Abstract
Gastric bypass surgery, an operation that restricts the stomach and bypasses the duodenum and part of the jejunum, results in major improvement or remission of type 2 diabetes. Duodenual-jejunal bypass was developed by one of the authors (FR) as an experimental, stomach-sparing variant of gastric bypass surgery to investigate weight-independent mechanisms of surgical control of diabetes. Duodenual-jejunal bypass has been shown to improve various aspects of glucose homeostasis in rodents and in humans, thus providing an experimental model for investigating mechanisms of action of surgery and elusive aspects of gastrointestinal physiology. Performing duodenual-jejunal bypass in rodents, however, is associated with a steep learning curve. Here we report our experience with duodenual-jejunal bypass and provide practical tips for successful surgery in rats. Duodenual-jejunal bypass was performed on 50 lean rats as part of a study aimed at investigating the effect of the procedure on the physiologic mechanisms of glucose homeostasis. During the study, we have progressively refined details of anatomic exposure, technical aspects of duodeno-jejunostomy and peri-operative care. We analysed the role of such refinements in improving operative time and post-operative mortality. We found that refinement of exposure methods of the gastro-duodenal junction aimed at minimizing tension on small visceral vasculature, technical aspects of duodeno-jejunal anastomosis and peri-operative management played a major role in improving the survival rate and operative time. Overall, an experimental model of duodenual-jejunal bypass was successfully reproduced. Based on this experience, we describe here what we believe are the most important technical tips to reduce the learning curve for the procedure.
Introduction
Although originally developed as a weight loss intervention, bariatric surgery has been found to improve various metabolic co-morbidities of obesity, particularly type 2 diabetes (T2DM). Indeed, bariatric surgery induces long-lasting remission of hyperglycaemia and reduces cardiovascular disease and mortality associated with T2DM.1–3 Currently, the two most commonly performed procedures in humans are the Roux-n-Y gastric bypass (RYGB) and the vertical sleeve gastrectomy (VSG). Both operations result in major clinical benefits and achieve durable normalization of plasma glucose and insulin levels in many patients. 4
However, both RYGB and VSG involve significant restriction of the volume of the stomach, thus potentially imposing mechanical restriction to food intake and consequent weight reduction. To rule out possible confounding factors from reduction of food intake and weight loss, a stomach-sparing form of gastric bypass (duodenal-jejunal bypass; DJB) was developed by one of the authors (FR) to investigate the hypothesis that gastrointestinal operations may exert direct and weight-independent effects on glucose homeostasis. 5 The procedure was designed to reproduce the same length of proximal intestinal bypass as in standard RYGB (which bypasses the duodenum and upper jejunum) while keeping the stomach intact; gastrointestinal continuity was restored by means of either a duodeno-jejunostomy or gastro-jejunostomy. Original experiments with DJB showed that the procedure improves glucose tolerance in both lean 5 and obese models 6 of T2DM despite inducing minimal or no weight loss. Various groups have reported on the use of DJB in both mouse7,8 and rat models. 9 DJB has also been shown to improve T2DM in humans. 10 Clinically, DJB is performed either as a standalone procedure, typically in non-obese patients with T2DM,10–13 or in combination with sleeve gastrectomy. The latter variant is usually preferred when greater weight reduction is desirable or to reduce risk of marginal ulcer formation.14,15
DJB is a well-characterized procedure in rodents. In its most common version, the entire duodenum and 10 cm of the proximal jejunum are bypassed and a duodeno-jejunostomy is performed. The excluded limb of small bowel (excluded from food passage but still carrying bile and pancreatic juice) is re-anastomosed downstream in the distal jejunum/proximal ileum, usually about 15 cm from the duodeno-jejunostomy (Figure 1).
