Abstract
Ketamine administration has been associated with controversial behavioural impairments and psychotic episodes. Even though ketamine alone and in combination with midazolam or dexmedetomidine are frequently used in laboratory animals, the side-effects of such protocols are not well known. Therefore, our aim was to evaluate the effects of ketamine alone and in combination with midazolam or dexmedetomidine on emotional reactivity, as well as the effects on learning and memory in adult rats at least 48 h after anaesthesia. The evaluation of the potential influence of 100 mg/kg ketamine administered alone and in combination with midazolam (5 mg/kg), or dexmedetomidine (0.25 mg/kg) on spatial learning and recognition memory was studied in adult Wistar rats using the radial maze as well as object recognition and location tests. The influence of these combinations on emotional reactivity was investigated using the new exploration test and the elevated plus maze. Results showed that ketamine alone or in combination with midazolam or dexmedetomidine affected neither spatial and recognition memory, nor emotional reactivity. These results reinforce the safe clinical use of ketamine and its combinations in rats in a research context since the administration of these anaesthetic combinations did not produce significant changes with regard to spatial and recognition memory or emotional reactivity. Furthermore, these results indicate that the quality of scientific data produced in adult rat neurobehavioural research is not jeopardized by the use of these anaesthetic protocols.
Anaesthesia is used during surgery and other interventions to control pain, anxiety and consciousness. In veterinary practice, anaesthesia is also essential in immobilizing animals for clinical examination.1,2 Likewise, anaesthesia is required for several procedures in research involving laboratory animals, especially in neurobiological research.
All experiments using laboratory animals should be based on the ‘three R principle’: refinement, replacement and reduction. 3 Refinement of anaesthesia should act as an important parameter in consideration of, not only the promotion of animal welfare, but also the avoidance of anaesthetics that may interfere with experimental results.
In research using laboratory animals the N-methyl-D-aspartate (NMDA) antagonist ketamine is probably the most frequently used anaesthetic. In humans, ketamine is described as a dissociative anaesthetic and is still used worldwide in clinical settings 4 and in emergencies. 5
The use of ketamine is limited because of its psychotic side-effects, 6 which can be overcome by finding a balanced anaesthesia. This technique allows for safe anaesthesia by decreasing the doses required for each individual drug and still achieving a clinical effect. Therefore, ketamine is frequently used in combination with midazolam or dexmedetomidine.7,8 Besides providing anterograde amnesia, the benzodiazepine midazolam is also used as a sedative and anxiolytic compound. 9 Dexmedetomidine is a recent α2-adrenergic agonist that also induces sedation, anxiolysis, and analgesia. 10
Acute and repeated sub-anaesthetic doses of ketamine have been reported to impair cognition in humans
11
and rodents.12,13 However, acute ketamine administration has also been shown to attenuate post-operative cognitive dysfunction.
14
Regardless of all the evidence, the literature is nonetheless scarce regarding the effects of ketamine in combination with dexmedetomidine or with midazolam on long-term memory. Studies in humans have reported that these combinations produce anterograde amnesia and anxiolysis, and reduce the side-effects related with ketamine.15,16 The ketamine
Therefore, in the present study, we evaluated the effects of the ketamine alone and in combination with midazolam or dexmedetomidine on emotional reactivity, and on learning and memory in rats – one of the most frequently used animal models in neuroscience research. This study attempts to replicate what happens in research and contributes to the refinement of anaesthesia in laboratory animals by evaluating this combination when rats are anaesthetized and used in neurobehavioural research.
Materials and methods
Animals
Eighty-two male Wistar rats (from the colony of the Institute of Molecular and Cell Biology, University of Porto, Portugal) aged 10–12 weeks were used in the study. Rats were housed in pairs in a controlled environment (20 ± 2℃, 45–55% humidity) with a 12 h light/dark cycle. Food and water were supplied ad libitum. All behavioural experiments were performed during the dark phase, except for the elevated plus maze (EPM) test. All procedures were carried out under personal and project licences approved by the national authority for animal protection, ‘Direção Geral de Alimentação e Veterinária’ (Lisbon, Portugal), and were performed in accordance with the European Directive 2010/63/EU on the protection of animals used for scientific purposes.
