Abstract
The aim is to create a new and safe experimental model of radiation-induced neurovascular histological changes with reduced morbidity and mortality for use with experimental microsurgical techniques. Seventy-two Sprague–Dawley rats (250–300 g) were divided as follows: Group I: control group, 24 rats clinically evaluated during six weeks; Group II: evaluation of acute side-effects (two-week follow-up period), 24 irradiated (20 Gy) rats; and Group III: evaluation of subacute side-effects (six-week follow-up period), 24 irradiated (20 Gy) rats. Variables included clinical assessments, weight, vascular permeability (arterial and venous), mortality and histological studies. No significant differences were observed between groups with respect to the variables studied. Significant differences were observed between groups I vs II–III regarding survival rates and histological changes to arteries, veins and nerves. Rat body weights showed progressive increases in all groups, and the mortality rate of the present model is 10.4% compared with 30–40% in the previous models. In conclusion, the designed model induces selective changes by radiotherapy in the neurovascular bundle without histological changes affecting the surrounding tissues. This model allows therapeutic experimental studies to be conducted, including the viability of microvascular and microneural sutures post radiotherapy in the cervical neurovascular bundle.
Treatment of soft-tissue sarcomas (STS) usually requires a combination of surgery, radiotherapy (RT) and, occasionally, chemotherapy. Common external irradiation protocols are neoadjuvant (preoperative) or adjuvant (postoperative). 1 Neoadjuvant RT allows the administration of lower doses (Gy) and can reduce tumor volumes preoperatively, facilitating the subsequent surgical resection. However, it has been related to poorer healing and to acute local postoperative complications such as wound dehiscence and infection.2 –4
After tumor resection, among reconstructive techniques, microsurgical free flaps offer a wide range of options for managing tissue defects, including structural, functional and coverage options.5 –8 Clinical conditions may require microanastomosis to be performed in irradiated vessels or nerves, a challenge for surgeons due to the histological changes caused within the vascular wall and nervous tissue.9 –11
The success of microsurgical reconstruction depends on several factors including the permeability of the pedicle vessels9,11 –13 and the timing between the neoadjuvant radiation, tumor resection and reconstruction surgery.14,15
In microsurgical experimental studies with animals, the cervical neurovascular (NV) bundle of the rat is optimal due to its accessibility, size, vessel characteristics and microanastomosis viability. 16 However, studying the effect of RT on vascular microanastomosis causes an increase in rats' morbimortality after cervical/cranial external irradiation.17 –20
The aim of the present study is to create a new and safe rat model of RT-induced NV histological changes for microsurgical research, reducing the morbimortality described in previous models.17 –20
Animals
Seventy-two adult male Sprague–Dawley rats weighing 250–300 g were obtained from Janvier Labs (Roubaix, France). None of the rats had been used previously in other studies and none were excluded from the present study. The rats received standard chow and water ad libitum. They were kept in cages in groups of three and on a 12 h light/dark schedule and constant temperature (22 ºC) for two weeks prior to the surgery, without environmental enrichment.
Materials and methods
Experimental procedures were performed at the Oncologic Radiotherapy and Pathology Departments of the Institut de Recerca Vall d’Hebron (VHIR) and Hospital Universitario Vall d’Hebron, Barcelona, Spain. The Institut de Recerca Ethics Committee and the Hospital Universitari Vall d’Hebron (Universitat Autònoma de Barcelona) Ethics Committee approved the experimental protocol. All animals received care in compliance with the Principles of Laboratory Animal Care formulated by the National Society for Medical Research and the Guide for Care and Use of Laboratory Animals prepared by the National Institutes of Health (publication no. 80–23, revised 1985) and the Spanish law for the protection of experimental animals (Real Decreto 223, 1988).
