Abstract
The streptozotocin (STZ)-induced diabetic mouse model has been extensively used as a model for diabetes and diabetic nephropathy, but it is still influenced by many off-target toxic effects and large variation in diabetes induction. Therefore, the aim of this study was to compare different STZ dosing regimens to optimise animal welfare and minimise unwanted effects of STZ measured by acute renal toxicity, impairment of stomach emptying and weight loss. Male 129/Sv mice were injected with 1 × 50, 1 × 100, 1 × 125, 1 × 150, 1 × 200, 5 × 50, 2 × 100 and 2 × 125 mg/kg STZ or vehicle and euthanized 24 hours after the last injection. All STZ doses were found to induce significant enlargement of the stomach. All multiple doses of STZ increased the albumin:creatinine ratio significantly, and immunohistochemical staining of KIM-1 and Ki-67 was increased by 5 × 50 and 2 × 100 mg/kg STZ. Renal gene expression of Cdkn1a, KIM-1, NGAL and MCP-1 was increased by most of the STZ doses. No difference was found between the double intermediate dose of 2 × 100 mg/kg and the multiple low dose of 5 × 50 mg/kg regarding either stomach enlargement or kidney injury. However, the reduced fasting periods and injections in the 2 × 100 mg/kg STZ group could have lowered the impact on the general condition measured as change in body weight. This shows that the double intermediate dose is a good alternative to the recommended multiple low dose for diabetes induction in these mice. The STZ-induced mouse model has again proven to be a model with large variations affecting both animal welfare and model robustness.
Introduction
For many years, the streptozotocin (STZ)-induced mouse model has been a extensively used to study both diabetes and diabetic nephropathy (DN). However, major challenges still exist with finding the optimal dosing regimen of STZ to keep toxicities to a minimum while obtaining a reliable and reproducible induction of diabetes. The toxic effects of STZ on various organs are associated with a high degree of animal suffering but might also have a significant impact on renal readouts and can thereby have a substantial effect on the qualities of studies using this model.1,2
Although first discovered as an antibiotic, 3 since the discovery of its diabetogenic effect,4,5 STZ has been extensively used to induce models of both diabetes and DN.6,7 Being a glucose analogue, STZ is transported into the cells by glucose transporters, mainly GLUT2, where it accumulates and causes necrosis particularly caused by alkylation of DNA, production of nitric oxide and production of reactive oxygen species. The pancreatic beta cells, which have a high concentration of GLUT2, are highly susceptible to STZ. By inducing beta cell death, STZ causes a reduction in insulin production and development of hyperglycaemia.8–10 Hyperglycaemia eventually results in STZ-treated animals developing modest renal changes with similarities to those seen in early human DN. 11
The cell toxicity of STZ is not limited to beta cells, but also induces direct acute damage to the kidneys12–14 as well as the liver and intestine.15,16 Transport via GLUT2 into proximal tubule cells has been considered the main entry for STZ into the kidneys. However, alternative uptake methods must exist, since direct toxicity towards cells that have no GLUT2 expression (e.g. cardiomyocytes 17 and immune cells 18 ) has been shown. Therefore, it has been speculated whether other active or passive glucose transporter such as GLUT1 could be involved. 18 Furthermore, it has been indicated previously that uptake of STZ into the kidneys could be partly through sodium/glucose cotransporter (Sglt) transporters. 14 In the same study, it was shown that high-dose injections of STZ can cause an impairment of gastric emptying, leading to a significant increase in stomach weight. 14 This could lead to distress and possibly pain, thereby compromising the welfare of the mice considerably. In particular, proteinuria has been found to be a renal readout that is partly caused by the toxic effect of STZ and not solely the long-term hyperglycaemia. 12 Renal changes caused by STZ can be hard to distinguish from the diabetic effect and should be taken into account when interpreting results from STZ models.12,13
A single high dose of STZ varying from 150 to 200 mg/kg in mice 19 has traditionally been a widely used way of inducing diabetes. With this dosing regimen, beta cells are quickly eliminated, and this is therefore particularly well suited for islet transplantation models where total eradication of beta cells is needed. 2 In contrast, the Diabetic Complications Consortium (DiaComp) has recommended the use of a multiple low dose of STZ for induction of diabetes in mice where 50 mg/kg STZ is given over five consecutive days.20,21 In addition to the two above-mentioned STZ dosing regimens, a double intermediate dose was also included in this study, where 100 or 125 mg/kg is given twice (days 1 and 4). This dosing regimen has previously been described to induce diabetes efficiently in 129/Sv mice, which is the strain used in this study.22,23
The main aim of these studies is to compare directly the acute effect of different doses of STZ on the kidneys as well as the stomach in male STZ-treated 129/Sv mice. With regards to renal toxicity, we especially wanted to investigate how some of the well-known markers used in models of DN are affected shortly after STZ injection. In the comparison of different STZ dosing regimens, a specific focus was to investigate whether a double intermediate dose of STZ would be a good alternative for the recommended multiple low dose regarding renal toxicity, stomach enlargement and weight loss.
Materials and methods
Ethics statement
Approval for the studies was given by the Animal Experiments Inspectorate, Ministry of Environment and Food of Denmark (license no. 2014/15-0201-00429). The studies were conducted according to the EU Directive 2010/63/EU on the protection of animals used for scientific purposes. Any mice showing signs of compromised health (inadequate activity/moribund) or with a maximum weight loss of 20% was terminated immediately.
Mice and housing
A total of 282 129/Sv (129S2/SvPasCrl) male mice were used. The mice were delivered from Charles River (Copenhagen, Denmark) and included in the study at 8–10 weeks of age and with a minimum body weight of 23 g. All mice were acclimatised for at least one week before the start of the study. The mice were ear punched for identification and housed in groups of five mice, with the conditions previously described in detail. 23 In short, the mice were housed in type IV open cages (595 mm × 380 mm × 200 mm) in cabinets (Scanbur, Karlslunde, Denmark). The mice had free access to a standard chow diet (Altromin 1324; Brogaarden ApS, Lynge, Denmark) and tap water through the automatic water system in the cabinets.
Acute toxicity and food intake study
Experimental design
Overview of dosing groups.
