Abstract
The streptozotocin (STZ)-induced diabetic mouse is a widely used model of diabetes and diabetic nephropathy (DN). However, it is a well-known issue that this model is challenged by high weight loss, which despite supportive measures often results in high euthanization rates. To overcome these issues, we hypothesized that supplementing STZ-induced diabetic mice with water-softened chow in addition to normal chow would reduce weight loss, lower the need for supportive treatment, and reduce the number of mice reaching the humane endpoint of 20% weight loss. In a 15 week STZ-induced DN study we demonstrated that diabetic male mice receiving softened chow had reduced acute weight loss following STZ treatment (p = 0.045) and additionally fewer mice were euthanized due to weight loss. By supplementing the diabetic mice with softened chow, no mice reached 20% weight loss whereas 37.5% of the mice without this supplement reached this humane endpoint (p = 0.0027). Excretion of corticosterone metabolites in faeces was reduced in diabetic mice on softened chow (p = 0.0007), suggesting lower levels of general stress. Finally, it was demonstrated that the water-softened chow supplement did not significantly affect the induction of key disease parameters, i.e. %HbA1C and albuminuria nor result in abnormal teeth wear. In conclusion, supplementation of softened food is refining the STZ-induced diabetic mouse model significantly by reducing stress, weight loss and the number of animals sacrificed due to humane endpoints, while maintaining the key phenotypes of diabetes and nephropathy.
Introduction
The streptozotocin (STZ)-induced diabetic mouse has for many years been one of the most used models in both diabetes and diabetic nephropathy (DN) research.1,2
STZ, which was originally discovered as an antibiotic, 3 was further developed in oncology as a chemotherapeutic agent against pancreatic islet cell cancer. 4 Since the discovery of the diabetogenic effect of STZ in the 1960s, 5 the STZ-induced diabetes model has been a widely used rodent model in both diabetes and DN research. 6 STZ is a glucose analogue which is transported into pancreatic beta cells via GLUT2 glucose transporters where it accumulates and triggers necrosis.7,8 The STZ-induced hyperglycaemia eventually induces the development of kidney damage resembling the early pathological changes in human DN manifested as albuminuria and glomerular mesangial expansion. 9
STZ-induced diabetes is typically induced in male mice and rats, since males are more sensitive to STZ than females.10–13 The susceptibility for STZ in mice is also affected by the mouse strain due to differences in GLUT2 expression. 12
The main advantage of the STZ-induced diabetes model is the cost effectiveness and the simple technical requirements needed to induce stable diabetes and signs of nephropathy in mice. However, despite its extensive use, STZ-induced diabetes is variable and no generally accepted standardized protocol exists regarding mouse strain, STZ administration, health monitoring or supportive treatment. One of the main issues with STZ-induced diabetes in mice is that the mice often suffer significant and acute weight loss resulting in high euthanization rates.9,14–16 This weight loss is likely to be the result of the acute onset and progression of diabetes, but the acute weight loss might also be caused both by the repeated fasting of the animals and the acute toxicity of STZ.14,17 In addition to being toxic to beta-cells, STZ has been found to have a direct toxic effect especially on the kidneys and other organs such as the liver and intestines.18,20–22 Originally, a single high dose of STZ (in mice 150–200 mg/kg) was used to induce diabetes. With this high dose, the STZ induces a direct cytotoxic effect on the β-cells, leading to fast induction of severe diabetes with undetectable levels of insulin. 23 But the toxic effect on especially the kidney is also more pronounced with this high dose of STZ.9,19
To overcome the acute toxicity of STZ, the Diabetic Complications Consortium (DiaComp) recommends multiple low doses of STZ, 5 × 50 mg/kg given on five consecutive days,24,25 resulting in a slower progression of hyperglycaemia driven by apoptosis of the β-cells.26,27 In this study, we use a protocol which we refer to as double intermediate dose, where we inject the mice with 100 mg/kg STZ on day 1 and 4, as previously published by Meier et al. 28 We have found this protocol to have the same consistency in induction of diabetes and similar or less loss in body weight compared with 5 × 50 mg/kg (unpublished data) and moreover this protocol has the advantage that the animals are only fasted and injected with STZ twice.
The high weight loss of the STZ-treated mice affects the overall animal welfare and requires larger group sizes to ensure appropriate power and extensive health monitoring throughout the study period. However, many reports do not account for weight loss and the loss of animals making the extent of this problem difficult to determine. It is nevertheless certain that there is a need for refining this animal model so fewer mice reach significant weight loss. Thus, initiatives to increase the animal welfare, reduce the number of animals required in studies as well as potentially improve the quality of the outcome should be pursued.
The aim of this study was to investigate if supplementing STZ-induced diabetic male mice with water-softened chow in addition to their regular chow pellets could result in (1) reduced acute weight loss, (2) reduced euthanization rate due to humane endpoints, (3) lower need for supportive treatment e.g. insulin injections, and (4) reduced stress level in the mice. Finally, it was investigated if the water-softened chow supplement would significantly affect the induction of diabetes and DN.
