Abstract
Fast recoveries are essential when looking for a safe anaesthetic protocol to use on mice. Propofol is a short-acting anaesthetic agent, which provides a smooth, fast recovery. A recent study carried out in our laboratory showed that the intraperitoneal (i.p.) administration of propofol combined with a fast-acting opioid does not provide a sufficiently stable anaesthesia. In this experiment, we hypothesized that the additional application of medetomidine would increase muscle relaxation and analgesia. Fifty-four male CD1 mice, divided into six groups of five and three groups of eight, were used to test nine different combinations of propofol, medetomidine and fentanyl administered i.p. and reversed with atipamezole 30 min after induction. These combinations were composed in the following manner: propofol 75 mg/kg, medetomidine 1 and 2 mg/kg and fentanyl 0.1, 0.15 and 0.2 mg/kg. The depth of anaesthesia, loss of righting reflex, loss of pedal withdrawal reflex, pulse rate and respiratory rate were recorded along with the duration and quality of the recovery. The combination of propofol and medetomidine provided a predictable induction, hypnosis and muscle relaxation, but surgical anaesthesia (loss of pedal withdrawal reflex) was not achieved. The addition of fentanyl increased analgesia leading to surgical anaesthesia. We concluded that a combination of 75/1/0.2 mg/kg of propofol, medetomidine and fentanyl, respectively, is a safe, easy and reversible technique for i.p. anaesthesia in mice, providing a surgical window of 15 min and restraint for 30 min with a fast recovery.
Most anaesthetic protocols used in mouse anaesthesia are based on the combination of two drugs, which are usually administered via the intraperitoneal (i.p.) or subcutaneous (s.c.) route. The widely used i.p. technique has the advantage of being simple while remaining a minor cause of distress to the animal. 1 Long anaesthetic recoveries are associated with hypothermia and hypoxia 2 making it essential to find a safe anaesthetic protocol with minimal complications and anaesthetic death rate in order to diminish the recovery period. In many research facilities, volatile anaesthesia is the main technique used when looking for fast recoveries. However, in some circumstances, this technique is not recommended, such as neurosurgical 3,4 or oropharyngeal procedures. This technique also requires the use of specific delivering equipment.
Most of the commonly used anaesthetic procedures for mice are based on combinations of dissociative drugs with α2-agonists and/or tranquilizers or sedatives. 5,6 Unfortunately, these procedures produce prolonged sedation and full recovery of consciousness can take several hours. Some anaesthetic combinations such as medetomidine/ketamine 6,7 and medetomidine/clonidine/droperidol/fentanyl 8 have the advantage of providing faster recoveries by reversing the medetomidine effect with a specific α2-antagonist such as atipamezole. 9 Nevertheless, combinations including ketamine raise safety issues. These are usually associated with low-quality recoveries, and recent studies showed that sub-anaesthetic treatment with ketamine can induce schizophrenic behavioural disturbances. 10
In order to determine the possibility of using a combination of propofol and a fast-acting opioid to produce anaesthesia in mice, we recently conducted a study where 28 anaesthetic combinations were tested. Propofol was administered on its own as well as in combination with fentanyl, sufentanil or remifentanil. The results obtained in these studies were unpredictable. The synergistic effect of propofol and an opioid was not sufficient to allow surgical procedures. Tail rigidity, limb shaking and head scratching with forefeet were continuously observed during anaesthesia. 11
The combination of fentanyl and medetomidine has been known to produce surgical anaesthesia in dogs 12 and rats. 1 Recently, a study described a successful combination of droperidol, medetomidine, clonidine and fentanyl to anaesthetize mice via the i.p. route. 8 The objective of associating fentanyl to an α2-agonist is to enhance the analgesic effect of fentanyl with minimal respiratory depression. 13
In this study, we hypothesized that the neuroleptanalgesic effect of fentanyl and medetomidine when associated with propofol would provide a suitable anaesthesia with good muscle relaxation, analgesia and reduce dosage requirements of all the drugs. After reversal with atipamezole, we also expect a faster recovery, providing a safer anaesthetic protocol.
Materials and methods
All procedures were carried out under personal and project licences approved by the national regulatory office (Direcção Geral de Veterinária – DGV, Ministério da Agricultura do Desenvolvimento Rural e das Pescas [MADRP], Largo da Academia Nacional das Belas Artes, No 2, 1200 Lisboa, Portugal).