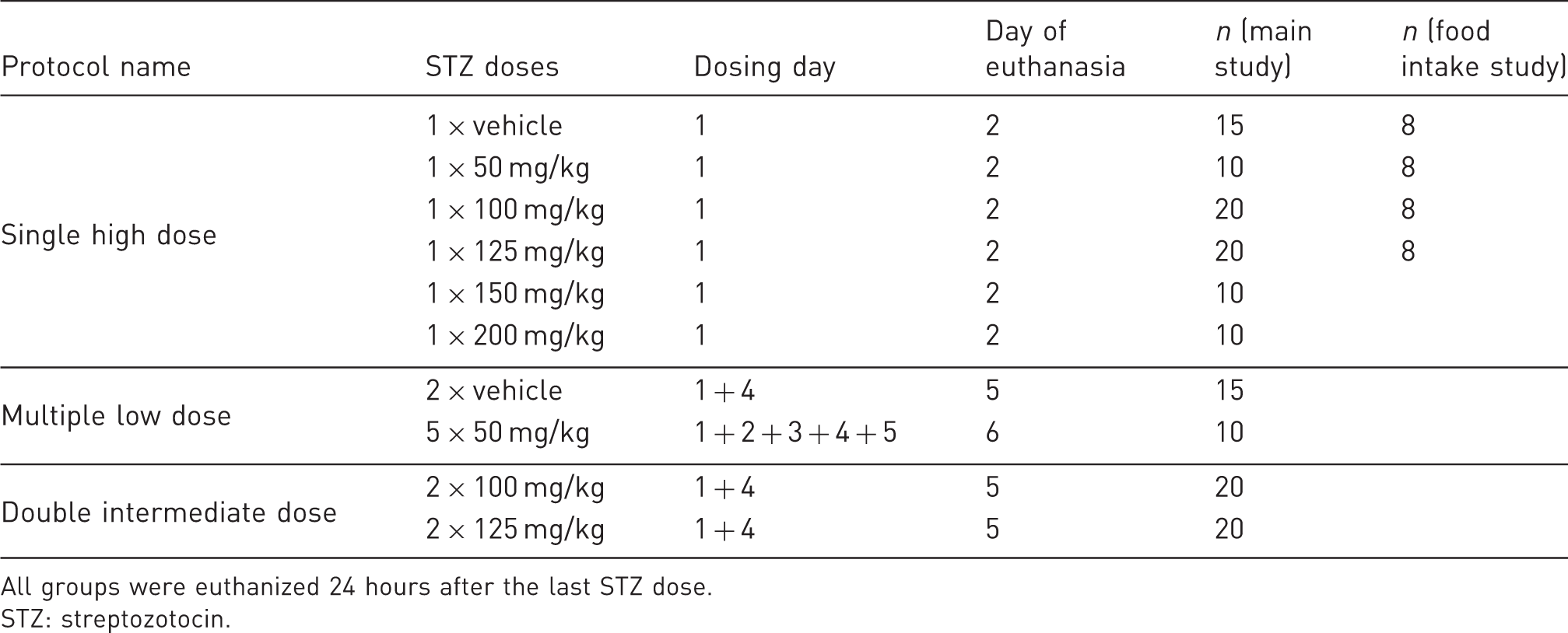
All groups were euthanized 24 hours after the last STZ dose.
STZ: streptozotocin.
For logistic reasons, the groups were split over different experiments using the same protocol. Furthermore, the 1 × 100, 1 × 125, 2 × 100, 2 × 125 and 1/2 × vehicle groups were repeated, due to unexpected large variation in the first run, and the results were pooled. This explains the groups sizes of 15/20 mice in these groups. The remaining groups were studied only in the first experiment and were not included in the repetition study. Therefore, there are only 10 mice in these groups (1 × 50, 1 × 150, 1 × 200 and 5 × 50 mg/kg).
Furthermore, for the immunohistochemical (IHC) staining and the gene expression analysis, five mice were chosen from each of the groups: 2 × vehicle and 1 × 50, 1 × 100, 1 × 200, 5 × 50 and 2 × 100 mg/kg. As 2 × 100 mg/kg has been found to be sufficient to induce diabetes and nephropathy in these mice (Figure 5), we chose not to include 2 × 125 mg/kg in these analyses.
As we had seen a large variation in the effect of STZ on stomach enlargement, it was decided to select mice with an even distribution of stomach enlargement.
In a separate study, food intake was measured after STZ treatment. The mice were placed individually in Biodaq cages (Research Diets, New Brunswick, NJ) one week before the start of the study for acclimation. As standard chow pellets cannot be used in these cages, the mice were fed D12450B pellets (Research Diets), with similar energy density and content of macronutrients as the chow diet. The mice were then randomly divided into groups receiving a single dose of 50, 100 or 125 mg/kg STZ or vehicle (n = 8; Table 1). Food intake was hereafter measured until the mice were euthanized 24 hours after STZ injection.
STZ administration
Before STZ treatment, the mice were fasted for four hours (from 7:00am to 11:00am). All mice were weighed, and STZ doses were calculated based on weight. Immediately before intraperitoneal injection, STZ (S-0130; Sigma–Aldrich, St Louis, MO) was reconstituted in a sodium-citrate buffer (pH 4.5, 1.06448; Merck, Billerica, MA) at a concentration of 10 mg/mL.
Body weight and blood glucose monitoring
The mice were weighed on day 0, before STZ injection on day 1 and then every day until they were euthanized. Blood glucose (BG) was measured at least on day 0 and on the day of euthanasia. For the groups receiving 5 × 50, 2 × 100 or 2 × 125 mg/kg STZ, BG was furthermore measured on day 3. Blood for BG was collected from the tail vein from non-fasted mice and diluted in glucose buffer, and the BG concentration was measured using a Biosen 5040 (EKF Diagnostics, Cardiff, UK).
Study termination
At study termination, the mice were anaesthetised using isoflurane. Urine (for albuminuria measurement) was aspirated directly from the bladder, and the mice were euthanized by intra-cardiac perfusion of heparinised saline. The left kidney was snap-frozen and stored at –80℃ for mRNA purification, and the right kidney was fixed in 10% neutral-buffered formalin. Moreover, the stomach was dissected at the gastroesophageal junction and the pylorus and weighed. The stomach was furthermore emptied, rinsed and weighed again.
