the mice were housed in a barrier facility.
All females were mated to the same male strains. Copulation was confirmed by visual detection of vaginal plugs the next morning and gestation was further confirmed by abdominal palpation at 10 dpc.
Euthanasia methods
The study assessed foetal time to death following the use of indirect or direct euthanasia methods. Indirect methods included cervical dislocation, CO2 inhalation and intraperitoneal injection of a sodium pentobarbital overdose. The direct method consisted of administering an overdose of pentobarbital by intraplacental injection.
Foetal time to death after pregnant dam euthanasia
Nine pregnant females (E16) per strain (sample = 9 for a confidence interval of 95%, alpha level 0.05) were euthanized using one of the following methods: cervical dislocation, CO2 inhalation, or an intraperitonal injection of a sodium pentobarbital (Dolethal®; Vétoquinol Ltd, Buckingham, UK) overdose at 120 mg/kg (diluted to 50% in phosphate-buffered saline [PBS]). One foetus per dam was monitored. Cervical dislocation was used as a control, since natural foetal death occurs as a result of hypoxia.
Foetal time to death after intraplacental pentobarbital injection
Nine pregnant females (E16) per strain (sample = 9 for a confidence interval of 95%, alpha level 0.05) were euthanized by cervical dislocation. Then, sodium pentobarbital diluted to 5% in PBS was administered at 600 mg/kg by intraplacental microinjection in the arterial blood region of the cotyledon (labyrinth) (Figure 1). Total volume per foetus was approximately 60 µL, based on an estimated average weight of 1 g/foetus. Weight estimation was based on previously published data.
7
At least two foetuses per dam were monitored: one following intraplacental injection of sodium pentobarbital and one non-injected (control). Any additional foetuses available were also intraplacentally injected and evaluated, with a maximum of three foetuses injected per dam.
Foetal cotyledon intraplacental microinjection of pentobarbital. (a) Pre-injected foetus: note the dark colour of the cotyledon. (b) Injected foetus: note the change to a lighter colour at the site of injection. (c) Post-injected foetus.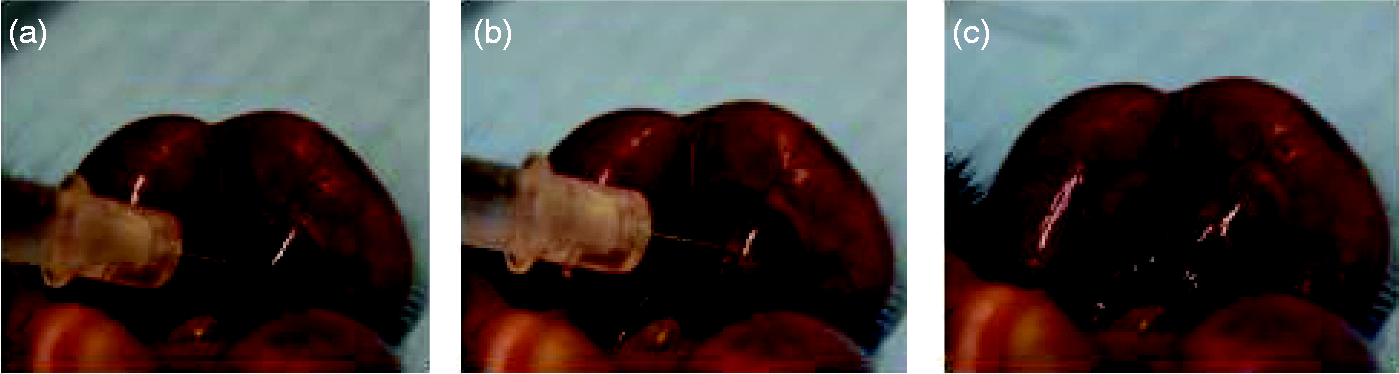
Each injection was visually checked for the expected colour change (from red to white) at the cotyledon injection site (Figure 1). Time between the death of the dam euthanized by cervical dislocation and the intraplacental injection did not exceed 30 s.
Confirmation of death
Death of the pregnant female was confirmed by cessation of blood circulation (absence of heartbeat) and the absence of breathing and pedal reflex. Foetal death was confirmed by ultrasound determination of absence of heartbeat. Euthanized pregnant females were quickly (i.e. less than 5 s) placed on a warmed (38.5℃) ultrasound plate. The uteri were quickly exposed after a longitudinal abdominal incision. The use of warmed ultrasound jelly (38.5℃) prevented foetal hypothermia. Pups were randomly selected for continuous ultrasound monitoring of heart activity. When hearts began to show abnormal activity, a pulsed wave Doppler (pointed towards the heart and the aorta) was performed. In the case of persistent blood flow, a B-mode scan was performed to further monitor heart activity. This cycle of alternative B-mode and Doppler scans was repeated until death was confirmed by at least one of the following: absence of heartbeat for at least 3 s or absence of measurable blood flow into the aorta.
