Abstract
Chinchillas are popular as laboratory models and companion animals, and they can be affected by a variety of infectious and non-infectious neurological diseases. Little information is available on making a neurological diagnosis in this species, in part because the neurological examination has not been standardized in chinchillas and the expected physiological findings in healthy chinchillas have not been reported. In this study, a standardized neurological examination was performed on 30 clinically normal chinchillas. The perineal reflex and the menace response were absent in all chinchillas evaluated and so should not be used as functional tests. Several tests were consistently positive, such as the oculocephalic reflex, maxillary, mandibular and auricular sensations, withdrawal reflexes, thoracic limb paw replacement, and the wheelbarrow test. Other tests, such as the cutaneous trunci reflex and pelvic limb paw replacement, had variable responses. Based on these findings, it is advised that clinicians prioritize tests that can be easily performed without undue stress to the animal, and give consistent results in a healthy patient.
Chinchillas (Chinchilla lanigera) are frequently used in laboratory animal research. They are also popular as companion animals. Chinchillas are susceptible to a range of infectious and non-infectious neurological diseases. These include bacterial otitis, herpes virus infection, listeriosis, lead toxicity and trauma.1–3 While existing literature have documented the clinical signs of these diseases, information on making a neurological diagnosis is lacking. In particular, there is little evidence-based information on how to perform a neurological examination in chinchillas, and what findings would be considered neurologically ‘normal’ in this species.
Performing a complete neurological examination in most rodent species remains challenging and obtaining a consistent and valid neurological assessment in rodents is considered to be difficult. 4 Small body size and aversion to handling can hamper attempts to perform tests, while stress can confound the results. It has therefore been recommended to observe the animal’s movement and to check for neurological abnormalities prior to handling.4,5 It has also been recommended to adopt neurological examination techniques that have been established in domestic rabbits, and to extrapolate these as much as possible for rodents. 4 While extrapolation is useful to some extent, one must be aware of natural species variations. The limited information available on normal neurological findings in chinchillas has arisen from ophthalmological research. In one study, 100% of chinchillas had a direct pupillary light reflex (PLR), but 90% lacked a dazzle reflex and 97% lacked a menace response. 6 The corneal reflex has been used to quantify corneal sensitivity in chinchillas but there is disagreement as to how their corneal sensitivity compares with that of other species. Spontaneous blinking is agreed to be infrequent; blinking rates of 2–4 blinks per 10 min have been reported.6,7 Chinchillas’ pupils are typically semi-constricted in ambient light and pupillary response to pharmacological dilation is generally poor. 7
In contrast to these detailed ophthalmological studies, there is little if any information on non-ocular neurological tests in chinchillas. Given this considerable gap in the literature, the present study aimed to establish baseline physiological parameters for neurological examination in healthy chinchillas.
Materials and methods
Ethical statement
This study was approved by the University of Wisconsin–Madison, School of Veterinary Medicine’s animal care and use committee. Animal use complied with the National Research Council Guide for the Care and Use of Laboratory Animals standard and the University of Wisconsin–Madison is AAALAC-accredited.
Animals
Thirty (16 male, 14 female) adult chinchillas (Chinchilla lanigera), ranging in age from one to five years, with body weights ranging from 0.46 to 0.84 kg (median 0.65 kg) were obtained from a commercial breeder (R & R Chinchilla Inc, Jenera, OH, USA). Animals were housed in a climate-controlled room with a 12 h light cycle, from 07:00 to 19:00 h. Room temperature was maintained between 21 and 23℃ and relative humidity ranged between 40 and 55%. The chinchillas were maintained in individual cages (Allentown 6-cage Rabbit Housing Unit; Allentown Inc, Allentown, NJ, USA) measuring 0.69 m × 0.69 m × 0.46 m with perforated plastic pans. Each cage contained a plastic hide box, as well as cardboard tubes and a piece of natural manzanita wood for foraging. The opportunity to exercise and socialize in a 1.8 m × 0.9 m playpen, which contained a dust bath, was provided at least once weekly. The chinchillas were offered a commercial pelleted rabbit diet (MannaPro Rabbit pellets; MannaPro Products, LLC, Chesterfield, MO, USA) and tap water from a rabbit ball-tipped water bottle. All chinchillas were acclimatized to the housing conditions for at least eight weeks prior to starting the experiments. A complete physical examination was performed on each animal after acquisition, and food intake, fecal output and body weight were monitored regularly. Only animals with normal physical examination findings were used in this study. The animals were part of a research colony and were previously used in various non-invasive experimental studies. Following completion of this neurological examination study, the animals were returned to the research colony.