Animals
Fifty-four, specific pathogen free outbred 9–10-week-old male mice (CD1, Harlan Ibérica, Barcelona, Spain), weighing between 28 g and 35 g were used. The animals were kept in a room with controlled temperature (21°C) and humidity (55%). Lights were on for a 12/12 h cycle, with lights off at 17.00 h. Water and rodent pellets (4RF25-GLP Mucedola, Società a Responsabilità Limitata [SRL], Italy) were provided ad libitum. The animals were housed in Makrolon type II cages, provided with corn-cob bedding material (Probiológica, Lisbon, Portugal), tissue nesting material and a cardboard tube. The cages were changed once a week. These animals, previously observed in behavioural studies in the same laboratory, were housed in groups of eight mice per cage.
Drugs
Propofol 2% (Lipuro®, Braun, Melsungen, Germany), fentanyl 0.05 mg/mL (B Braun, Queluz de Baixo, Portugal), medetomidine 1 mg/mL (Domitor®, Pfiser, Seixal, Portugal) and atipamezole 5 mg/mL (Antisedan®, Pfiser) were administered in this study. Standard physiological saline, 0.9%, (Soro Fisiológico, Paracelcia, Porto, Portugal) was used for dilution.
Apparatus
A pulse oximeter (S&W 9040, Athena, Germany) was used for monitoring oxygen saturation (SaO2) and pulse rate in the main study. The environmental temperature was monitored and mouse body temperature was maintained between 36°C and 38ºC during anaesthesia using a homoeothermic blanket (N-HB101-S-402, Panlab, Cornellà, Barcelona, Spain). One hundred percent oxygen (2 L/min) was delivered to the animal via a face mask connected to a coaxial circuit.
Experimental procedures
The study consisted of two parts. A preliminary study was carried out to identify the most suitable drug combinations and dosages. Three combinations were thereafter evaluated in a more detailed study.
General
The mice were weighed using an electronic scale (EMB 200-1, Kern & Sohn GmbH, Balingen, Germany). The drugs were freshly mixed in a multidose container and administered i.p. as a mixture in a single injection.
The i.p. administration was performed lateral to the midline next to the umbilicus. A short 25-gauge needle was inserted at an angle of 45° to the abdominal wall in the lower left quadrant of the abdomen. The maximum volume administered was 0.52 mL. Each animal was restrained and anaesthetized always by the same investigator. Anaesthetic behaviour (Table 1) was recorded by a researcher blinded to the doses administered.
Study protocol
After administration, the animal was placed alone in a cage with bedding material until loss of righting reflex (RRef; defined as the lack of the ability to return to sternal recumbency), and this period of time was recorded. These animals were then moved to a homoeothermic blanket and the body temperature was monitored with a rectal probe. A facial mask was placed to provide oxygen (100%) throughout the procedure.
Animal breathing was clinically assessed by monitoring thoracic movement including the type of movement observed (thoracic or abdominal) during anaesthesia. Respiratory depression was defined as the absence of thoracic muscle movement or the respiratory movements that became shallower and more abdominal leading sometimes to apnoea and death.
The anaesthesia was reversed by atipamezole administration via the i.p. route in a dose five times that of the medetomidine. The onset of recovery of the RRef was recorded. Necropsies were performed in the animals that did not survive in order to determine the cause of death.
Preliminary study
Thirty animals were randomly divided into six groups (n = 5) and the preliminary study was divided into two parts. All the animals in each group were anaesthetized together and the body temperature was monitored in only one animal in each group. Six different anaesthetic combinations were used:
Propofol 75 mg/kg + medetomidine 1 mg/kg (M1P75); Propofol 75 mg/kg + medetomidine 2 mg/kg (M2P75); Propofol 75 mg/kg + medetomidine 1 mg/kg + fentanyl 0.1 mg/kg (M1P75F0.1); Propofol 75 mg/kg + medetomidine 2 mg/kg + fentanyl 0.1 mg/kg (M2P75F0.1); Propofol 75 mg/kg + medetomidine 1 mg/kg + fentanyl 0.15 mg/kg (M1P75F0.15); Propofol 75 mg/kg + medetomidine 2 mg/kg + fentanyl 0.15 mg/kg (M2P75F0.15).
The depth of anaesthesia was evaluated by assessing the pedal withdrawal response (PWR; assessed by pinching the tip of the tail and the metacarpal region of the hind foot between the index finger and the thumb). Reflexes were tested every 2 min after loss of RRef during anaesthesia, alternating between the left and right limbs.