Ultrasound examinations were carried out using a Vevo 770 ultrasound machine (Visualsonics, Toronto, Canada). A RMV707B probe at a frequency of 45 MHz was used. The probe enabled the acquisition of B-mode images of foetal hearts, the visualization of their heartbeats and of the atrial and the ventricular movements (Figure 2).
Doppler ultrasound images of mice foetal hearts. (a) Circulation is active. (b) Circulation has ceased.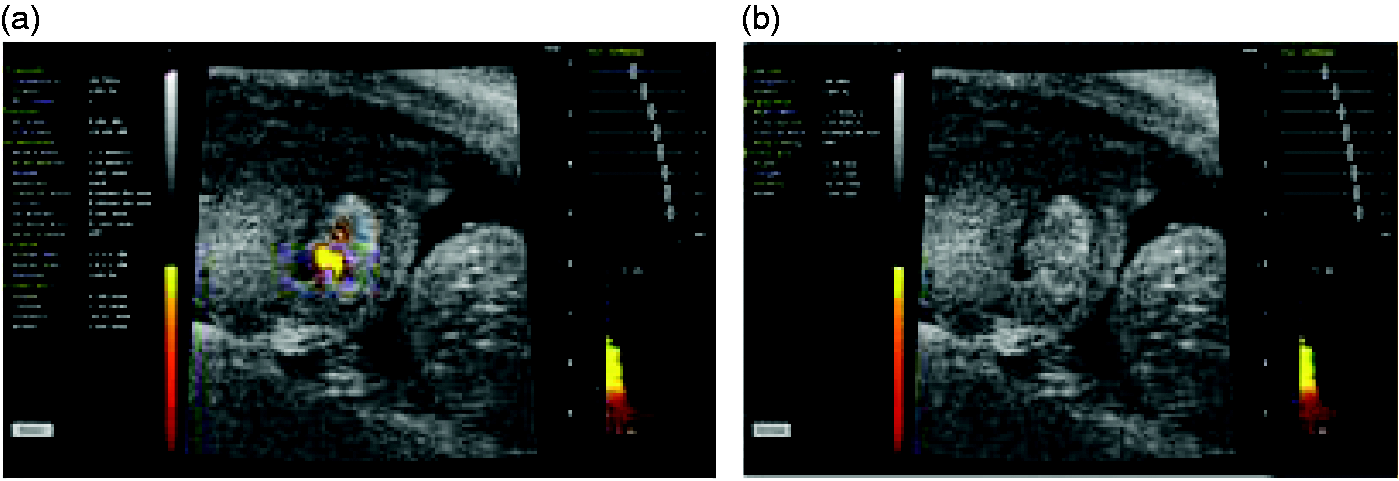
Statistical analysis
Data are presented as the mean ± SEM and all the results were analysed using Prism software (GraphPad Software Inc, San Diego, CA, USA). Data were analysed using either one- or two-way analysis of variance (ANOVA). A post-hoc analysis was carried out using a Bonferroni test. Student’s t-test was used in order to compare the two groups. The level of significance considered was P < 0.05.
Results
Foetal time to death after pregnant dam euthanasia
Foetuses remained alive for an average of 46 min (SEM = 2.901; n = 9) after cervical dislocation of the dam (control group). Foetuses euthanized by using CO2 inhalation of the dam remained alive for an average of 37.6 min (SEM = 2.57; n = 9) showing no significant differences when compared with the control group. Interestingly, foetuses euthanized by intraperitoneal sodium pentobarbital overdose of the dam remained alive for an average of 29.8 min (SEM = 2.45; n = 9). A one-way ANOVA revealed a decrease in the foetal time to death when using the intraperitoneal sodium pentobarbital overdose of the dam euthanasia method when compared with the control group. Subsequently, post-hoc tests revealed significant differences between these two groups (P = 0.00, Figure 3).