Experimental design
Animals were randomly assigned into three groups, each consisting of 24 rats: Group 1 (non-irradiated, six-week follow-up period); Group 2 (irradiated, two-week follow-up period); and Group 3 (irradiated, six-week follow-up period).
Anesthesia protocol
The rats were anesthetized with an intraperitoneal injection of 2.5 mL (1 mL every 100 g) of a mixture of ketamine hydrochloride (50 mg/kg, Ketalar; Parke Davis, Eczacibasi, Istanbul, Turkey) and xylazine (5 mg/kg, Rompun; Bayer AG, Leverkusen, Germany), and weighed. After anesthesia, all rats were injected with 5 mL of Ringer's lactate subcutaneously to avoid dehydration. Analgesia was administered twice daily after irradiation to all animals (buprenorphine subcutaneously, 0.01 mg/kg).
Irradiation
Under anesthesia, a computed tomography (CT) was performed to study the cervical topography (Figure 1), to delimitate the planning target volume (PTV), and to calculate the time of irradiation (Figure 2). Due to the shallowness of the PTV a cobalt unit was used, ensuring that the maximum dose was 0.5 cm deep.
(a) Computed tomographic study of the cervical topographical. Depth, characteristics and localization study of the cervical neurovascular bundle. (b) Coronal section. (c) Sagittal section. (d) Three-dimensional reconstruction. Radiation dosimetry calculation of interaction with the cervical neurovascular bundle. (a) Coronal section. (b) Axial section. (c) Sagittal section.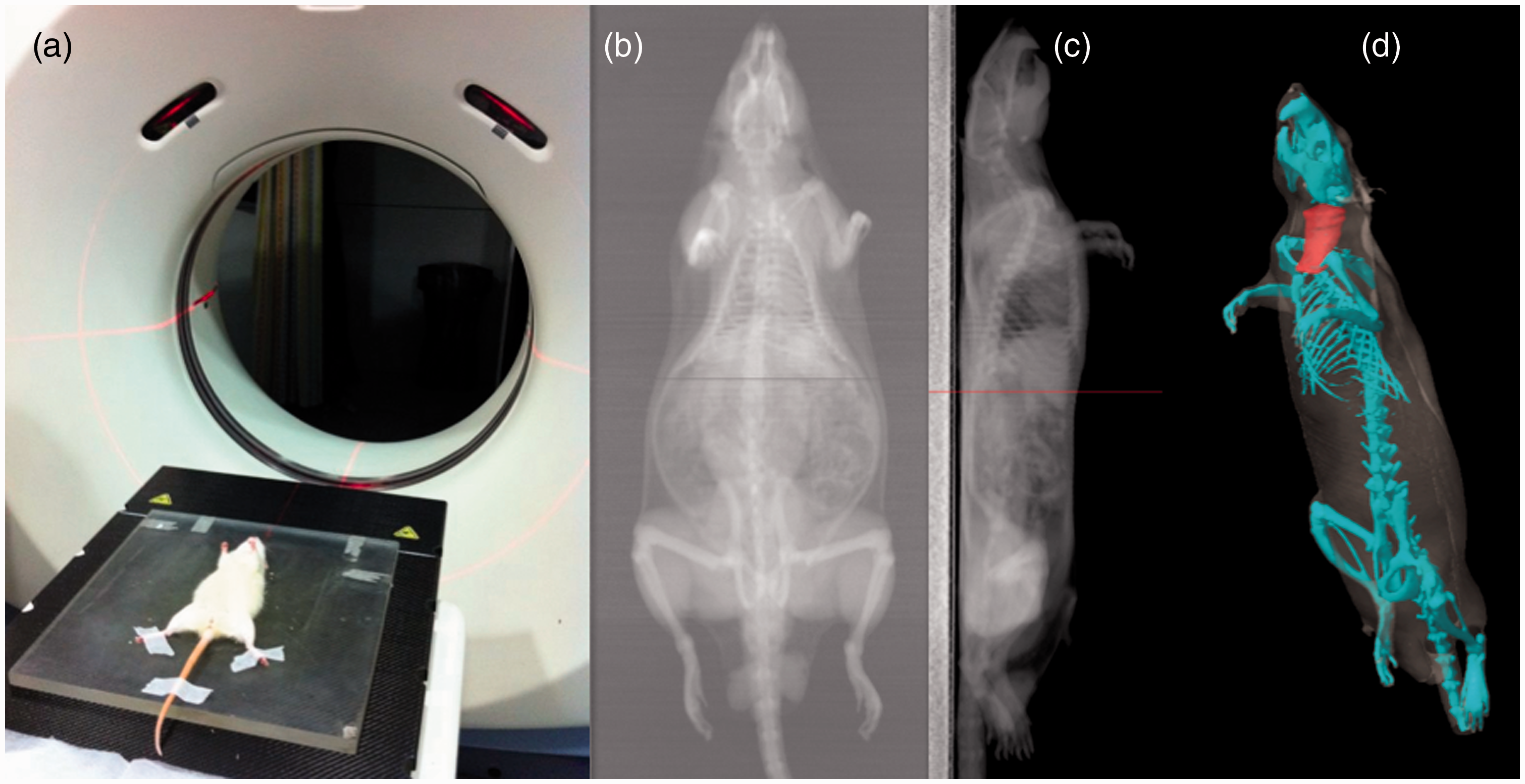
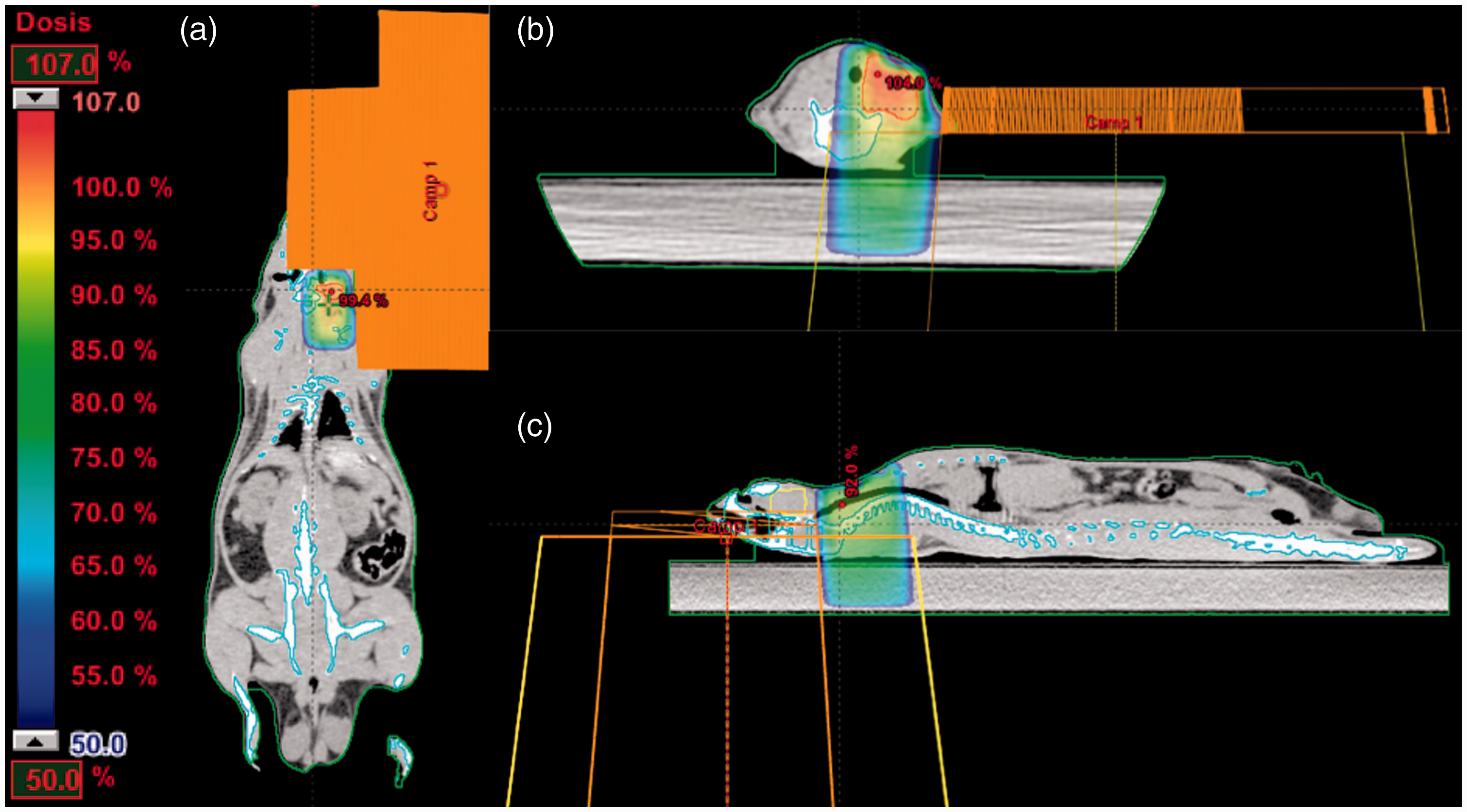
A cross-shaped Cerrobend plate was designed to irradiate four rats simultaneously (2 × 2 field irradiation), preserving structures of the midline cervical such as the esophagus and trachea (Figure 3). A 2 × 2 cm frontal field covered only the left hemicervical region, which was treated with gamma rays from cobalt-60 with a beam energy of 1.25 MeV at a distance of 20 cm from the source field to the skin (Figure 3).
(a) Cobalt-60 radiotherapy treatment unit Theraton. (b) Simultaneous four 2 × 2 cm field irradiation. (c) 4 cm2 frontal field over the left hemicervical region. *Crossed-shaped Cerrobend plate placed in the middle of the radiation beam.
The calculation was done using the pencil beam convolution algorithm (version 7.0.5) in the Eclipse treatment planning system (Varian Medical Systems, Palo Alto, CA, USA). The prescription was 100% in the center of the PTV.
The irradiated group exposure was done in subgroups of four rats simultaneously at a single fraction of 20 Gy, irradiating at an average dose rate of 2.0 Gy/min. A radiographic verification of each rat's positioning was done before starting RT. The animals in the control group were anesthetized, but did not receive any radiation.
Clinical evaluation
Independent veterinarians from VHIR evaluated all the animals clinically. The study was blind for all authors. The rats were evaluated daily during their first post-RT week and weekly thereafter until the conclusion of the study. Animal observation was based on the general appearance of the animal, body posture, mobility, feces appearance and quantity, body weight, rectal temperature, type of breathing, heart rate, ocular appearance, body and irradiated area skin and fur condition, behavior and sociability, and indications of self-harm. All the rats were weighed at the beginning of the study and weekly subsequently until the end of the study. The clinical endpoints considered were as follows: according to the expected data for the viability and pathological study, intraoperative or postoperative hypovolemic shock that did not respond to recovery measurements, septic process, loss of animal welfare, and irreducible intraoperative fractures that were expected to cause pain to the rat.
