Abstract
Alfaxalone is a neuroactive steroid used as a general anaesthetic in several species including dogs, cats, rabbits and ferrets. It has a wide margin of safety and a similar anaesthetic profile to propofol. To increase its aqueous solubility, a new formulation with cyclodextrins has been marketed recently. The objective of this study was to evaluate the anaesthetic effect of several doses of alfaxalone alone, considering differences between sexes, and alfaxalone combined with dexmedetomidine and fentanyl in the rat administered by the intraperitoneal route. A total of 40 Sprague Dawley rats, involved in three studies, were used. Firstly, 25, 35 and 45 mg kg–1 of alfaxalone alone were tested. In a second study, alfaxalone (25 mg kg–1, females; 75 mg kg–1, males) was combined with dexmedetomidine (0.05 mg kg–1). Finally, alfaxalone (20 mg kg–1, females; 60 mg kg–1, males) was combined with dexmedetomidine (0.05 mg kg–1) and fentanyl (0.1 mg kg–1). Times of onset and duration of anaesthesia, and analgesia, deemed as losing of withdrawal pedal reflex, were recorded. Alfaxalone alone produced a 2 - to 3-fold longer time of anaesthesia in females, although surgical anaesthesia was not achieved in either sex. The addition of dexmedetomidine and fentanyl to alfaxalone produced a similar time of analgesia as well as increased time of anaesthesia in both sexes. In conclusion, alfaxalone produces light anaesthesia in rats, and males required a higher dose. The combination with other sedatives or analgesics, such as dexmedetomidine or fentanyl, allows a more prolonged anaesthesia with analgesic effects, potentially suitable for invasive procedures.
Although inhalational anaesthesia has been increasingly employed in rodents, injectable anaesthetics should also be considered since they may provide advantages over the former technique. These may include avoidance of (1) lower cardiovascular depression derived from high doses of inhalational anaesthetics, (2) safety concerns due to potential environmental pollution, and (3) limitations in the use of bulky equipment. A suitable injectable anaesthetic combination should involve short-acting anaesthetic drugs with a wide safety margin and that are, preferably, reversible. Most commonly, the use of injectable anaesthetic procedures for rodents is based on combinations of dissociative drugs such as ketamine with alpha-2 adrenergic agonists and/or tranquilizers or sedatives. Unfortunately, these procedures produce prolonged sedation, and ketamine-based combinations may cause a relatively prolonged recovery. 1 Another short acting anaesthetic drug, the GABAA agonist propofol, has been proposed as an alternative to ketamine-based anaesthetic combinations. However, this drug does not provide surgical anaesthesia when administered alone intraperitoneally (IP) and has been combined with the alpha-2 adrenergic receptor agonist medetomidine and the opioid fentanyl in rodents to attain such an effect.1–3
Alfaxalone is a neuroactive steroid with anaesthetic properties and a wide safety margin, which modulates the inhibitory GABAA receptors. 4 This anaesthetic has been reformulated solubilised with 2-hydroxypropyl-b-cyclodextrin (Alfaxan). The anaesthetic effects of alfaxalone in the former commercial presentation, containing polyethoxylated castor oil (Cremaphor) as surfactant, have been compared with the new formulation of alfaxalone, confirming that both presentations retain the same anaesthetic profile. The allergic sensitivity to the former formulation limited its use to veterinary anaesthesia. 5
Alfaxan has been licensed for use in cats and dogs and reported recently in rats, where relevant differences between the sexes have been observed.6–8 These differences were observed previously with the former formulation of alfaxalone and alphadalone. 9 The new formulation has also been employed in rabbits and ferrets, producing light anaesthesia lasting some 20–30 min, depending on the dose employed, and fast recovery with minimal or limited cardiovascular effects.10, 11 However, a lack of analgesia was observed, and analgesic support should be provided in painful procedures. 12 In addition, alfaxalone produces hypoventilation and oxygen support may be advisable. The anaesthetic profile of alfaxalone resembles that of propofol, and its effects may be potentiated when combined with sedatives and analgesics. 12 We hypothesised that this anaesthetic may produce dose-related anaesthesia when administered IP to rats and may be potentiated by the coadministration of sedatives and analgesics. The aim of this study was to assess the anaesthetic and analgesic properties of Alfaxan administered alone to rats IP, and to determine a suitable dose range in this species. In addition, differences in Alfaxan anaesthesia between the sexes were evaluated. Finally, the combined effects of Alfaxan with dexmedetomidine and fentanyl IP were characterised.