Anaesthesia
Ketamine (Imalgene 1000, Merial, Lisbon, Portugal; 100 mg/mL), midazolam (B Braun Medical, Barcelona, Spain; 5 mg/mL), and dexmedetomidine (Dexdomitor, Esteve Farma, Lda, Carnaxide, Portugal, 0.5 mg/mL) were dissolved in a sterile saline solution (Braun Vet, Queluz de Baixo, Portugal) before intraperitoneal injection at 1 mL/kg of body weight. Rats were randomly distributed into four groups: CONTROL (saline solution, n = 18), 100 mg/kg ketamine (KET, n = 19), 100 mg/kg ketamine and 5 mg/kg midazolam (KET/MID, n = 19), and 100 mg/kg ketamine and 0.25 mg/kg dexmedetomidine (KET/DEX, n = 19); the combination of drugs was administered in a single injection. In addition to the groups previously referred to, and in order to evaluate emotional reactivity using the EPM test and to avoid prolonged periods of anaesthesia, KET/DEX (100 mg/kg and 0.25 mg/kg) anaesthesia was reverted using atipamezole (Antisedan, Pfizer, Oeiras, Portugal; 5 mg/mL) one hour after loss of righting reflex (KET/DEX+A).
After anaesthetic administration, the animals were isolated until righting reflex was lost. Subsequently, animals were placed on a homeothermic blanket, connected to a rectal thermal probe (50-7061-F; Harvard Apparatus Ltd, Kent, UK) to maintain a body temperature at 37 ± 1℃. During anaesthesia, rats received oxygen through a facemask, and their eyes were kept humid with sterile saline solution. Oxygen saturation was maintained between 90 and 99% using a pulse oximetry (Vitalstore Multimonitor base unit; Vetronics, Devon, UK) with a sensor placed in the hindlimb of each animal. Respiratory and pulse rates were measured using the same equipment every 5 min. Time to lose and time to gain the righting reflex were also recorded.
Experiment 1: Evaluation of the potential influence of ketamine alone and combined with midazolam or dexmedetomidine on spatial learning and recognition memory
The cognitive status was assessed in the eight-radial maze and in the object recognition and location (OR/L) tests. Forty-eight animals were randomly distributed between the radial maze and the OR/L tests. Six animals were allocated to each of the experimental groups (CONTROL, KET, KET/MID and KET/DEX). The radial maze training and the habituation to OR/L started 48 h after the anaesthesia protocols.
Radial maze test
Working and reference memories were assessed simultaneously through a fixed position of reward task, in which three of the arms were baited and their positions were fixed throughout the training trials. The entry into the never-baited arm was considered to be a reference memory (RM) error. Within a training session, the re-entry into one of the arms was considered a working memory (WM) error. 21 The response latency was defined as the total duration of the experiment and the total number of visited arms.
The radial maze was made of black Plexiglas (Panlab, SLU, Barcelona, Spain) and elevated 50 cm from the ground. It consisted of a central area with eight sliding doors giving access to eight equally-sized arms. For reinforcement, three sugar pellets (Bio-Serv, Frenchtown, NJ, USA) were placed in the distal end of each arm. External-maze cues were added to aid navigation. The amount of time a rat took to complete a trial was recorded. During experiments, animals had restricted access to food, and were being fed only following testing. The body weight loss of animals was kept below 10%.
Habituation began five days before the training period. On three consecutive days, sugar pellets were distributed and rats were allowed to individually explore the maze for 5 min or until the pellets were consumed. Twenty-four hours after the habituation period, animals were anaesthetized.
Training started 48 h after anaesthesia, with daily sessions (five trials per session) over a nine-day period. At the beginning of each trial, rats were placed in the centre of the maze and always facing the same arm, and were allowed to explore the maze for 5 min or until all the pellets were consumed. An arm entry was confirmed when all four paws were inside the arm of the maze. After each session animals were returned to their home cage and were fed. Between sessions, the maze was cleansed with absorbing paper and a 30% ethanol solution to minimize the olfactory cues. In addition to minimizing these cues, maze rotation was made between trials.
