Abstract
The existing animal models used for the simulation of acute gastrointestinal bleedings are usually non-survival models. We developed and evaluated a new porcine model (domestic pig, German Landrace) in which the animal remains alive and survives the artificial bleeding without any cardiovascular impairment. This consists of a bleeding catheter which is implanted into the stomach, then subcutaneously tunnelled from the abdomen to the neck where it is exteriorized and fixed with sutures. Using the injection of porcine blood, controllable and reproducible acute upper gastrointestinal bleeding can be simulated while maintaining normal gastrointestinal motility and physiology. Depending on the volume of blood applied through the gastric catheter, the bleeding intensity can be varied from traces of blood to a massive haemorrhage. This porcine model could be valuable, e.g. for testing the efficacy of new bleeding diagnostics in large animals before human use.
An acute upper gastrointestinal bleeding is a life-threatening condition and remains one of the most common reasons for urgent hospital admission. Major haemorrhage occurs when arteries of a larger diameter are involved. A dependable animal model for controllable bleeding is mandatory for research, technical development, and medical training purposes. It should be able to simulate recurrent bleedings in the stomach of variable intensity while normal gastrointestinal motility is maintained. Unfortunately, no live model which meets these needs has been described in the literature so far. There are only non-survival porcine models for gastrointestinal bleedings which produce spurting bleedings, e.g. by surgical implantation of gastric arteries into the stomach.1–3
We adapted the technique used in the ex vivo Erlangen Active Simulator for Interventional Endoscopy model (EASIE model) and transferred it to a porcine survival model. In the EASIE model an infusion catheter is tunnelled into the stomach wall and connected to a flow regulator which diffuses artificially stained liquids to mimic arterial bleeding. 4 We used the same gastric implantation technique with a bleeding catheter which was further subcutaneously tunnelled from the abdomen to the neck of the animal where the end of the catheter was exteriorized and fixed with sutures. The bleeding catheter enabled the injection of blood into the stomach to simulate an acute onset of gastrointestinal bleeding. The pig remained alive and survived the artificial bleeding without cardiovascular impairment.
In the following we describe the surgical implantation technique of the bleeding catheter as well as the experience gained with this new model.
Local ethical committee approval was obtained for the experimental trial. The study, including animal husbandry, was conducted at the Experimental Surgical Unit at the University of Tübingen, Germany.
The procedure was carried out on 10 healthy animals (domestic pigs, German Landrace, female, 3–4 months of age, mean weight 42 kg [range 34–48 kg]). The preoperative health status of the pigs was determined from the physical examination by the responsible veterinarian in the surgical facility. They were allowed a 10-day acclimatization period before surgery.
General anaesthesia was induced by intramuscular injection of atropine (0.05 mg/kg) and azaperone (4.0 mg/kg) followed by midazolam (1.0 mg/kg) and ketamine (14 mg/kg). The animals were then endotracheally intubated and connected to an anaesthetic machine – mechanical ventilator. Anaesthesia was maintained by the administration of isoflurane (0.8–2.0%) and propofol (2.0–5.0 mg/kg), supplemented by fentanyl (30–100 µg/kg/h).
For the operation, the animals were positioned in dorsal recumbency. First, a suitable position in the stomach for implantation of the bleeding catheter (silicon tube, inner diameter 1.0 mm, wall strength 0.6 mm) was identified via an endoscopy. Once a good position was found, it was marked endoscopically by a coagulation spot using a high-frequency (HF) probe inserted through the working channel of the endoscope. A 10 cm upper midline laparotomy was made. Using a diaphanoscope, the stomach wall was incised at the previously defined location. Approximately 2 cm of the catheter tip was inserted into the stomach (Figure 1). A purse-string suture was placed and tied around the catheter at the gastrotomy site. Using the Witzel gastrostomy technique, a seromuscular tunnel of 4–6 cm in length was created, embedding the catheter by interrupted sutures, to minimize leakage and dislocation. To avoid any accidental or incidental removal of the catheter by the animal, an exit site at the posterior neck was chosen. Therefore, the subcutaneous layer of the skin was tunnelled stepwise from the abdomen to the neck of the pig where the end of the catheter was exteriorized and fixed with sutures. The midline laparotomy was then closed. The patency of the catheter was tested by injection of 10 mL of sodium chloride solution, monitored using an endoscope. Implantation of the catheter into the stomach was performed without problems in any of the animals. The mean duration of surgery was 137 min (range 75–220 min).