Materials and methods
Ethics statement
The study was approved by The Animal Experiments Inspectorate, Ministry of Environment and Food of Denmark (license no. 2014/15-0201-00429). Any mice in this experiment that showed signs of compromised health (inadequate activity/moribund) or had lost more than 20% body weight was terminated immediately according to the EU Directive 2010/63/EU on the protection of animals used for scientific purposes.
Mice
Fifty-five 129S2/SvPasCrl male mice (6–8 weeks old, min. 21 g) were purchased from Charles River, Germany. The mice were housed in groups of 6–10 mice in type IV open cages (595 mm × 380 mm ×200 mm) from Scanbur, Karlslunde, Denmark, located in cabinets (NOVOtainer, Scanbur, Karlslunde, Denmark) with up to 16 cages in each. The mice had free access to tap water (from the Scantainer’s watering system as well as extra water bottles in cage lid) and to standard mouse chow pellets (Altromin 1324; Brogaarden ApS, Lynge, Denmark). Furthermore, as enrichment they were given paper nesting material, wooden blocks, a cardboard tube (31008, 30968, 31005, Brogaarden ApS, Lynge, Denmark), a plastic hide (special order, Bach Ventilation ApS, Søborg, Denmark), and a hemp rope (4101022006, Fyns Kran Udstyr A/S, Vallensbæk Strand, Denmark) hanging from the cage lid. The mice were kept in a room with 25℃, 30–60% relative humidity, and a 12 h day/night cycle. Ear punches were used as an identification method. Before study start the mice were acclimatized for 2 weeks.
Experimental design
The mice were allocated to either STZ or vehicle groups and further divided into groups receiving either normal chow pellets or both chow pellets and water-softened non-pelleted chow (Altromin 1324, Brogaarden ApS, Lynge, Denmark).
At study week 6 the mice receiving the softened food (both non-diabetic and diabetic) were further divided into two groups, being housed either with or without red plastic igloos (Mouse Igloo, K3327, Bio-Serv, Flemington, USA) during the metabolic cage housing described below. The rationale for this was to test if giving the mice a hide in the metabolic cages could reduce weight loss or the stress hormone response seen when housing mice in metabolic cages. Since we found no difference for either of these readouts, data from the two groups was merged in all analysis and this test was not repeated at week 14 (see Supplementary Figures 1 and 2 online). All the mice only receiving pellets were given an igloo.
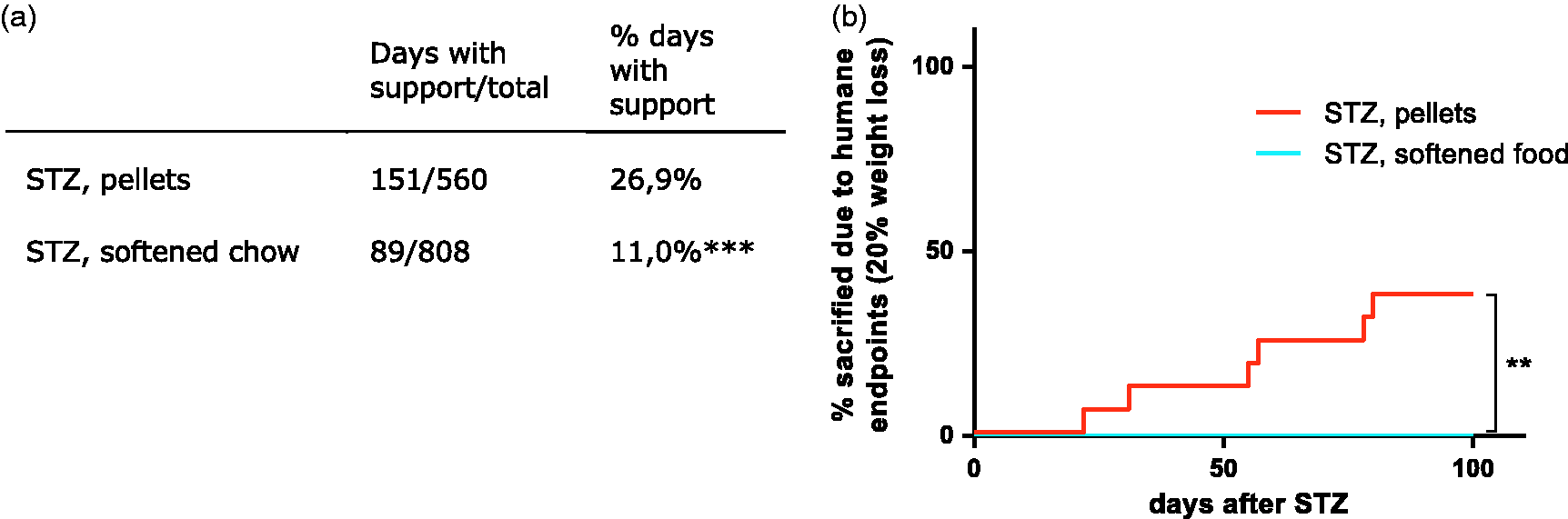