Duodenal jejunal bypass.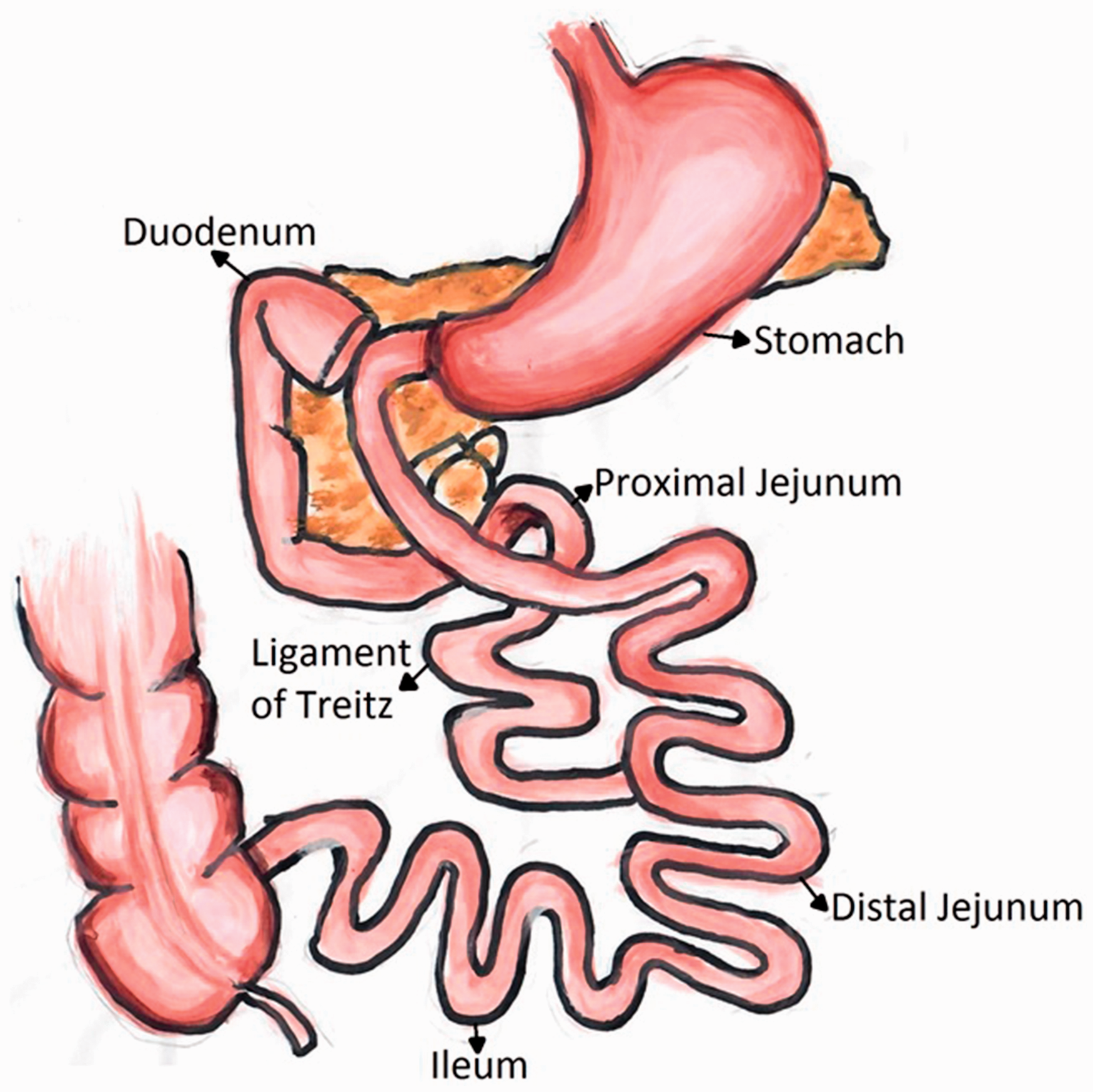
Technical variants in rodents include modifications of the limb length 16 or performing anastomosis between the distal stomach and jejunum (rather than between the duodenum and jejunum). 17 DJB has also been performed in combination with sleeve gastrectomy (vertically oriented resection of two-thirds of gastric volume). 18
Since its first description in 2004, 5 DJB has been used as an experimental model for investigations of mechanisms of gastrointestinal physiology as well as effects of gastrointestinal surgery on T2DM,5,19–21 hepatic metabolism,22–25 kidney 26 and cardiovascular disease. 27 DJB has been shown to cause changes in gut hormones,28,29 glucose transporters,30–32 bile acid33–35 and gut microbiota16,36 among other mechanisms of metabolic regulation.
The development of a DJB model in rats has advanced knowledge on the mechanisms of action of bariatric/metabolic surgery. However, DJB is a multi-anastomotic procedure and therefore technically challenging to perform in small animal models. The mortality rates have been rarely reported in previous DJB studies; available data show up to 35% mortality but rates vary depending on technical variants of the procedure.17,37 Long-term post-operative survival has also not been reported in previous studies.
This study provides insight on the development of an experimental model of DJB in rats, with particular focus on the aspects of surgical technique and peri-operative management that can improve long-term survival rates.
Animals, materials and methods
Materials
Animals: adult (2–5 months) male Wistar rats weighing 300 to 500 g and Sprague Dawley rats (2–7 months) weighing 500 to 700 g (Charles River Ltd, Margate, Kent, United Kingdom) were maintained under controlled temperature (21–23℃), humidity (30–70%) and light (12:12 hour light-dark schedule, lights on at 7:00 a.m.). Animals were double housed in a polycarbonate cage containing appropriate bedding, a hiding tube and chewing object, and equipped with a watering bottle. The animals were fed PMI Nutrition International Certified Rodent Chow No. 5CR4 ad libitum.
Male rats were selected due to their larger size facilitating the operation, whereas the species allows for rapid adipose tissue accumulation. This should pose no limitations on the surgical technique as the gastrointestinal anatomy does not differ significantly between genders. Then 3 days prior to surgery, animals were exposed to a liquid diet, consisting of normal chow and water to facilitate gastric manipulation and recovery. Animals were then fasted for 18 hours before the procedure, while allowing ad-libitum access to water. This timeframe allowed the gastrointestinal tract to empty completely before the procedure, thus ensuring no food spillage occurred in the abdomen during gut resection. Moreover, it improved the visibility of the gut lumen and anastomosis suturing. Body weight and food intake was monitored pre- and post-operatively. All animal procedures were performed as part of studies of mechanisms of action of DJB on glucose metabolism, approved by the British Home Office under the United Kingdom Animal (Scientific Procedures) Act 1986 under Home Office Licence Authority with approval from the King’s College London Animal Welfare Ethical Review Body. The Animals (Scientific Procedures) Act (ASPA) has recently been revised to change the European Directive 2010/63/EU on the protection of animals used for scientific purposes.