Materials and methods
Animals
Forty Sprague-Dawley rats from our institutional breeding colony (originally purchased from Harlan Laboratories, San Pietro Al Natisone, Udine, Italy), weighing 230–300 g (females) and 300–400 g (males), aged between 6 and 8 months old (both sexes), were housed in groups of three to four (Models 2154F and 1291H, Type III H, Techniplast, Buguggiate, Italy; bedding Ultrasorb, Panlab, Cornella, Spain) and were kept in a 12 h:12 h light:dark cycle at room temperature between 21–24℃. Food (A04 SAFE, standard diet for rodents, Panlab, Cornella, Spain), and water were provided ad libitum. All animals were considered healthy based on veterinary physical examination, with no history of health abnormalities, and were not handled before their inclusion in the study except for routine cage cleaning maintenance. All the animals were used according to European and national regulations and the study was approved by the Ethics Committee (CEEA 001 b/14; Getafe University Hospital, Madrid, Spain).
Drugs
Alfaxalone (Alfaxan 10 mg mL–1, Vétoquinol, Lure, France); Dexmedetomidine (Dexdomitor 0.1 mg mL–1, Esteve, Barcelona, Spain), Fentanyl (Fentanest 0.05 mg mL–1, Kern Pharma, Terrasa, Spain). Dilutions were made with saline 0.9 % (Grifols, Parets del Vallès, Spain).
Study design
Experimental design of the three consecutive studies indicating the doses of Alfaxan, dexmedetomidine and fentanyl administered intraperitoneally to male and female Sprague-Dawley rats. The n value within each study is shown in parenthesis.

Randomisation was performed within each study, with the same number of male and female rats being used with a random generated table (Excel, Microsoft). Randomisation was blocked by sex, variables were recorded in a non-blinded fashion, and the experimental unit was the individual rat. Rats were anaesthetised between 10.00
Determination of a suitable dose range of Alfaxan
A crossover study (randomised block design) was performed administering three doses of Alfaxan IP (25, 35 and 45 mg kg–1) to eight rats (four females and four males), at least at 1-week intervals. The lowest dose was based on a previous study. 4 Although relevant differences between the sexes were found, the results were considered consistent enough (duration of anaesthesia) to choose a suitable dose of Alfaxan to be combined with dexmedetomidine in Study 2.
Combination of Alfaxan with dexmedetomidine
Since differences in the response between sexes were determined, a dose of Alfaxan of 25 mg kg–1 was selected for females (n = 8) and 75 mg kg–1 for males (n = 8), and were combined with dexmedetomidine 0.05 mg kg–1 and administered IP in random order. A total of 16 rats were used.
Combination of Alfaxan with dexmedetomidine and fentanyl
Alfaxan (20 and 60 mg kg–1, females (n = 8) and males (n = 8), respectively), dexmedetomidine 0.05 mg kg–1 and fentanyl 0.1 mg kg–1 were combined and administered IP in random order. A total of 16 rats were used.
Monitoring
Once animals were induced to anaesthesia, a pulse oximeter probe (UT100V Veterinary Handheld Pulse Oximeter, Utech Chongqing, China) placed in a hind limb was used for the continuous monitoring of the heart rate (HR) and haemoglobin oxygen saturation (SpO2), and recorded every 5 min. Observation of thoracic wall movement was employed to monitor and record respiratory rate (fR) every 5 min, using a 20-s interval to count respiratory movements. A digital rectal thermometer (Henry Schein Inc., Melville, NY, USA) was used to record body temperature (BT) every 10 min. Rats were allowed to breathe room air in study 1. However, in studies 2 and 3, 100% oxygen (0.5 L/min) was delivered via a face mask (Rodent Circuit Set, Harvard Apparatus, Holliston, MA, USA) (see Study design, below).
The following times were also recorded: Time to Induction, defined as the time taken from injection to the loss of the righting reflex; Duration of Analgesia, defined as the time of loss of pedal withdrawal reflex (lack of movement), assessed by digital pinching of the toes with the nail; and Duration of Anaesthesia, defined as the total time of loss of the righting reflex.
Anaesthesia
No previous acclimatisation to the procedure was performed, and rats were weighed and individually transferred to a single cage with bedding material just before the drugs were administered. Volumes of Alfaxan alone or combined with dexmedetomidine or dexmedetomidine and fentanyl were calculated according to the doses, freshly prepared and adjusted to equal volumes within each study with sterile saline, and administered IP into the right caudal quadrant of the ventral abdomen by an experienced researcher (MA). A cloth was employed to reduce the stress on handling. After injection, the rat was monitored closely in the cage until loss of the righting reflex was achieved. Then, it was placed on a warming blanket (Aposan, Cofares, Madrid, Spain) to maintain body temperature between 37.0℃ and 38.5℃, and a protective ointment (Lubrithal 10 g, Dechra Veterinary products, LLC, Northwich, UK) was applied to the eyes. The pedal withdrawal reflex was tested every 5 min alternating between limbs.
