Abstract
Interstitial fluid (ISF) has recently garnered interest as a biological fluid that could be used as an alternate to blood for biomedical applications, diagnosis, and therapy. ISF extraction techniques are promising because they are less invasive and less painful than venipuncture. ISF is an alternative, incompletely characterized source of physiological data. Here, we describe a novel method of ISF extraction in rats, using microneedle arrays, which provides volumes of ISF that are sufficient for downstream analysis techniques such as proteomics, genomics, and extracellular vesicle purification and analysis. This method is potentially less invasive than previously reported techniques. The limited invasiveness and larger volumes of extracted ISF afforded by this microneedle-assisted ISF extraction method provide a technique that is less stressful and more humane to laboratory animals, while also allowing for a reduction in the numbers of animals needed to acquire sufficient volumes of ISF for biomedical analysis and application.
Introduction
Advances in extraction of interstitial fluid (ISF) for biomedical applications and biomarker identification have gained robust interest.1–3 ISF represents 80% of extracellular fluid, yet remains incompletely characterized due to limitations in sampling techniques. 4 ISF is in contact with blood through capillary walls and ISF analysis may have implications in diverse areas of research.5–8 Glucose concentrations in ISF correlate well with blood, leading to interest in ISF monitoring in disease.9,10 It has also been demonstrated that ISF can be used as a means of therapeutic drug monitoring.5,11 Additionally, tumor ISF is a rich reservoir of biomarkers and shows promise in elucidating markers of tumor microenvironment and metastatic potential. 6
Various sampling techniques for ISF extraction have been reported,1–3 yet these have been limited by volume and have induced tissue damage through blistering or negative pressure that may be painful to animals. Moreover, these methods do not produce adequate volumes of ISF, from single animals, for downstream analytical techniques and, thus, numerous animals are required.10,12,13 Our extraction technique overcomes these challenges by allowing for the collection of 40–60 µL of ISF from a single animal while also providing a less invasive method, compared with previous techniques, thus reducing the number of animals needed for analysis while minimizing pain and distress.
Animals, materials, and methods
The animal care and use program of the University of New Mexico (UNM) is accredited by AAALAC International and the UNM's animal care and use committee approved all experiments. A total of 21, 6–8-week-old, female CD hairless, Crl:CD-Prss8hr, rats (Charles River Laboratories, Wilmington, MA), were anesthetized with 2.0% isoflurane using a nose cone. Three rats were used for anatomical position studies and 18 for exosome isolation and volume optimization. Formal power calculations to prespecify sample size were not possible due to the