Before performing DJB on living animals, the surgical team underwent extensive training on sacrificed animals under tutoring by a surgeon experienced with the procedure in this rodent model.
Operating theatre setup: cotton swabs with optimal stiffness (cotton tipped applicators; Hardwood Products Company, Guilford, ME, USA) were prepared. A warming blanket was used during surgery to minimize health dissipation. Animals were housed in a warm chamber (37℃) immediately after surgery. General anaesthesia was performed with a rodent anaesthesia apparatus (VetEquip Inc., Pleasanton, CA, USA) that included an evacuation canister and induction chamber.
Analgesia and hydration: 50 mg/mL of Carprofen (Norbrook Pharmaceuticals Ltd, UK) was injected subcutaneously for analgesia immediately before the operation. Carprofen was used due to the low heart rate observed in post-operative animals injected with opioid analgesics. Rescue analgesia was used post-operatively, according to the instructions of the Animal Care and Welfare Officer and the Named Veterinary Surgeon overlooking the protocol of the study, upon assessment of the post-operative charts for signs of pain. Subcutaneous injections of 0.9% sodium chloride were administered pre-operatively to maintain adequate hydration. Packed non-wetting sterile HydroGel (ClearH2O, ME, USA) was placed in the cages before and after the operation.
Surgical instruments and sutures: basic instruments required for small animal surgery were used for the anastomoses (one Weitlaner-Locktite retractor, curved forceps, one pair of straight and one pair of angled Iris scissors, one Castroviejo needle holder, one ultra-fine curve haemostat and two bulldog clamps (Fine Science Tools, Heidelberg, Germany)).
Closure of the duodenal stump and anastomoses were performed using a Vicryl Absorbable Coated Braided, Round Needle (5-0 W9105; Ethicon, Inc., Somerville, NJ, USA). The intraperitoneal cavity was closed using a Vicryl Absorbable, Round Needle (3-0 W9114; Ethicon, Inc., Somerville, NJ, USA) and the skin was closed using a Vicryl Rapide, Cutting Needle (3-0 W9919; Ethicon, Inc., Somerville, NJ, USA).
Anaesthesia
All operative procedures were performed under general anaesthesia. Inhalational anaesthesia was induced and maintained using isoflurane and oxygen. The anaesthesia and operative time were recorded in all cases. For induction, isoflurane and oxygen flow was set at 5 L/min and then reduced to 0.5–2.0 L/min for the maintenance phase.
Operation
Laparotomy: after induction of anaesthesia, the abdomen is shaved using electric clippers and prepped with betadine. A 3 cm midline skin incision is performed, extending from the xiphoid process to the umbilicus along the linea alba. To this end, a small incision is initially performed just above the umbilicus while lifting the abdominal wall to separate it from the intestinal loops. Then, the incision is carried on cranially under direct view, thus avoiding injury to abdominal structures (liver and mesenteric damage). During the procedure, warm saline solution is dripped onto the intraperitoneal organs to prevent drying during laparotomy. Retraction of the abdominal wall provides maximal exposure of the abdominal cavity. Care must be taken to avoid liver and mesenteric damage while performing the laparotomy.
Exposure of the duodenum: the left lobe of the liver is retracted upwards to show the stomach. Next, the stomach is retracted to the left to expose the pylorus and the proximal duodenum. Using ultra-fine haemostats and right-angled forceps, careful dissection is performed along the upper and lower border of the duodenum approximately 3 mm distal to the pylorus and close to the wall of the intestine. This allows separation of the proximal duodenum from pancreatic tissue on the lower border and identification of the gastroduodenal artery and its branches at the upper border. Next, the posterior aspect of the duodenum is carefully exposed by gently lifting the lower duodenal border and retracting it cranially. The angled scissors are then used to incise the duodenum, taking care to avoid any injury to the gastroduodenal artery and other small mesenteric vessels (Figure 2a). Care should also be taken to avoid damaging the pancreas. A cotton swab should be available before the duodenum is divided and used to promptly control any bleeding from the duodenal wall at the margin of section, should it occur. The distal stump of the duodenum is closed by means of a full thickness, running suture with 5-0 Vicryl (Figure 2b). At this point, two full-thickness sutures are placed in the proximal duodenal stump, one at the upper and one at the lower border, leaving the needle in place. The sutures are left loose and secured using two bulldog clamps.