Foetus survival time when using cervical dislocation, sodium pentobarbital or CO2 inhalation for dam euthanasia. Significant differences were found between sodium pentobarbital administration and cervical dislocation.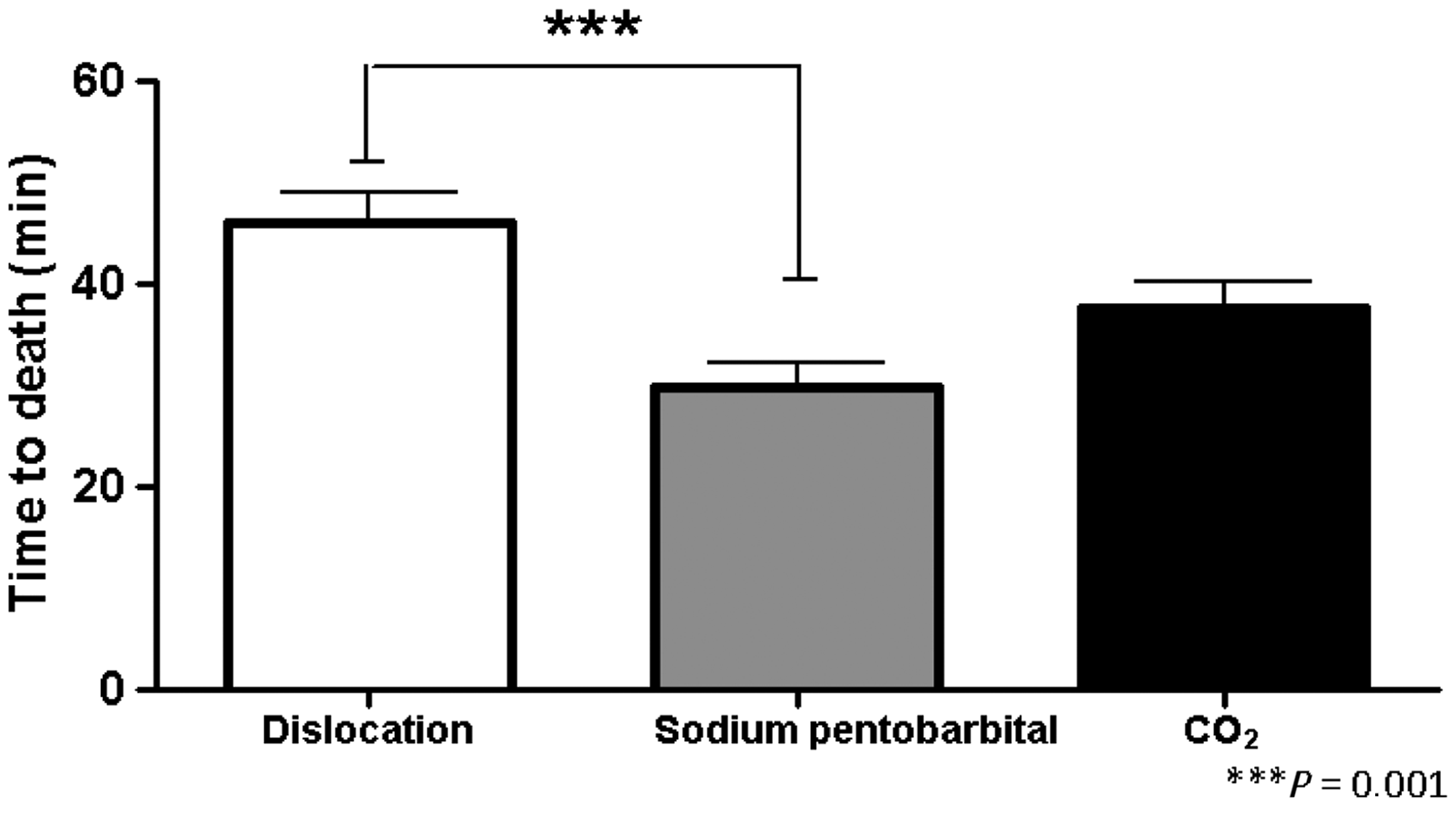
Two-way ANOVA did not reveal any effect on survival time results for foetuses from the different strains when using the same euthanasia method on the pregnant females (P = 0.43, Figure 4).
