Abstract
Tissue fixation methods are well established for rodents, but not for large animals. We present a simple technique for in situ brain perfusion fixation in a male porcine model, using cervical vessels for inflow and outflow and achieving a closed system. Thirty-four pigs, aged 4.7 ± 0.6 months and weighing 60.7 ± 10.9 kg, were anaesthetised and mechanically ventilated. The ipsilateral common carotid artery and external jugular vein were dissected and constituted the inflow and outflow access, respectively. The brains were perfused and fixed in situ with heparinised saline followed by buffered formaldehyde. Then, specimens (brain, cerebellum and brainstem) were extracted and processed for histology. Fixative fluid leakage was avoided, achieving a closed system. This technique minimises the exposure to toxic chemicals such as formaldehyde and associated hazards (inherent toxicity, eye irritation), thereby increasing operators’ safety. Perfusion was performed with a peristaltic pump for 20–30 minutes at an optimum rate of 0.20 l/min and required only 5 litres of the fixative. The specimens were sufficiently hardened to be extracted. High-quality tissues were available for histology analysis. This technique offers a user-friendly closed system for brain perfusion fixation which can be adapted for other tissues of the head, face and neck.
Introduction
The porcine model is one of the best amongst the non-primate large animals. The pig’s brain is comparable to the human brain with regards to the overall neuro-anatomy (gyral pattern, grey/white distribution) and its growth pattern.1,2 Moreover, the brain size and its biomaterial properties are suitable for computer modelling and simulation.2,3
Fixation perfusion is the gold method for studying the central nervous system and the peripheral nerves. The fixative agent penetrates the tissues, ensuring a better fixation in addition to maintaining the original geometry of soft tissues and limiting distortion while sampling.4,5 Although protocols of perfusion fixation are well established for rodents, particularly whole-body perfusion fixation via transcardial access, 6 fixation methods for large animals are yet to be established.
Whole-body perfusion fixation in larger animals via transcardial access has several limitations. Firstly, this technique requires massive fluid volumes. Furthermore, it is performed using an open system.7–9 Most fixative agents are known for their toxicity, and ordinary ventilating systems, such as extractor hoods, are not adapted to the size of large animals. In addition, setting a vascular access requires a heavy surgical setup and a well-experienced operator. 5
Therefore, we present a simplified technique of in situ brain perfusion fixation in a male porcine model, using the common carotid artery and the external jugular vein as inflow and outflow, respectively, and achieving a closed system.
Thirty-four castrated male domestic pigs, with an average age of 4.7 ± 0.6 months and average weight of 60.7 ± 10.9 kg, were used. These were Landrace, conventional and not genetically modified pigs, supplied by the Centre for Animal Husbandry, KU Leuven, Belgium. All the animals were delivered by trucks adapted for large animal transportation at least five days before the beginning of the experiments.
The pigs were housed in individual pens of 2 m2 with concrete floors and wood shavings as bedding material in an open system (no special microbiological protective measures). All cages had a hanging toy. The following parameters were checked and documented daily. Firstly, 14-hour light/10-hour dark cycle from 7:00 am to 9:00 pm. Secondly, temperature was maintained at 22 ± 2℃. Thirdly, humidity ranged between 45% and 70% but could vary with weather conditions, especially in winter. All animals had free access to tap water through an automated drinking system. The pigs received Optivo Pro 9041® (AVEVE, Leuven, Belgium) pellets twice daily.
Our study was approved by the Ethical Committee for Animal Experimentation, KU Leuven (P160/2014, since 1 September 2014) and all the animal experiments were carried out at the Laboratory Animal Centre, KU Leuven, Belgium.
General anaesthesia was induced with intramuscular 20% lidocaine (0.05–0.3 mg/kg) and tiletamine and zolazepam (7–10 mg/kg), and then maintained with a continuous perfusion of intravenous (iv) 20% propofol at 15 ml/min via the auricular veins. Subsequently, the animals were intubated and mechanically ventilated (50% oxygen, tidal volume: 600 ml, frequency: 16/min) to maintain peripheral oxygen saturation above 90% and end-tidal carbon dioxide below 40 mm Hg. Analgesia was achieved with iv buprenorphine (10–20 µg/kg).