Neurological examination
The neurological examinations were performed by a board-certified veterinary neurologist (HR). Materials used for the examination included a counter with a non-slippery surface, cotton-tipped applicators, a pair of hemostats, a pen torch and a reflex hammer.
The neurological examination comprised assessment of mentation, cranial nerve tests, spinal reflexes and postural reactions. The tests were extrapolated from the companion animal neurological examination 8 and were assessed as to their feasibility in chinchillas during a pilot study. Tests deemed unfeasible due to anatomical or handling problems (e.g. the gag reflex) were not included in the current study reported in this paper. Gait analysis was not evaluated in this study.
Each animal was handled carefully with the aim of minimizing stress. If an animal was struggling excessively or appeared stressed during the examination, 20–30 s breaks were given, before the examination recommenced. Restraint by an assistant was deemed necessary in order to assess cranial nerves and spinal reflexes. For postural reactions, the animal was restrained by the examiner on or over a non-slippery surface. The neurological tests below were generally performed as for companion animals. 8 However, certain chinchilla-specific differences were noted and are described below.
Cranial nerve tests included the menace response; the pupillary light, palpebral, corneal (Figure 1a) and oculocephalic reflexes; and auricular, maxillary and mandibular sensations. The oculocephalic (vestibulo–ocular) reflex was evaluated by raising the upper eyelid in order to visualize the sclera, which enabled detection of eye movement (Figure 1b). Maxillary, mandibular and auricular sensations were assessed by touching the upper lip, lower lip and the inside of the ear in turn with the end of a cotton-tipped applicator and observing for local twitching, head shaking or movement of the head away from the stimulus.
(a) Assessment of the corneal reflex. The corneal surface is touched with a moistened cotton-tipped applicator. The chinchilla should respond by blinking and retracting its eye. (b) Assessment of the oculocephalic reflex. The upper eyelid is raised with two fingers to visualize the margins of the iris. The assistant is moving the chinchilla sideways. A physiological nystagmus should be apparent.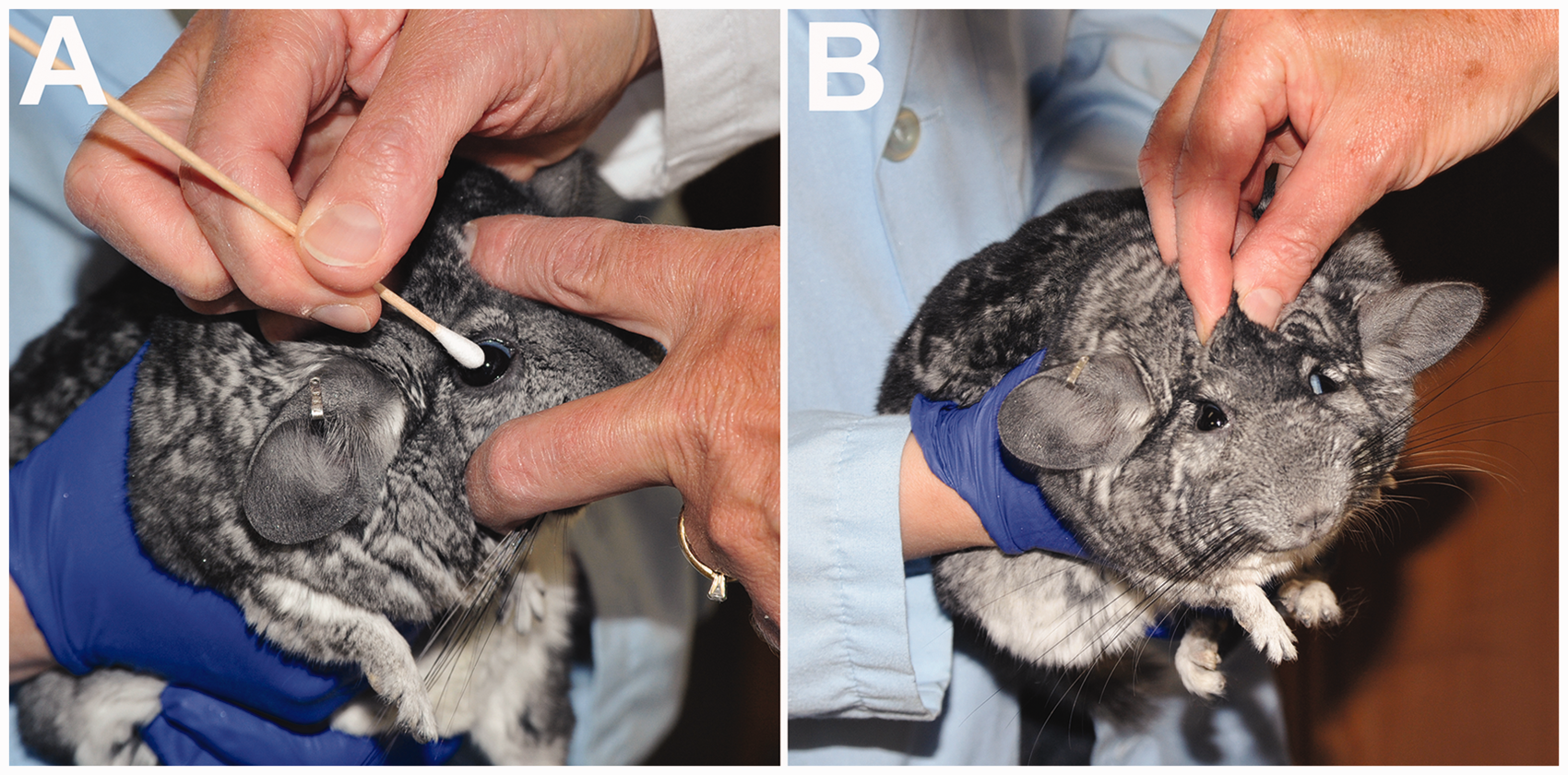