Vascular permeability test and organ harvesting
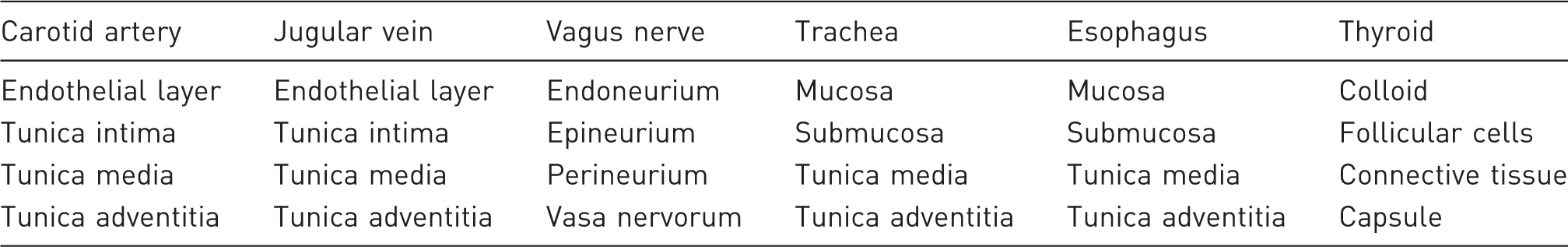
At the end of the study, all animals were anesthetized and subsequently weighed. Analgesia was used prior to organ harvesting. The rats were placed in decubitus supine to perform a medial cervical longitudinal incision, and transversal following the clavicle axis incision. A section over the platysma following the same direction of the skin incision was performed, elevating the two triangular flaps of the upper external vertices and so allowing visualization of the submandibular glands underneath. In the center of the field, the sternothyroid muscle and laterally the sternomastoid muscle were visualized, externally surrounded by the external jugular vein. Between both muscles the common carotid artery next to the vagus nerve could be dissected. Once these three structures were dissected, anterograde and retrograde permeability tests were done in both common carotid arteries, and an anterograde test in both external jugular veins. Finally, all rats were sacrificed with CO2 inhalation. Rats were euthanized in their own cage. The CO2 tank was opened to initiate flow of gas, filling it slowly (30% of chamber volume per minute, approximately 7.5 L/min).
Pathology and histology
Histological study.
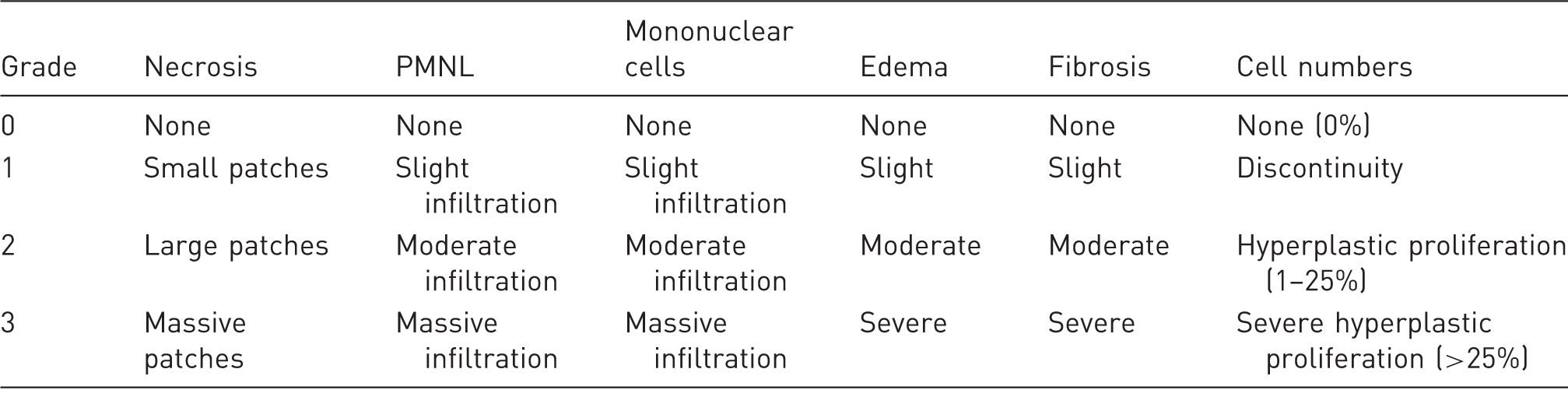
PMNL: polymorphonuclear leukocyte.
Organ harvesting.
Statistical analysis
Statistical tests were performed using SPSS software version 18.0 (SPSS Inc, Chicago, IL, USA). All data are presented as mean ± standard deviation (SD), analyzed with a Kruskal–Wallis one-way analysis of variance. The non-parametric Mann–Whitney U-test was used to identify differences between groups. Statistical significance was set at a P value of < 0.05.
Results
Survival rate
Group I: All rats (100%) survived the 6-week study period. Group II (two weeks): a 91.7% survival rate, as two rats were lost during the acute period post-RT. Group III (six weeks): a 87.5% survival rate, as two rats were lost during the acute period post-RT (two weeks) and another rat was lost during the subacute period post-RT (2–4 weeks). Significant differences were observed between groups I and II–III (P < 0.05).
Weight
Mean body weight (g).

Clinical evaluation
Acute complications were considered to happen during the first two weeks after surgery, whereas subacute complications occurred during two to six weeks after surgery. Complications were exitus, postoperative pain, weight loss, dermatitis of the head and neck regions, skin desquamation, oropharyngeal mucositis, incisor malocclusion, tongue ulcers and neoplasms. Of all these complications, only dermatitis was observed.
Group I
No rats developed acute complications (during the two-week follow-up period) or subacute complications (during the six-week follow-up period). Rats showed soft fur, without local necrosis, skin dermatitis or skin desquamation throughout the six-week follow-up period. No signs of oropharyngeal mucositis or mouth ulcer were found. All animals showed normal behavior without objectifying acute alterations.
Group II
Two animals developed acute post-RT complications (two weeks post-RT). Rats showed soft fur, without local necrosis, skin dermatitis or skin desquamation throughout the two-week follow-up period. Signs of oropharyngeal mucositis and mouth ulcers were found in two rats. All animals showed normal behavior without objectifying acute alterations after RT.
Group III
Two rats developed acute post-RT complications. One rat developed subacute post-RT complications (2–6 weeks after RT). Rats showed soft fur, without local necrosis, skin dermatitis or skin desquamation throughout the first two-week follow-up period. After this period, between two and six weeks, all rats (24/24) developed skin dermatitis in the irradiated region without local necrosis. Signs of oropharyngeal mucositis and mouth ulcers were found in two rats. All animals showed normal behavior without objectifying acute alterations after RT. Skin dermatitis was found in the neck region of all the rats examined. Histologically, the lesion was compatible with RT-induced dermatitis. The extents of the skin lesions were divided into cm2: < 1, 1–2, 2–3, 3–4, > 4. All skin lesions were <4 cm2; 7/24 (29%) < 1 cm2; 11/24 (46%) 1–2 cm2; 5/24 (21%) 2–3 cm2; 1/24 (4%) 3–4 cm2.
Vascular permeability
All common carotid arteries studied presented a positive anterograde and retrograde permeability test (144/144 = 100%). Under the microscope, no arterial thrombosis, aneurysm or pseudoaneurysm was observed. Macroscopically, higher levels of intraoperative bleeding were observed during dissection of irradiated arteries (Groups II, III, left) in comparison to contralateral arteries (Groups II, III, right) and the non-irradiated arteries (Group I, left and right). Moreover, a higher degree of tissue fibrosis was observed in the long-term follow-up irradiated group (III) than in the short-term follow-up group (II) and in the control group (Group I and Groups II–III right vascular bundle).
All external jugular veins presented a positive anterograde permeability test (144/144). Under the microscope, no venous thrombosis, aneurysm or pseudoaneurysm was observed. Intraoperative macroscopic observations were comparable to those observed in arteries. Non-significant differences were found between groups (chi-square test, P > 0.05).