The pigs were positioned in dorsal recumbency and their necks set in extension. Cervical vessels were dissected to set the perfusion connection as illustrated in Figure 1(a)–(e). Two transparent extension tubes were used as cannulae (diameter = 3 mm, length = 150 cm, volume = 1.18 ml). The external jugular vein
10
(5 cm below the fork at the junction of the linguofacial vein and maxillary vein) was used for the outflow as illustrated in Figure 1(b). The liquids were drained by gravity into 5 litre plastic containers set on the floor (Figure 1(e)). The common carotid artery,
10
running deeper along the trachea with the internal jugular vein and the vagus nerve branch (Figure 1(a)), was used as inflow and was connected to a peristaltic pump (Figure 1(d)). The perfusion tubes were inserted up to a length of 4–5 cm inside the vessels and restricted by surgical strands (2-0 Mersilene® Ethicon, Somerville, NJ, USA) as shown in Figure 1(c).
(a)–(e) Closed system of perfusion developed in-house – fixation for tissues of the head (brain and face) using cervical neck vessels by means of a unilateral perfusion. (a) The common carotid artery (arrow down) with the internal jugular vein and the vagus nerve. (b) The external jugular vein (arrow up). (c) Perfusion connections: common carotid artery as inflow and external jugular vein as outflow. (d) Inflow sustained by a pump. (e) Liquid drainage into a plastic container. PFA: phosphate-buffered formaldehyde; NaCl: sodium chloride.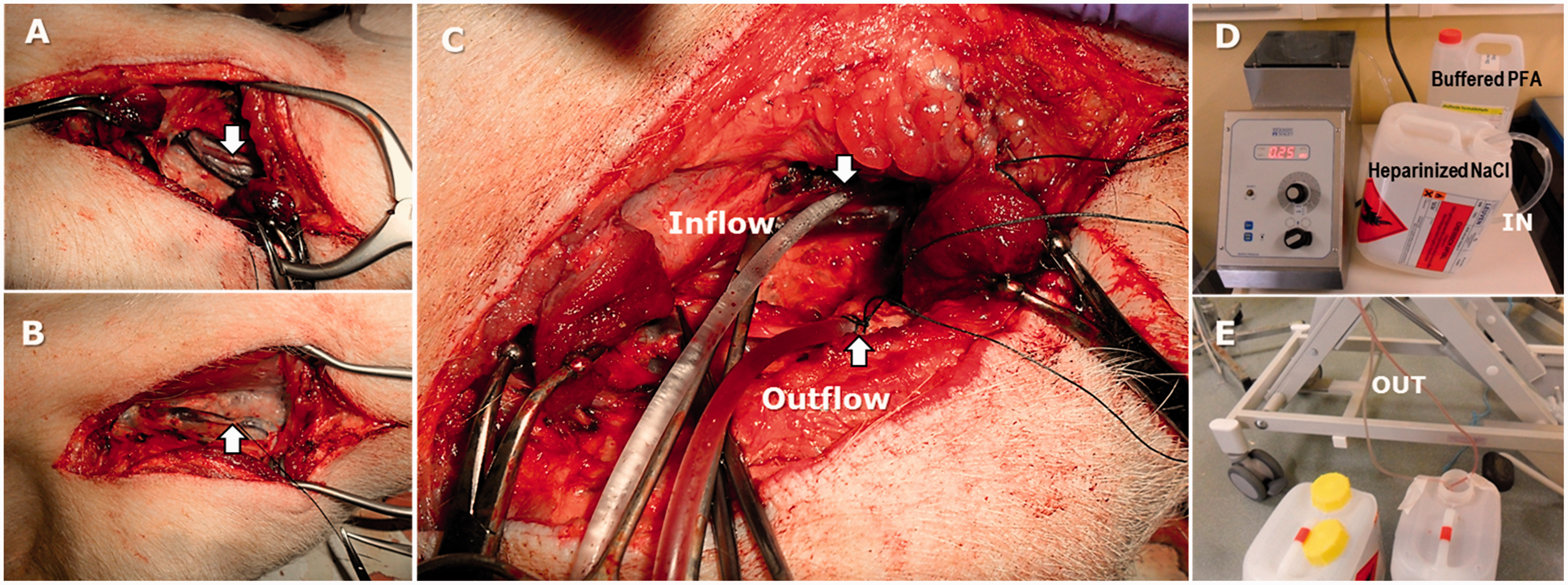
Following this, iv heparin (10,000 IU) was administered and the animals were euthanised with an excess of barbiturates. Death was confirmed after the oxygen saturation and end-tidal carbon dioxide curves flattened on the monitoring device. The brain was immediately perfused with a rinsing solution of 3 litres of heparinised saline and then fixed in situ with 5 litres of 4% solution of phosphate-buffered formaldehyde (neutral pH) at 0.20 l/min (Figure 1(b)). Next, the inflow and outflow tubes were clamped to let formaldehyde penetrate the tissues. The pig head was then cut off. All the muscles and connective tissue were removed and the skull was carefully sawed allowing the specimen (brain, cerebellum and brainstem) to be delicately removed along with the meningeal coverings (dura and pia mater). Finally, the brain tissues were processed for histology analysis.