Water-softened chow reduces acute weight loss following STZ injection. (a) Percent change in body weight from day 1 (prior to STZ injection) until the first occurrence of a euthanization at day 22. The mice were injected with either 100 mg/kg STZ or vehicle at day 1 and 4, marked by vertical dotted lines in the figure. Data are shown as mean ±SD. (b) Area under the curve for the body weight change of individual mice in the two STZ groups. *p = 0.045, by two-tailed students t-test. Data are shown as individual points as well as mean ±SD. Water-softened chow reduced frequency of supportive treatment and humane endpoints. (a) Table showing the frequency of days with supportive insulin and saline injections given to the diabetic mice when reaching 10% weight loss of total possible monitoring days. ****p < 0.0001, RR = 2.45 (CI: 1.928–3.108), by two-tailed Fisher’s exact test. (b) Graph showing the total number of mice euthanized during the study due to 20% weight loss. No mice were euthanized in the softened chow group due to 20% weight-loss. **p = 0.0027, by log-rank test.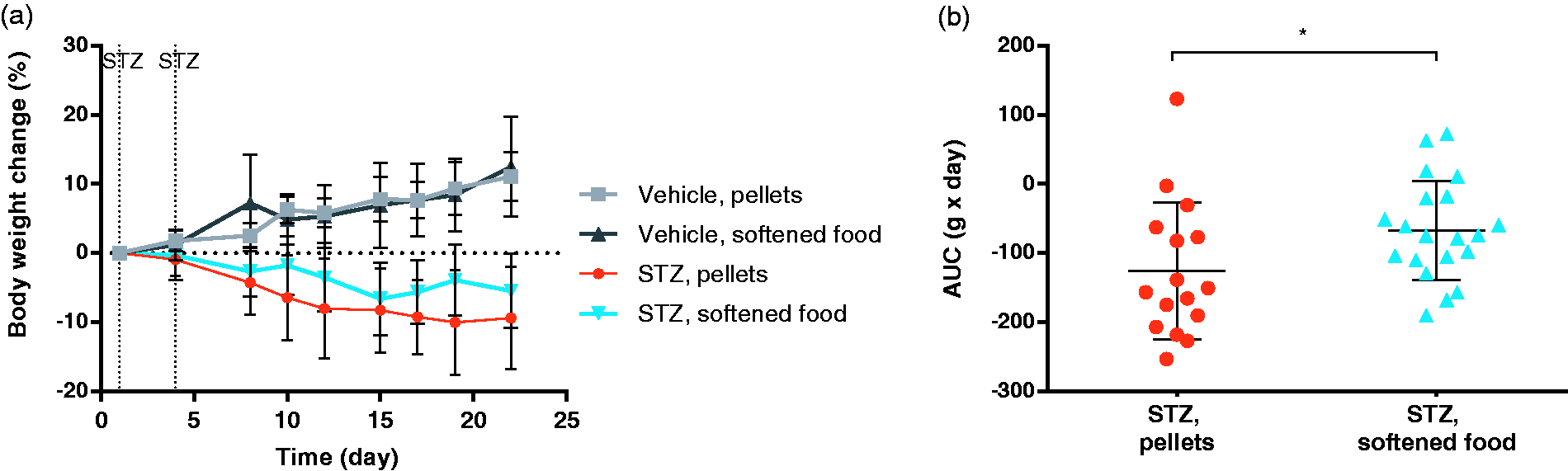
The group sizes for the merged groups were at the beginning of study then as follows: vehicle, pellets: n = 7; vehicle, softened food: n = 12; STZ, pellets: n = 16; STZ, softened food: n = 20. The group sizes were chosen based on previous studies and experience. Since loss of animals in the diabetic groups was expected more animals were assigned to the STZ groups. Furthermore, the groups receiving softened food had more animals to make it possible to test the hypothesis of mouse igloo lowering stress in the metabolic cages.
The softened food was given once every morning in the bottom of the cage in a 100 mL plastic weighing boat (HEA1424C, Heathrow Scientific, Vernon Hills, USA) from the day of the first STZ injection. A portion contained approximately 25 g of non-pelleted chow and enough tap water to moisten all the food (around 35 mL).
The animals were weighed three times a week. If an animal had lost more than 10% body weight compared to start weight, 1 mL 0.9% saline was administered intraperitoneally (i.p.) once a day. In addition, Lantus (insulin glargine, commercially available), was administered at 6 U/kg using a NovoPen Echo. This supportive treatment was given three times a week for at least a whole week and until the weight loss was no longer exceeding 10%.
Any animal that experienced more than 20% weight loss was euthanized immediately.
The mice were euthanized under isoflurane anaesthesia by intra-cardiac perfusion with 0.9% NaCl with heparin.
STZ administration
STZ (S-0130, Sigma-Aldrich, St Louis, MO, USA) was reconstituted in a sodium citrate buffer (pH 4.5, 1.06448, Merck, Billerica, USA) in a concentration of 10 mg/mL. Immediately after reconstituting the STZ, the mice (8–10 weeks of age, >22 g) were injected i.p. with either 100 mg/kg STZ or 10 mL/kg sodium citrate buffer on study day 1 and 4 after being fasted for 4 h (from 7 to 11 a.m.).