IHC staining (Ki-67 and KIM-1) and real-time quantitative polymerase chain reaction (genes analysed CCL2, CDKN1A, HAVRC1 and LCN2) were performed on the kidneys. For details of these analyses, as wells as the albumin, creatinine and unacylated ghrelin measurements, see the Supplemental Data.
Long-term diabetic and nephropathy induction
One hundred 129/Sv male mice were randomly allocated to vehicle (n = 20), 5 × 50 mg/kg STZ (n = 40) or 2x100 mg/kg STZ (n = 40). BG was measured once weekly. STZ dosing and BG measurements were performed as described for the acute studies. Albuminuria was measured on urine collected in metabolic cages at 8, 13, 16 and 20 weeks after STZ dosing from 20 mice from both STZ groups and 10 mice from the vehicle group. Fifteen weeks after STZ dosing, half of the mice were euthanized, and the remaining continued until 22 weeks after STZ dosing. At both time points, the mice were euthanized by intra-cardiac perfusion of heparinised saline, and the kidneys were weighed.
Statistical analysis
GraphPad Prism (Version 7.04, GraphPad Software, Inc., San Diego, CA) was used for statistical analysis. The specific statistical tests used for each data set are mentioned in the respective figure legend. A p-value of 0.05 was considered statistically significant.
Results
Acute toxicity and food intake study
Body-weight change and BG
To evaluate the general condition of the mice shortly after STZ injections, the mice were weighed before injection and daily until being euthanized 24 hours after the last STZ injection. As illustrated in Figure 1(a), none of the single doses of STZ affected the change in body weight. When comparing the full dosing regimen of 5 × 50, 2 × 100 and 2 × 125 mg/kg, the 5 × 50 and 2 × 125 mg/kg groups had a significantly lower increase in weight during the study compared to the 2 × 100 mg/kg group. Before euthanasia, 24 hours after the last STZ dose, unfasted BG was measured in all mice. Only the 1 × 200 mg/kg (Figure 1(c)) and 2 × 125 mg/kg (Figure 1(d)) dosing groups had significantly increased BG compared to the vehicle groups. However, many mice in these groups were still not diabetic at this time point.
Body-weight change and blood glucose (BG) levels. (a) and (b) Mice were weighed before the first streptozotocin (STZ) injection and at euthanasia 24 hours after the last STZ injection. (c) and (d) BG was measured from the tail blood of unfasted mice at euthanasia. *p < 0.05; **p < 0.01; ***p < 0.001. All groups compared by one-way analysis of variance (ANOVA) using Tukey’s multiple comparisons test. n = 10–20. Data are shown as individual points as well as the mean ± SD.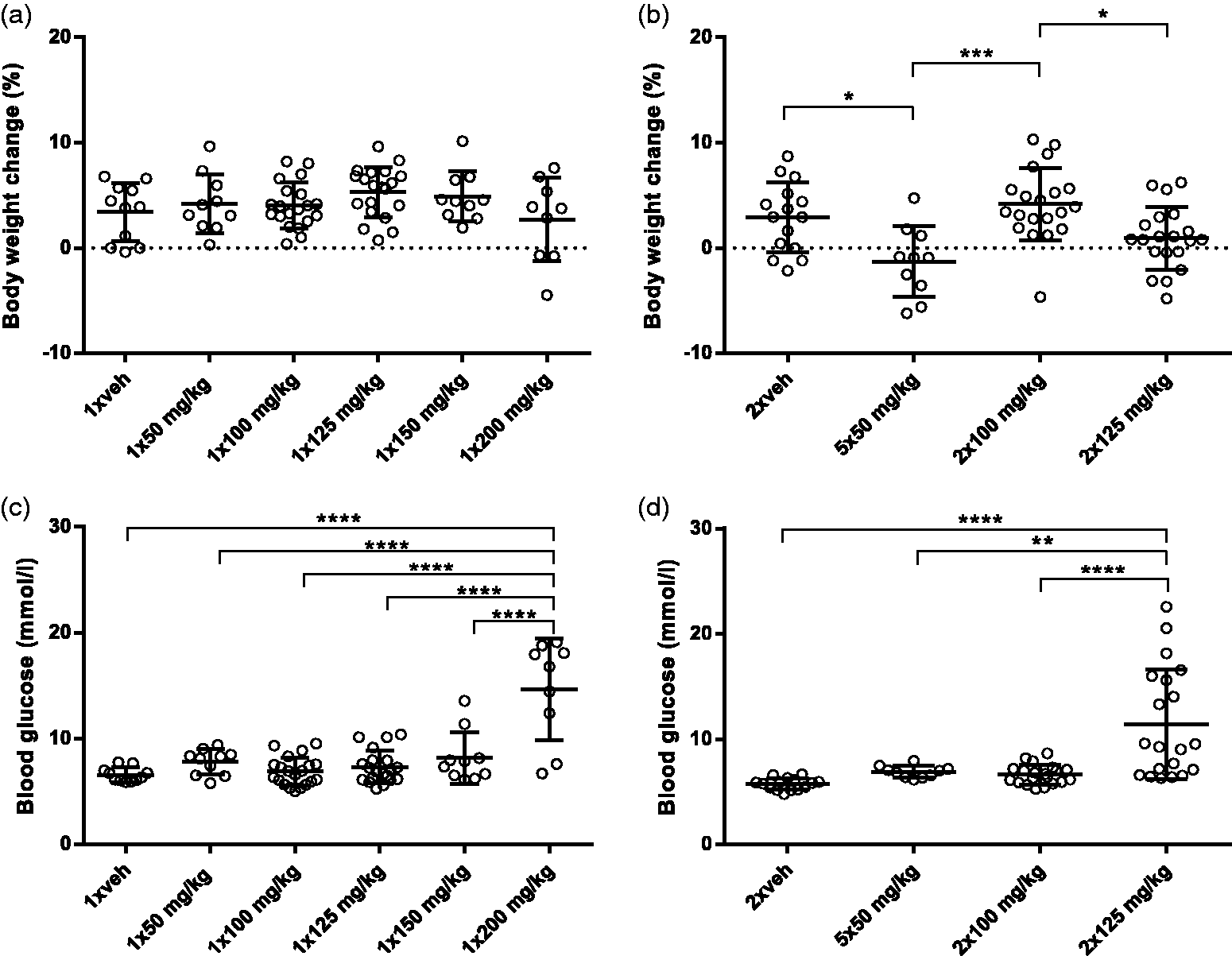
Stomach weight
Since it has previously been described by Brouwers et al.