(a) Closure of duodenal stump using a running suture; (b) exposure of duodeno-jejunostomy; (c) completed duodeno-jejunostomy; (d) exposure of jejuno-jejunostomy; (e) completed jejuno-jejunostomy.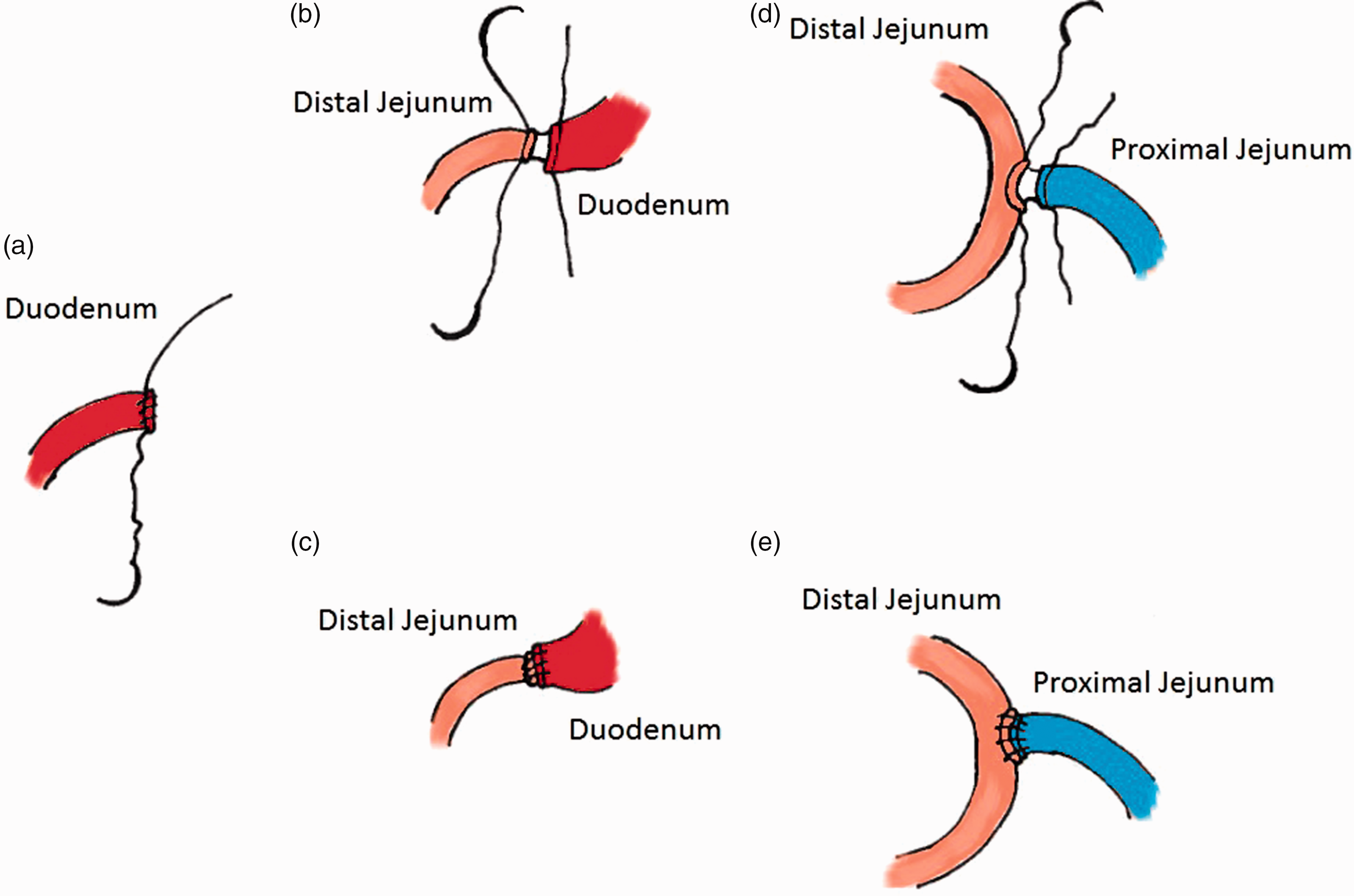
Duodeno-jejunostomy: the omentum is retracted cranially while jejunal loops are gently retracted down and to the left to identify the transverse colon. The latter is then retracted upward thus exposing the ligament of Treitz, which marks the duodenal-jejunal junction. The biliary (excluded) limb of the DJB is prepared by measuring downward 10 cm of jejunum starting from the ligament of Treitz. After gentle dissection of its mesenteric border, the jejunum is divided at this site using the angled scissors. The distal end of the jejunum is then brought up to the duodenum to perform a duodeno-jejunostomy. Care must be taken to ensure no torsion of the mesentery occurs during this step. The sutures previously placed at the upper and lower borders of the duodenum are then used to secure the first two stitches of the anastomosis, which will be performed by two semi-continuous running sutures. Specifically, the upper border of the duodenum is stitched to the anti-mesenteric side of the jejunum, whereas the lower duodenal border is stitched to the mesenteric side of the jejunum (Figure 2c). The anterior aspect of the anastomosis is sutured first from the anti-mesenteric to the mesenteric side (top to bottom) using continuous, full thickness suture, tied to the tail of the stitch previously placed at the inferior border of the anastomosis. Next, the tail of the suture at the upper border is passed posteriorly and gently pulled downward to help flip the limbs towards the operator to expose the posterior portion of the anastomosis. At this point, the posterior aspect of the anastomosis is sutured from the mesenteric to the anti-mesenteric side (Figure 2d). The top and bottom corners, where the sutures are placed and tied, should be completely sealed.