Monitoring diabetes and DN
Mice were weighed three times per week to monitor their body weight and general well-being. Blood glucose was measured two times per week and %HbA1c every third week throughout the study to investigate the effect of the softened food on diabetes induction and development. Blood samples for BG measurements were obtained from non-fasted, conscious mice by puncturing the tail vein with a lancet. 5 µL blood was collected into heparinized capillary tubes and transferred to 250 µL glucose buffer (0201-0002-024, EKF diagnostics, Cardiff, UK). Blood glucose concentration was measured on a Biosen 5040 (EKF Diagnostics, Cardiff, UK).
Blood (5 µL) for %HbA1c measurements was collected into heparinized capillary tubes from the tail vein from non-fasted, conscious mice, transferred to 500 µL HbA1c assay buffer (11488457, Roche Diagnostics GmbH, Mannheim, Germany), frozen at −20℃ until analysis and analysed on a Cobas 6000 (Roche Diagnostics Ltd, Rotkreuz, Switzerland).
At study week 6 and 14 the mice were placed individually in metabolic cages (Techniplast, Buguggiate, Italy) for 19 h where urine was collected for albuminuria measurements and faeces for corticosterone measurement (only week 6). When the mice were taken out, the urine was weighed and samples for albumin measurements were diluted 20× in 50 mM TBS pH 8.0, 1% BSA, 0.05% Tween20 and stored at −20℃ until analysis. Albumin concentration was measured using an assay based on an ELISA kit from Bethyl Laboratories Inc. (E90-134, Bethyl Laboratories, Inc., Montgomery, USA). Calibrator and controls was made using albumin from Sigma-Aldrich (A3139-10 mg, Sigma-Aldrich, St Louis, MO, USA). Coating buffer, wash solution and blocking buffer was additionally purchased at Sigma-Aldrich (C3041, T9039, T6789, Sigma-Aldrich, St Louis, MO, USA). The 19 h sample was used to calculate the 24 h urinary albumin excretion rate.
Quantification of corticosterone metabolites in faecal samples
The faecal samples were kept at −20℃ until analysed. The corticosterone metabolites were quantified using a protocol modified from a previous description. 29 The faecal samples were added to 96% ethanol (3 mL/g faeces) and placed on a tipping table overnight. After centrifuging twice in 20 min at 4000 r/min, 200 µL of the supernatants were evaporated to dryness and the residues dissolved in 200 µL PBS. The samples were then analysed using a corticosterone ELISA (EIA-4164; DRG Diagnostics GmbH, Marburg, Germany).
Statistical analysis
Statistical tests were done using GraphPad Prism software. The specific statistical tests used for each data set are mentioned in the respective figure legend. A p-value of 0.05 was considered statistically significant. All mice were included in every analysis but since several mice were lost during the study, data from these mice are missing in some of the analysis.
Results
Weight loss, supportive treatment and humane endpoints
The water-softened chow supplement was initiated from the first injection of STZ and soon after introduction the mice were observed to eat the softened food in addition to their regular chow pellets. The weight of the mice was monitored throughout the study. During the first three weeks, no mice were euthanized from any of the four groups. Thus, to evaluate the effect of the water-softened chow on acute weight loss following STZ injection, mean body weight change for the first 3 weeks is shown in Figure 1(a). Compared to the vehicle groups, which gained weight, both STZ-injected groups lost weight as expected. However, when comparing the AUC of the weight change curve for the two STZ groups (Figure 1(b)), the STZ mice given water-softened chow in addition to chow pellets lost significantly less weight (p = 0.045) during the first 3 weeks.
Beyond the first three weeks, individual mice had to be euthanized due to reaching the humane endpoint of 20% weight-loss, making statistical comparisons of mean body weight change for the whole study biased by the loss of mice with the most significant weight loss. The body weight change for the entire study can be found in Supplementary Figure 3.