Histological study
Arterial: all post-RT groups showed similar findings (Figure 4, Table 4)
Cellular discontinuity and decrease were observed in the endothelial layer. An absence of endothelial cells was identified in some samples. There was an absence of cellular proliferation from week 2 (Group II) to week 6 (Group III). No histological changes in Group I (bilateral) and Groups II and III (non-RT right carotid) were identified.
Arterial pathological section. Masson's trichrome staining. Longitudinal section, 4×. (a) Group I, non-irradiated artery section: no histological alterations in the vascular structure are observed. (b) Group II, irradiated artery section: tunica intima hyperplasia. (c) Group III, irradiated artery section: increased tunica intima hyperplasia (compared with Group II) and edema, fibrosis and polymorphonuclear leukocyte infiltration, as well as a progressive decrease in muscular cells. Mean histological results from the common carotid artery. Safe model of cervical radiotherapy. RT: radiotherapy; PMN: polymorphonuclear cells.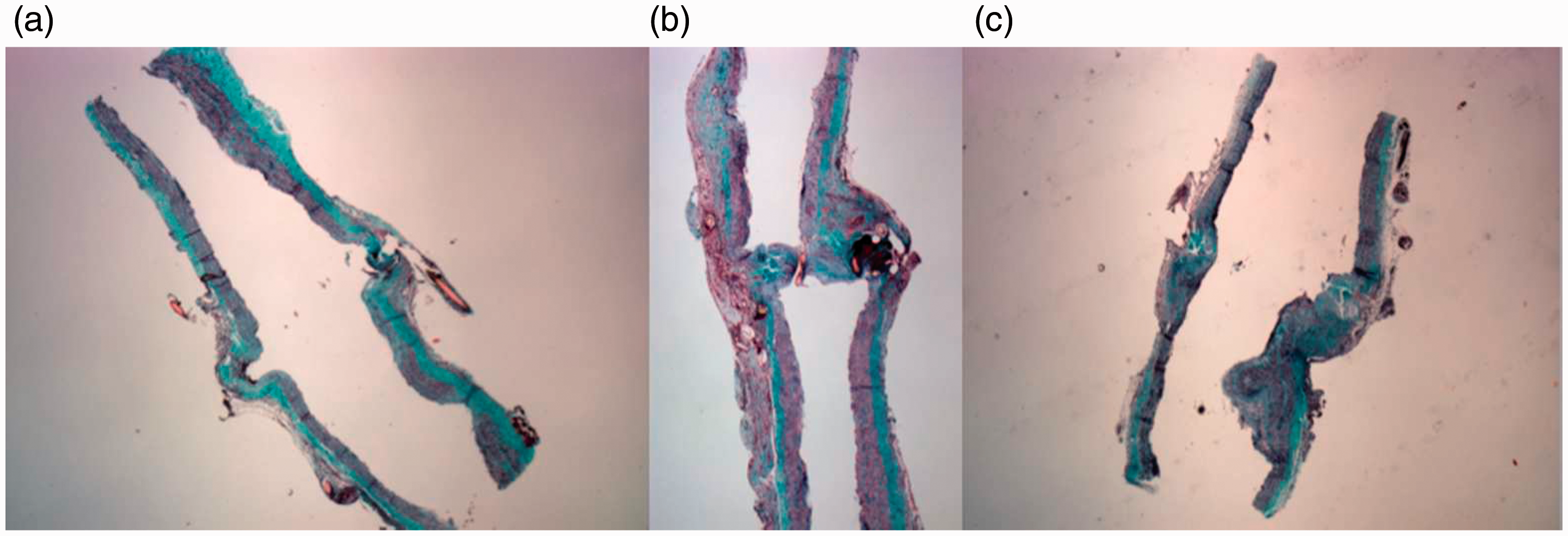
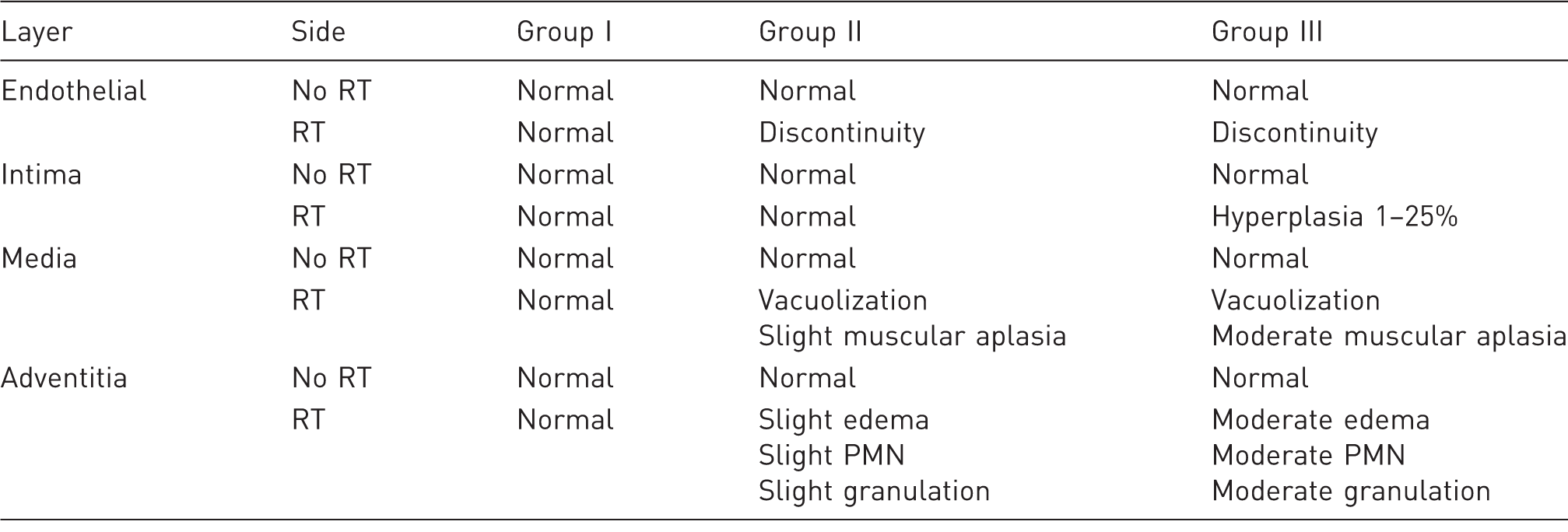
The tunica intima showed a progressive hyperplastic proliferation, increasing from 0% at week 2 (Group II), to 1–25% in week 6 (Group III). No histological changes in Group I (bilateral) and Groups II and III (non-RT right carotid) were observed.
Tunica media showed a progressive decrease in muscular cell numbers and their vacuolization, decreasing from week 2 (Group II) to week 6 (Group III). No histological changes in Group I (bilateral) and Groups II and III (non-RT right carotid) were observed.
Tunica adventitia showed edema, fibrosis and polymorphonuclear leukocyte (PMNL) infiltration, which increased from 0% in week 2 (Group II) to 1–25% in week 6 (Group III). No histological changes in Group I (bilateral) and Groups II and III (non-RT right carotid) took place.
Venous: all post-RT groups showed similar findings (Figure 5, Table 5)
Cellular discontinuity and decrease were observed within the endothelial layer. An absence of endothelial cells was observed in some samples. No cellular proliferation from week 2 (Group II) to week 6 (Group III) occurred. No histological changes in Group I (bilateral) and Groups II and III (non-RT right jugular) were identified.