Spinal nerve tests included the thoracic limb withdrawal, pelvic limb withdrawal, patellar, perineal and cutaneous trunci reflexes. The patellar reflex was elicited by tapping the tendon with the handle of the reflex hammer (Figure 2). When testing the cutaneous trunci reflex, care was taken to grasp skin rather than fur with the hemostats.
Assessment of the patellar reflex. The stifle is slightly flexed and the patellar tendon gently tapped with the handle of a reflex hammer. The stifle should extend cranially. The chinchilla can be held by the examiner (a) or by an assistant (b).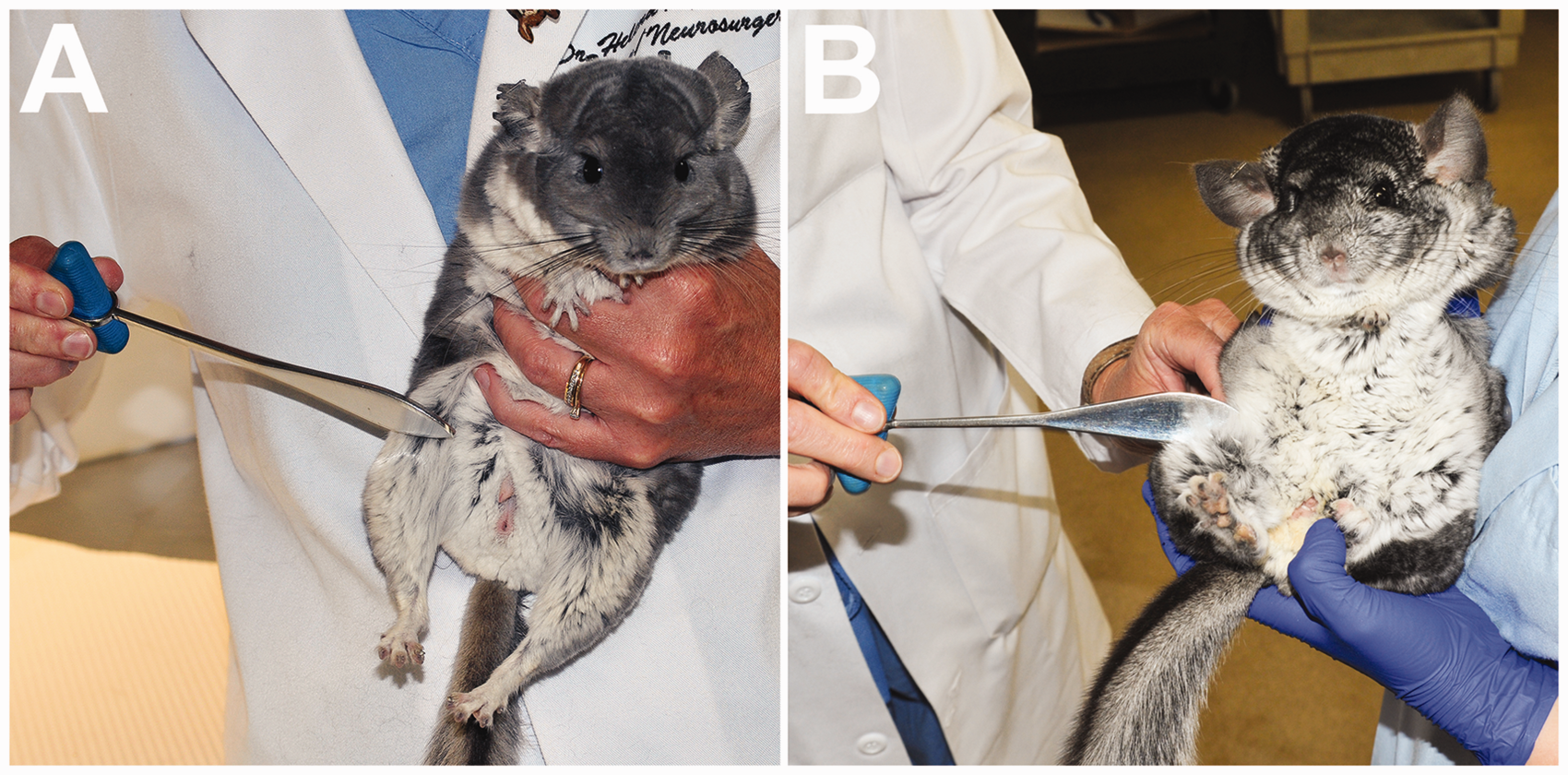
Postural reactions included paw replacement, visual placing, tactile placing, hopping, hemiwalking, wheelbarrowing and extensor postural thrust. To assess paw replacement, the chinchilla was restrained with one hand under the ventrum while using the other hand to place the dorsum of each paw on the examination table (Figure 3a). Both visual and tactile placing were tested in the thoracic limbs by supporting the chinchilla’s body and bringing it towards the edge of the examination table, with the eyes covered if testing tactile placing (Figures 3b and c). In the pelvic limbs, visual placing was impossible but tactile placing was attempted (Figure 3d). Hemiwalking (Figure 3e), hopping (Figure 3f), wheelbarrowing (Figure 3g) and extensor postural thrust (Figure 3h) were also tested similarly to dogs and cats.