Water-softened chow reduces faecal corticosterone metabolite excretion. (a) Total weight of faeces samples taken from the metabolic cages at week 6 after STZ injections. Data are shown as individual points as well as mean ±SD, *p = 0.0156, by one-way ANOVA. (b) Corticosterone metabolite concentration in faeces in ng/g faeces. Data are shown as individual points as well as mean ±SD, *p = 0.0449, by one-way ANOVA. (c) Total corticosterone metabolites excreted in the faeces during 19 h in metabolic cages. Data are shown as individual points as well as mean ±SD, ***p = 0.0007, by one-way ANOVA.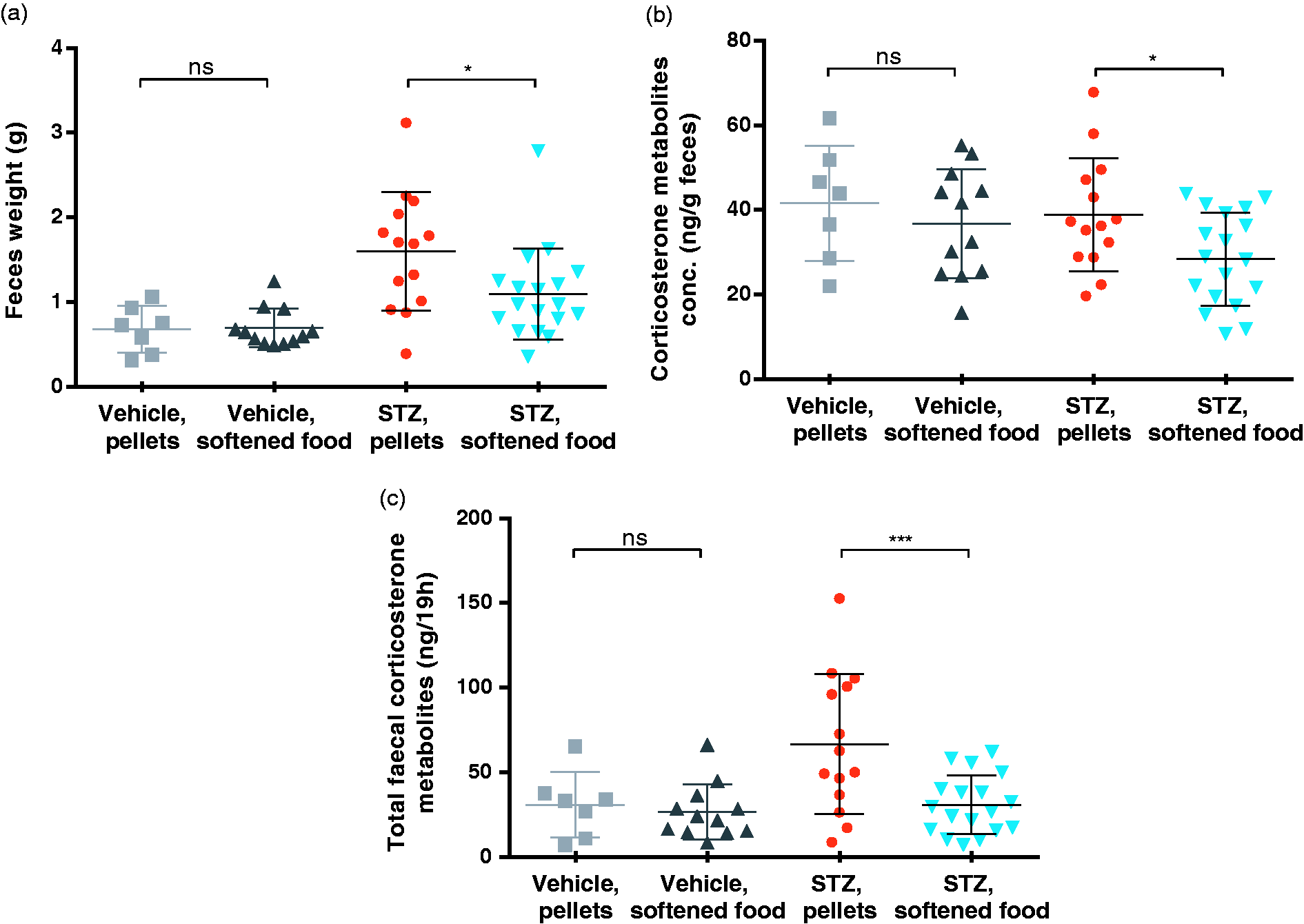
Any mouse experiencing weight loss of 10% or more received supportive treatment with insulin and saline according to protocol (see Materials and methods). The STZ-induced diabetic animals given water-softened chow in addition to normal chow experienced a significantly lower frequency of days with supportive injections of insulin and saline per total number of monitoring days (Figure 2(a); p < 0.0001).
Any animal experiencing more than 20% weight loss compared to start weight was euthanized immediately as a humane endpoint. As shown in Figure 2(b), no mice in the STZ-injected group given softened chow reached this humane endpoint. In contrast, 37.5% of the mice in the pellet group were euthanized due to 20% weight loss and this was despite the increased frequency of supportive measures. Most of these mice were euthanized between day 50 and termination. It should be mentioned that three diabetic mice receiving softened chow were found dead (on day 30, 95 and 97) without showing any increased weight loss or sign of discomfort in the preceding days. No unambiguous reasons were found for these spontaneous deaths during necropsy. One mouse had a slightly increased spleen and another one signs of mild ascites. In the last case, the decay process made in impossible to reach a diagnosis. We find it unlikely that the softened food caused these spontaneous deaths, although we cannot completely rule this out.
As a behavioural measure and to investigate if the mice receiving softened chow were still normally wearing their teeth eating pellets or gnawing wooden blocks, the length of the upper and lower incisors was measured at termination (Supplementary Figure 4). No significant difference was found in the length of either the upper or the lower incisors, suggesting that gnawing behaviour and teeth-wear was intact despite the supplement of water-softened chow.