Venous histological section. Masson's trichrome staining. Longitudinal section, 4×. (a) Group I, non-irradiated venous section: no histological alterations observed. (b) Group II, non-irradiated venous section: mild histological alterations in the vascular structure are observed. (c) Group III, irradiated venous section: endothelial discontinuity (*), muscular layer disorganization (black arrow) and adventitial fibrosis (black arrowhead). Mean histological results from the external jugular vein. Safe model of cervical radiotherapy. RT: radiotherapy; PMN: polymorphonuclear cells.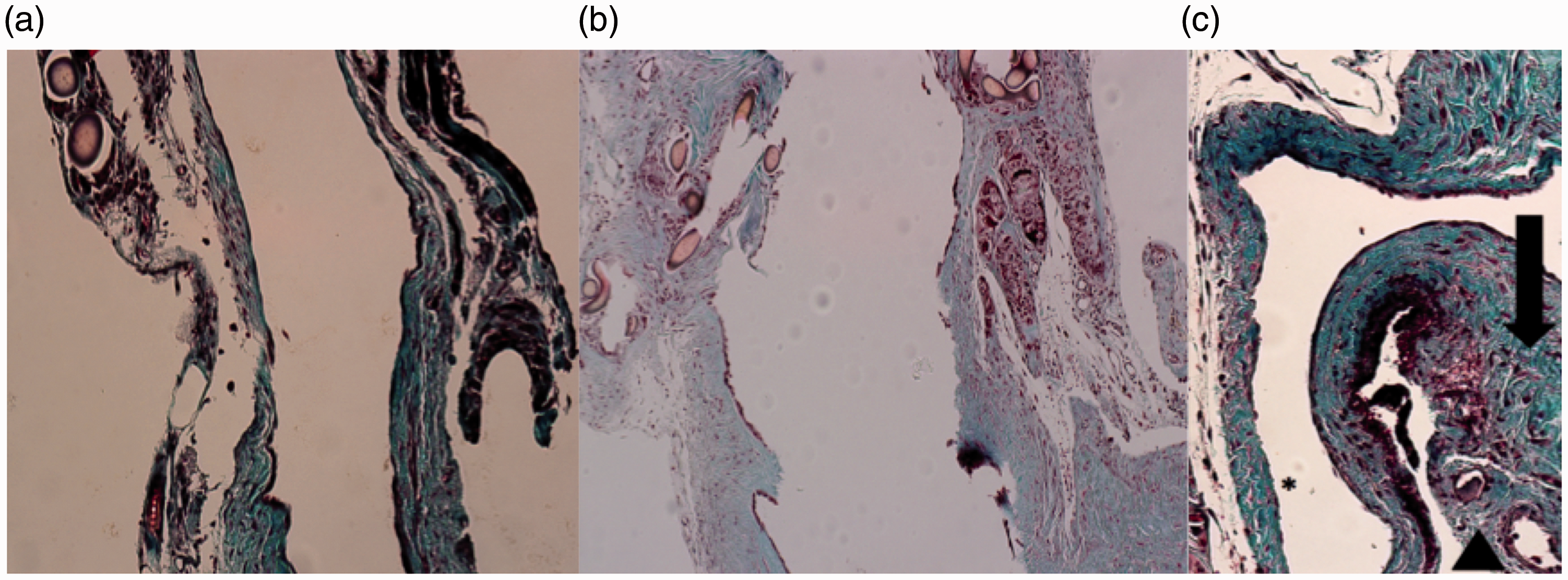
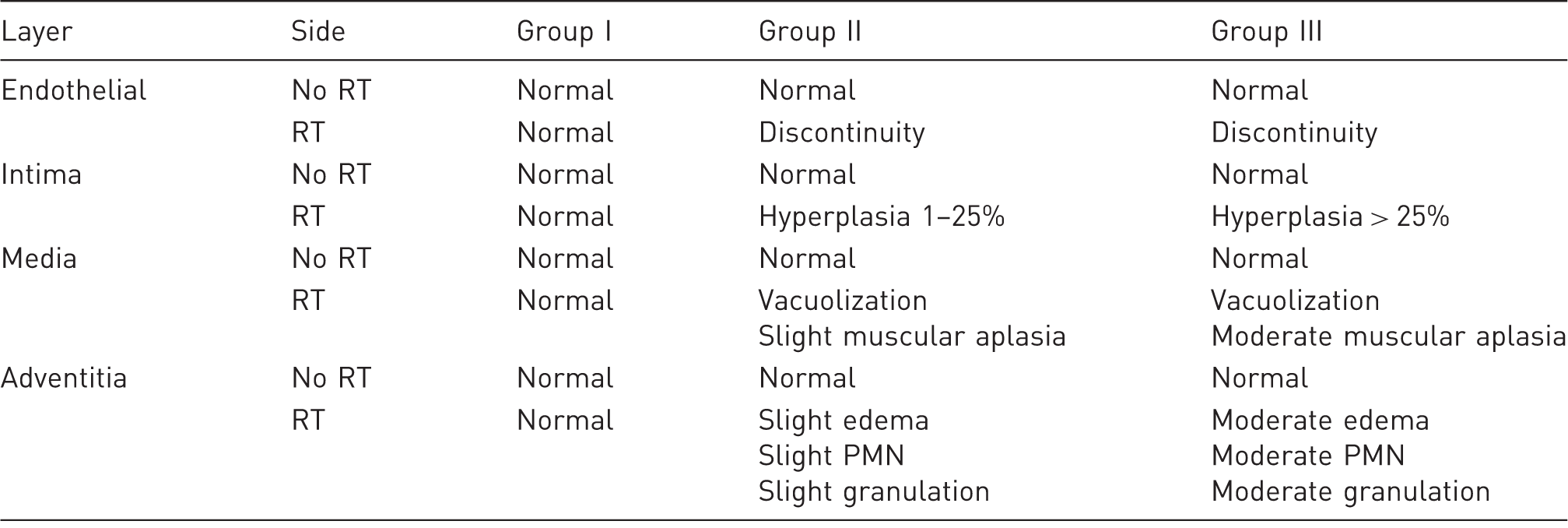
Tunica intima showed progressive hyperplastic proliferation, increasing from 0% in week 2 (Group II) to 1–25% in week 6 (Group III). No histological changes in Group I (bilateral) and Groups II and III (non-RT right jugular) were observed.
Tunica media showed a progressive decrease on the muscular cell numbers and their vacuolization, progressively decreasing from week 2 (Group II) to week 6 (Group III). No histological changes in Group I (bilateral) and Groups II and III (non-RT right jugular) were observed.
Tunica adventitia showed edema, fibrosis and PMNL infiltration, increasing from 0% in week 2 (Group II) to 1–25% in week 6 (Group III). No histological changes in Group I (bilateral) and Groups II and III (non-RT right jugular) were observed.
Vagus nerve (Figure 6)
The epineurium showed fibrosis that increased from 0% in week 2 (Group II) to 1–25% in week 6 (Group III). No identifiable alterations in the vasa nervorum or histological changes in Group I (bilateral) and Groups II and III (non-RT right vagus nerve) were observed.