The noxious stimulus was performed always by the same researcher (MA) by pinching the web of the skin between the toes with the finger nail. Movement of the hind limb was considered as a positive response and the lack of response as negative. Once the rat recovered the righting reflex, it was placed isolated from social group in a clean cage in a quiet and warm area (24–28℃) and closely monitored until total recovery approximately 1 h later. The animal was transferred to its former cage with the social group 12 h later.
Statistical analysis
A hypothesis of differences between doses and sexes was considered in Study 1, whereas no differences were considered between sexes in Studies 2 and 3 by adjusting the dose. Data are expressed as mean ± SD.
Power calculation
Duration of anaesthesia was considered the principal variable for power calculation in Study 1. A power of 0.8 with an α value of 0.05, an effect size of 10–20 min of duration of anaesthesia, and SD of 9.4 min (obtained from the first three rats, 2-tailed), gave a sample size of eight rats per group (three groups: 25, 35 and 45 mg kg–1), with both sexes balanced, in Study 1 (Experimental Design Assistant, NC3Rs; https://eda.nc3rs.org.uk/eda). Rats from this study were included in a crossover design, and every animal received three doses of Alfaxan in a random fashion. In Studies 2 and 3, the duration of analgesia was considered as the main variable with an effect size of 5–10 min and SD of 5.5 min (2-tailed) suggesting a sample size of eight rats per group (two groups: eight females and eight males).
Group comparison