Diabetes induction. (a) Percent HbA1c measured in tail vein blood every third week. Data are shown as mean ±SD. p = 0.2356, by comparing AUC in a two-tailed students t-test. (b) BG measured from tail blood twice weekly. Data shown from day 1 (prior to STZ injection) until the first occurrence of a euthanization at day 22. Mean ± SD. **p = 0.0095, by comparing AUC in a two-tailed students t-test. (c) BG for entire study. Mice euthanized during the study are excluded completely from these data. Mean ± SD. ***p = 0.0003, by comparing AUC in a two-tailed students t-test.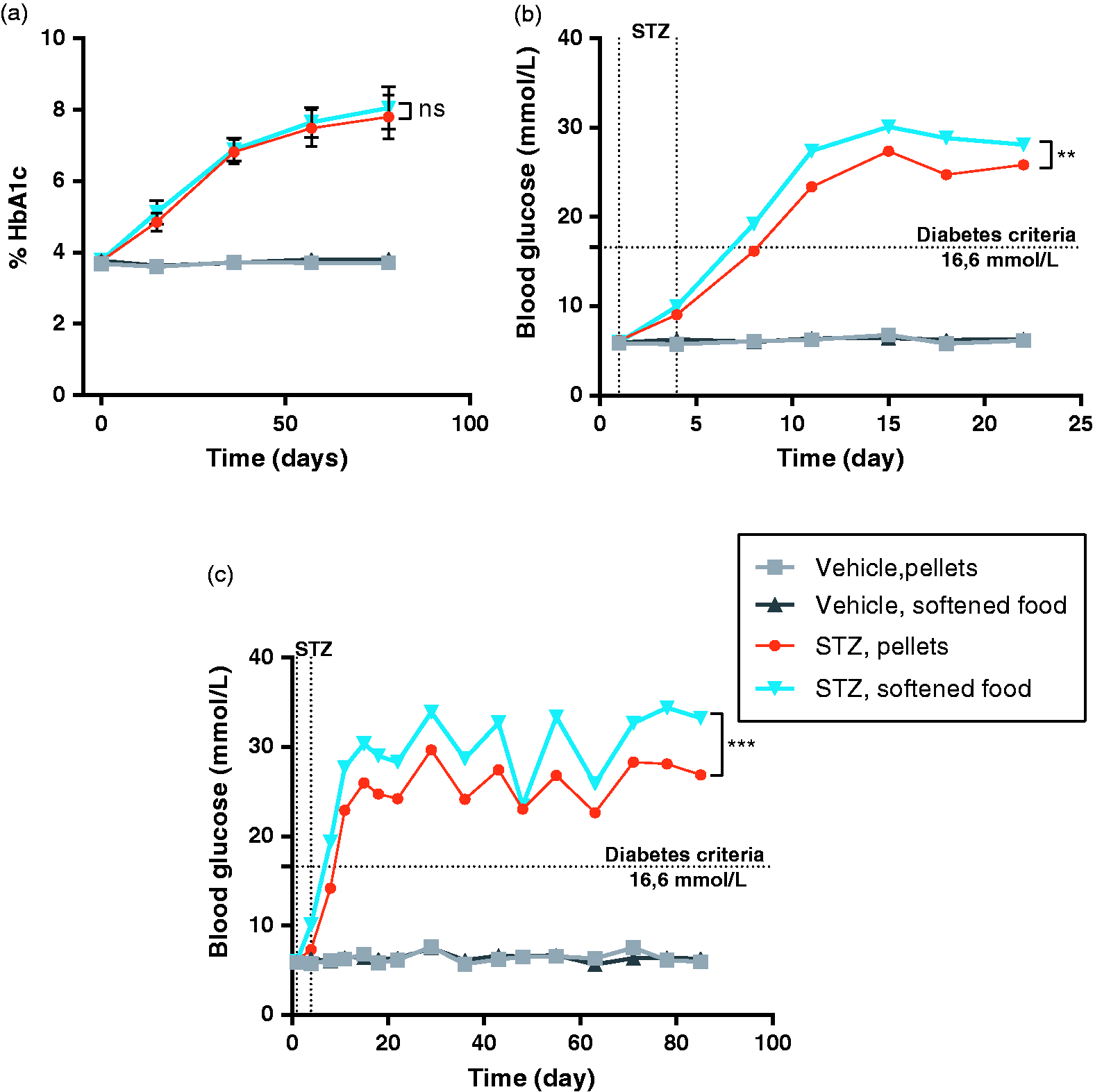
Corticosterone excretion
To assess the general level of stress experienced by the mice, faecal corticosterone metabolites were measured in the mice. Faecal samples were taken at week 6 from the metabolic cages as a collection of the total excretion over 19 h and this was analysed for corticosterone content. The total weight of the faeces samples showed that the diabetic mice only receiving pellets defecated significantly more in the 19 h compared to the diabetic mice given softened chow (Figure 3(a)). The concentration of corticosterone per g faeces was calculated and showed that the diabetic mice receiving softened chow had a significantly lower faecal corticosterone concentration compared to the group only receiving pellets (Figure 3(b)). When looking at the total amount excreted during the 19 h, the diabetic mice receiving softened chow excreted significantly lower total amount of corticosterone metabolites in their faeces compared to the diabetic mice only eating pellets (Figure 3(c)).
Diabetic and DN disease readouts
To investigate if the supplementation of water-softened chow might affect the induction of diabetes, BG was measured twice weekly, and%HbA1c every third week (Figure 4). As expected both BG and %HbA1c was increased in both STZ-treated groups compared to the vehicle groups. When comparing the two STZ-treated groups, a significant higher BG is found in the group receiving softened food both in the first 3 weeks and the entire study (Figure 4(a) and (b)). This difference is not seen when comparing the HbA1c for these groups, and both results suggest an intact induction of diabetes (Figure 4(c)).