Nerve histological section. Hematoxylin and eosin staining. Longitudinal section, 10×. (a) Group III, non-irradiated nerve section: no histological alterations in the neural structure are observed. (b) Group III, irradiated nerve section: epineural fibrosis (black arrow).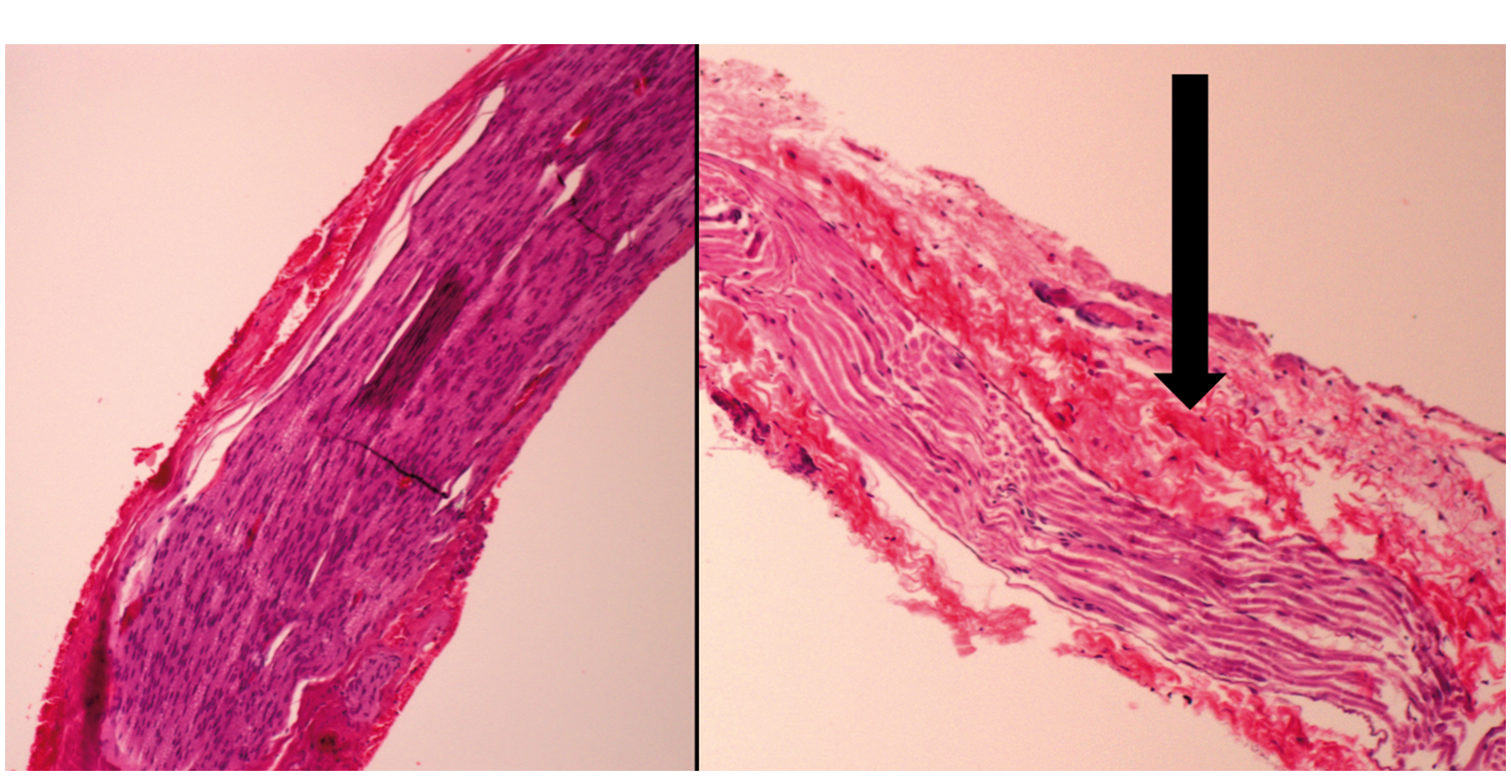
Perineurium and endoneurium did not show histological identifiable changes. No histological changes in Group I (bilateral) and Groups II and III (non-RT right vagus nerve) occurred.
Larynx/trachea/esophagus/thyroid (Figures 7 –9)
Control samples from Group I showed normal histological findings. No injuries secondary to manipulation or surgical techniques were identified.
Trachea histological section; Group III irradiated. Transversal section, hematoxylin and eosin staining. (a) 4×. (b) 10×. (c) 20×. No histological changes with respect to the non-irradiated samples.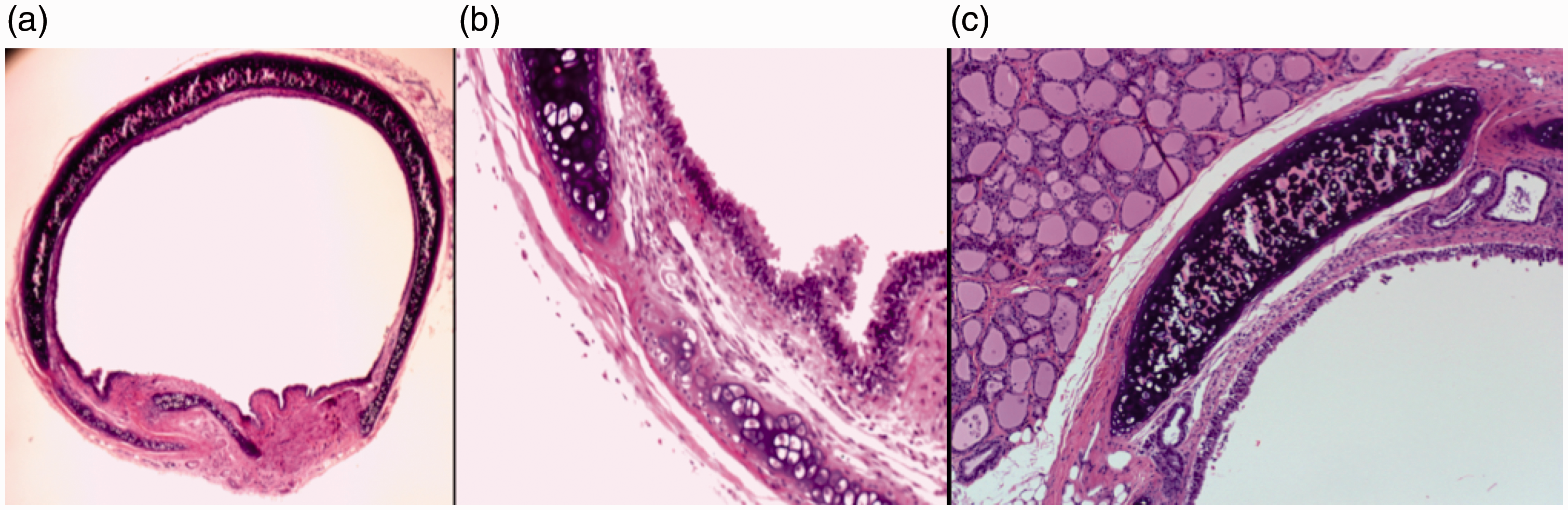
No significant changes between the non-irradiated (Group I) and irradiated groups (Groups II and III) were observed.
Discussion
Limb salvage has become the standard treatment in patients with STS, as a result of advances in adjuvant treatments, surgical techniques and reconstructive implants.13,22,23 In addition, a higher number of patients survive and for a longer period of time, due to systemic improvements in adjuvant therapy, thus increasing the quality demands with reconstructive techniques. Protocols for sarcoma treatment are based on a multidisciplinary approach; this generally requires a combination of wide surgical resection and RT and, occasionally depending on the histological subtype, chemotherapy.