Foetal time to death after employing cervical dislocation, sodium pentobarbital or CO2 procedures of euthanasia of the dam in the indicated strains. No significant differences were found when comparing time to death of the different strains.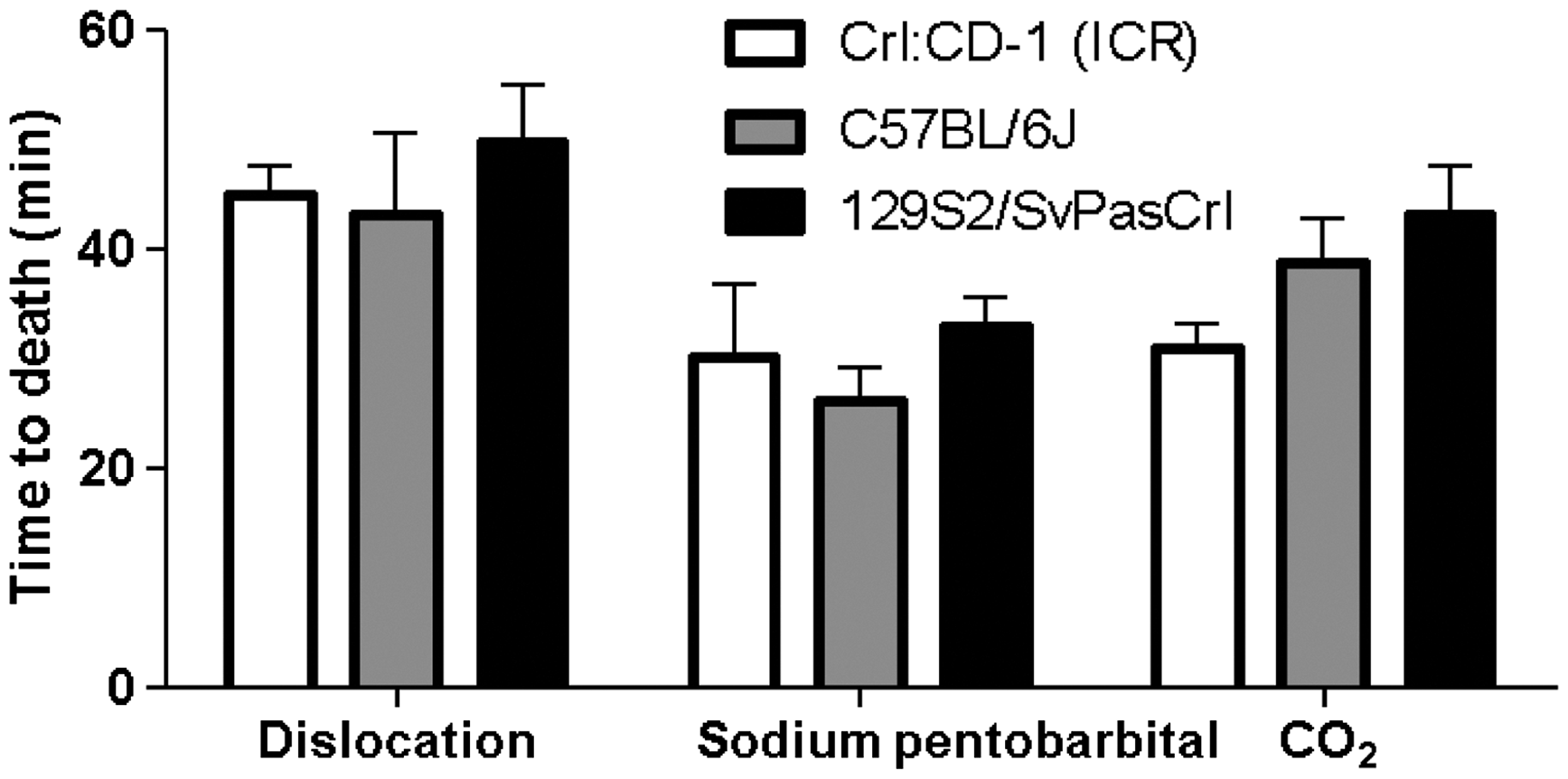
Foetal time to death after intraplacental pentobarbital injection
The average time to death for the non-injected foetuses was 42.96 min (SEM = 0.95; n = 27). The average time to death for injected foetuses after cervical dislocation of the dam was 13.93 min (SEM = 1.29; n = 57). Student’s t-test revealed that the time to death of the injected foetuses was significantly lower compared with the non-injected foetuses (P = 0.0003, Figure 5).