Assessment of postural reactions. (a) Paw replacement: the chinchilla is supported with one hand under the ventrum and 1–2 fingers between the thoracic limbs. The other hand is used to place the dorsal aspect of the paw on the countertop. A positive response is seen if the chinchilla immediately returns the paw to its natural position. (b) Visual placing: the chinchilla should place its thoracic limbs on the countertop on seeing the edge of the counter. (c) Tactile placing of thoracic limbs: one hand is used to cover the eyes while the other brings the chinchilla forwards so that the dorsal aspect of the paw meets the edge of the counter. The chinchilla should place its paws on the counter. (d) Tactile placing of pelvic limbs. (e) Hemiwalking: the body is supported and two ipsilateral limbs are lifted off the ground. (f) Hopping: the chinchilla’s body is supported with three limbs off the ground. For hopping and hemiwalking the animal’s weight should be gently shifted laterally to the weight-bearing limb(s). The test is positive if the animal hops to take the weight. Note the non-slip surface taped to the countertop. (g) Wheelbarrow test: the trunk is supported ventrally with one hand, and the hindquarters are elevated to enter the wheelbarrow position. The chinchilla should respond by running forward on its thoracic limbs. Placing one finger on either side of the neck can help to slow down the chinchilla and prevent escape. (h) Extensor postural thrust: the chinchilla is lifted off the ground and then lowered, pelvic limbs first, to the surface.
Data analysis
For the purposes of this study, a ‘positive’ result was one that would be considered positive if the test were performed in a dog or cat, e.g. pupillary constriction for PLR and physiological nystagmus for the oculocephalic reflex. The data are reported as frequencies of total numbers as well as percentages unless otherwise specified.
Results
Summary of neurological findings in clinically healthy chinchillas.
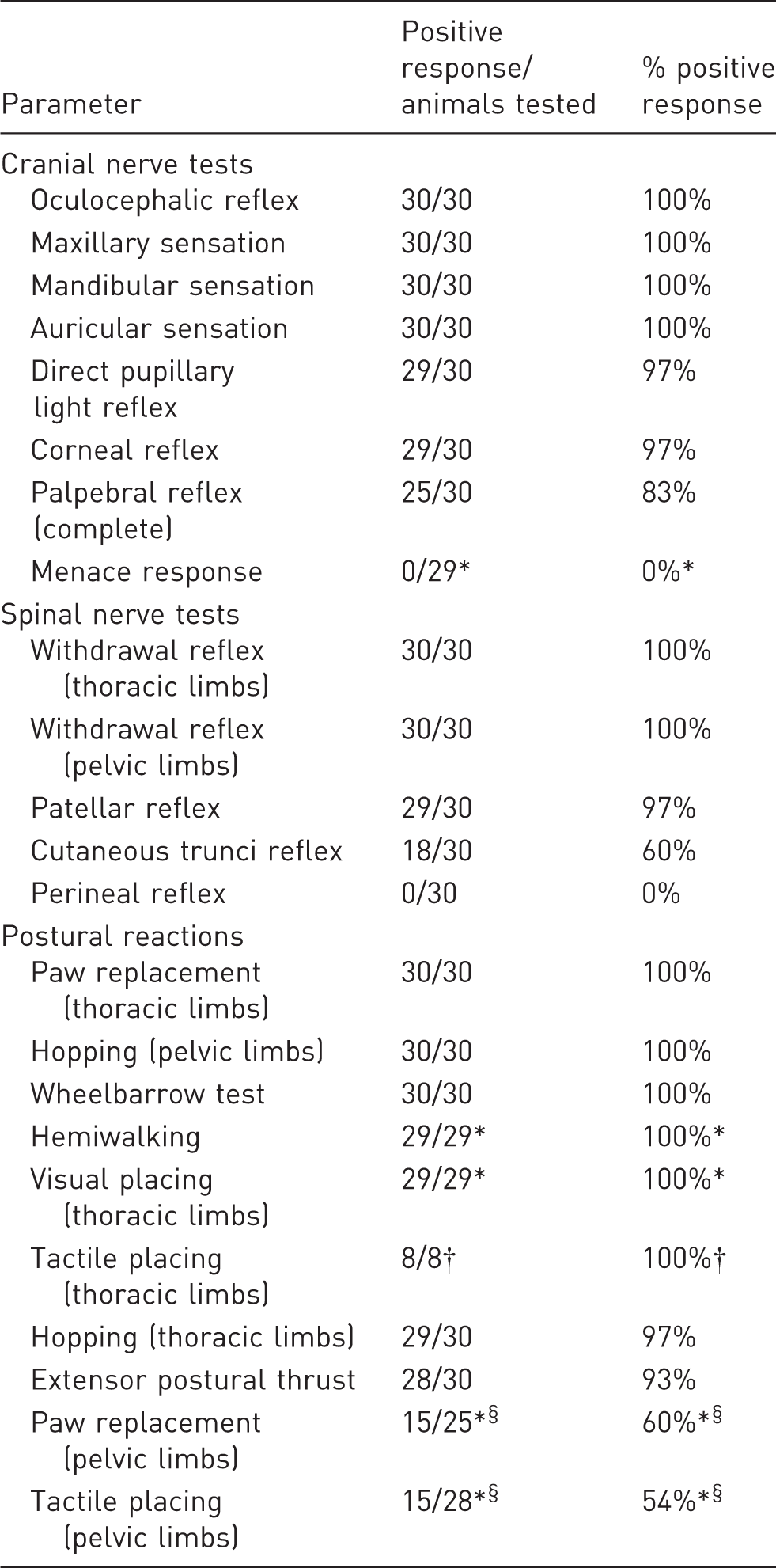
Data missing for one animal. †Tactile placing was only tested in eight animals. §One or more animals could not be assessed due to their temperament.