Anaesthesia was reversed 20 min after PWR loss. When the animals did not loose the PWR, anaesthesia was reversed 30 min after loosing the RRef or at the moment of the first movement (FM). The animals were continuously observed during the entire procedure. When the animals recovered, they were returned to their home cage and observed for the next 2 h for any occurrence of abnormal behaviour.
Main study
Twenty-four animals were randomly divided into three groups (n = 8). Each animal received one of the following three combinations. The combinations used were
Propofol 75 mg/kg + medetomidine 2 mg/kg + fentanyl 0.1 mg/kg (M2P75F0.1); Propofol 75 mg/kg + medetomidine 1 mg/kg + fentanyl 0.15 mg/kg (M1P75F0.15); Propofol 75 mg/kg + medetomidine 1 mg/kg + fentanyl 0.2 mg/kg (M1P75F0.2).
After RRef loss, the animals were placed on the homoeothermic blanket with a rectal probe, a pulse oximeter and a facial mask for O2 administration. The pulse oximeter was placed on the upper right hind leg. The pulse rate was recorded at 5 min intervals and at the moment of the FM after the administration of atipamezole. The PWR (assessed by extending the leg and pinching the interdigital web of the foot using the nails of the forefinger and thumb) was the primary method for assessing depth of anaesthesia. The responses were rated according to Antunes et al.
14
based on a scale from 1 to 5, with 1 indicating no response and 5 a marked response. The respiratory rate, rhythm and pattern, eye blinking and whisker movements were also used in the overall evaluation of depth of anaesthesia. These reflexes were tested every 5 min, alternating between the left and right limbs after the RRef was lost.
Anaesthesia was reversed 30 min after the RRef was lost and the mouse placed in a recovery cage, underlying with a piece of paper marked with a 15 cm diameter circle. The mouse was placed in the centre of this circle and observed continuously until complete recovery. Full recovery time was defined as the moment when the animal was upright, making normal, coordinated movements, and having moved its entire body (with the exception of the tail) out of the circle. The animals were then returned to their home cage and observed for another 2 h for abnormal behaviour. During anaesthesia, the respiratory rate was recorded in intervals of 5 min after the RRef was lost. Pulse rates and respiratory frequencies were always recorded prior to PWR evaluation in order to avoid alteration of these values by noxious stimulation. The animals were also observed during the following week to detect any postanaesthetic complications.
Data analysis
The data were recorded using Microsoft® Excel (Microsoft Corporation, Redmond, WA, USA) with a developed macro. The measured anaesthetic times (Table 1) were calculated and are shown as mean and standard deviation in minutes. The data were tested for normal distribution and homogeneity of variance. One-way analysis of variance (ANOVA) was used to compare induction time, latency to achieve PWR loss, period of surgical anaesthesia and total anaesthetic period. Repeated measurements ANOVA with Bonferroni corrections for multiple comparisons were used for pulse rate and respiratory rate comparison among groups and within groups.
For depth of anaesthesia, normal distribution or homogeneity of variance was not assumed. A Kruskal-Wallis ANOVA was used for data comparison between groups, the Mann-Whitney test applied for pairwise comparisons. Wilcoxon Signed Ranks test was applied to compare data within groups. Statistical analysis was calculated with Leadtools® SPSS® 13.0 (SPSS Inc, Chicago, IL, USA).
Results
Preliminary study
The results of the preliminary study are shown in Table 2. Loss of RRef was observed in all animals, in all combinations. No signs of abdominal pain or discomfort were observed after i.p. administration.
Preliminary study
Number of mice that lost the righting reflex (RRef) and pedal withdrawal reflex (PWR) with the respective induction, achievement of surgical anaesthesia, surgical anaesthesia, anaesthetic period and recovery time with the respective number of animals used for each calculation in parenthesis and the death rate of mice when tested with different anaesthetic protocols. Time is expressed in minutes (average ± standard deviation). When animals recovered RRef before administration of atipamezole, recovery time was considered as zero
Propofol/medetomidine: In these combinations, loss of PWR was only observed in one animal, one to which the highest dose of medetomidine (2 mg/kg) was administered, and which did not recover from anaesthesia.
Propofol, medetomidine and fentanyl: PWR loss was observed in all the groups with these combinations; however, only in groups M1P75F0.15 and M2P75F0.15 did all the animals loose the PWR. It was also only in these groups that the average time between loss and recovery of the PWR would allow a reasonable time to perform minor surgery (at least 15 min). Combination M2P75F0.15 was not significantly different from combination M1P75F0.15 in any of the anaesthetic parameters (Table 2). Death was always associated with marked respiratory depression. A mild lymphangiectasis and an image compatible with chyloperitoneum were observed in the dead animals upon necropsy, no other alterations were detected.