preliminary nature of this study. Ultra-fine Nano PEN needles (BD, Franklin Lakes, NJ) were placed into 3D-printed microneedle array holders. Each needle was attached to a 1–5 µL calibrated pipet capillary tube (Drummond Scientific Co., Broomall, PA) (Figure 1(a)). The array assembly was then pressed into the dermal tissue of the rats (Figure 1(b)). Arrays were held in place until sufficient quantity of ISF was collected. ISF was recovered from capillary tubes, pooled, and immediately analyzed or flash frozen (Figure 1(c) and (d)). All animals had a terminal cardiac puncture performed at the conclusion of each experiment.
Microneedle array ISF extraction and downstream analysis. (a) Five-needle, 3D-printed microarray holders and fully assembled apparatus. (b) Microneedle array application in a rat and (c) collection of ISF. (d) Downstream analysis options.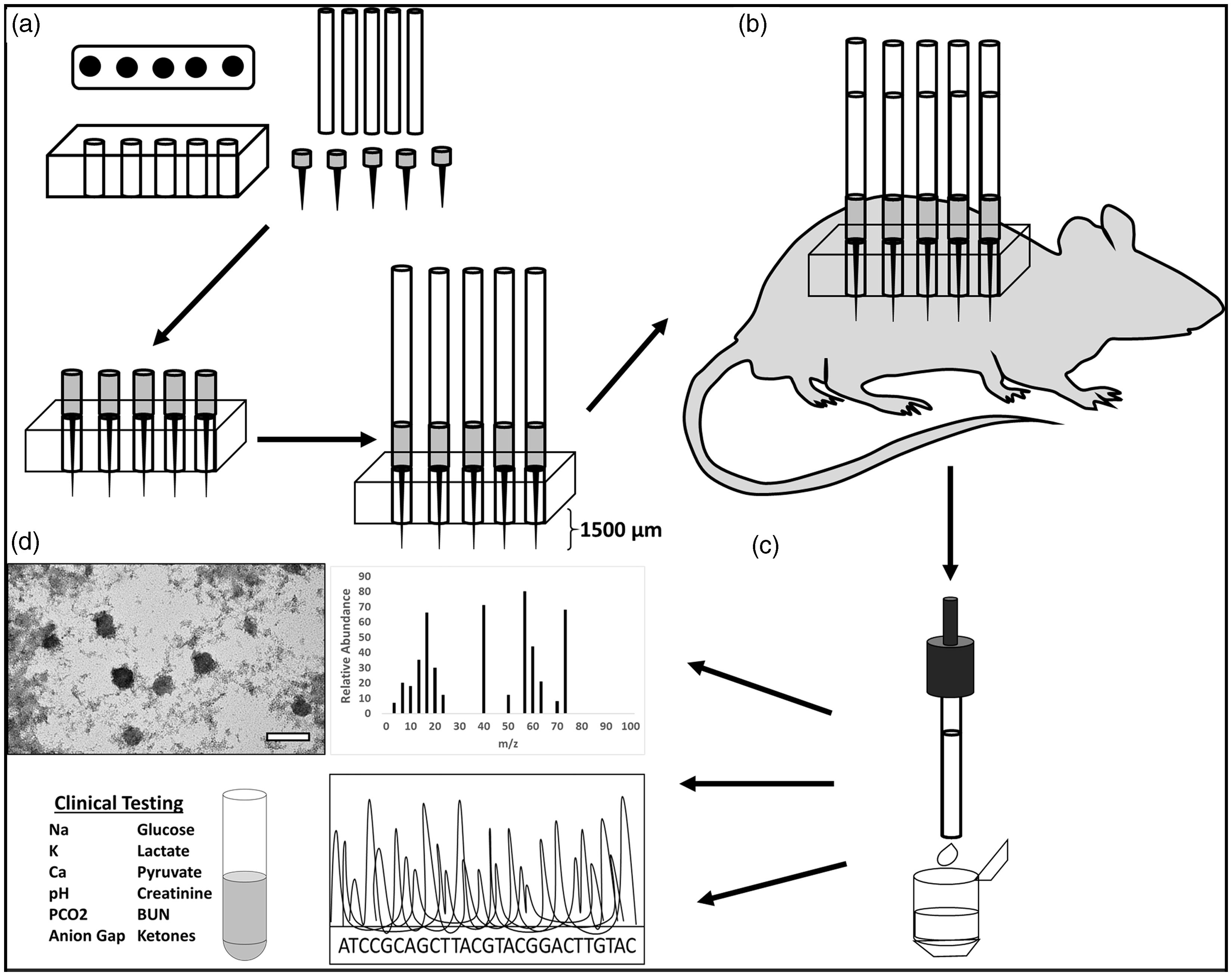
To determine optimal anatomical position, microneedle arrays were tested in the flank, abdomen, and back. We compared extraction when needle bevels were either vertically facing up or down, as well as when needles were removed or not removed between insertions. Arrays were left in place 4 min per insertion, removed, and ISF in each tube was measured using a 20 µL pipette. Extractions were repeated four times.
Exosomes were purified using a Total Isolation Kit from Other Fluids (Invitrogen, Carlsbad, CA). For transmission electron microscopy (TEM), 5 µL of exosome suspension was applied to a carbon-coated grid and stained with 4% uranyl acetate. Exosomes were visualized using a Hitachi 7700 TEM with an 80 kV acceleration voltage.
Discussion
Previous methods of ISF extraction are limited by volume and induce tissue damage.1–3 These methods rely on suction or blistering and extract small volumes of ISF (<10 µL).10,12,13 Our microneedle-array technique overcomes these challenges by allowing for larger volumes of ISF while also providing a less invasive method. 3D-printed microneedle array holders precisely control insertion depth by allowing the microneedles to only insert into the dermis to a pre-specified depth (1360 ± 210 µm) (Figure 1). Hairless rats were used to ensure hair did not contaminate the samples. ISF typically rose up the capillary within 1–2 min. The abdomen produced highest volumes of ISF (Figure 2(a) and (b)). In the bevel down position, both rate of extraction (0.56 ± 0.33 µL/min) and volume extracted per insertion (2.2 ± 1.3 µL) were significantly greater (p = 0.0005), compared to the bevel up position (0.1 ± 0.2 µL and 0.03 ± 004 µL/min, respectively). We also measured extraction when needles were exchanged or not exchanged between the replicates. A non-significant (p = 0.75) increase was achieved when needles were not exchanged. We extracted 42.5 ± 7.1 µL of ISF from single rats. We purified exosomes (Figure 2(c)) and performed genomic and proteomic analysis (data not shown) with ISF from these rats. This technique could be further optimized by increasing array density and adjusting microneedle length for species.