Cranial nerve tests
Tests for the oculocephalic reflex and the maxillary, mandibular and auricular sensations were positive in all the chinchillas evaluated. A direct PLR was demonstrated in 29/30 animals, but could not be assessed in one animal as the pupils were very constricted and the act of constricting could not be observed. Corneal stimulation produced blinking in all the animals, and corneal retraction in 29/30 animals. The palpebral reflex was present to some degree in all animals: 25/30 animals showed a complete blink and five an incomplete blink. The menace response was absent in all animals evaluated.
Spinal reflexes
Withdrawal reflexes were positive in all four limbs of every chinchilla. The patellar reflex was present bilaterally in 29/30 and unilaterally in 1/30 chinchillas. The cutaneous trunci reflex was found to be bilateral in 18/30, unilateral in 2/30 and absent in 10/30 animals, but was also occasionally noted to be variable within the same individual. By contrast, the perineal reflex was very clearly absent in all the chinchillas evaluated.
Postural reactions
Thoracic limb paw replacement was positive in all the chinchillas. Pelvic paw replacement could not be assessed in 4/29 animals due to temperament. Of the remaining 25 animals, only 15 were positive. For the thoracic limb, visual and tactile placing were positive in 8/8 animals tested. Tactile placing of the pelvic limbs was positive in 15 animals, negative in 13 and could not be assessed in one as the animal withdrew its limbs.
Hopping reactions were positive in all four limbs in 29/30 chinchillas; the remaining animal showed a deficit in one thoracic limb. Hemiwalking and wheelbarrowing were positive in all animals evaluated, while extensor postural thrust was positive in 28/30 animals.
Observations on the neurological examination
During the study, the following observations were made regarding the practical aspects of the neurological examination in chinchillas:
Direct PLR was difficult to observe as chinchilla pupils constricted extremely quickly; complete darkness prior to testing was essential. For the same reason, testing the consensual PLR was deemed infeasible. The palpebral reflex was incomplete at first attempt in many animals, but became complete on further stimulation (4 to 5 times). The presence or absence of the withdrawal reflex was simple to assess; however, subtle variations in reflex quality were challenging to detect due to the speed and small size of the movements. Eliciting the patellar reflex was feasible but could take time and effort due to the limb being small and occasionally tense. By slightly abducting the coxofemoral joint the reflex could be more easily produced. The skin twitch of the cutaneous trunci reflex was challenging to assess due to the thick fur coat and the tendency of the animals to fidget. Paw replacement was relatively easy to test in the thoracic limbs; however, pelvic paw replacement was difficult to test and to interpret. Several chinchillas withdrew their pelvic limbs and objected to further manipulation, perhaps because the position required for this test is highly abnormal for a semi-plantigrade limb. Tactile placing was also more challenging to assess in pelvic limbs as several animals wriggled their legs in objection to handling. Hopping, hemiwalking and wheelbarrowing were simple to test, as the small body size enabled easy support of the body and non-tested limbs, and were well tolerated. Extensor postural thrust could take 3 to 4 attempts to elicit.