Recovery was excellent and very fast in the surviving animals (Table 2). They showed no spontaneous movements during anaesthesia. No abnormal behaviour was detected during the 2 h that followed anaesthesia and no animal showed any sign of postanaesthetic complication during the seven subsequent days.
Main study
The results obtained in the main study are shown in Table 3. No deaths were observed in the main study.
Main study
Data for pulse rate (PR) in beats per minute (bpm), respiratory rate (RR) in movements per minute (mpm) and depth of anaesthesia (DA) in intervals of 5 min and at the moment of first movement (FM) after induction of anaesthesia (0 min), for each anaesthetic combination
Within each parameter, superscript letters a, b and c indicate statistically significant differences (P < 0.05) between time points within groups. Within each parameter, superscript symbols * and † indicate significant differences (P < 0.05) between groups at the same time point. Values are expressed as average ± standard deviation. Induction time, recovery time and time for achievement of complete recovery of mice tested with different anaesthetic protocols of propofol (P), medetomidine (M) and fentanyl (F). Time is expressed in minutes (average ± standard deviation)
No significant differences were identified between the groups M2P75F0.1, M1P75F0.15 and M1P75F0.2 in the induction time, recovery time or complete recovery time.
The behaviour of the animals during anaesthetic induction was very similar. A smooth induction was observed with an increasing reduction in motility. After that, RRef was lost with no signs of respiratory distress, muscle spasms or ataxia.
The recovery of RRef was very fast in all the animals after atipamezole administration. The complete recovery too was very fast in all the animals and no behavioural differences were observed between the groups during this period. No abnormal behaviour was detected in the 2 h following anaesthesia, and no animal presented any sign of postanaesthetic complication during a seven-day follow-up. No deaths were observed in any group in the main study.
A regular pulse rhythm was observed throughout anaesthesia in all the animals. Changes in pulse rate are shown in Table 3 revealing statistically significant differences within the groups. In group M1P75F0.2, the mean pulse rate decreased by 14% between 5 min and 15 min (P < 0.01) and increased by 105% between 30 min and FM (P < 0.01). In group M1P75F0.15, mean pulse rate increased by 48% between 30 min and FM (P < 0.01). In group M2P75F1, mean pulse rate increased by 63% between 30 min and FM (P < 0.01). There were no significant differences between the groups.
Breathing was clinically evaluated during anaesthesia and the pattern was found to be predominantly thoracic. Apnoea was not observed. Oxygen saturation was maintained above 95% in all the animals. Respiratory rates were significantly different between the groups (P < 0.01). Group M1P75F0.2 presented lower respiratory rate values when compared with groups M1P75F0.15 and M2P75F0.1 (P < 0.05). Significant differences within the groups were observed between groups M1P75F0.2 and M1P75F0.15. In group M1P75F0.2, mean respiratory rate increased by 8% at 20 min (P < 0.05). In group M1P75F0.15, mean respiratory rate increased by 28% between induction and 15 min (P < 0.05). Figure 1 shows respiratory rates measured in mice at 5 min intervals.
