Abstract
Timely and accurate diagnosis of pregnancy in the laboratory rat is an invaluable endpoint in embryological and reproductive toxicology studies. The purpose of the present study was to assess the feasibility, sensitivity, accuracy and safety of laparoscopic examination of the uterus for detecting pregnancy at an early stage, and defining the number of fetuses in surviving rat models. Female Wistar rats were subjected to a two-port laparoscopic examination of their uterus from day 6 to day 10 post-coitum (pc). Mean recovery time from anesthesia was 148 ± 66 s. A postoperative 4.0 ± 1.8% body weight loss was restored in 3.9 ± 1.3 days. Uterine bulges suggestive of underlying embryonic vesicles could be detected by day 7 pc with a 0% false-negative diagnosis. Comparison of the number of laparoscopically defined uterine bulges (fetal counts) with litter size revealed a 0% underestimation rate of fetal counts, and a 42.9–87.5% overestimation rate from day 7 to day 10 pc. In conclusion, the present study introduced the laparoscopic examination of the uterus and showed it to be a feasible, reliable and safe method of early pregnancy diagnosis in the laboratory rat by day 7 pc.
Keywords
Accurate and timely pregnancy diagnosis of the laboratory rat offers an invaluable tool for biomedical researchers to optimize fertility and reproductive toxicology studies, as well as to better manage rat breeding colonies. In practice, pregnancy can be detected by transdermal palpation of the uterus after days 10–12 post-coitum (pc) with variable sensitivity, 1 and can be confirmed by the birth itself. Indications of pregnancy include increased body weight and food consumption, abdominal and nipple enlargement, as well as mammary development.1,2 Recently, ultrasonographic examination was described as a safe and reliable method of pregnancy diagnosis in the rat by day 11 pc, but without offering an accurate assessment of the number of fetuses. 3 Although exploratory laparotomy could be used for counting fetuses, it would cause extended trauma and could pose a serious risk of adverse effects on fetal development in surviving models.
Laparoscopy is a minimally invasive surgical procedure used in human and veterinary medicine for the diagnosis of a variety of diseases. Under general anesthesia, a very small incision (3–10 mm) is made to the abdominal wall to allow introduction of a trocar to the peritoneal cavity which has been previously inflated with an inert gas. Through that trocar, a specially designed rigid camera is inserted intraperitoneally enabling images to be sent to a video monitor. This method offers the advantage of inspecting abdominal organs in real-time under direct magnified vision, avoiding the need for laparotomy and consequent exposure of organs to environmental conditions. Compared with exploratory laparotomy, diagnostic laparoscopy is significantly less traumatic. It reduces postoperative pain which results in a shorter recovery time, while preserving immune function. 4 According to clinical studies, laparoscopic procedures during pregnancy are considered to be safe.5,6 The laparoscopic approach has been adapted for a number of procedures in rodents as well, such as in partial hepatectomy models, 7 serial parenchymal organ biopsies, monitoring of disease progression by visualization of intra-abdominal organs, and neoplastic cell inoculation into target organs. 8 To this end, the present project was undertaken in order to evaluate the feasibility, sensitivity, accuracy and safety of a laparoscopic examination of the uterus for early pregnancy diagnosis in the laboratory rat.
Animals
Sixty female Wistar rats (Rattus norvegicus), aged 7–8 months and weighing 270–300 g, provided by our laboratory in-house breeding colony were used in this study. They were the 35th inbred generation of Wistar rats obtained from the conventional breeding facility of the Democritos National Center of Physics Research (Athens, Greece). Colony health status was monitored semiannually, and rats were found to be free of Mycoplasma spp., adventitious viruses, respiratory and enteric bacteria, as well as ecto- and endoparasites. The rats were housed in polycarbonate cages (Techniplast, Buguggiate, Italy) in an enriched environment, three rats per cage, at 20–22℃, on a 12:12 h light:dark cycle. They were provided with nest building material and were given a commercial pelleted diet (4RF25; Mucedola, Milan, Italy) and tap water ad libitum. The facilities were operated in accordance with Directive 2010/63/EU which refers to ‘the protection of animals used for scientific purposes’.