14
that emptying the stomach is affected by STZ, the weight of the stomach was measured at the point of euthanasia Figure 2(a) and (b) illustrates that the stomach was found to be enlarged in mice from all groups injected with STZ. In the single doses of STZ (Figure 2(a)), it was found that stomach enlargement increased with increasing STZ dose up to 125 mg/kg, suggesting a direct link to STZ dose. When comparing the multiple doses of STZ, no difference was found between groups (Figure 2(b)). In most groups, some mice seem to be unaffected by the injection of STZ. There was no difference in the weight of the stomach without content.
STZ increases stomach weight. (a) and (b) Stomach weight at euthanasia 24 hours after the last STZ injection. (c) Photos of mice at euthanasia: vehicle (left) and 1 × 125 mg/kg (right). Arrow illustrates enlarged stomach. (d) Total food intake was measured in Biodaq cages from a single STZ injection to euthanasia after 24 hours. (e) Levels of unacylated ghrelin in plasma from euthanasia. χp < 0.05, χχp < 0.01, χχχp < 0.001 and χχχχp < 0.0001 compared to vehicle. *p < 0.05, **p < 0.01 and ****p < 0.0001 STZ groups compared to each other. All groups are compared by ANOVA using Tukey’s multiple comparisons test. n = 10–20 (stomach weight); n = 8 (food intake and unacylated ghrelin). Data are shown as individual points as well as the mean ± SD.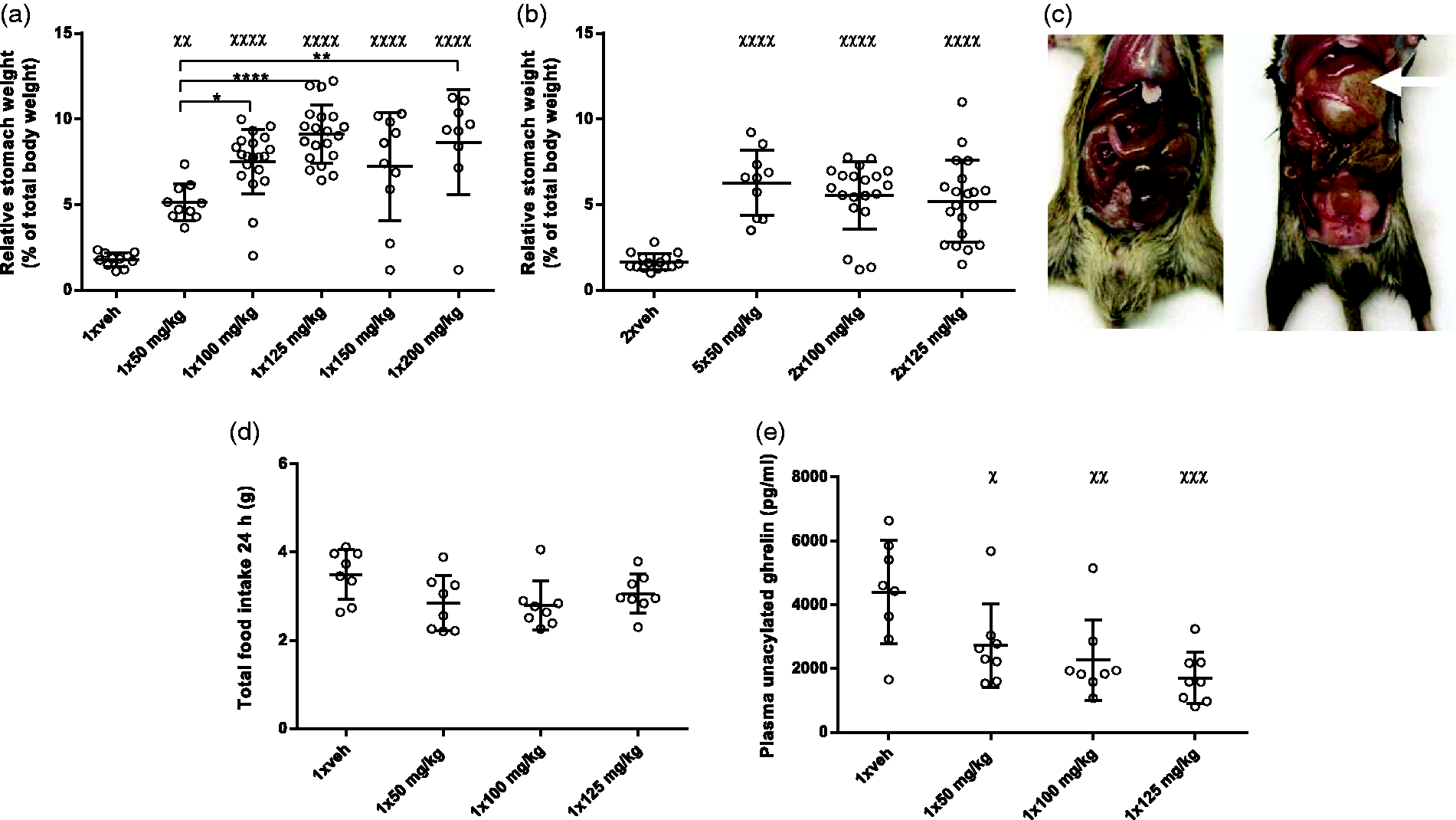
Figure 2(d) shows the result of the food intake study where it was determined that the enlargement was not caused by an increase in food intake. Levels of unacylated ghrelin in plasma were found to be decreased by STZ injection (Figure 2(e)).