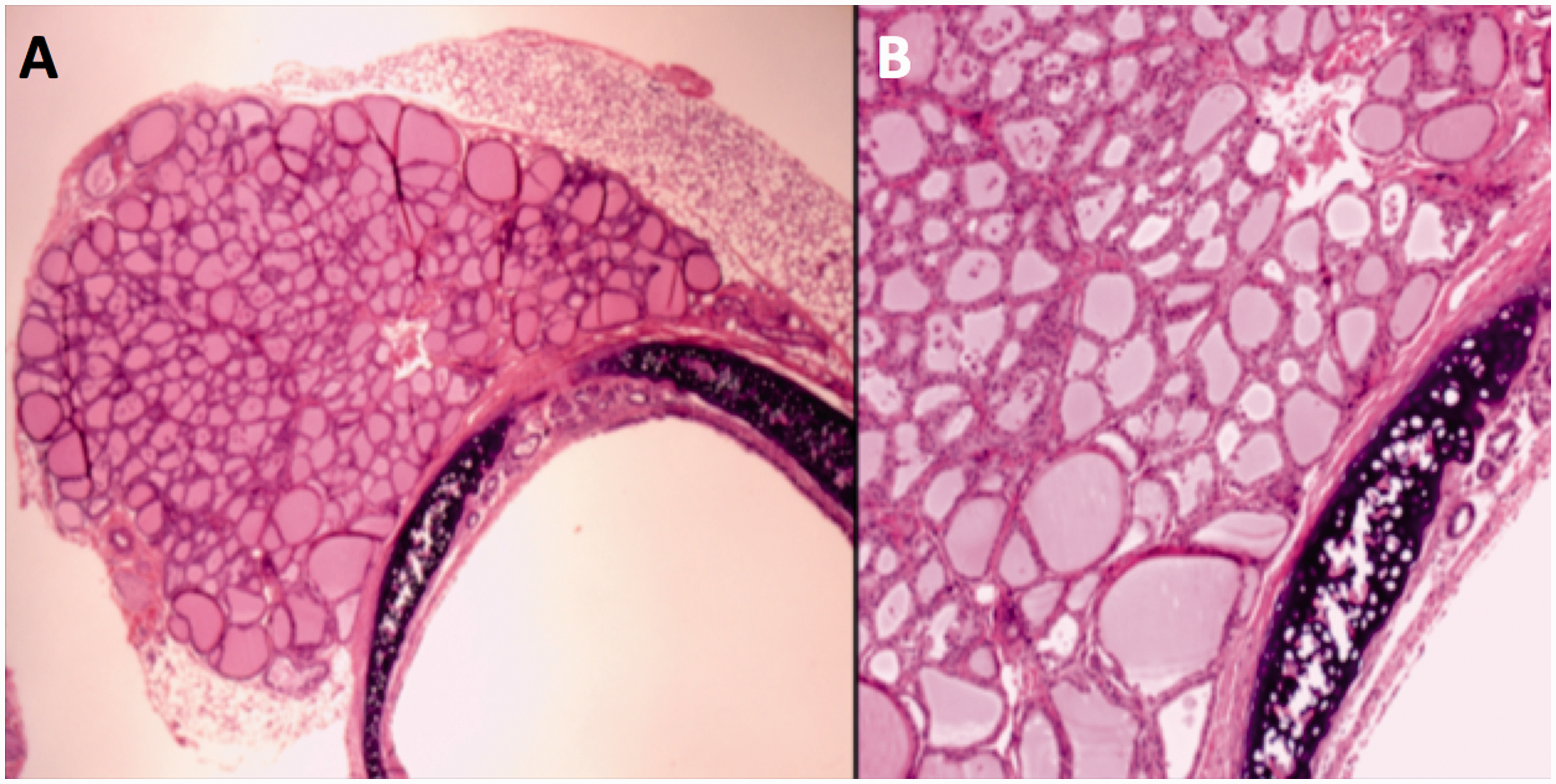
Esophagus histological section, 10×. Transversal section, hematoxylin and eosin staining. (a) Group I, non-irradiated. (b) Group II, irradiated. (c) Group III, irradiated section. No histological changes with respect to the non-irradiated samples. Thyroid histological section, Group III irradiated. Transversal section, hematoxylin and eosin staining. (a) 4×. (b) 10×. No histological changes with respect to the non-irradiated samples.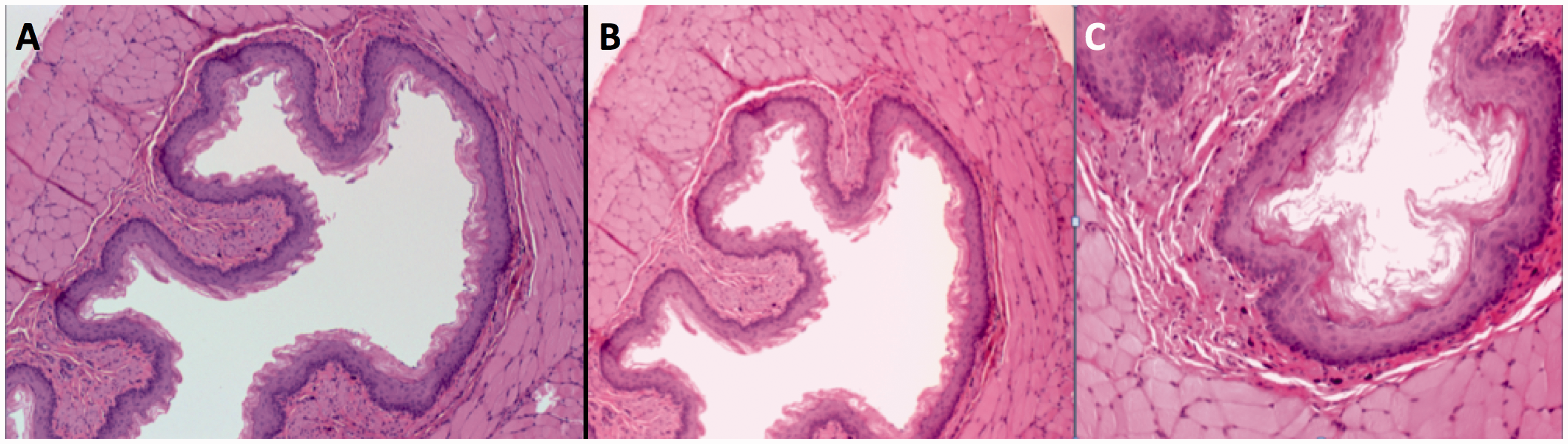
Previous experimental models of microanastomosis on irradiated population.