Materials and methods
Experimental design
Female rats were mated with male rats in a 1:1 mating scheme. The uterus of the mated female rats were subjected to laparoscopic examination under general anesthesia from day 6 to day 10 pc (6 rats per day of examination) in order to detect uterine horn bulges, suggestive of the presence of embryonic vesicles. The sensitivity of the method was evaluated based on confirmation of pregnancy at birth. The accuracy of the method was evaluated based on a comparison of the number of uterine horn bulges (fetal count) with the number of neonates at the end of the day of parturition (litter size). During the course of the procedure, the duration of anesthesia, pneumoperitoneum, laparoscopic examination and then recovery from anesthesia (righting reflex restoration) were recorded. Postoperatively, body weights were recorded daily to assess the time needed to regain the weights to preoperative levels, thus providing an index of recovery. The experimental protocol was approved by the animal care and use committee of the local veterinary service as complying with the requirements set by the national legislation on the use of animals for scientific purposes (PD 56/2013) which is in accordance with Directive 2010/63/EU.
Mating
Each female rat was caged with a single male rat of verified fertility (with a history of fertile mating) until successful copulation. Detection of sperm in vaginal smears, collected between 09:00 and 11:00 h the following morning, was considered to be evidence of mating. Smears were examined at a magnification of ×100 (KF2; Carl Zeiss, Hamburg, Germany). The day of sperm detection was defined as day 0 pc.
Preparation of animals
Rats were anesthetized by administration of the inhaled anesthetic sevoflurane which was initially provided in a plexiglas chamber (4% in oxygen) and then through a face mask (2% in oxygen). They were secured in a supine position on a heated-surface operating table. Their abdomen was clipped and disinfected with a 10% povidone iodine solution. A 21 G intravenous catheter was inserted intra-abdominally through the umbilicus and connected to an insufflation tube to introduce carbon dioxide up to an intra-abdominal pressure of 5 mmHg.
Instruments – equipment
The following were used in the procedure:
Trocars: Two 5 × 90 mm single incision laparoscopic surgery (SILS) port cannulae (Covidien, Norwalk, CT, USA). Laparoscopic instrument: One 5 × 310 mm duck-jaw laparoscopic forceps (Endo Grasp 5 mm, Auto Suture; Covidien). Endoscope: 4 × 150 mm 30° telescope (A7595 A; Olympus, Center Valley, PA, USA). Laparoscopic tower (Lemke Vision; World of Medicine AG, Ludwigsstadt, Germany).
Laparoscopic procedure
Two 5 mm trocars were introduced transdermally into the abdominal cavity of the rat after performing a stamp incision with a No. 11 blade at the insertion site (Figure 1). The first trocar with an insufflation side port was introduced into the left hind quadrant of the abdomen. The insufflation tube was then connected to the insufflation side port, whereas the intravenous catheter used for pneumoperitoneum installation was left in place to facilitate the relief of gas and equilibration of the intra-abdominal pressure. The second trocar was introduced into the right hind quadrant of the abdomen at the level of the first trocar. The endoscope was introduced from the left hand side trocar, while the laparoscopic grasper was introduced from the right trocar in order to facilitate intestine and body fat manipulation, which was necessary for uterine horn exposure. Each uterine horn was inspected throughout its entire length, from the ovary to the uterine body. Grasping of uterine horns was avoided to minimize the risk of potentially adverse effects on fetal development. After the examination, the trocars were withdrawn, the peritoneal cavity was deflated and the abdominal wall was closed in two layers using 2-0 polyglycolic sutures. At the end of the operation, each rat received antibiotic (a single subcutaneous injection of enrofloxacin, 10 mg/kg) and analgesic prophylactic (single subcutaneous injection of meloxicam, 2 mg/kg) treatments. Pain was assessed and recorded on a pain assessment score sheet based on food consumption, body weight, activity and grooming.