The pigs were perfused successfully in situ and good quality brain fixation was obtained in a reproducible way.
Leakage of fixative fluid was avoided by properly connecting the perfusion tubes to the vessels of the neck, thereby achieving a nearly closed system (Figure 1(a)–(e)). Because the system was powered by a peristaltic pump, a relatively rapid perfusion at an optimum rate of 0.20 l/min could be performed. This technique minimises exposure to chemical substances such as formaldehyde and health hazards (inherent toxicity, eye irritation) associated with it, thereby increasing operators’ safety.
A good perfusion was indicated by homogeneous brain tissue firmness and a change of colour from orange to yellow.
4
Indeed, the brain tissues were hardened well enough after the in situ perfusion fixation to allow the required manipulation during brain extraction (Figure 2(a)–(d)). Thus, high-quality brain tissues were available for histology analysis as illustrated in Figure 2(e)–(h).
(a)–(h) High-quality histology material produced after in situ perfusion fixation. (a) Frontal, parietal, temporal and occipital bone pieces removed, exposing a fixed brain covered by dura mater. (b) Specimen removed from the skull. (c) and (d) Pig brain tissues fixed and processed for paraffin embedding. (e) and (f) Fronto-parietal sample of porcine dura mater. (g) and (h) Frontal cerebral cortex. Magnification: (e) and (g): × 12.5; (f) and (h): × 400.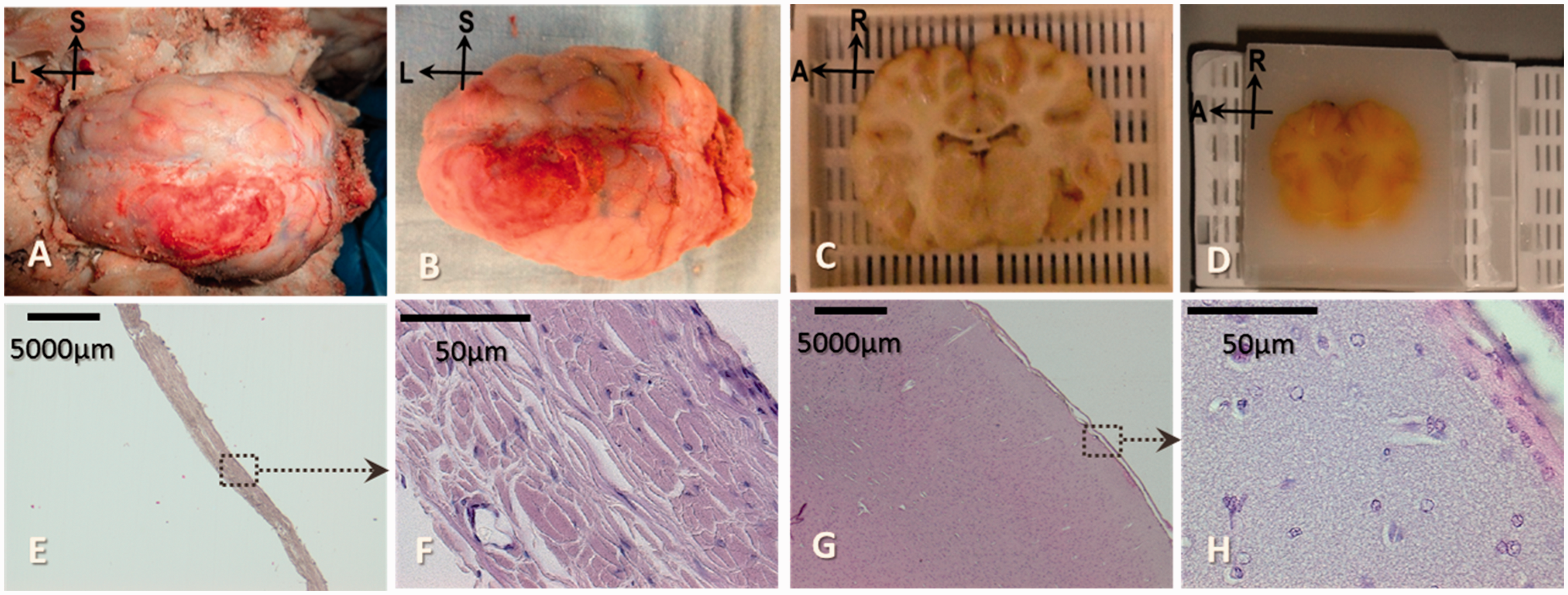
We have presented a simplified and user-friendly closed system in a porcine model for large-animal brain perfusion fixation. To the best of our knowledge, this is the first time that successful in situ brain perfusion fixation has been reported in a porcine model using the unilateral common carotid artery and external jugular vein. So far, perfusion fixation in large animals has been performed with a bilateral vascular access and massive fixative volumes.7–9