Effect of the intraplacental injection of sodium pentobarbital on foetal time to death. Significant differences were found when comparing time to death of the injected foetuses to non-injected foetuses.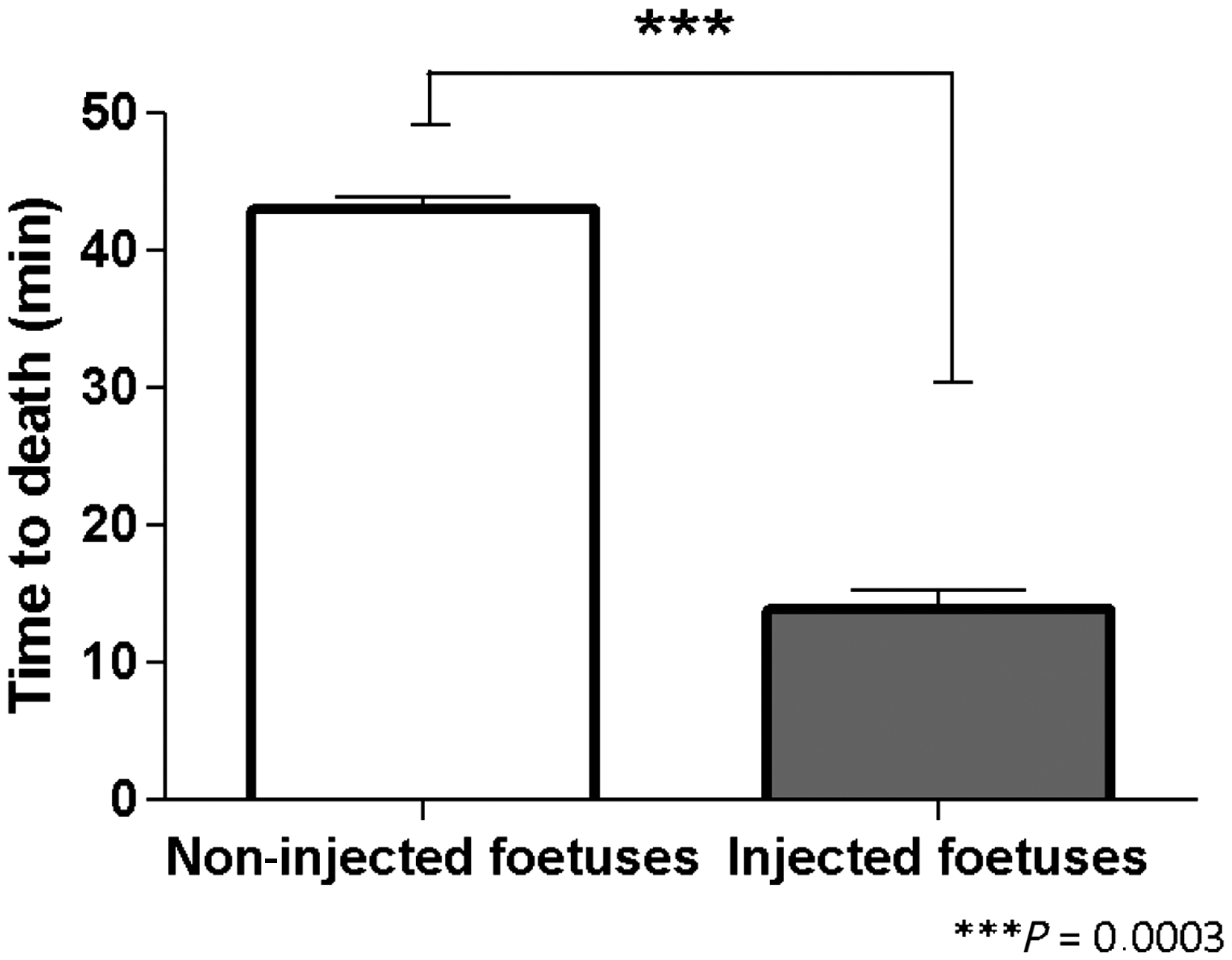
One-way ANOVA revealed significant differences between the strains. The average time to death after intraplacental injection for Crl:CD-1 (ICR) foetuses was 16.27 min average (SEM = 2.045; n = 22); 7.7 min for C57BL/6J foetuses (SEM = 1.916; n = 17); and 14.9 min for 129S2/SvPasCrl foetuses (SEM = 2.1; n = 18). Bonferroni’s multiple comparison tests revealed that there were significant differences between the C57BL/6J and Crl:CD-1 (ICR) strains (P = 0.011, Figure 6).