Trocar placement for two-port laparoscopic examination of the rat uterus.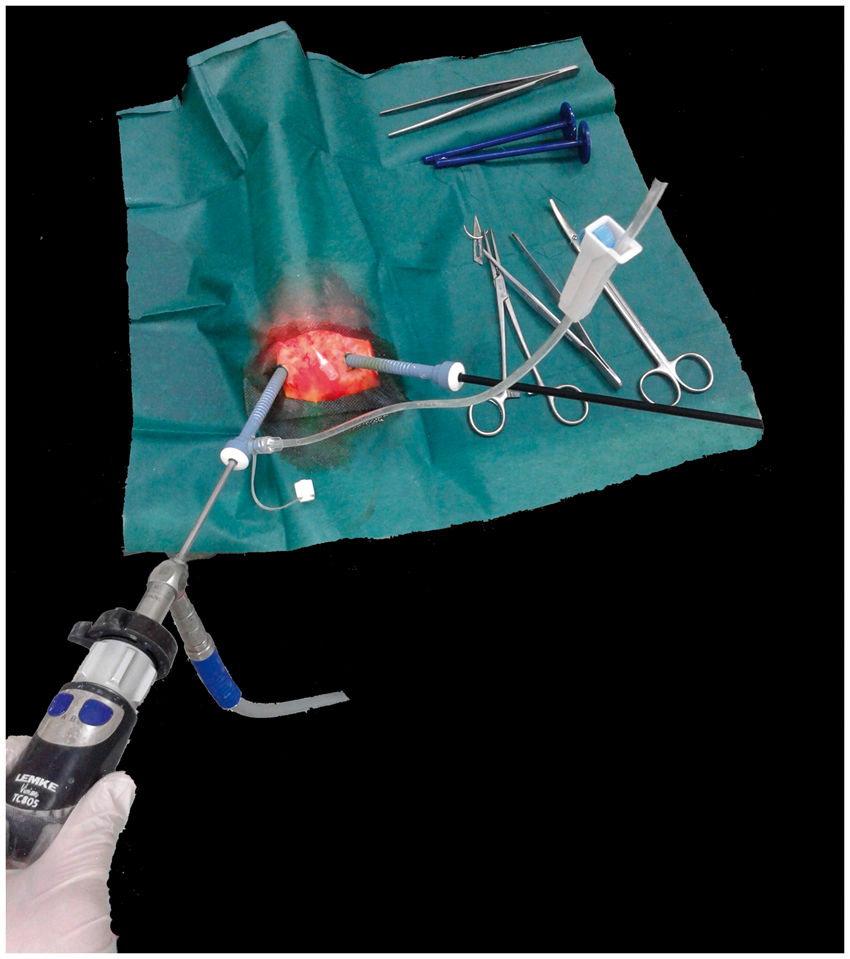
Results
All the rats that were found to be pregnant after laparoscopic examination of their uterus gave birth. The anesthesia lasted 40 ± 7 min (average ± standard deviation), pneumoperitoneum 21 ± 6 min and laparoscopic examination (from insertion of the endoscope to deflation of the peritoneal cavity) 10 ± 5 min. Recovery from anesthesia was achieved after 148 ± 66 s. The maximum postoperative body weight loss was 4.0 ± 1.8%, while body weight was restored in 3.9 ± 1.3 days from the day of operation.
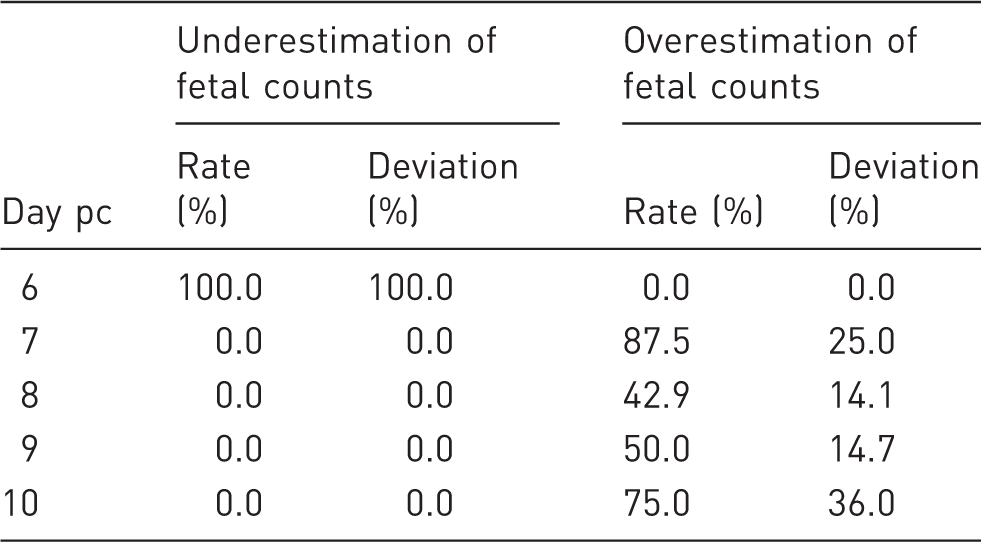
Uterine horn bulges were detected by day 7 (Figure 2). The percentage of false-negative pregnancy diagnosis was 100, 0, 0, 0 and 0% on days 6, 7, 8, 9 and 10, respectively. Fetal counts, when compared with litter size, were found to be underestimated in all animals on day 6, but in none of the animals on days 7–10 (Table 1). On the other hand, fetal counts were higher than litter size on days 7–10. Fetal counting revealed an unequal distribution of fetuses in the uterine horns (data not shown).