To analyse data, the normality of each group sample was ascertained with the Shapiro-Wilk test. In Study 1, the repeated measures (RM) ANOVA for repeated data was employed for monitored and recorded data during anaesthesia (HR, fR, SpO2 and BT). Also, this RM test was employed to compare studied times (onset of anaesthesia, duration of analgesia, duration of anaesthesia) where the dose was considered the repeated factor within the same individual, and sex the second factor. Student’s t-test was employed to determine differences between sexes when the same dose was tested.
In Studies 2 and 3, Student’s t-test was employed for single values (weight and times) and the RM ANOVA (HR, fR, SpO2 and BT). The Bonferroni post hoc test was employed to compare doses and times when appropriate. A p value < 0.05 was considered as significant.
Results
All animals recovered fully from anaesthesia and were included in the data analysis.
Study 1: Alfaxan
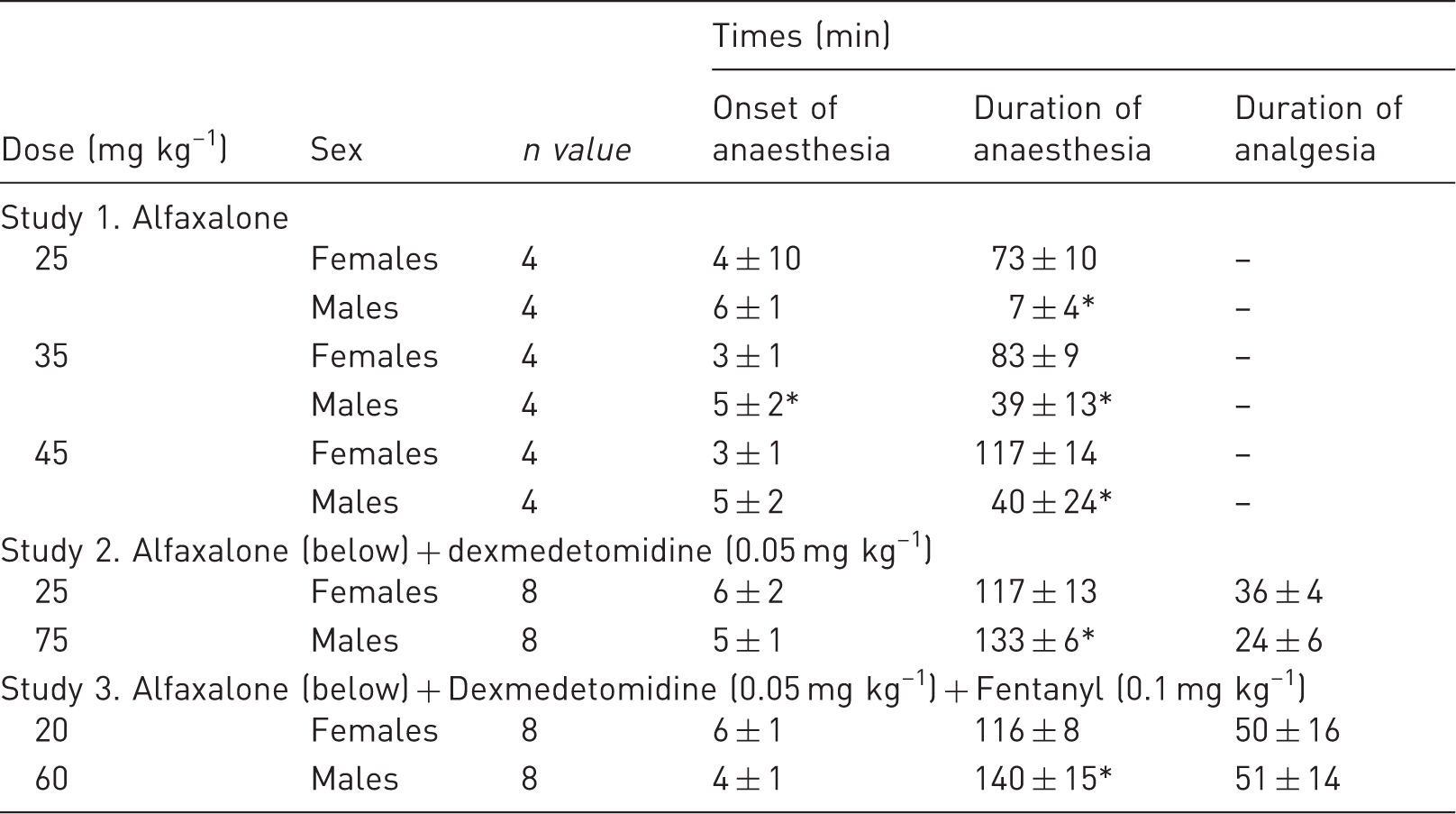
Onset of anaesthesia, defined as the time taken from injection to the loss of the righting reflex; Duration of Analgesia, defined as the time of loss of pedal withdrawal reflex (lack of movement), assessed by digital pinching of the toes with the nail; and Duration of Anaesthesia, defined as the total time of loss of the righting reflex, in rats anaesthetised with three different doses of Alfaxan administered intraperitoneally (IP) (Study 1), Alfaxan and dexmedetomidine IP (Study 2) and Alfaxan, dexmedetomidine and fentanyl IP (Study 3).
Indicates differences between sexes (Student’s t-test; see text for p-values)
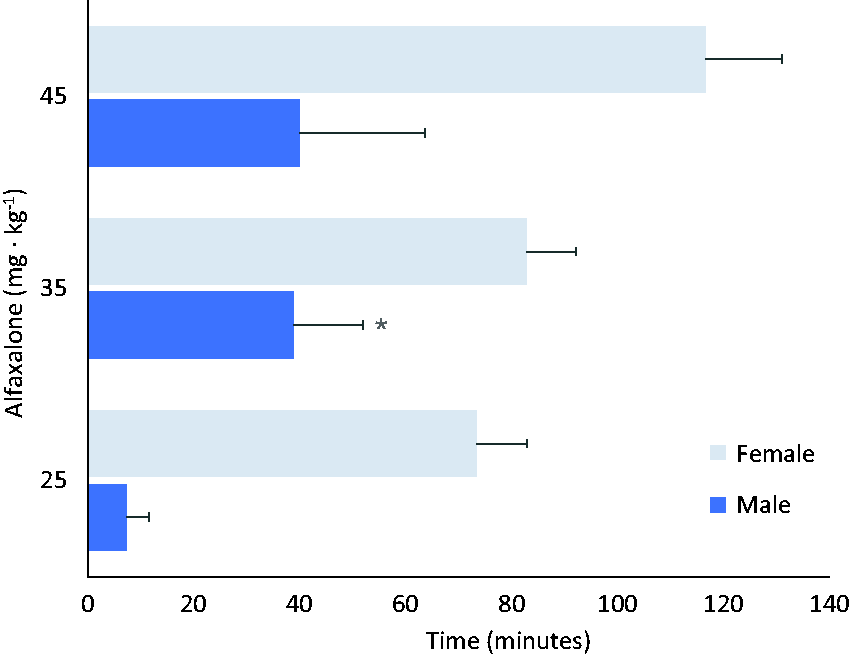
Duration of anaesthesia from male (n = 4) and female (n = 4) rats anaesthetised with three doses of Alfaxan (25, 35 and 45 mg kg–1) administered intraperitoneally. Significant differences were found between both sexes (RM ANOVA; p < 0.05). * indicates significant differences compared with the lowest dose, same sex (Bonferroni test; p < 0.05).