Comparison of foetal times to death of different strains after performing euthanasia by intraplacental injection of a euthanasia agent. Significant differences were found between the Crl:CD-1 (ICR) and C57BL/6J strains.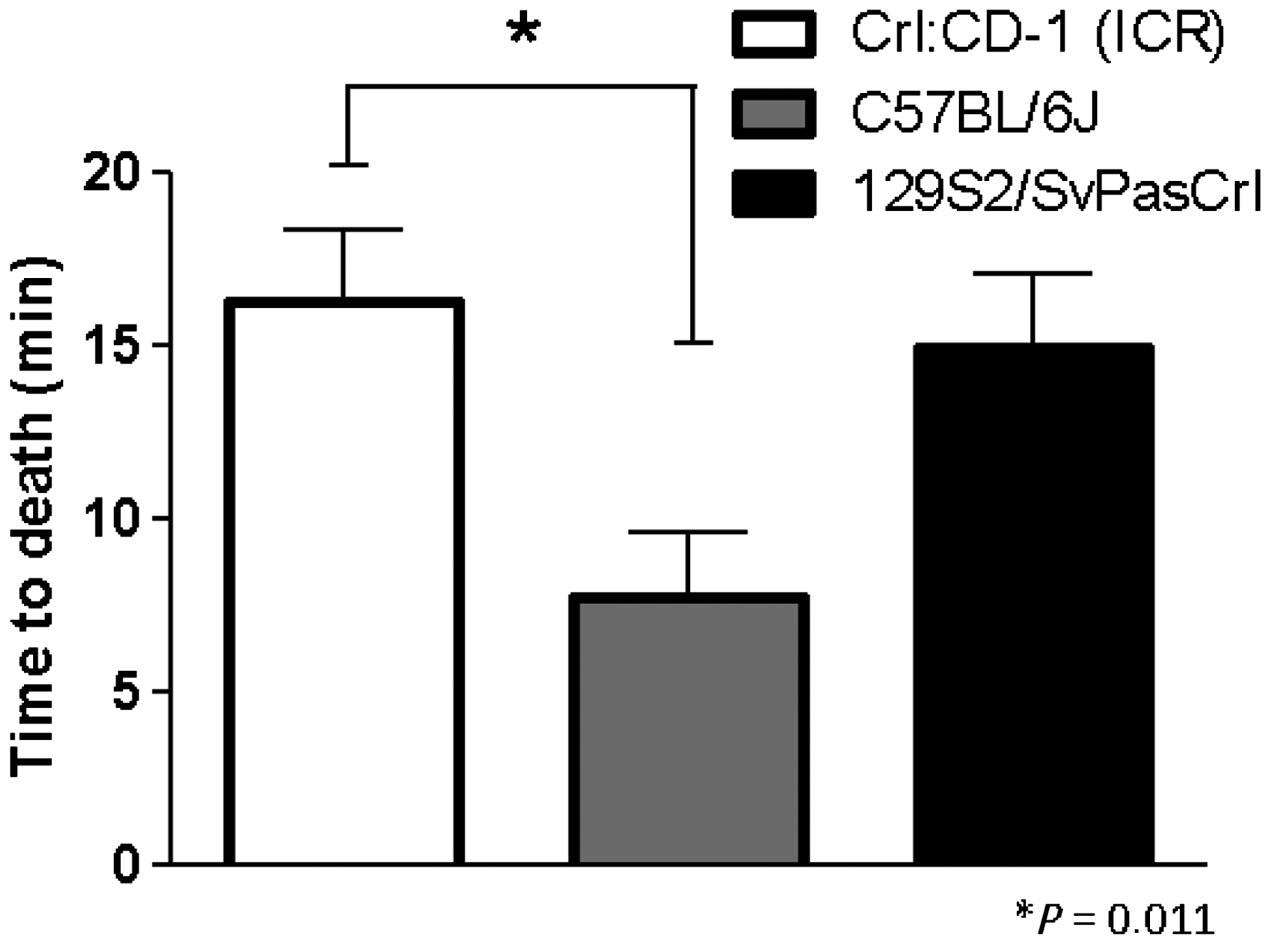
Foetal time to death comparison between direct and indirect euthanasia methods
One-way ANOVA showed statistically significant differences when comparing foetus average time to death after each direct method (intraplacental injection) to three indirect methods (cervical dislocation of the dam, intraperitoneal sodium pentobarbital overdose of the dam and CO2 inhalation of the dam). Dunnett’s multiple comparison tests between the direct method and the three different indirect methods revealed significant differences between the direct method and cervical dislocation of the dam (P: 0.009). No statistically significant differences were found when comparing the foetuses using the direct euthanasia method and the intraperitoneal sodium pentobarbital overdose of the dam or CO2 inhalation of the dam.