Laparoscopic view of the pregnant rat uterus on days 6–10 post-coitum (pc). Uterine horn bulges (arrows) denote fetuses. NP: not pregnant. Accuracy of laparoscopic fetal counting. Deviation %: % deviation of laparoscopic fetal counts from litter size; pc: post-coitum.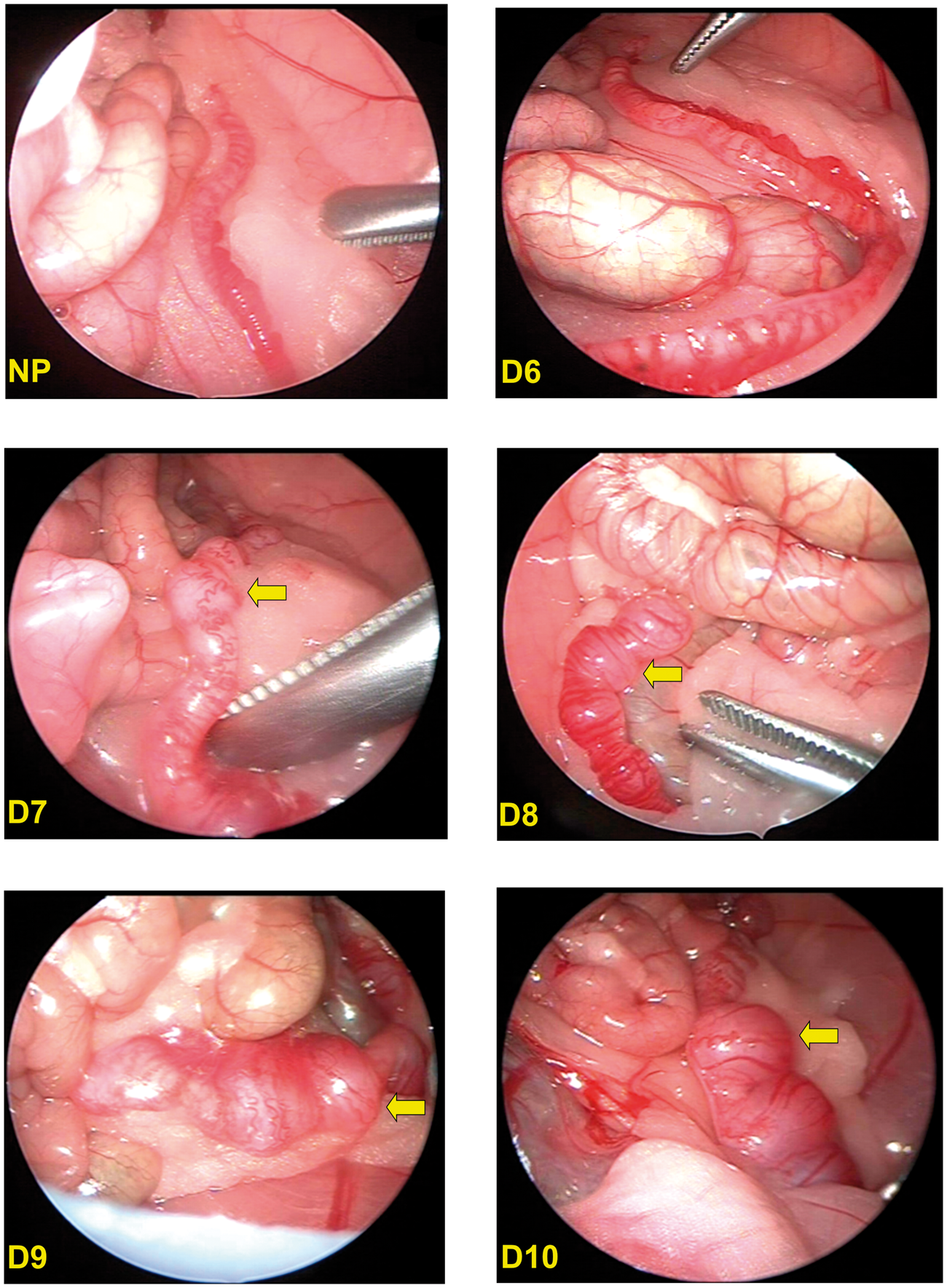
Discussion
The present study introduced laparoscopic examination of the uterus for early pregnancy diagnosis in the laboratory rat. It provided sufficient evidence on the technical feasibility, sensitivity, accuracy and safety of the method for detecting pregnancy and counting of fetuses by day 7 pc.