Jejuno-ejunostomy: after completing the duodeno-jejunostomy the jejunum is measured downward for approximately 15 cm (alimentary limb or Roux-limb) to select the site for the jejuno-jejunostomy, which re-creates intestinal continuity between the biliary limb and the small bowel. Next, a 7 mm enterotomy is performed along the anti-mesenteric margin of the jejunum. After ensuring no torsion is present on the mesenteries, the biliary limb is anastomosed to the alimentary limb. The anastomosis is started by placing a first stitch between the anti-mesenteric side of the biliary limb and the upper corner of the enterotomy on the alimentary limb. A second stitch is placed between the mesenteric side of the biliary limb and the lower corner of the enterotomy (Figure 2e). Next, a running suture (five full-thickness stitches) is performed top to bottom (anti-mesenteric to mesenteric) to close the anterior portion of the anastomosis (Figure 2f). The tails of the two stitches are then used to flip the anastomosis towards the operator to facilitate suturing of the posterior side (mesenteric to anti-mesenteric). Appropriate exposure of the posterior aspect of the anastomosis is essential during this step to ensure the anastomosis is sealed and no gaps are left between the stitches.
Abdominal closure: the peritoneal cavity and organs are washed with warm saline and the duodeno-jejunostomy is covered with the greater omentum to contain and facilitate sealing of potential micro-leaks. The peritoneum, fascia and skin are closed separately with running sutures. The final knot of the skin suture is buried under the skin (Figure 3).
Timeline of the procedure (min) with average operating times.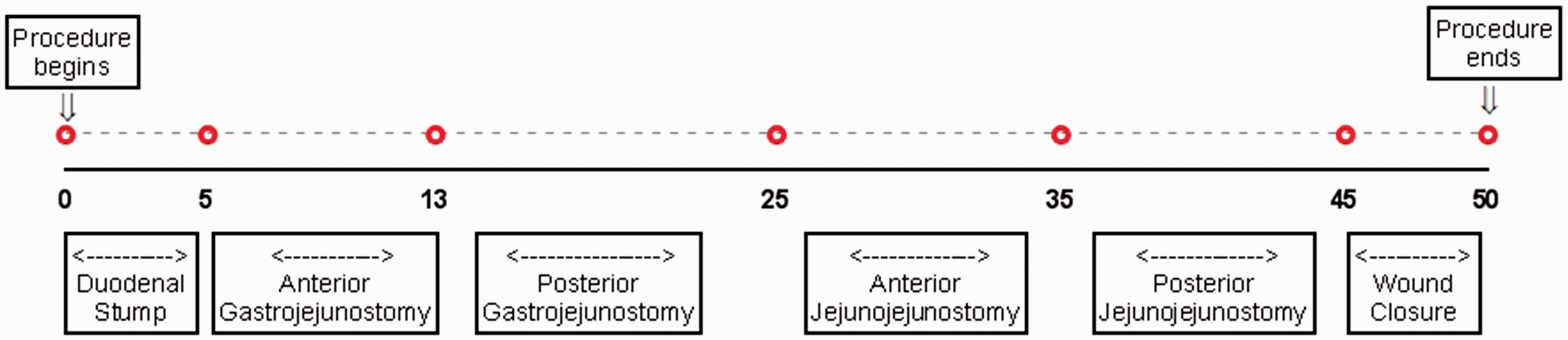
Post-operative care and observations
Following the operation, the rat is placed in a warm chamber (37℃) for 30 minutes to allow a faster recovery. Only liquid diet should be supplied for the first 2 days post-operatively to prevent potential obstruction by food at the level of duodeno-jejunostomy. Survival checks are made every 2 hours for the first 3 days after surgery looking for typical signs of distress in rats. Signs of pain are recorded in a post-operative chart, including piloerection, posture, eye and nasal discharge, respiration, activity, interaction with other animals and alertness. At this stage, it is important to check the eating and drinking behaviour of the animal immediately after the surgery, as well as its ability to urinate and defecate after the first 24 hours. Body weight is recorded daily. Animals demonstrating prolonged (>48 hours) signs of pain such as failure to eat, drink, urinate and defecate were euthanized and post mortem examination was performed.
Statistical analysis
Data are presented as mean ± SD. Prism 5.01 (GraphPad Software Inc. San Diego, USA) statistical software was used for graphs and nonparametric, unpaired Mann-Whitney t-test was used for statistical analysis. The level of statistical significance was set at a p value of less than 0.05.
Learning curve
The initial training in our study was done in cadavers, in accordance to the British Home Office under the United Kingdom Animal (Scientific Procedures) Act 1986, which does not allow training in living animals or animals under terminal anaesthesia. In our experience, performing 15–20 procedures in dead animals is necessary before the operator becomes proficient with this technique (Figure 4). As expected with any surgical procedure, the outcomes improve with experience with the procedure. We found indeed that operative time decreased in our studies with living animals from an initial 157.6 ± 4.7 min to 53.75 ± 4.7 min.