Following a skin incision, 10 the targeted vessels can be quickly found, dissected and cannulated, allowing for a fast procedure. Moreover, this closed system increases the safety of the operators handling toxic fixative solutions. Lastly, this technique has the potential to be used for brain fixation of other large animals.
Mathematical and computer models offer a valuable alternative to animal experiments (in silico approach). Once a mathematical model is validated, computer simulation not only allows significant reduction in the number of animals used in experimentation but also speeds up what can be performed in the lab. However, the accuracy of such models depends on the data used as input. 11 The ultimate goal of our work is to provide detailed histology data that can be implemented into such computer models.
Footnotes
Acknowledgements
We would like to thank the Laboratory Animal Centre, KU Leuven, Leuven, Belgium, as the animals were housed and taken care of by their staff during the experiments. Next, we highly appreciate the support provided by the Departments of Pathology and Forensic Medicine, Leuven University Hospitals. Especially, we acknowledge Dr Wim Develter for sharing his precious expertise in handling brain autopsy. We are also grateful to Sijn Massart and all his colleagues at the Centre for Animal Husbandry in Lovenjoel for their collaboration in breeding, choosing and delivering the animals. Lastly, Marc Verbeek, from the Medical Technical Services at Leuven University Hospitals, has also been of great help in adapting some of the devices we needed to process the pig brain histology.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This technique was developed for a multidisciplinary project supported by the Fonds voor Wetenschappelijk Onderzoek – Vlaanderen (Research Foundation-Flanders) grant number: G.0C6713.N) and by the KU Leuven (grant number: OT/12/071).
Contributions
GUM: conception and design, acquisition, analysis and interpretation of data, drafting and reviewing the article, final approval. SDV: conception and design, reviewing the article, final approval. RS: analysis and interpretation of data, reviewing the article, final approval. EV: conception and design, final approval; and BD: conception, final approval.