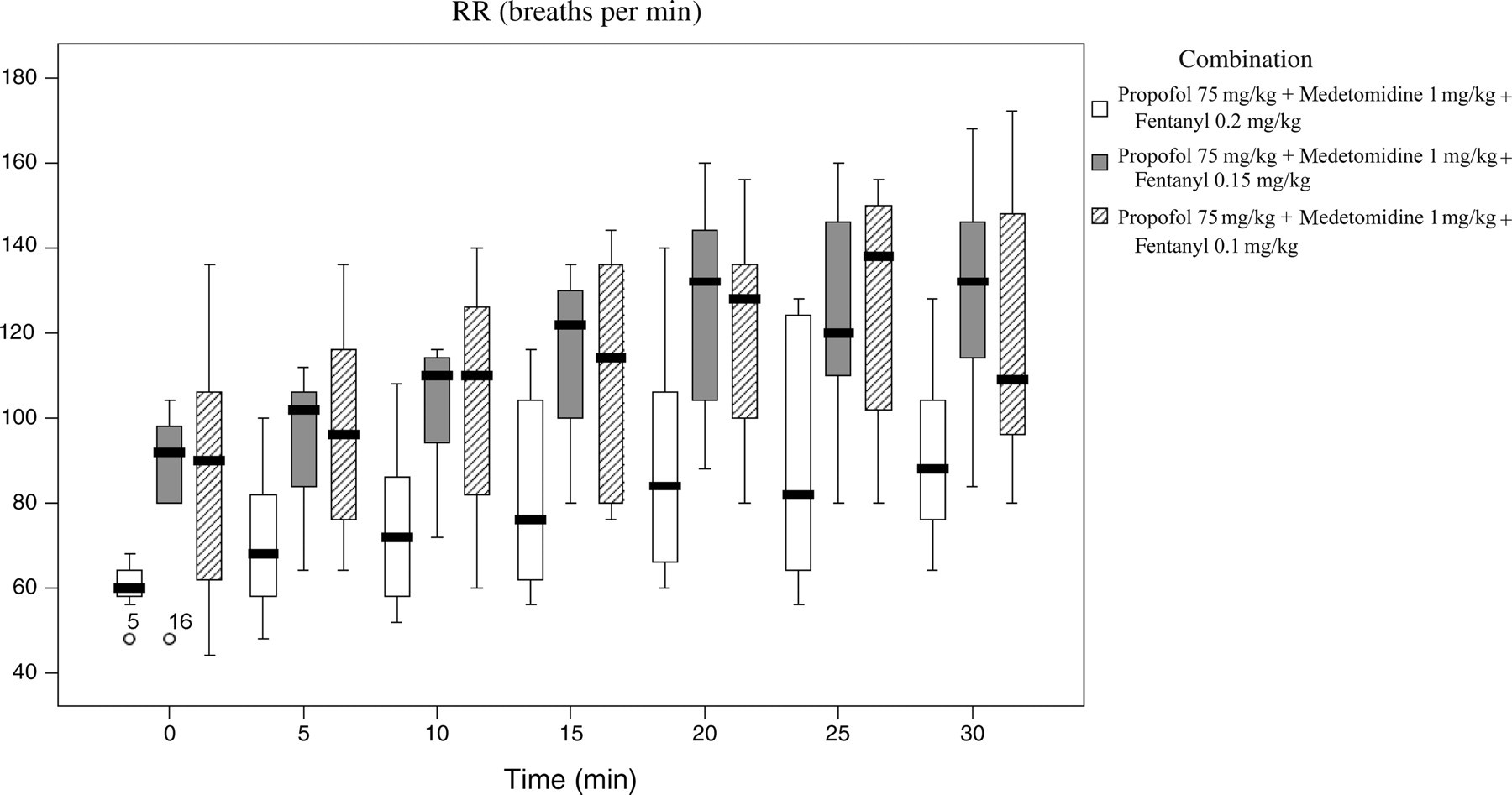
Box plots for respiratory rate (RR) measured in mice, in intervals of 5 min, anaesthetized with three different combinations of propofol, medetomidine and fentanyl
The depth of anaesthesia varied significantly from group to group at 20, 25 and 30 min (P < 0.01). The animals in group M1P75F0.2 were at a significantly deeper anaesthetic level at 20 and 25 min than the animals in groups M1P75F0.15 and M2P75F0.1 (P < 0.01) and also at 30 min when compared with group M2P75F0.1 (P < 0.01). The depth of anaesthesia in group M1P75F0.15 was also significantly lower than that of group M2P75F0.1 at 30 min (P < 0.01). In group M1P75F0.2, depth of anaesthesia increased between 0 and 5 min (P < 0.05) and between 15 and 30 min (P < 0.02). In group M1P75F0.15, an increase was observed between 10 and 15 min, 15 and 20 min and 20 and 30 min (P < 0.05). In group M2P75F0.1, a decrease was observed between 0 and 5 min (P = 0.01), and an increase observed between 5 and 20 min, 20 and 25 min and 25 and 30 min (P < 0.05).
Discussion
In this study, we evaluated the combination of propofol, medetomidine and fentanyl and its capacity to provide surgical anaesthesia when administered via the i.p. route in mice. An average anaesthetic period of 30 min, including 15 min of surgical anaesthesia was achieved with a smooth induction and a very fast, side-effect-free recovery. The use of volatile anaesthetics overcomes these problems; however, this may not be available in all facilities, and this technique has the disadvantage of providing no perioperative analgesia and concerns with gas scavenging and operating room pollution should be considered. 15
The practical aspects of drug administration in mice limit the number of protocols suitable for anaesthetizing these animals. Most protocols use combinations of dissociative agents combined with drugs such as benzodiazepines and α2-agonists administered via the i.p., intramuscular or s.c. routes. 6,16–20 In addition to the unpleasant behavioural effects associated with the administration of ketamine, 10 these protocols usually require large anaesthetic/sedative doses, leading to long recoveries and a higher risk of hypothermia and hypoxia.