Discussion
Our collected data revealed that the method of euthanasia of the pregnant female did affect the foetal time to death, this being at its highest when the method of choice was cervical dislocation and its lowest when the method was intraperitoneal sodium pentobarbital administration. CO2 administration euthanasia method had an in-between foetal time value to death. The present study demonstrated that intraplacental injection might be an effective euthanasia method that also allows for the preservation of anatomical and histological mouse foetal structures and avoids death by hypoxia.
Previous studies, conducted by Klaunberg et al., assessed different euthanasia methods on pregnant mice and foetuses. 11 Intravenous potassium chloride injection under anaesthesia, CO2 inhalation, and cervical dislocation alone or under anaesthesia were found to be excellent methods of euthanasia for pregnant mice. None of the methods used (halothane inhalation, CO2 inhalation, intraperitoneal sodium pentobarbital injection, intravenous potassium chloride, and cervical dislocation with and without anaesthesia) were however adequate for euthanizing foetuses. 11 Since Klaunberg et al only determined whether or not foetal cardiac arrest occurred within 20 min after euthanasia of the dam, it was not possible to perform a quantitative comparison of time to death of foetuses based on different euthanasia methods. In our study, we have quantified the time until foetuses undergo cardiac arrest after applying three of those indirect euthanasia methods employed by Klaunberg et al., which are considered to be the most commonly used procedures in animal facilities all over. Following cervical dislocation of the pregnant female the foetuses remained alive for an average of 46 min, when CO2 inhalation was performed the foetuses survived for an average of 37.6 min, and after administration of an intraperitoneal sodium pentobarbital overdose the foetuses remained alive for an average of 29.8 min. Our results thus pointed to pentobarbital intraperitoneal overdose as the fastest indirect method for euthanizing foetuses. Nevertheless, we consider that even 29.8 min to death is too long a period of time for it to be considered clinically efficient or ideal in any of the three indirect methods of euthanasia assessed. Rodent foetuses have been documented as resistant to hypoxia with a heartbeat activity lasting for up to 50 min after their mothers’ death. 5 These findings are in agreement with the results we obtained after performing euthanasia by cervical dislocation of the dam (considered as a control), where we recorded an average time to foetal death of 46 min.
Our results matched those of published data demonstrating that when euthanasia is accomplished by any agent causing death by hypoxia, such as CO2, it takes more than 30 min for foetuses or neonates 4 to die. Even if the time to death at E16 after CO2 inhalation through the dam was to be the same as the one observed when P0 neonates were directly exposed to CO2, 5 the difference in time to death was not significant when compared with the control group (cervical dislocation).
Our results have confirmed that none of the three indirect methods of euthanasia that we assessed, cervical dislocation, CO2 inhalation or intraperitonal sodium pentobarbital overdose, are effective and humane methods for the euthanasia of foetuses.
Although not statistically significant, we found differences between outbred and inbred strains in our tests when euthanizing the dam via CO2 inhalation, cervical dislocation and sodium pentobarbital. We observed that the outbred strain (CD-1) had a faster time to death as compared with the other two strains examined. Pritchett et al. have found differences between outbred and inbred preweanlings (0 days of age) 5 when exposed to CO2.
The use of warm ultrasound jelly and homeothermic blanket to reduce the temperature effect was considered to be important. Temperature control was applied throughout both phases of our study. Foetal exposure outside the placenta could cause haemorrhage and hypothermia, which could in turn trigger the uncontrolled death of the exposed foetuses. As survival may be directly affected by hypothermia, any euthanasia methods used should attempt to keep the foetus outside of the dam for the shortest amount of time possible, and the temperature should be controlled. It has been shown that after suffering hypothermia, a foetus may be revived through gradual warming. 12 We did not find any differences in foetal time to death after the cervical dislocation of the dam (first part of our study) and the non-treated foetuses after exposing the uterus (second part of the study), so it may be assumed that temperature was appropriately controlled and did not affect our data.
In the second phase of our study, an intraplacental injection was performed. A technique for indirect placental injection has already been described for vascular corrosion casting of fetoplacental vasculature. In this technique, a catheter placed in the ascending thoracic aorta of a pregnant mouse permits the introduction of a methyl methacrylate casting compound into the lower body vasculature, including the uterus and placenta. 12 We did not attempt to employ this procedure due to the inability to control the extent of the agent reaching the foetus and the need to maintain the anaesthesia of the dam.