(a) Gastric implantation of the catheter. (b) Endoscopic view of the inserted tip of the bleeding catheter (arrow). (c) Seromuscular tunnelling of the catheter (Witzel gastrostomy technique) to minimize leakage and dislocation. (d) Stepwise subcutaneous tunnelling of the catheter from the abdomen to the posterior neck. (e) Exteriorization and fixation of the end of the catheter with sutures at the neck of the animal. (f) Patency testing of the catheter by injection of sodium chloride solution monitored using an endoscope.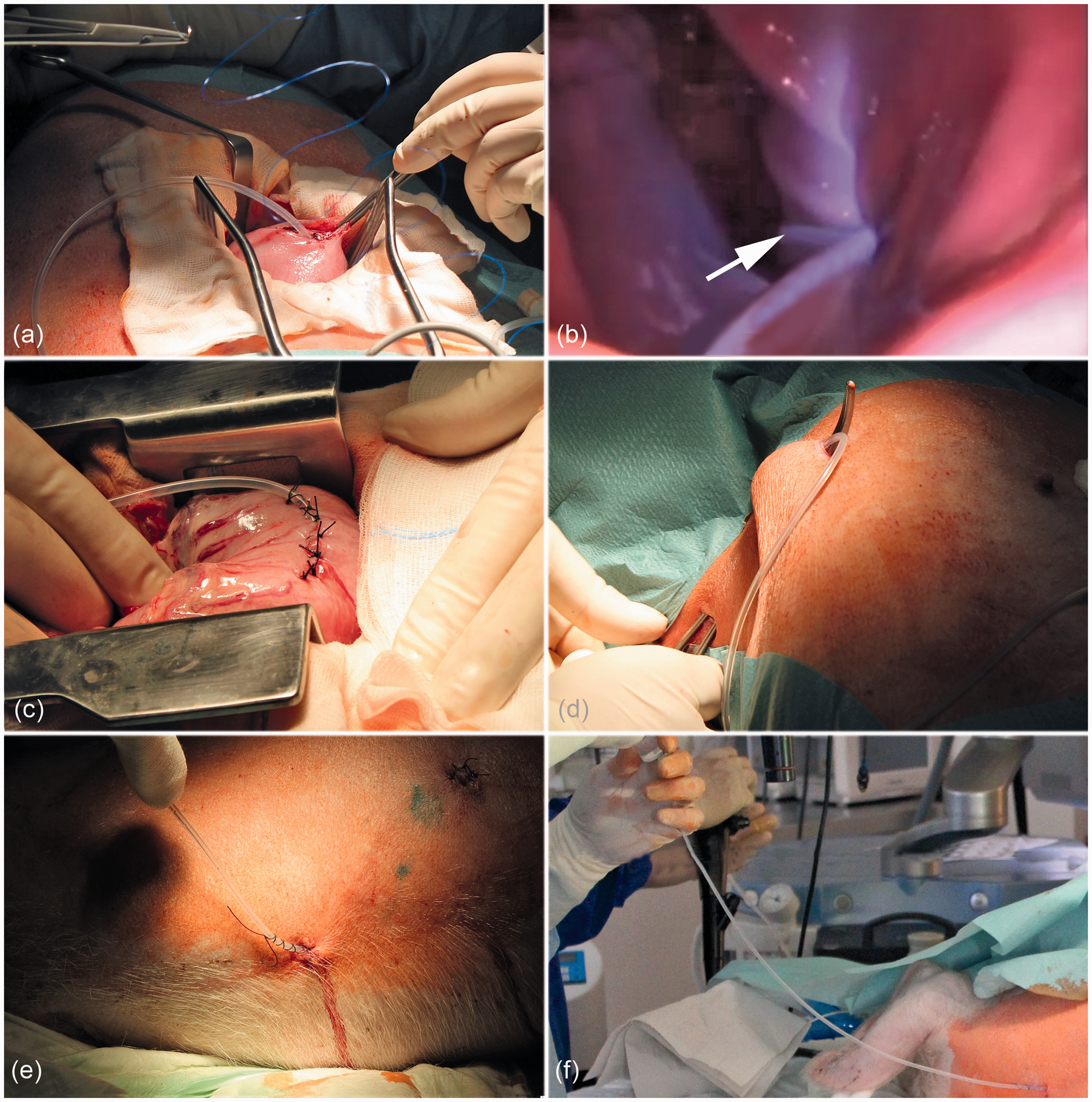
After surgery, the animals were kept in a specific pathogen free (SPF) facility. They were housed individually in pens with a minimum of 13 m2 of space. The bedding material was replaced by rubber mats to prevent the pigs from eating the litter as this would have interfered with the experiment. Lights were on a 12:12 h light–dark cycle, room temperature was 19.5℃, and air humidity was 61% (rH). The animals were monitored and examined twice a day by the responsible veterinarian in the surgical facility over an observation period of 14 postoperative days. The surgical wounds at the abdomen, flank and posterior neck healed without any complications in all cases. There were no signs of local or systemic infection or wound healing problems. The animals had free access to drinking water and were fed a custom-mixed high calorie semi-liquid diet. The mean daily weight gain was 0.59 kg (range 0.28–1.21 kg) and the final mean weight after 14 days was 51 kg (range 41–59 kg).