Combinations with propofol and medetomidine provided predictable induction, hypnosis and muscle relaxation. Surgical anaesthesia (loss of PWR), however, was difficult to achieve as was observed in these studies. This combination provided adequate restraint for non-painful procedures for an average period of 24 min.
In this study, we observed that combining propofol with medetomidine and fentanyl, muscle relaxation and analgesia were increased, avoiding opiate-induced catalepsy 21 and providing a suitable anaesthetic combination for mice. Kanto 22 reported the hypnotic effects of propofol administration; however, we have previously demonstrated that it is difficult to induce an adequate hypnotic level in mice using propofol administrated i.p. alone or associated with fast-acting opioids. 11 The potent sedative and analgesic effect of medetomidine has been demonstrated in several species. 23–25
In the main study, the most suitable combinations of the preliminary study and a new combination M1P75F0.2 were tested in order to evaluate differences in pulse rate, haemoglobin saturation, respiratory rate and depth of anaesthesia. The choice of an extra combination with 0.2 mg/kg of fentanyl was supported by the bibliography. 8 Combination M2P75F0.15 was not used in the main study since no significant differences were observed when compared with group M1P75F0.15.
The combination of propofol, medetomidine and fentanyl administered via the i.p. route in mice provided a smooth induction with a very fast recovery. Adequate muscle relaxation and hypnosis were also observed in all dose combinations. Surgical anaesthesia could be achieved in all the groups; however, only in groups M1P75F0.15 and M2P75F0.15 the period between loss and recovery of PWR provided enough time to perform minimal surgical procedures. The PWR was lost in all the animals only in these groups. It is important to notice that i.p. administration is described as being associated with a high failure rate. In order to reduce variability due to technical problems, well-controlled injection should always be performed by the same operator. 26
The lymphangiectasis and chyloperitoneum observed at necropsy may be associated with the administration of propofol by the i.p. route. The complications observed during anaesthesia in the preliminary study were most likely due to problems relating to temperature control. The animals were anaesthetized in groups of five on the same homoeothermic blanket and their body temperature controlled accordingly to the temperature of just one animal. In the main study each animal was anaesthetized separately and temperature control was specific to that animal's body temperature in order to maintain physiological values.
A significant increase in pulse rate was observed after atipamezole administration in all the groups. No significant differences in pulse rate were observed from group to group. Some of the pharmacodynamic effects of medetomidine include hypothermia, hypotension and cardiovascular depression due to α2-agonists. 9 Transitory hypertension can usually be observed in the initial phase of medetomidine combination anaesthesias due to the effect on peripheral α2-adrenoreceptors. 27,28 The low pulse rate observed throughout the anaesthetic period is a common medetomidine effect. 29,30 Fentanyl is also associated with a decrease in blood pressure and heart rate. 31
The respiratory rate decreased significantly with the increasing fentanyl dosages from 0.15 mg/kg to 0.2 mg/kg. The respiratory depression of fentanyl on ventilation is well documented in previous studies. 8,32–35 Haemoglobin saturation values were always above 95% during anaesthesia in all the animals. Nevertheless, the administration of oxygen is recommended to avoid hypoxia.
The increase in fentanyl dosage to 0.2 mg/kg provided a more stable anaesthesia and a longer surgical period. Group M1P75F0.2 showed significantly lower values in the depth of anaesthesia after 20 min when compared with groups M1P750.15 and M2P75F0.1.
The fast recovery observed with this technique is a major benefit. A fast and smooth recovery is essential for the animal's wellbeing, experimental standardization and a decrease in postanaesthetic complications associated with hypothermia and cardiorespiratory depression.
We have used the same combination in other strains of mice (C57BL/6J and NMRI) in our laboratory. An increase on propofol dose until 100 mg/kg has been used, when the initial 75 mg did not induce RRef loss.
We conclude that, a freshly prepared combination of 75/1/0.2 mg/kg of propofol, medetomidine and fentanyl is a safe, easy and reversible technique to anaesthetize mice via the i.p. route. It provides a surgical window of 15 min and restraint for 30 min with a fast recovery.
Footnotes
ACKNOWLEDGEMENTS
Portuguese Foundation for Science and Technology funded by POCTI, co-funded by FEDER, under project POCTI/CVT/59056/2004; and Dr Sílvia Gradim for assistance with the English review.