Optimal ISF extraction methods and exosome isolation. ISF volume per insertion (a) and rate of extraction (b) for the various anatomical positions in the bevel up and down configurations. (c) TEM image of exosomes isolated from rat ISF.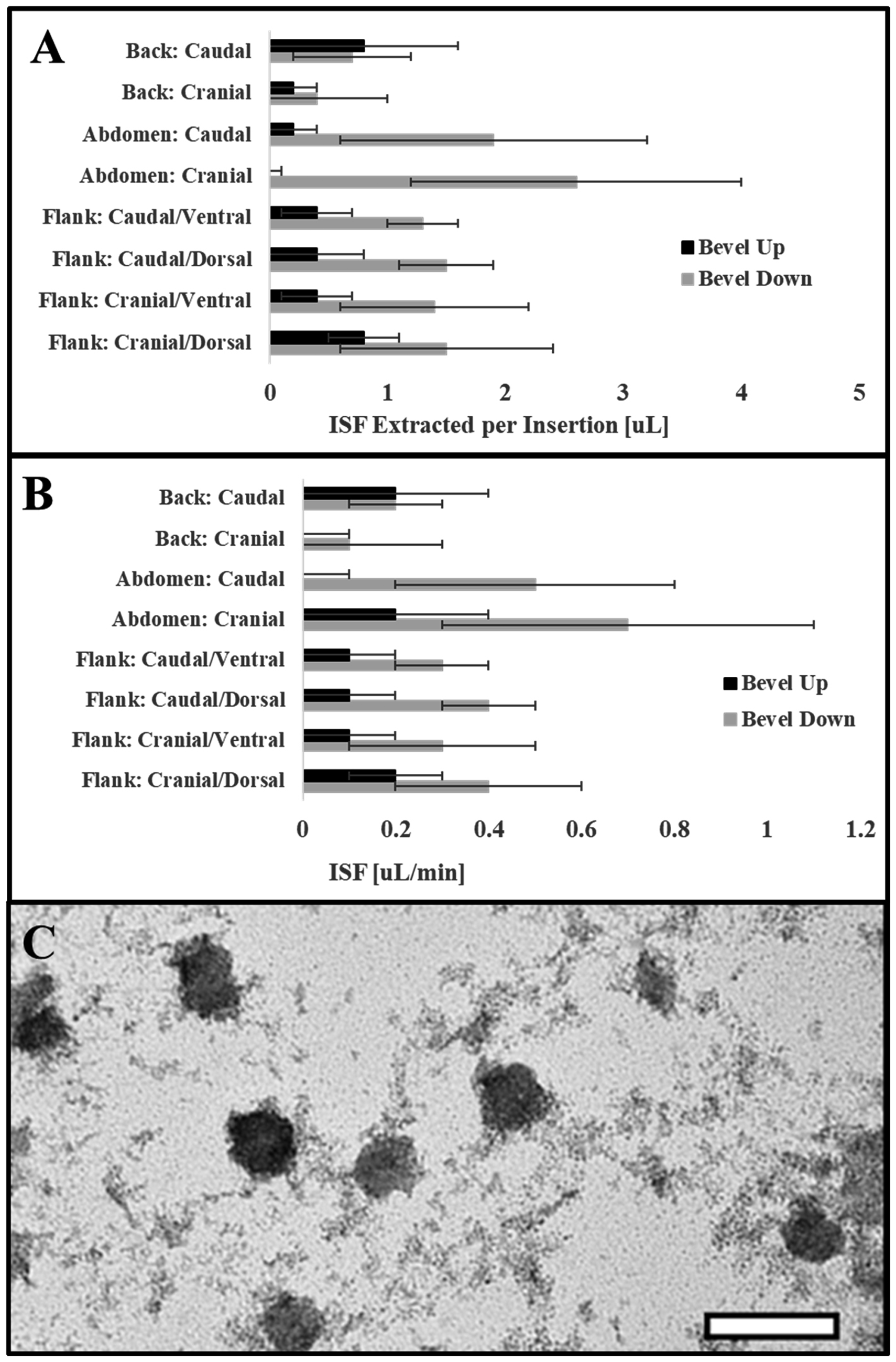
This minimally-invasive, microneedle array technique provides an effective way of collecting larger volumes of ISF than previously reported and reduces the number of animals needed for downstream analysis. It offers exciting new research avenues using ISF for mechanistic biomedical studies and biomarker identification.
Footnotes
Acknowledgments
Sandia National Laboratories is a multimission laboratory managed and operated by National Technology and Engineering Solutions of Sandia LLC, a wholly owned subsidiary of Honeywell International Inc. for the US Department of Energy’s National Nuclear Security Administration under contract DE-NA0003525. The authors thank Victor H Chavez of Sandia National Laboratories for assistance with printing microneedle array holders.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was made possible by funding from the Defense Threat Reduction Agency–Joint Science and Technology Office for Chemical and Biological Defense: Project 189920 to Sandia National Laboratory. Doctors Baca and Taylor received infrastructure support from NIH CTSC (grant number UL1TR00449).
Notes
Conclusions and opinions presented here are those of the authors and are not the official policy of the US Government.