The 24 h urinary albumin excretion rate (UAER) was calculated from urine samples after 19 h in metabolic cages 14 weeks after STZ (Figure 5(a)). This readout showed no significant difference when the water-softened chow supplement was compared to chow pellets alone in the STZ groups, suggesting an intact induction of albuminuria. Furthermore, to determine the gross pathological impact on kidneys following diabetes induction, the kidney-to-body weight ratio was measured at termination in all groups (Figure 5(b)). Compared to vehicle groups, both STZ-treated groups showed a significant increase in relative kidney weight. The kidney weight at termination could therefore further indicate if the softened chow affects the induction of DN. The softened chow did not affect the kidney weight in either the vehicle- or STZ-treated mice.
Disease readouts. (a) 24 h urinary albumin excretion rate calculated from urine collected in metabolic cages 14 weeks after STZ injection. Data are shown as individual points as well as mean ±SD. **p = 0.0013, ***p = 0.0002, by one-way ANOVA. (b) Kidney weight measured at termination and presented as relative kidney weight as a percentage of body weight. Data are shown as individual points as well as mean ±SD. ***p = 0.0006, ****p < 0.0001, by one-way ANOVA.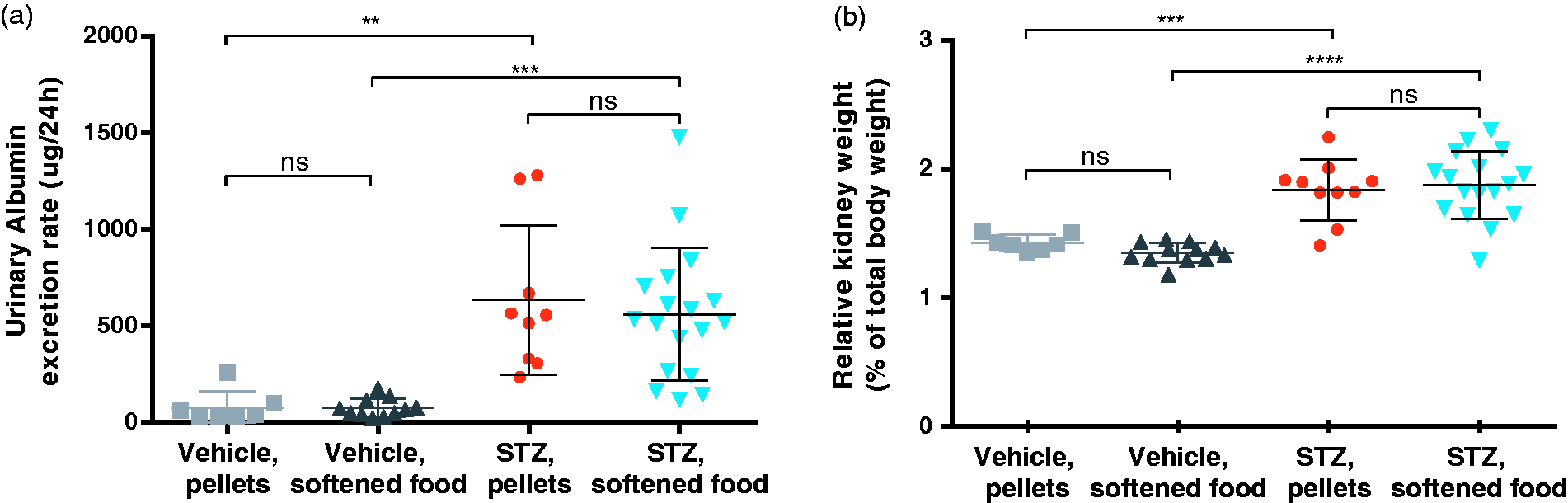
Discussion and conclusion
The STZ-induced diabetic mouse model is one of the most used models for studying diabetes and DN. However, challenges regarding significant weight loss and euthanization rates still calls for optimization and refinement of this model to improve overall animal welfare and study quality. In this study, we found that giving the mice the opportunity to eat water-softened chow in addition to the regular chow pellets could reduce weight loss in STZ-injected male mice. We found a significant reduction in both the acute weight loss (first 3 weeks) and in the number of mice that reached the humane endpoint of 20% weight loss. The latter was found even though the STZ-injected mice receiving softened chow received a significantly lower frequency of supportive treatments with insulin and saline throughout the study, without which, the difference might have been even more significant. Overall, this study demonstrates that supplementing STZ-injected male mice with water-softened chow once daily in addition to regular chow pellets could significantly improve animal well-being. Since only male mice were included in this study, it is therefore only possible to conclude on the effect of softened food on diabetic male mice. In this study, we looked primarily on weight loss but in future studies it would be interesting to make a thorough and systematic investigation of the nature of the weight loss, behaviour, appearance, body condition score etc.