Operating time learning curve in cadavers. Each point is reported as average operative time in minutes ± SD.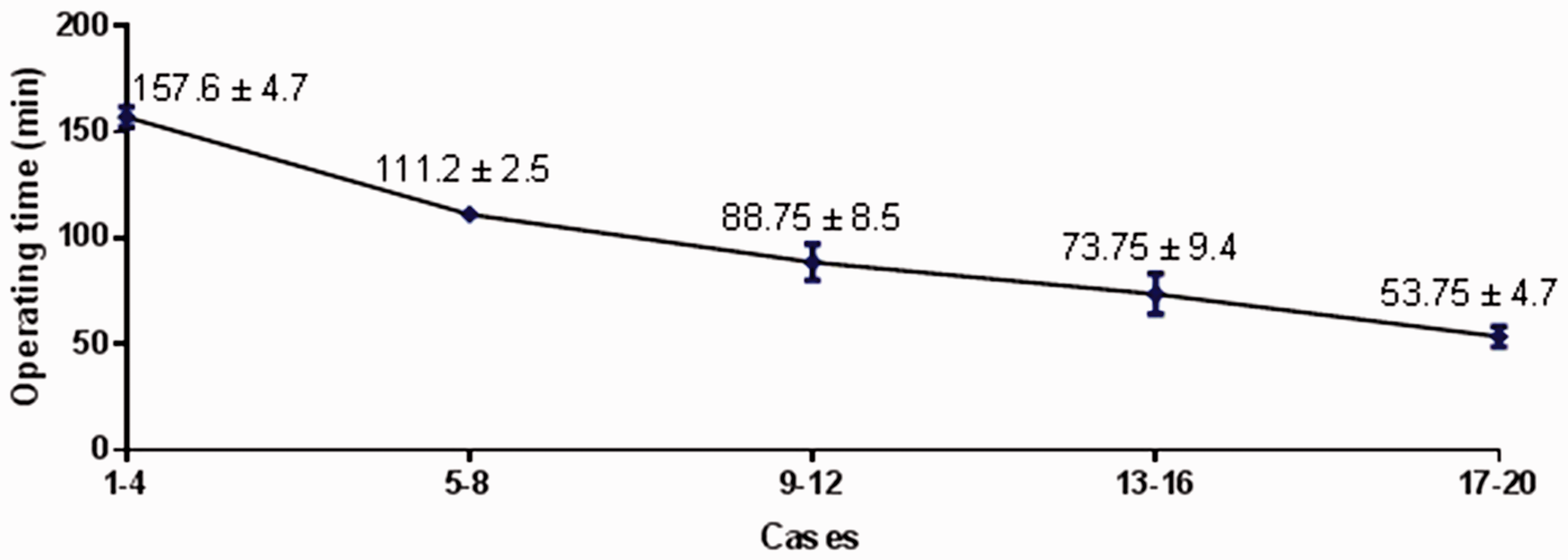
Post-operative mortality
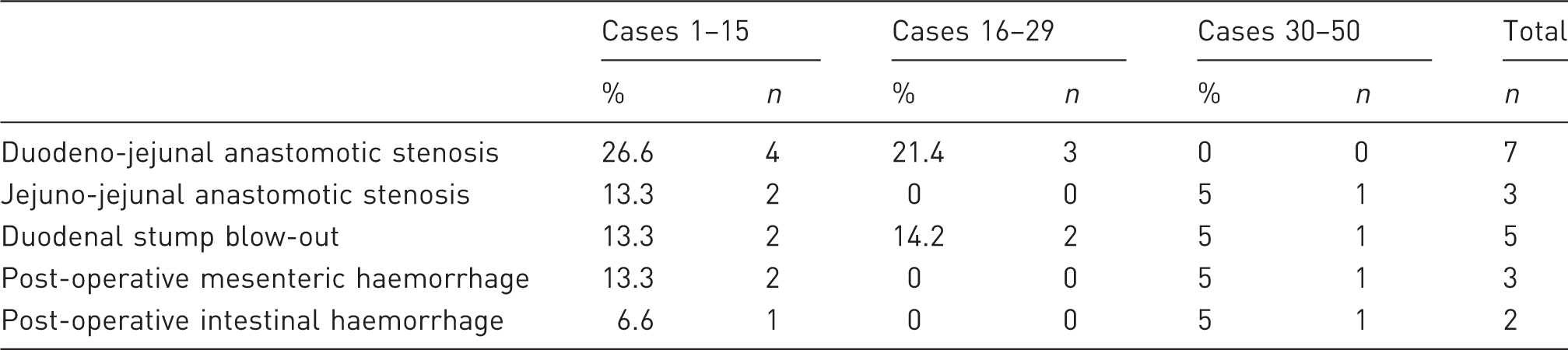
Survival for 3 days after surgery with no sign of significant distress can be considered confirmation of successful surgery. We noticed that the mortality rate after this time point is negligible. Overall survival rates increased from about 30% in the first 15 cases to 80% in the final group of 20 animals (Figure 5). The primary causes of short-term mortality (<3 days) that lead to low survival rates in the first group of animals were obstructions at the level of the duodeno-jejunal anastomosis, or leak of the duodenal stump (which resulted in bile collection in the peritoneal cavity). The intestinal anastomoses can cause stenosis if the sutures are placed too closely, or leakage if the sutures are placed too widely. In the case of duodeno-jejunal anastomosis, the difference in lumen width between the duodenum and the jejunum is a risk factor for stenosis. Indeed, duodeno-jejunal anastomotic stenosis was the cause of the majority of overall deaths (n = 7) in all three cohorts, followed by Duodenal stump blow-out (n = 5), which can be a result of a partial jejuno-jejunal anastomotic stenosis. Further causes included intra-peritoneal haemorrhage due to injury at the level of the mesenteric vessels and endoluminal haemorrhage following enterotomy (Table 1). Nonetheless, overall mortality was significantly lower in cohort 30–50 compared to cohort 1–15 (p = 0.03).