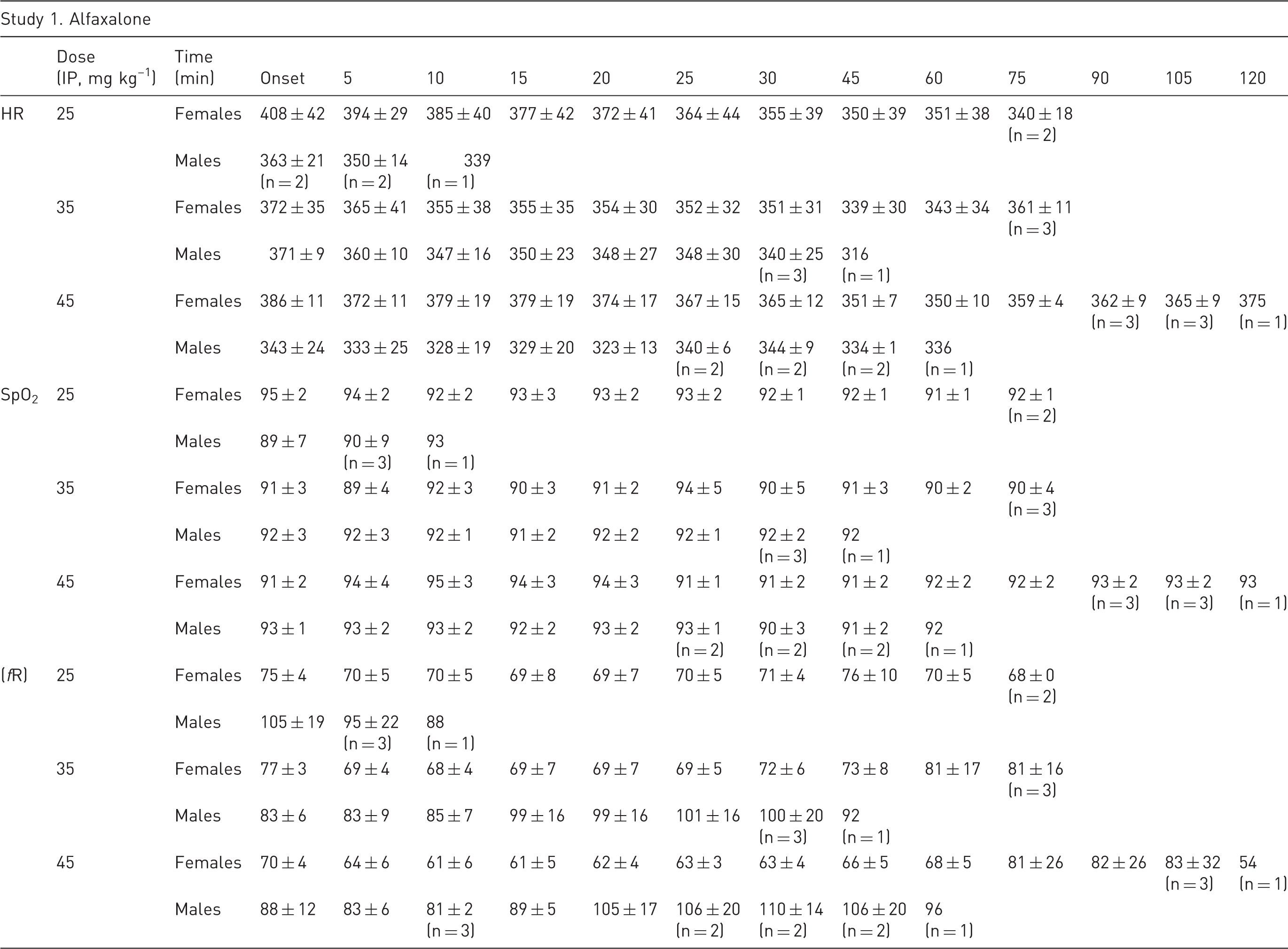
Heart rate (HR) in beats per minute (bpm), respiratory rate (fR) in breaths per minute and haemoglobin oxygen saturation (SpO2) in %, from female and male rats anaesthetised with Alfaxan intraperitoneally at 25, 35 and 45 mg kg–1 (n value is always 4 unless otherwise indicated; see Materials and Methods for details).