It is difficult to get a comprehensive picture of the extent of the issues with weight loss and loss of animals in this model, since many publications do not account for the number of animals lost during these studies. 14 As described in other publications, we in this study used injections with insulin and saline as supportive treatment. We have found this to minimize the loss of animals especially during longer-term studies such as for development of measurable nephropathy. Other researchers have suggested that liquid nutrition supplements could be used in combination with insulin in order to prevent significant weight loss in this model, 9 but how this supplement affect the diabetic mice is unknown and would be an interesting topic for further studies. In the current study, we present an initiative that could potentially reduce weight loss and euthanization rate by simply giving the mice a water-softened chow supplement in addition to their regular chow. Importantly, giving the mice water-softened chow once daily did not affect the induction or magnitude of diabetes or change their DN phenotype, suggesting an intact disease model. Our results indicate that giving the mice the opportunity of eating softened chow helps stabilize their weight. And by reducing weight loss, the need for supportive treatment is also lowered. This significantly lowers the risk of stressing the animals due to frequent weighing and injections and the work load of monitoring and handling them.
Measuring faecal levels of corticosterone metabolites is widely used as a biomarker of stress and has some advantages compared to quantifying blood levels of corticosterone, since it gives a measure of stress levels over a longer period and is not affected by the potential stress response of the blood sample procedure.30–33 While the concentration of corticosterone metabolites in the STZ-injected mice receiving softened chow supplement was only slightly lower than in those getting only chow pellets, this difference was even greater when looking at the total corticosterone metabolites excreted during the 19 h, suggesting a lower stress response in the water-softened chow group. It has previously been argued that expressing the corticosterone metabolites as total amount excreted during a time period is more accurate than concentration.29,34,35 Therefore, one could interpret the data as if the STZ-injected mice receiving softened food were less stressed than the STZ- injected mice receiving pellets. However, since the vehicle groups differ in proportion to the STZ groups in terms of corticosterone levels expressed as concentration (Figure 3(b)) versus excretion (Figure 3(c)), this should be interpreted with caution. It seems, though, that the softened food supplement to STZ-treated animals under no circumstances are resulting in higher corticosterone levels, and does thus not seem to inflict any negative impact to the welfare of the animals. Furthermore, the mice were housed in the metabolic cages for 19 h to obtain urine samples for albuminuria measurement and faeces samples for measurements of corticosterone metabolites. Since the gastrointestinal transit time is only around 6 h in mice, 36 the higher total excretion of corticosterone in the STZ-injected mice only eating pellets could both be an effect of an increased stress level in both the home cage and in the metabolic cages. It has previously been shown that being housed in metabolic cages significantly increases the excretion of corticosterone metabolites, 37 although that study investigated mice constantly housed in metabolic cages for several weeks. Since the mice in this study were only housed in the metabolic cage for 19 h, it is not possible to conclude on the effect of the metabolic cages on the level of stress in the different groups. The increased level of stress hormone in the STZ-injected mice only eating pellets could also be the result of increased stress prior to placement in the metabolic cage.
The addition of the water-softened chow does not interfere with the disease induction or progression. We detect slightly higher BG levels throughout the study in the STZ injected group receiving water-softened chow, but this increase is not large enough to be reflected in the HbA1c measurements. Also the UAER increase was comparable in the two STZ groups displaying an intact induction of both diabetes and albuminuria.
From our experience the STZ-induced diabetes model is a model prone to variability; also when it comes to weight loss. Therefore, even though the results of this study show a clear effect of giving the STZ-injected mice softened chow it might be appropriate to test the reproducibility of these results in other research environments. Furthermore, it would be relevant to explore the effect of the softened food on other strains used in DN research as well as female mice and additionally when using a different STZ injection protocol. Currently, this initiative has only been assessed in the STZ-induced diabetes/DN model but could potentially be considered in other mouse models where high weight loss is an issue.
After first being described in 1959 by Russell and Burch, the concept of the 3Rs now has become the most important ethical principle when working with animal welfare in research. In this study we have been implementing both the refinement and reduction part of these principles. 38 By reducing weight loss and lowering the need for supportive insulin and saline this initiative is significantly refining this animal model. Furthermore, by giving the mice softened chow we reduce number of animals needed in studies as fewer animals reach significant weight loss and the humane endpoint. While the supplement of water-softened chow increased the overall animal welfare; it did not affect the measured readouts for diabetes or DN which indicates an unchanged disease model.
In conclusion, this study suggests that giving STZ-injected male mice water-softened chow once daily in addition to normal chow pellets could significantly reduce weight loss and reduce the risk of losing animals to humane endpoints without interfering with the development of diabetes or DN.
Footnotes
Acknowledgements
The authors would like to thank the laboratory technicians in Diabetes Complications Pharmacology for assistance in the in vivo part of the experiment as well as the Laboratory Animal Science group at Novo Nordisk A/S for assistance with mouse husbandry. Also, the authors thank Helle Runchel Porsdal and Trine Marie Ahlman Glahder for assistance with measurements of corticosterone metabolites.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this news article: This work was a part of a PhD project funded by Innovation Fund Denmark (grant number 4135-00008B) and Novo Nordisk A/S.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