(a) Mortality and survival rates. The initial 50 trial surgeries are shown in terms of overall mortality rate (b) and survival time in days. Causes of mortality in duodenal-jejunal bypass.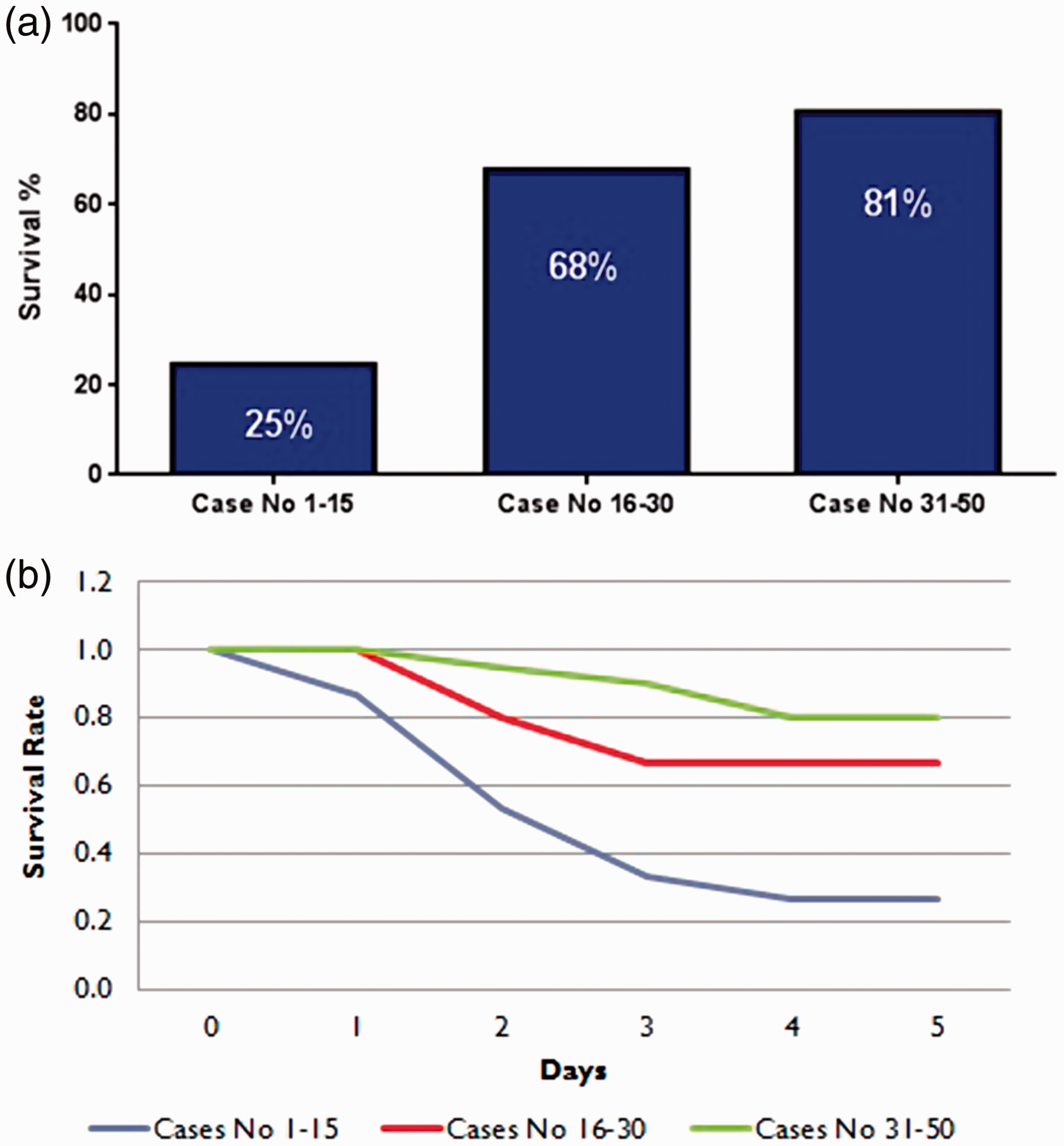
Discussion
Mechanistic research over the last decade has provided evidence suggesting that changes in gastrointestinal physiology can play a role in the effects of surgery on diabetes. 38 These studies have shown that the improved glucose homeostasis observed following bariatric surgery is not due to weight loss alone.39,40
The use of DJB has allowed investigating mechanisms of action of bariatric/metabolic surgery and specifically the effect of anatomic manipulation of the small intestine, without the weight loss caused by gastric resection. Original experiments with DJB provided the first evidence of a direct effect of gastrointestinal surgery on T2DM, 5 a knowledge that provides a biological rationale for the use of surgery as a specific therapy for the disease (metabolic or diabetes surgery). The DJB has also been used in research on other aspects of the metabolic syndrome and its relationship with changes in gut microbiota22,24,36 and has facilitated investigations linking dorsal and ventral striatal regions with different intestinal segments through the nigrostriatal dopamine pathways. 41 Hence, DJB represents an excellent experimental model for investigations aimed at elucidating elusive mechanisms of metabolic regulation.