During the observation period week, medium and heavy bleeding with a corresponding injection speed of 12 mL/h, 60 mL/h and 300 mL/h, respectively, was simulated using either an infusion pump or a standard syringe filled with porcine blood (Figure 2). Bleeding simulations were performed twice per day (in the morning and in the evening) for 60 min each over 13 days, starting the day after the surgery. Bleeding simulations were conducted for the testing of a new telemetric sensor system for real-time detection of gastrointestinal bleeding.
5
Simulation of gastrointestinal bleeding by injection of blood through the bleeding catheter using a standard syringe.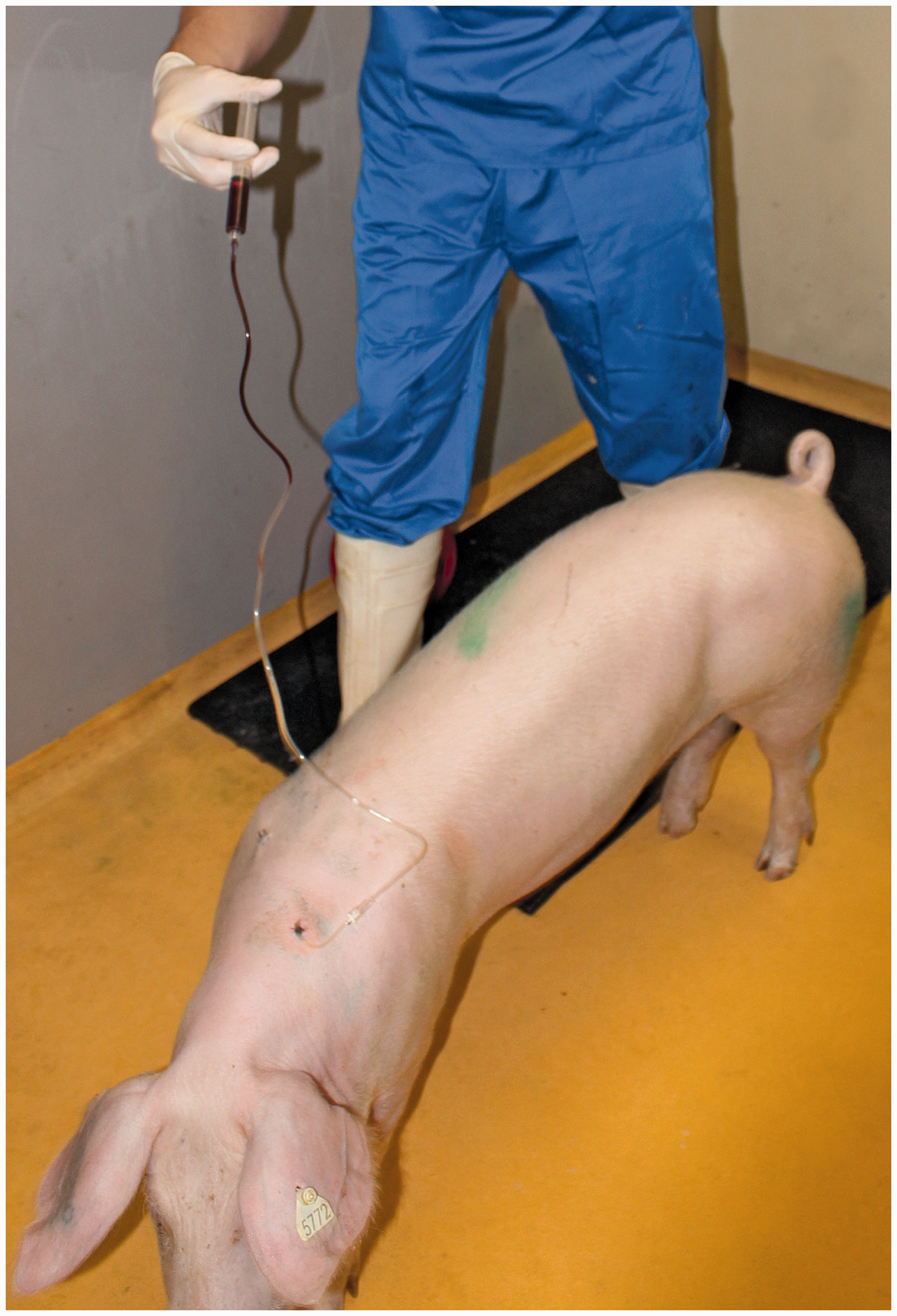
At the end of the observation period, the situation of the bleeding catheter was assessed by relaparotomy of the sacrificed animals.
In four of the 10 animals problems with the bleeding catheter were observed. In three animals the tip of the catheter had slipped out of the stomach and dislocated into the abdominal cavity, presumably due to the fast growth of these animals (20–26% of weight gain in two weeks). In one case the catheter became occluded with clotted blood. However, in only two of these cases, both with a dislocated bleeding catheter, the simulation of bleeding had to be stopped early (postoperative days 9 and 12, respectively) as the other two events occurred on postoperative day 13. Overall, 228 bleeding simulations were performed in all 10 animals and all animals could be observed over the entire period of 14 postoperative days.
In one of the three animals in which the accidental dislocation of the bleeding catheter had occurred, the catheter had caused large adhesions of the left hepatic lobe to the abdominal wall and stomach without life-threatening consequences. No intra-abdominal abnormalities were seen in the other two animals as the wounds in the stomach had already healed.
As a consequence, the catheter was implanted with a loop in the abdominal cavity in all the remaining animals to compensate for the relative shortening of the catheter due to animal growth and to prevent the problem of catheter dislocation. The loop was placed between the liver and the chest wall to avoid interference with the bowel.
In conclusion, we developed and evaluated a new porcine model use for the simulation of controllable acute upper gastrointestinal bleeding. The survival model is feasible and is useful for creating reproducible artificial bleeding while maintaining normal gastrointestinal motility and physiology. Depending on the volume of blood applied through the gastric catheter, the bleeding intensity can be varied, from traces of blood to a massive haemorrhage. This porcine model could be valuable, e.g. for testing the efficacy of new bleeding diagnostics in large animals before human use. Beyond the simulation of bleedings, the implanted catheter allows the experimental application of other fluids into the upper gastrointestinal tract under defined circumstances.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