Discussion
As shown in this study, it is feasible, to some extent, to extrapolate the standard veterinary neurological examination to chinchillas. Several tests, such as the gag reflex or the PLR, were ruled out or were complicated by the animal’s anatomy and temperament; but other tests, such as hopping, were found to be simple to perform. Based on our findings we recommend prioritizing tests which are easily performed without undue stress to the chinchilla, and which give a consistent result in the healthy patient. For example, the possibility of a neurological lesion affecting a pelvic limb is best evaluated by observing the animal’s movement and testing its withdrawal reflex and hopping reaction; time spent on pelvic limb paw replacement and tactile placing is likely to be less useful. Tests that cause stress are more difficult to perform and interpret, while tests that have variable results in a healthy patient are less likely to be diagnostic in a sick one.
A consistent and notable result in this study was the lack of a perineal reflex in chinchillas. To the best of our knowledge this has not previously been reported; indeed no record could be found of any other mammalian species lacking the perineal reflex. The innervation of the region has been documented in the chinchilla. The pudendal nerve arises from S1–S2 and splits into superficial and deep branches to innervate the skin and muscles of the anal and perianal regions, while the caudal rectal nerve emerges from S2–S3 to innervate the sphincter muscles of the anus and the skin of the anal region. Curiously, these origins and routes of innervation are more similar to dogs and cats than to guinea pigs and rabbits. 9 However, there is currently no clear anatomical or physiological reason for an absence of the perineal reflex in chinchillas. Further studies will be needed to verify our result in other chinchilla populations, to determine whether the reflex is truly absent or merely suppressed by stress, and to determine why chinchillas in particular, should lack it.
The other consistently negative test result was the menace response; this supports previous work in which 97% of chinchillas lacked a menace response. 6 The reason for this is unclear, but as the menace response is learned rather than reflexive, it is possible that chinchillas have different means of evading threats. A perhaps more likely possibility is obliteration of the response by stress: in rabbits, the common failure of the menace response is attributed to nervousness and ‘freezing’ on clinical examination. 10 The palpebral reflex was also found to have different ‘normal’ limits, with 17% of chinchillas showing only a partial blink in this study. In some animals that blinked partially at first, a complete blink could be elicited on subsequent attempts. This too could suggest stress-induced inhibition. However, a partial failure of the palpebral reflex, coupled with the lack of a menace response, raises the question of how chinchillas protect their eyes from sudden threats.
Some of the data obtained were inevitably limited by handling difficulties. Although great effort was made to distinguish between being unable to perform a test (i.e. due to the animal’s anatomy or temperament) and being able to perform a test but getting a negative result, it is possible that our less consistent results are due to practical limitations rather than, or in addition to, individual variation. Such results include the cutaneous trunci reflex (60% positive), pelvic limb paw replacement (60% positive) and pelvic limb tactile placing (54% positive). With the cutaneous trunci reflex, it could be difficult to see a clear skin twitch. For both paw replacement and tactile placing, it was difficult to position the pelvic limbs appropriately for the test, and movement of the limbs may have been an objection to handling rather than a true postural reaction. In addition, with the degree of manipulation required to test pelvic limb paw replacement, it is possible that reactions observed had withdrawal components rather than truly indicating paw proprioception. However, anyone attempting to perform these tests in a clinical setting will face similar limitations, and so recognizing that these challenges exist is arguably more important than determining the exact proportion testing positive. Finally, as the chinchillas in this study came from one source, results ought to be verified using other chinchilla populations.
In conclusion, this study set out to define ‘normal’ neurological parameters in healthy chinchillas. The neurological tests performed varied in their feasibility and in the consistency of their results, and it is hoped that the data will enable clinicians to prioritize the least stressful, most reliable tests in their neurological examinations. ‘Normal’ findings of particular note included the lack of a menace response and the lack of a perineal reflex. These tests therefore cannot be used to evaluate cranial nerve, cerebral or sacral nerve function in chinchillas. The mechanism by which these responses are lacking should be investigated further.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