Study 2: Alfaxan + Dexmedetomidine
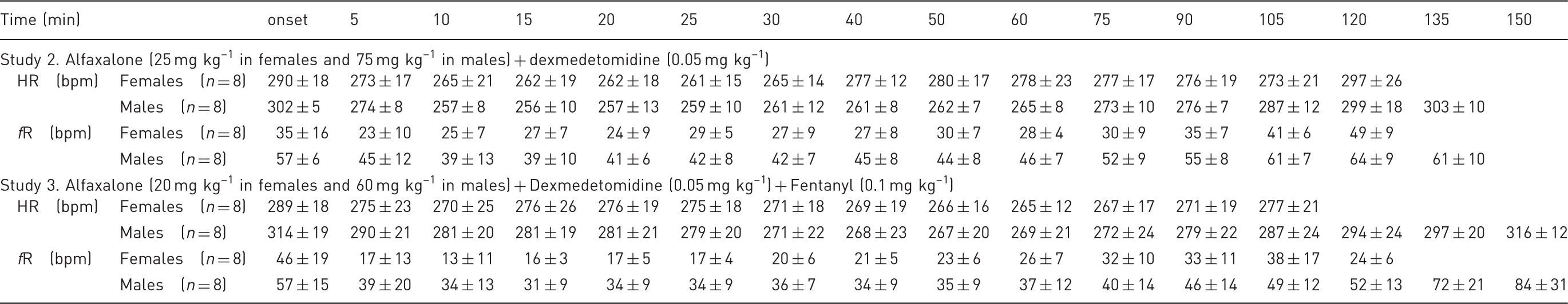
Onset of anaesthesia was similar and below 7 min with both sexes (t-test, p = 0.378). Duration of anaesthesia was shorter in females (117 ± 13 min) than males (133 ± 6 min; t-test, p = 0.006), whereas the duration of analgesia was shorter in males (24 ± 6 min) compared to females (36 ± 4 min; t-test, p = 0.0003, Figure 2).
Duration of anaesthesia, defined as the total time of loss of righting reflex, and duration of analgesia, defined as the time of loss of the pedal withdrawal reflex, from male and female rats anaesthetised with a) alfaxalone (25 and 75 mg kg–1, eight females and eight males, respectively) and dexmedetomidine (0.05 mg kg–1) intraperitoneally; or b) alfaxalone (20 and 60 mg kg–1, eight females and eight males, respectively), dexmedetomidine (0.05 mg kg–1) and fentanyl (0.1 mg kg–1) intraperitoneally. * indicates significant differences compared to female rats (Student’s t-test p < 0.05).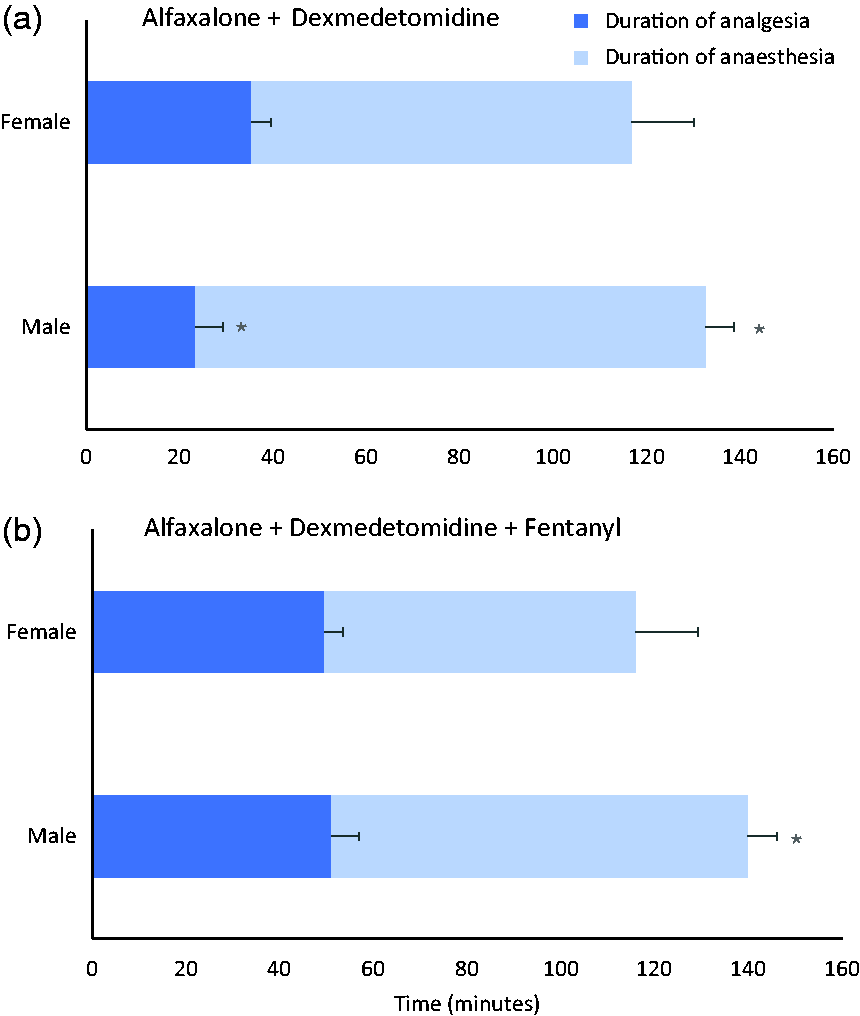
HR and fR in female and male rats anaesthetised with Alfaxan combined with dexmedetomidine or dexmedetomidine and fentanyl. Doses administered IP to females and males are indicated (see Materials and Methods for details).