According to embryological data, rat blastocysts descend to the uterine lumen by day 4 pc, are distributed to the sites of implantation by the end of day 5, and begin to increase in size on day 6. 9 Bulges of the uterine horns resulting from intraluminal blastocyst enlargement were laparoscopically detectable as early as day 7 pc. The laparoscopic approach offers a considerably earlier accurate pregnancy diagnosis compared with the formerly described ultrasonographic examination of the uterus with which embryonic vesicles could be detected by day 9 with a 25% false-negative diagnosis, whereas it has a 100% accuracy by day 11. 3
Laparoscopic examination of the uterus enabled accurate fetal counting, an important endpoint in embryological and reproductive toxicology studies, without necessitating post-laparotomy or post-mortem assessment. Interestingly, the employment of this method, which offers the advantage of indisputable fetal counting under direct magnified vision of the uterus through a video monitor, revealed a high incidence of early embryonic deaths as documented by laparoscopic overcounting of fetuses compared with litter size. Furthermore, it emphasized the importance of fetal counting at an early stage of pregnancy which can be used as a reliable index of the conception rate. To the authors’ knowledge, there are no available studies on assessing spontaneous embryonic death in the laboratory rat. This would require comparison of fetal counts at an early stage of pregnancy with the number of live embryos at mid and/or late pregnancy using a non-invasive technique. Direct injury, as a potential cause of selective fetal loss, was excluded since manipulations of uterine horns during laparoscopic examination were avoided. On the other hand, anesthesia, pneumoperitoneum and/or post-operative stress might have induced a more generalized effect (e.g. abortion). Apart from fetal absorption during middle or late pregnancy, which warrants further investigation, one should also consider cannibalism of stillborn puppies as a potential reason for the discrepancy noted between laparoscopically-based fetal counts and litter size.
Diagnostic laparoscopy, although an interventional method, is a minimally invasive surgical procedure. The number of trocars used defines the extent of trauma and hence affects the time needed for recovery. 10 The one-port access to the peritoneal cavity was evaluated in a series of pilot experiments, which highlighted the need for the use of a laparoscopic grasper at the expense of an additional trocar insertion, to facilitate manipulation of intestines and abdominal fat in order to expose uterine horns over their entire length and hence enhance sensitivity and accuracy of the method. Although a working laparoscope with a built-in working channel could have been an alternative to minimize invasiveness of the procedure, this would substantially increase the cost of the instruments needed and would also eliminate the advantage offered by the two-port access with a side view of the uterine horns while performing laparoscopic manipulations.
There is compelling evidence that laparoscopy is a considerably less harmful procedure compared with laparotomy in terms of postoperative pain, recovery time and immune response. 4 Yet, it requires general anesthesia, intraperitoneal insufflation of gas, abdominal wall penetration and essential manipulations of splanchnic organs. Inevitably, these procedures result in pain and distress for the animal under study. To this end, the use of inhalation anesthetics that warrant short recovery times, the establishment of pneumoperitoneum at low intraperitoneal pressure, the use of small diameter trocars that necessitate tiny abdominal wall incisions, the performance of delicate manipulations of splanchnic organs, and the application of efficient analgesic treatment contribute to a reduction in animal suffering. According to our findings, the laparoscopic procedure had minimal adverse effects on the general health of the rats as indicated by the short recovery times recorded. Recovery from sevoflurane anesthesia was rapid and a 4% body weight loss was restored in approximately four days, suggestive of full postoperative recovery.
The advantages of not only diagnosing pregnancy at an early stage and accurately determining the number of fetuses, but also of causing only minimal and reversible consequences in the health status of rats, while avoiding unnecessary experimentation on animals to be euthanized, contribute to the promotion of two of the 3R principles of reduction and refinement in the use of laboratory animals for scientific purposes. Nevertheless, one must also take into account the transient, although mild, deterioration of the general health status of animals when designing toxicological studies.
Rodent diagnostic laparoscopy offers a promising tool for biomedical researchers who incorporate technological advances in surgical equipment into experimental protocols. The present study introduced laparoscopic examination of the uterus and demonstrated it to be a safe, reliable and accurate method of early pregnancy diagnosis in surviving rat models.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