To perform the intraplacental injection, we took advantage of the capillary network surrounding an arteriole, which takes oxygenated blood to the umbilical cord. This area forms a lobular or cotyledonary structure with a densely packed villous tree. 13 Assuming that maternal blood is not flowing due to cardiac arrest (confirmed death of the dam), studies carried out by Coan et al. have found that the small fetoplacental arterioles have smooth muscle layers, and, therefore, may play a role in regulating local fetoplacental perfusion to match perfusion and/or oxygenation of adjacent maternally perfused sinusoids. 13 This could explain the observed perfusion capacity of the placental foetal side, which may also be assisted by the pumping effect of the foetal heart.
A dose of 600 mg/kg was used in our study. A study by Klaunberg et al. has found successful results in neonates (P8–P14) with doses of 400 mg/kg and 800 mg/kg. 11 In that study, an intraperiotenal injection of 800 mg/kg sodium pentobarbital administered to mixed genetic background P8 neonates achieved death of mice after ≥20.00 min (n = 7). Our results showed an average of 13.93 min after intraplacental injection in E16 foetus. The difference in developmental age and resistance to hypoxia and/or the route of administration may explain the differences.
The significant differences among strains observed in the intraplacental injection study may also be clinically explained by placenta study. Most available data related to foetal size, development, and behaviour during pregnancy has been obtained in CD-1 mice. Rennie et al. 14 included in their foetal studies C57BL/6 mice and compared them with CD-1 mice. E17.5 foetal body weight continues to increase in both C57BL/6 mice and CD-1 mice. From this stage, the increase is blunted in C57BL/6 foetuses, which weigh approximately 20% less than CD-1 foetuses. Anatomically, foetal blood in capillaries is separated from maternal blood by an interhemal membrane, which gets thinner as gestation progresses, reducing the barrier to materno–foetal exchanges. 15 Rennie et al. have also found a decreased capillarization of the labyrinth during late gestation, and a greater interhemal thickness in C57BL/6 strains (membrane thickness is 34% greater and foetal blood space volume is reduced by 60% in C57BL/6 compared with CD-1 strains). 14 Differences between C57BL/6J, Crl:CD-1(ICR), and 129S2/SvPasCrl strains in the thickness of this membrane could, at least partially, justify the differences found in foetal time to death after intraplacental injection. The membrane is thicker in C57BL/6J and, therefore, permeability with the mother could be lower compared with the other two strains. We hypothesize that the exchange rate and the anatomical features in the 129S2/SvPasCrl strains are similar to those of the Crl:CD-1(ICR) strains.
Differences in foetal time to death in different strains may also be explained by size differences (body mass). At birth, inbred mice appear to be less mature than outbred mice and they develop more slowly during the period from birth to eye-opening. 3 Ultrasound studies show that foetal size is greatly affected by strain line: CD-1 foetuses are bigger than C57BL/616 ones. In our experience, as seen when using ultrasound, the spinal column and bones of the C57BL/6 mice appear to be less ossified than in the other two strains (data not shown). The differences in bone development may be particular to the skeletal system and do not imply differences in metabolic or cardiovascular function. Therefore, as our doses were estimated based on the average published weight,13,14,17 which tends to use CD-1 as the mouse model, the possibility that the C57BL/6 strain could have been overdosed should be considered.
In our study direct and indirect methods of euthanasia of the foetuses were compared. The results showed that even if the differences in the time to foetal death are not statistically significant when the intraperitoneal sodium pentobarbital and CO2 methods are compared with the intraplacental injection method, this direct method of foetal euthanasia is clearly more efficient, achieving the objective of reducing the time to death of foetuses.
Although the technique of intraplacental injection that we have described may be useful, it poses several challenges for the euthanasia of a larger number of foetuses. In that instance we would recommend its use only for those specific procedures that require the preservation of anatomical and histological mouse foetus structures and the avoidance of death by hypoxia. In conclusion, we propose that the intraplacental euthanasia technique may be useful when decapitation is not an appropriate or recommended option. Nevertheless, where possible, we would still consider foetal decapitation to be a method of choice for euthanasia. Additionally, we also propose using the intraplacental route as an alternative to toxicology and pharmacology studies of foetal metabolism, allowing direct access to foetal circulation and offering the possibility of measuring drug biodistribution and/or metabolism.
We believe that our results have provided a first step to refining the available methods available when euthanasia of foetuses is required.
Footnotes
Acknowledgments
We wish to acknowledge Ms Elena Andrés for her technical imaging support during the development of the techniques; the entire CNIO Animal Facility Unit staff for their predisposition to take on new challenges; and Luis Donate, Lidia Bravo, Alan Hoberman and Aleksandar Popovic for their comments and revision during the preparation of this manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