Study 3: Alfaxan + Dexmedetomidine + Fentanyl
Onset of anaesthesia was more prolonged in females compared to males (6 ± 1 and 4 ± 1 min, respectively; t-test, p = 0.004), whereas the duration of anaesthesia was shorter (116 ± 8 and 140 ± 15 min, respectively; p = 0.002). There were no differences in the duration of analgesia between both sexes (50 ± 16 and 51 ± 14 min in females and males, respectively; p = 0.831).
No differences in HR were observed between both sexes (RM ANOVA, p = 0.471) although a decrease in HR was observed with time (RM ANOVA, p =8 × 10–7). The fR was lower in females (RM ANOVA, p = 0.002) decreasing with time (p = 2 × 10–8). All rats received oxygen via facemask and had a SpO2 > 95%.
Recovery from all alfaxalone and alfaxalone-based combinations was smooth and uneventful.
Discussion
Alfaxan produced relevant differences in the duration of anaesthesia between males and females when given alone, resulting in a more prolonged effect in females when the same doses were administered IP. Compared to females, a three-fold higher dose of Alfaxan was required in males to attain a similar duration of anaesthesia. This effect was observed with three different doses, none of which provided analgesic effects to prevent the response to a standard noxious stimulation. Although alfaxalone has no known antinociceptive properties, coadministration with dexmedetomidine increased anaesthetic duration and produced short-term analgesia, which increased further when fentanyl was also administered.
Sex-related differences in the anaesthetic effects of alfaxalone have already been described with the former formulation with a mixture of alfaxalone and alphadalone solubilised in castor oil (Althesin). 9 Almost a four-fold IP higher dose was required in males compared to females. However, such a difference was not observed when this drug was administered by the IV route.9, 13 Similarly, we found a three-fold dose difference between sexes with the new cyclodextrin-based formulation of alfaxalone to achieve a similar effect. Differences in anaesthesia duration between the former and the currently marketed formulation may involve the use of alphadalone in the former commercial presentation but also the different age and strain of rats employed. Anaesthetic dose and duration of anaesthesia using the former formulation required higher doses of alfaxalone in female rats under 30 days old, although no further differences were observed in rats aged above 30 days. 9 However, in a study using the cyclodextrin-formulated alfaxalone in young (7–10 weeks) Wistar female rats, a dose of 20 mg kg–1 IP produced a shorter and highly variable duration of anaesthesia (42 min) compared to a slightly higher dose of 25 mg kg–1 (73 min) employed in the present study. 4 The strain and age of the rats, but also absorption from the site of IP injection may account for the difference, suggesting a relatively high variability in the effects of alfaxalone when administered alone. 14 Furthermore, differences in the pharmacokinetics of intravenous cyclodextrin-formulated alfaxalone be-tween sexes have been identified in Sprague Dawley rats in plasma clearance, half-life and mean residence time when administered as a constant rate of infusion. 8
A relevant clinical sex difference in the effects of Alfaxan by the IP route in rats is a limitation for its use since no other anaesthetic drug employed in this species produces such an effect, and doses provided to male and female rats are the same. Reasons for such differences have been attributed to a higher concentration of the endogenous GABA modulator (3α-hydroxydihydroprogesterone) in females. 5 Alfaxalone, as well as naturally occurring steroids, enhance GABA-mediated inhibition in the brain, suggesting a potentiation of their effects. 15
Regardless of the dose or depth of anaesthesia, Alfaxan did not provide any analgesic effect when administered alone, thus its combination with analgesic drugs would be required for painful procedures. In rats, alfaxalone-based anaesthetic combinations have not been reported previously. In mice, surgical anaesthesia has been determined with alfaxalone alone but also in combination with medetomidine and butorphanol, administered subcutaneously. Interestingly, surgical anaesthesia was not achieved with either anaesthetic regimen when given by the IP route. 16 However, the definition of surgical anaesthesia was different from our study and while we evaluated the lack of pedal withdrawal reflex, other reflexes have also been considered. 16 Withdrawal of the hind limb following toe pinching is a commonly employed method suggesting surgical anaesthesia in rodents. Reduced, non-surgical, anaesthetic effects of alfaxalone may result from reduced drug availability due to a first pass elimination by hepatic metabolism when administered IP.