In our experience, approximately 20 DJB cadaver practices are required to master the technique for an experienced microsurgeon. Although cadaver training is a vital part of the learning curve, it has certain limitations. It does not provide a realistic representation of the bleeding risks during mesenteric handling and intestinal resection. In addition, it cannot completely replicate the anastomotic tension caused by solid food during bowel movement. These observations were made during the first cohort of animals operated on, resulting in the very high mortality rate of 75%, which then allowed us to improve the surgical technique and peri-operative care as described. Following cadaver practice, approximately 30 DJBs are required to substantially reduce mortality rate and obtain long-term survival (>3 days post-operatively) for the vast majority of operated rats.
In our experience presented here, the mortality rate dropped 53.1% between the first group and the third group, and the majority of the animals survived in the long term.
There are several modifications of the experimental model of DJB in terms of surgical technique, methods of anaesthesia and peri-operative care. Many different anaesthesia methods have been described, varying from ketamine injections to halothane inhalation. Moreover, different pre- and post-operative diets are used, including Regular Ensure from Abbott Laboratories and glucose infusions. There are also differences among laboratories regarding pre-operative fasting, size and material of sutures, with nylon, silk and PDS used more often. Technical variants of DJB include anastomoses between the stomach and jejunum (gastro-jejunostomy) or between the duodenum and jejunum (duodeno-jejunostomy as in the technique presented here). Both interrupted and running suturing have been described for the upper anastomoses, which can be performed either side-to-side (as we describe here) or end-to-side. Most importantly, different limb lengths have been reported, with a range extending from 12 cm to 35 cm for the alimentary limb.16,17 Here we reported the DJB procedure as originally described by Rubino and Marescaux, involving a 10 cm biliary limb and 15 cm alimentary limb. 5 These lengths were chosen to proportionally resemble a standard gastric bypass in humans.
In terms of anaesthesia, we used isoflurane inhalation to better control anaesthesia levels and time. We also found that fasting the animals overnight can be helpful when creating the anastomoses, as it allows a clearer exposure of the intestinal lumen. Pros and cons of different suture materials were thoroughly considered. We chose to use Coated Vicryl, which we believe provides a better seal of the anastomoses compared to nylon. Continuous suturing for the anterior and the posterior side of each anastomosis also improved the procedure time when compared to interrupted suturing.
During both the cadaver practice and the operations on living animals, a number of critical technical factors emerged. An important factor for the success of the procedure was the careful handling of the mesentery, avoiding unnecessary stretches that could cause blood loss either intra- or post-operatively. Moreover, ensuring gaps are no bigger than 1 mm in each anastomosis is essential to avoid leaks. Due to the differences in the width of the lumen between the pylorus and the jejunum, it is also important to place a maximum of four sutures on each side, to avoid inducing stenosis on the intestinal side.
A stenosis at the jejuno-jejunostomy can also result in increased intraluminal pressure in the biliary limb, which can cause a leak at the duodenal stump. Hence, an adequate enterotomy at the jejunum must be performed and care must be taken to minimize stenosis.
Factors that can facilitate recovery include pre-operative liquid diet and maintaining appropriate hydration peri-operatively. As reported before, 42 it is useful to expose the animals to the same diet before and after surgery as they might refuse to eat due to the combination of pain and a new diet in the first days post-operatively. One advantage of using a liquid diet for the first post-operative days is that even in the case of a partial anastomotic stenosis or pain, the animals will still be able to consume a sufficient amount of nutrients. Hydration through subcutaneous 0.9% sodium chloride injections can facilitate recovery, but placement of HydroGel inside the cage can be less stressful to the animals that are not able to stretch their abdomen to reach their water bottle due to pain. Additionally, appropriate levels of anaesthesia and oxygen delivery, correct anaesthetic mask and operating time can all affect survival and recovery. Temperature and heart rate should be recorded throughout the operation to anticipate potential post-operative complications.
In summary, the DJB is a challenging operation in rodents. A standardized protocol, as proposed here, can allow more researchers from different disciplines to use DJB for mechanistic investigations about metabolic/ bariatric surgery and the role of the gastrointestinal tract in metabolic regulation.
Footnotes
Author contribution
EA, LG and FV participated in all the experiments, coordinated peri-operative care and prepared the first draft of the article. EA, LG and FV performed surgical operations. IC provided assistance with experiments and contributed to drafting the article. SC and RW supervised the studies and contributed to the article. FR provided mentorship for the surgical technique, supervised experiments and revised the article. All authors have read and approved the content of the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Foundation for Liver Research and the National Institute for Health Research. LG was supported by the Institute of Cardiometabolism and Nutrition, Société Française de Chirurgie Digestive, Fondation Obélisque, APPERT Institute and Société Française et Francophone de l'obésité et des maladies métaboliques.