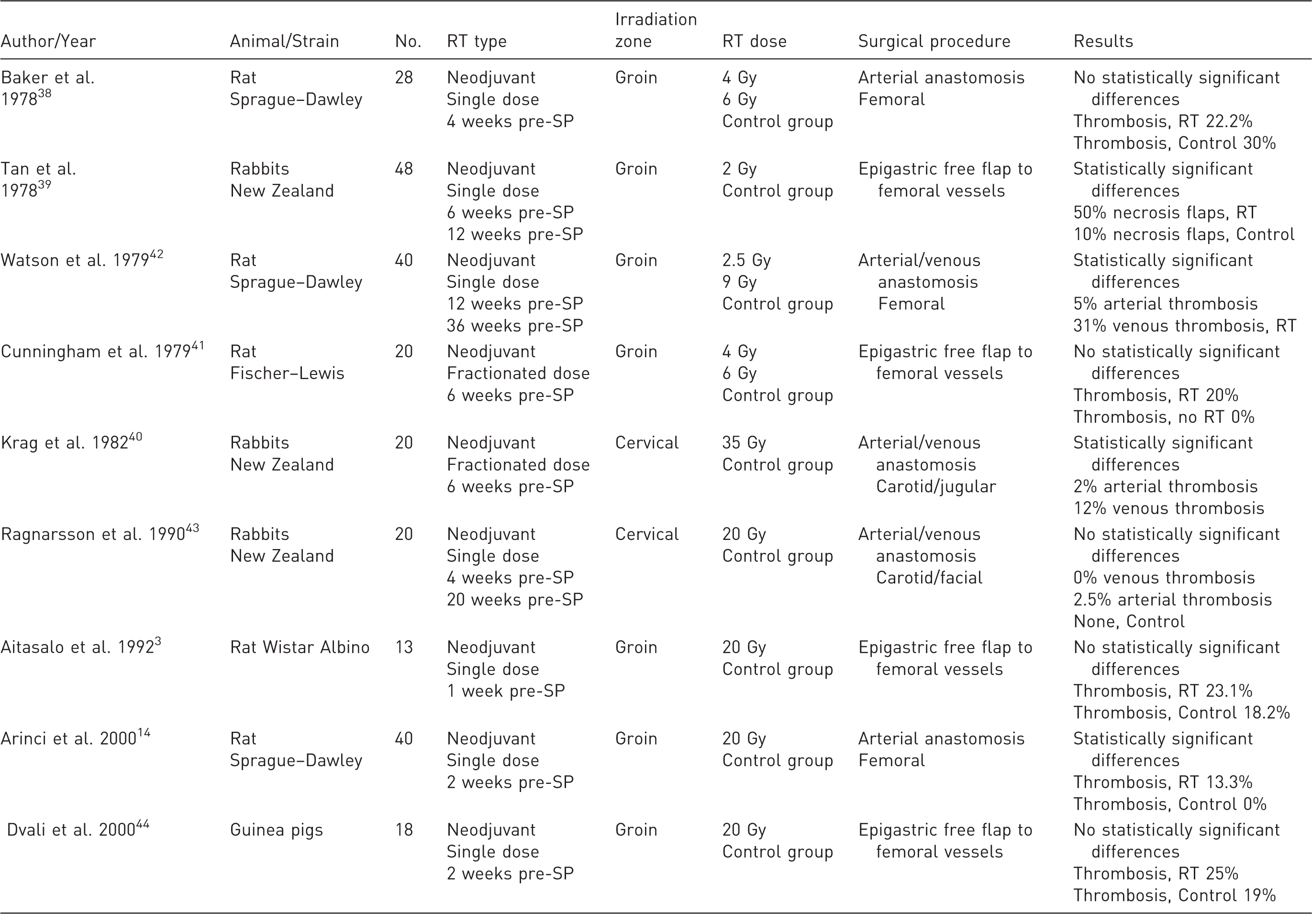
RT: radiotherapy; SP: surgical procedure.
Occasionally, it is necessary to use microsurgical techniques to complete a comprehensive treatment. Currently, microsurgical techniques are thoroughly extended in functional defect reconstruction (osseous and muscular) and skin coverage generated during oncological surgical resection.2 –5,11,15 Success rates of microsurgery procedures in non-irradiated tissues range from 90% to 99%.7 –11,14 –16,36 Whether these results are the same in previously irradiated tissues remain controversial.11,15,16 It is widely agreed that the majority of failures of microsurgical procedures are due to vascular phenomena such as arterial or venous thrombosis, or both.2 –6,8,10,12,37
Some previous experimental models have attempted to examine radiosensibility on microvascular anastomoses, showing high variability in their results (Table 6).3,14,38 –42 Furthermore, this permeability variability increases with its association with RT.2 –6,10,12,14,38 –42 The general opinion is that microanastomosis suture permeability rate decreases significantly in vessels post-RT.11,14,15 Notwithstanding, some studies have achieved greater permeability rates in irradiated vessels,10,14 although differences in surgical and RT techniques used among studies impede a direct and objective comparison.3,14,38 –42
In terms of vascular microsurgical permeability, the most stable and satisfactory results have been obtained in the cervical region 16 in relation to the inguinal region.33,34 This may be due to the size and the accessibility of the cervical vascular bundle (the common carotid artery and internal jugular). However, previous experimental models of irradiated heads and necks of rats have shown devastating local RT effects in comparison with the local effects in the inguinal region.16 –20 For that reason, a majority of experimental microsurgical studies in irradiated fields have been made in the inguinal region (Table 6),3,14,38 –42 although without obtaining comparable results in terms of viability of the vascular anastomosis to those obtained in the cervical region.4,6 –8,10,12,14,32
For all these reasons, there is a need to create a safe irradiated NV bundle model, with reproducible metrics and low local side-effects. This model, in contrast to previous described models in which the head and neck are irradiated indiscriminately,16 –20 is based on selective irradiation of the left hemicervical region by means of a 2 × 2 cm frontal field that covers the cervical NV bundle and avoids unnecessary irradiation of the surrounding vital structures.
Nagler et al.17 –19 evaluated in an experimental study the acute, subacute and chronic effects from cervical irradiation in rats. They distributed rats in six groups with 18–20 rats each with a one-year follow-up. A non-irradiated control group was compared with irradiated groups with 2.5, 5, 7.5, 10 and 15 Gy, finding a dose-dependent decrease in survival and body weight.
Acute post-RT effects (two weeks) are due to severe oropharyngeal mucositis. Nagler et al.17 –19 described this event in 66% of irradiated animals with 15 Gy. This can cause the deceleration of body weight increase, mouth ulceration, 30% mortality in animals irradiated with 10 Gy, and 40% mortality in those irradiated with 15 Gy.27 –30 Stiubea-Cohen et al. studied the effect of cranial RT in submandibular glands in an experimental study with rats, 20 and concluded that a single dose of 15 Gy in a head and neck rat model reduced salivary production by 50%, decreased body weight gain by 25%, and increased mortality by 32%. Our model shows an acute post-RT mortality of 10.4% (5 deaths/48 cases), a subacute post-RT mortality of 2% (1 death/48 cases) and a total mortality of 8.3% (6 deaths/72 cases), which are lower than in other available studies.
Other side-effects, shown in the literature to be highly prevalent,16 –20 and related to exposed tissue and organs inside the cervical irradiated field, such as xerostomia, osteoradionecrosis, swallowing disorders, vascular aneurysms or thyroid hypofunction, have also been reduced in our experimental model. All these facts were indicated by animal body weight, which recorded a progressive increase, and without significant differences between irradiated and non-irradiated groups. These data do not correspond to previous studies,16 –20 where body weight loss is considered to be an irradiation dose-dependent side-effect, objectifying statistical significance between RT groups (15 Gy).
The 20 Gy cervical RT model achieves the induction of changes in the NV bundle without altering the surrounding tissues. Thus, a safe and reproducible model with lower morbimortality rates than previous models was achieved. This model will allow experimental microsurgical therapeutic studies with the NV bundle with the benefit of dimensional (vascular diameter and pedicle length), mechanical (vascular wall thickness) and physiological characteristics similar to those in human structures. 16
Footnotes
Acknowledgements
We would like to thank Vall d'Hebron Research Institute (VHIR), A Rojo and M Rosal for their precious help in the care of the animals. We would also like to thank the Radiotherapy and Pathology Departments of Hospital Universitari Vall Hebron for their practical suggestions and encouragement. Grant sponsors are from the Sociedad Española Cirugía Ortopédica y Traumatología (SECOT; Grant number: 0012‐2012) and the Societat Catala Cirurgia Ortopèdica i Traumatologia (SCCOT; Grant number: 0347‐2012).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