In cats and rabbits, the intramuscular administration of Alfaxan alone produced moderate-to-deep sedation whilst anaesthesia was achieved only in the former species when medetomidine was co-administered.11,17,18 In rats, both analgesic and anaesthetic effect potentiation was found when dexmedetomidine was combined with equipotent alfaxalone doses (3:1, males:females). Although a longer duration of anaesthesia was observed in males, duration of analgesia was slightly shorter and fR was less reduced compared with females, suggesting that the doses of drugs providing analgesia such as dexmedetomidine, would need to be increased. Females showed a reduced time of anaesthesia associated with a longer analgesic action. Such a finding was unexpected and further studies should be conducted to determine whether it was an actual, rather than just statistical, finding.
Analgesic potentiation may be produced by administering opioids such as fentanyl. The combination of this opioid with alfaxalone and dexmedetomidine produced an increase in the duration of surgical anaesthesia, deemed as the loss of pedal withdrawal reflex. Again, an alfaxalone dose ratio of 3:1 (males:females) provided equivalent timing of both analgesic and anaesthetic effects. A similar propofol-based combination with medetomidine and fentanyl administered IP to rats demonstrated loss of the pedal withdrawal reflex during at least 30 min, when the alpha-2 adrenergic receptor antagonist atipamezole was administered. 3 As expected, this combination provided better anaesthetic and analgesic effects compared to propofol given alone. Similar results were obtained when the same combination was used in mice. 1
Anaesthetic drugs are usually associated with cardiovascular and respiratory depression closely related with the drugs involved but also their interactions. These effects are a safety concern during anaesthesia. Dexmedetomidine has potent alpha-2 adrenoceptor activity characterised by vasoconstriction, leading to an initial increase in blood pressure followed by hypotension, and bradycardia.19,20 Not surprisingly, when combined with alfaxalone, dexmedetomidine produced bradycardia. Besides, fentanyl, a potent mu opioid receptor agonist, also produces vagally mediated bradycardia.21,22 Thus, both alfaxalone-dexmedetomidine and alfaxalone-dexmedetomidine-fentanyl combinations produce a marked decrease in HR.
Ventilatory depression, determined by marked decreases in both fR and SpO2, was observed when alfaxalone was given alone, with the latter values suggesting mild to moderate hypoxemia within a normal BT range. Oxygen supplementation was then employed with the two alfaxalone combinations, where a decrease in fR was also observed. A greater decrease in fR was observed in females, which would suggest an increased depth of anaesthesia. However, the duration of analgesia was similar between sexes. The combination of medetomidine and fentanyl produces respiratory depression in rats. 23 Dexmedetomidine might induce relative minor ventilatory depression in rats, suggesting that most of the depressant effects might be due to fentanyl alone or its combined effects with dexmedetomidine.24,25 Therefore, it is advisable to provide oxygen to anaesthetised rats when these alfaxalone-based combinations are employed.
This study has some limitations. Complete cardiovascular and respiratory monitoring was not considered in the present study and only basic parameters were assessed. Therefore, the effects on relevant parameters such as blood pressure or capnography should be considered to better assess the true impact of alfaxalone and alfaxalone-based combinations. Besides, pedal withdrawal reflex was considered as a surrogate for surgical stimulus. Therefore, these drugs and combinations should be tested in clinical conditions where surgery is performed. Although the anaesthetic and analgesic variables were not blindly assessed and may have biased the results, the same researcher (MA) performed all assessments thus reducing variability. Rats were anaesthetised between 10.00
In conclusion, Alfaxan is a suitable drug for light anaesthesia in rats where analgesia is not required and produces minor adverse effects. However, this anaesthetic should be combined with other sedatives and opioids to provide surgical anaesthesia when administered intraperitoneally. A relevant sex difference in the dose required to produce anaesthetic effects was determined, where males required a dose three times higher. This may be a practical limitation for its use. Finally, clinical studies should evaluate the feasibility of these anaesthetic combinations in surgical procedures.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