Renal toxicity
The acute toxic effect of different dosing regimens of STZ on the kidneys was investigated. Urinary excretion of albumin was measured on urine aspirated directly from the bladder at the point of euthanasia and is expressed as the albumin:creatinine ratio (ACR; Figure 3). Of the single injections of STZ, 1 × 125, 1 × 150 and 1 × 200 mg/kg was found to increase the ACR significantly (Figure 3(a)). When comparing the multiple doses of STZ, all dosing regimens increased ACR to a similar degree (Figure 3(b)).
Multiple doses of STZ increased the albumin:creatinine ratio (ACR). (a) and (b) ACR measured on urine aspirated from the bladder during euthanasia. χχp < 0.001 and χχχχp < 0.0001 compared to vehicle. **p < 0.1, ***p < 0.001 and ****p < 0.0001, STZ groups compared to each other. All groups were compared by one-way ANOVA with Tukey’s multiple comparisons test on log transformed data. n = 10–20. In some groups, data points are missing due to a lack of urine in the bladder at euthanasia. Data are shown as individual points as well as the mean ± SD.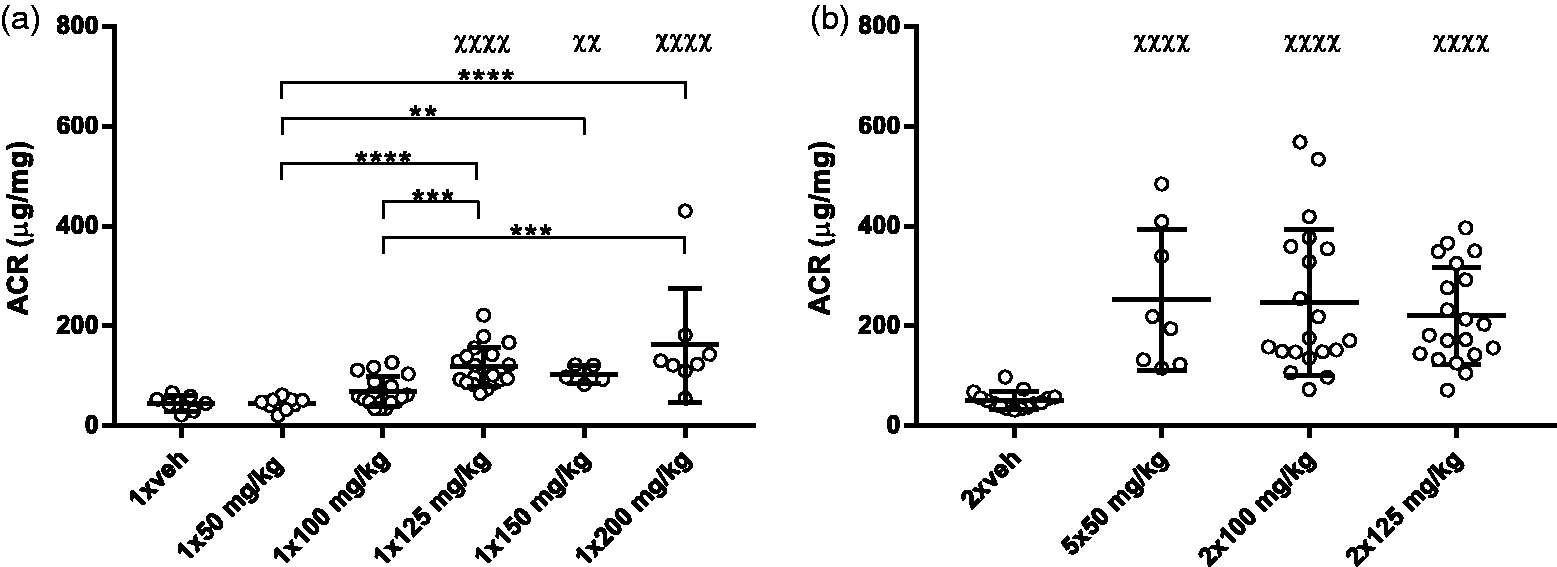
To investigate the acute damage to the kidney further, IHC staining of the kidney injury marker KIM-1 and the proliferation marker Ki-67 was performed and quantified by automated image analysis (Figure 4(a–f)). Both markers were found to be significantly increased in the groups injected with 5 × 50 and 2 × 100 mg/kg, with no difference observed between these two groups. KIM-1 staining was found to be located in the proximal tubules, and Ki-67-positive cells were additionally primarily proximal tubular cells as well as inflammatory cells. The toxic effect of STZ on the kidneys was further investigated by gene expression. As illustrated in Figure 4(g–j), STZ significantly increases the expression of genes coding for the injury marker Cdkn1a, KIM-1 and NGAL, as well as the pro-inflammatory cytokine MCP-1. No significant difference in expression of the chosen genes could be detected between 5 × 50 and 2 × 100 mg/kg STZ.
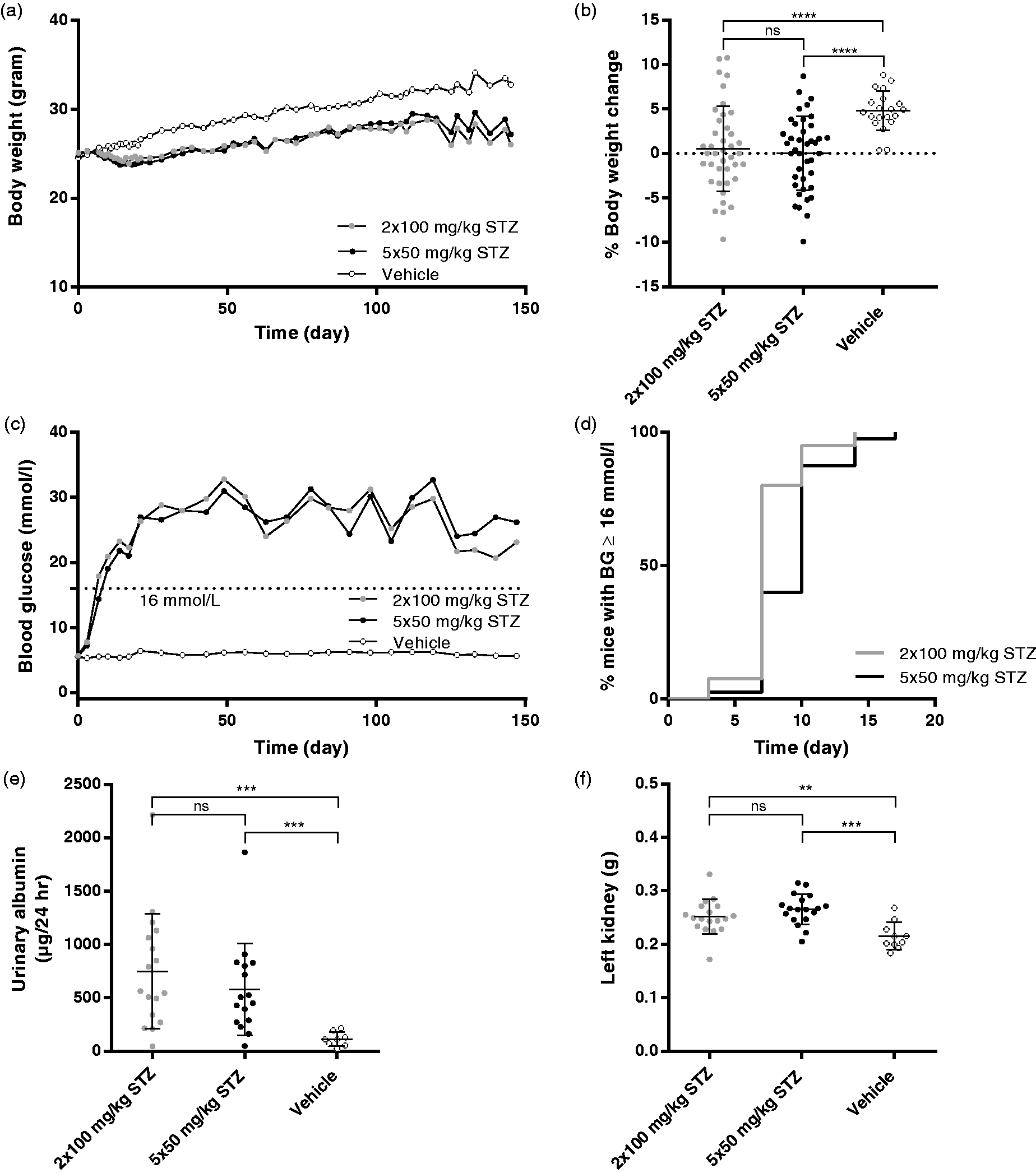
Multiple doses of STZ increase levels of renal injury markers. Immunohistochemistry (IHC) staining of (a) KIM-1 and (d) Ki-67 on kidney slides. Representative pictures of IHC staining of KIM-1 (b) 5 × 50 mg/kg STZ, (c) vehicle and Ki-67, (e) 5 × 50 mg/kg and (f) vehicle. The analysis of Ki-67 was limited to the cortex. **p < 0.01; ***p < 0.001; ****p < 0.0001. All STZ groups were compared to the vehicle group and 5 × 50 mg/kg STZ with 2 × 100 mg/kg by one-way ANOVA with Tukey’s multiple comparisons test. n = 5. Data are shown as individual points as well as the mean ± SD. Scale bars = 50 µm. mRNA levels of (g) HAVRC1, (h) CDKN1A, (i) LCN2 and (j) CCL2. Protein names shown in parentheses. Gene expression analysed by real-time quantitative polymerase chain reaction. Data are shown as fold change. Statistical analysis performed on ΔΔCt values. χp < 0.05, χχp < 0.01, χχχp < 0.001 and χχχχp < 0.0001 compared to vehicle. All STZ groups were compared to vehicle group and 5 × 50 mg/kg STZ with 2 × 100 mg/kg by one-way ANOVA with Tukey’s multiple comparisons test. n = 5. Data are shown as individual points as well as the mean ± SD. STZ dosing regimens 5 × 50 and 2 × 100 mg/kg are equally capable of inducing hyperglycaemia, albuminuria and kidney hypertrophy. (a) Body weight from day 1 (prior to STZ injection) until study termination at day 147. No difference was found between the STZ groups when comparing AUC in a two-tailed Student’s t-test. Data are shown as the mean. (b) Change in body weight from first STZ dose on day 1 until day 8. ****p < 0.0001 by two-tailed t-test with Welch correction. Data are shown as individual points as well as the mean ± SD. (c) BG measured weekly from the tail blood of unfasted mice from day 1 (prior to STZ injection) until study termination. No difference was found between the STZ groups when comparing area under the curver (AUC) in a two-tailed Student’s t-test. Data are shown as the mean. (d) Incidence of hyperglycaemia (>16 mmol/L). Diabetes was induced in 100% of all mice in both STZ groups. (e) Twenty-four hour urinary albumin excretion was measured on urine sampled in metabolic cages for 24 hours at 13 weeks after STZ injections. n = 20 (STZ), n = 10 (vehicle). ***p < 0.001 by two-tailed t-test with Welch correction. Data are shown as individual points as well as the mean ± SD. (f) Weight of left kidneys measured at euthanasia 22 weeks after STZ. n = 20 (STZ), n = 10 (vehicle). **p < 0.01 and ***p < 0.001 by two-tailed t-test with Welch correction. Data are shown as individual points as well as the mean ± SD.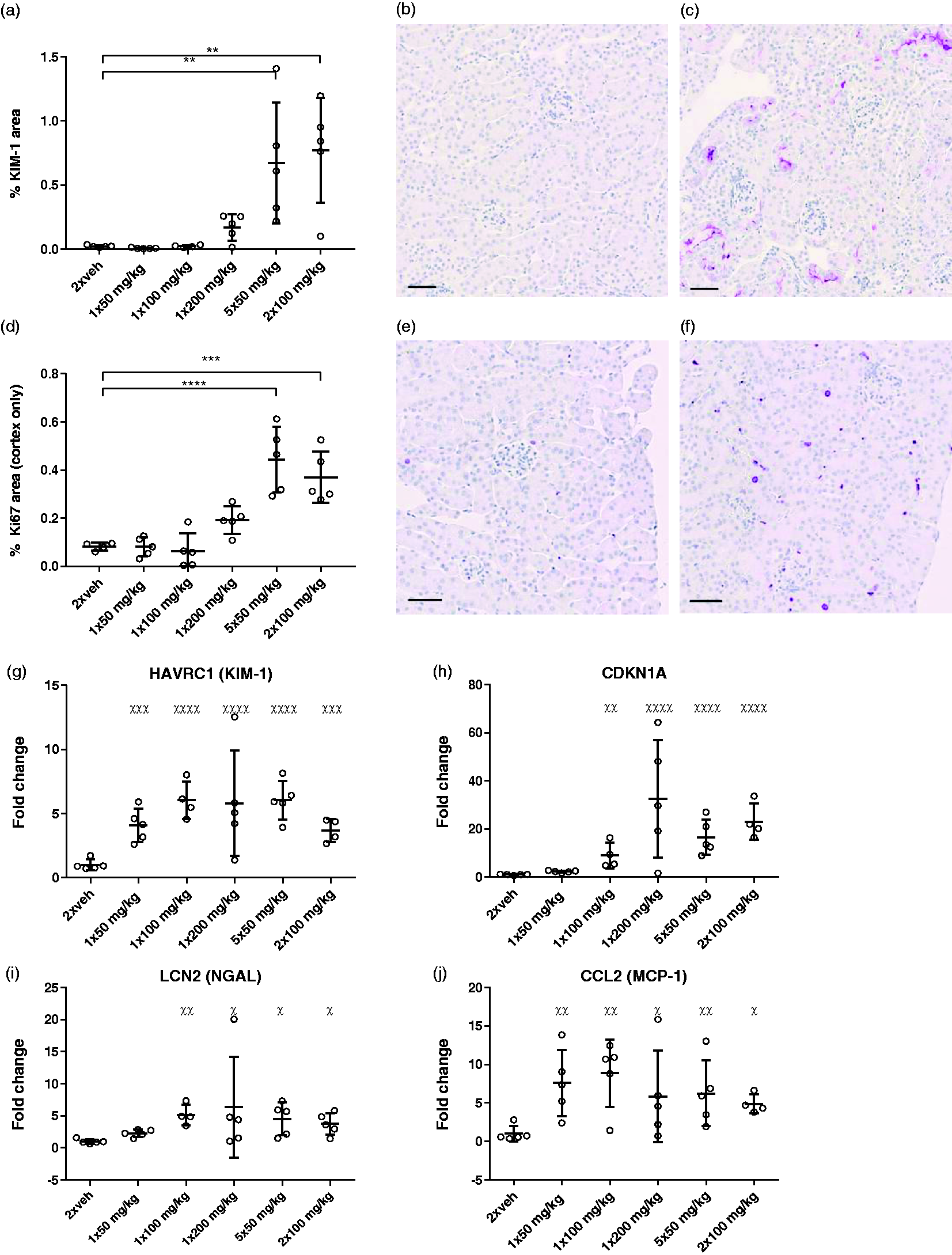
Long-term diabetic and nephropathy induction
The long-term effect of the two STZ dosing regimens, 5 × 50 and 2 × 100 mg/kg, on induction of diabetes and DN was investigated in a 22-week study. In this study, we found that both dosing regimens were well tolerated by the mice. Contrary to the short study, no difference was found between the two STZ groups when analysing weight change during the entire study calculated by area under the curve (Figure 5(a)) or the weight change shortly after the STZ injections (Figure 5(b)). The two dosing regimens were equally capable of inducing hyperglycaemia, albuminuria and kidney hypertrophy (Figure 5(c–f)).
Discussion and conclusion
Even though STZ has for many years been used to induce diabetes in animal models, research is still needed in order to understand the effect of STZ fully, find the optimal dosing protocol, reduce variation and minimise unwanted effects.
The DiaComp has recommended using a multiple low dose (e.g. 5 × 50 mg/kg) when inducing diabetes in mice.20,21 The rational for this dosing protocol is that lower doses of STZ cause fewer toxic effects on susceptible organs. However, there is not much published data directly comparing the different dosing regimens of STZ with respect to off-target toxicity. By directly comparing different STZ dosing protocols, we were able to investigate if the double intermediate dose could be an alternative for diabetes induction with a minimum of unwanted effects on the animals. Given the focus to analyse acute toxic effects, the outcome was analysed 24 hours after the last STZ injection. The kidneys were chosen, since there is a substantial reason for optimising STZ treatment in order to keep the renal toxicity to a minimum when working with STZ-treated mice in DN research. Since the previously published finding that stomach emptying 14 could significantly affect the welfare of these mice after STZ injection, this was included as another focus point.
When looking at the stomach, the 1 × 50 mg/kg dose was sufficient to cause significant enlargement. The increasing effect on stomach weight with increasing STZ dose, at least up to 125 mg/kg, suggests that this effect can be directly linked to the dose of STZ. Although lower when given in a single dose, five consecutive doses of 50 mg/kg were not found to be different from the two double intermediate doses (2 × 100 or 2 × 125 mg/kg) regarding stomach weight. A substantial variation in the effect of STZ, both diabetogenic and toxic, is often described. When looking at stomach enlargement in this study, it was also seen that there were mice in almost all dosing groups that did not respond at all to the STZ injection.
In this study, enlargement of the stomach was not caused by an increase in food intake, and no pathological changes to the stomach were found. The levels of unacylated ghrelin were decreased by STZ, which is in contrast to the findings by Brouwers et al. 14 Among other effects, unacylated ghrelin has been found to inhibit stomach emptying, 24 but our study indicates that other mechanisms might be involved in the observed stomach enlargement. In this study, we have not been able to investigate the underlying mechanisms for the enlargement of the stomach further, and neither have we investigated when the enlargement returns to normal. Whatever the mechanisms behind this effect of STZ on the stomach are, we consider this to cause substantial distress and potentially pain to the mice and thereby have a significant impact on animal welfare in this model. Therefore, it would be highly relevant to investigate if this unwanted effect of STZ can be avoided or reduced in any way.
As it has been indicated previously that direct effects of STZ, and not only long-term hyperglycaemia, can cause urinary protein leakage, 13 ACR was measured in this study. Since only the 1 × 200 and 2 × 125 mg/kg groups showed a significant increase in BG, the marked increase in ACR in the 5 × 50 and 2 × 100 mg/kg groups is likely to be directly linked to STZ toxicity.
To investigate the renal toxicity of STZ further, levels of different injury markers were examined using IHC staining and gene expression analysis. KIM-1 is a transmembrane protein which is known to be upregulated in proximal tubule cells in both DN and acute kidney injury.25,26 In the histological analysis, staining of KIM-1 was found to be significantly increased in the 5 × 50 and 2 × 100 mg/kg groups, with no difference between the two. The increase in Ki-67 staining found on IHC in the multiple doses of STZ indicates that regeneration processes are already initiated in the kidneys shortly after STZ injection. 27
In the gene expression analysis, KIM-1 was again found to be increased in all STZ groups, with no difference between the groups. Expression of genes coding for Cdkn1a (also known as p21),28,29 NGAL26,30,31 and MCP-1, 32 which have all been described to be increased in both acute kidney injury and DN, was increased in almost all of the STZ groups compared to the vehicle group. Again, for these genes, there was no difference between the STZ groups.
Overall, as with stomach enlargement, no difference was found between the 5 × 50 and 2 × 100 mg/kg groups for either of the renal readouts. Furthermore, we illustrated that many of the well-known markers used in models of DN are already increased shortly after STZ injection. This is relevant to consider when using the STZ model in DN research, since acute toxic renal injuries seem to be hard to distinguish from the diabetic effect.
As seen with stomach enlargement, significant variation is seen in the different readouts of acute renal injury, which makes it difficult to find potential differences between the STZ groups. However, our data clearly confirm the toxic effect of STZ to the kidneys and indicate that the double intermediate dose (e.g. 2 × 100 mg/kg) might be a good alternative to the multiple low dose (5 × 50 mg/kg), since no difference could be found between the two groups for any of the renal readouts.
Compared to the multiple low dose, the double intermediate dose has the considerable advantage of only having to fast, handle and inject the mice twice. This might explain the difference found in body-weight change in the short studies. Even though this difference was not present in the long study, the reduced number of STZ injections could significantly lower the stress placed on the mice, as well as decrease the workload, and it should therefore be taken into account when choosing the dosing regimen.
One of the weaknesses when assessing the results from these studies is that the time from the first STZ dose is different, although all groups were euthanized 24 hour after the last STZ dose. This is why the two single high doses (150 and 200 mg/kg) were not statistically compared to the other dosing regimens which can be used for inducing diabetes in mice (2 × 100, 2 × 125 and 5 × 50 mg/kg). In the mice injected with 1 × 200 mg/kg, the signs of acute kidney injury only had 24 hours in total to develop, and we expect that the readouts for renal damage would increase during the following days.
It has previously been described that the effect of STZ varies significantly with both strain33,34 and sex. 35 In this study, we only use male 129/Sv mice. Therefore, when using other strains or female mice, the dose might have to be adjusted. Furthermore, it has previously been described that significant variation in both diabetogenic and toxic effects, which cannot be explained by strain or sex, is often seen when working with STZ, even when working with inbred mice from the same vendor.2,36 The large variability of STZ, with respect to both efficacy/toxicity and consistency between repetitions of the same protocol, was confirmed in these sets of studies. We cannot conclude on the reason for this large variation, but it could potentially be caused by injection variations. Intraperitoneal injection is an often-used method for administrating STZ, but there is a considerable risk of accidentally injecting into the bowel or subdermis which could reduce the diabetogenic effect as well as impact the toxic effect. Previously, intravenous administration has been shown to be a more reliable administration route of STZ when inducing diabetes in mice, 12 which indicates that this route should be considered for possibly reducing variability. On the other hand, when handling larger cohorts of animals, simplicity is important, and given that the subcutaneous route of administration can be used for Alloxan, 37 another glucose analogue used to induce diabetes, and has also proven successful for STZ in rats 38 and guinea pig, 39 this route could also be taken into consideration.
In the study investigating the long-term effect of the two STZ dosing regimens, 5 × 50 and 2 × 100 mg/kg, it was found that the two dosing regimens are equally capable of inducing stable diabetes and nephropathy measured as albuminuria and kidney hypertrophy. This confirms that 2 × 100 mg/kg is a useful dosing regimen for DN studies in 129/Sv mice.
In conclusion, these studies show that the double intermediate dose of 2 × 100 mg/kg is a good alternative to the recommended multiple low dose for inducing diabetes in male 129/Sv mice. No difference could be found between the two dosing regimens regarding impaired stomach emptying and acute kidney injury. Furthermore, the advantages of having to fast, handle and inject the mice fewer times include both lower workload as well as a potential reduction in the stress induced on the mice. The double intermediate dose could thereby refine the STZ-induced diabetic mouse model. These studies once again prove the large variability in the diabetogenic and unwanted effects of STZ, emphasising the need for further optimisation of the STZ-induced diabetic mouse model. Finally, the results presented here suggest that when used to study elements of diabetic kidney injury, this model requires careful consideration of readouts.
Supplemental Material
Supplemental material for Optimising streptozotocin dosing to minimise renal toxicity and impairment of stomach emptying in male 129/Sv mice
Supplemental Material for Optimising streptozotocin dosing to minimise renal toxicity and impairment of stomach emptying in male 129/Sv mice by Sisse A Nørgaard, Henrik Søndergaard, Dorte B Sørensen, Elisabeth D Galsgaard, Constanze Hess and Fredrik W Sand in Laboratory Animals
Footnotes
Acknowledgements
The authors thank Hanne Nord Søndergaard, Nicolai Hansen and Mie Berndorff and technicians in Diabetes Complications Pharmacology for help in the in vivo part of the study, as well as the Laboratory Animal Science Unit at Novo Nordisk A/S. The authors also thank Malik Nygaard Nielsen for assistance with the IHC staining and analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This project is a part of a PhD project funded by Innovation Fund Denmark (4135-00008B) and Novo Nordisk A/S.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