Object recognition/location test
The OR test was performed as described in a previous work. 13 Briefly, the test apparatus consisted of an open box and the objects used were made of plastic, glass or metal in three different shapes: cubes, pyramids and cylinders. The test consisted of three phases.
Habituation began 48 h after anaesthesia and rats were allowed to explore the apparatus for 10 min, over five consecutive days. In all sessions, the arena contained a single object (a familiar object) placed in the centre. The following day, sample phase OR1 started by placing each rat in the apparatus with two identical objects (familiar) for 3 min and then the rat was removed to its home cage. The choice phases were performed after a 15 min (OR2) and 24 h (OR3) delay. In this phase the apparatus contained a novel object and a copy of the previously seen familiar object, and animals were allowed to explore the objects for 3 min. The time spent exploring the novel object served as the measure of recognition memory for the familiar object. Exploration was defined as follows: rat touched the object with its nose or the rat's nose was directed toward the object at a distance shorter than 2 cm. Circling or sitting on the object was not considered to be exploratory behaviours. The index of discrimination was calculated as the difference between exploration time of the novel and familiar objects. 22
OL testing was performed 24 h after the OR test. The sample phase of the OL1 test was exactly the same as for the recognition test. After a retention interval of 15 min, the rat was returned to the apparatus for 3 min with both objects, one of which was placed in the previously used location, and the other placed near the adjacent corner, a new location – choice phase (OL2). This task assessed the ability to discriminate the novelty of the OL. 22 Data from the OR/L tests were analysed using the Observer 10 software (Noldus Information Technology, Wageningen, The Netherlands).
Experiment 2: Evaluation of the potential influence of ketamine alone and combined with midazolam or dexmedetomidine on emotional reactivity
The emotional behaviours were assessed in the new object exploration (NOE) test and the EPM test 48 h after anaesthesia. Thirty-four animals were randomly distributed to each of the experimental groups (KET, KET/MID, KET/DEX and KET/DEX+A, n = 7, except CONTROL with n = 6) in the EPM test. The 24 animals used in the NOE were the same as those used during the first day of habituation to OR/L. Tests began 48 h after anaesthesia.
All behaviours were video-recorded with a camera placed above the apparatus and saved to a computer by the multi-camera vigilance GeoVision system (GV-800/8; GeoVision Inc, Taipei, Taiwan). Behaviours were analysed using the Observer 10 software.
New object exploration test
In this test, each rat was placed in the open box facing away from the test object, which was placed in the centre of the open box, and allowed to explore the environment for 10 min. The pattern of space occupation, frequency and duration of behaviours towards the object were recorded.
Elevated plus maze test
The EPM was made of dark grey plastic and positioned 50 cm above the floor. The maze consisted of four arms arranged in the shape of a cross, two arms had surrounding walls (closed arms) and the other two opposing arms had no walls (open arms). A test session was started by placing each rat on the central platform, facing an open arm. Over a period of 5 min, each rat's exploration was video-recorded. The maze was cleansed after each training session. The number of open and closed arm entries, and the time spent in the different compartments of the maze (central platform, open and closed arms) were scored. As mentioned previously, the KET/DEX+A group was added to this test in order to clarify the potential influence of anaesthesia duration on anxiety.
Statistical analysis
The number of animals used per group was based on previous work23,24 with a power of 0.8 and α = 0.05. Parametric data were analysed with one-way analysis of variance (ANOVA) and the paired Student's t-test (OR/L data for comparisons between groups and for differences within groups, respectively), and ANOVA for repeated measures with Bonferroni post hoc tests (random access memory (RAM) data with anaesthesia groups as a between-subject factor and day of testing as a within-subject factor). These were expressed as mean + SEM. Non-parametric data, such as anaesthetic parameters and NOE data, were analysed with the Kruskal–Wallis test followed by the Mann–Whitney U-test and expressed as median and 95% confidence intervals. A value of P ≤ 0.05 was considered to be statistically significant.
All results were analysed using Prism 6 for data management and SPSS 16.0 (Apache Software Foundation, Forest Hill, MD, USA) for statistical analysis.
Results
Anaesthesia
Physiological parameters during 60 min of anaesthesia.
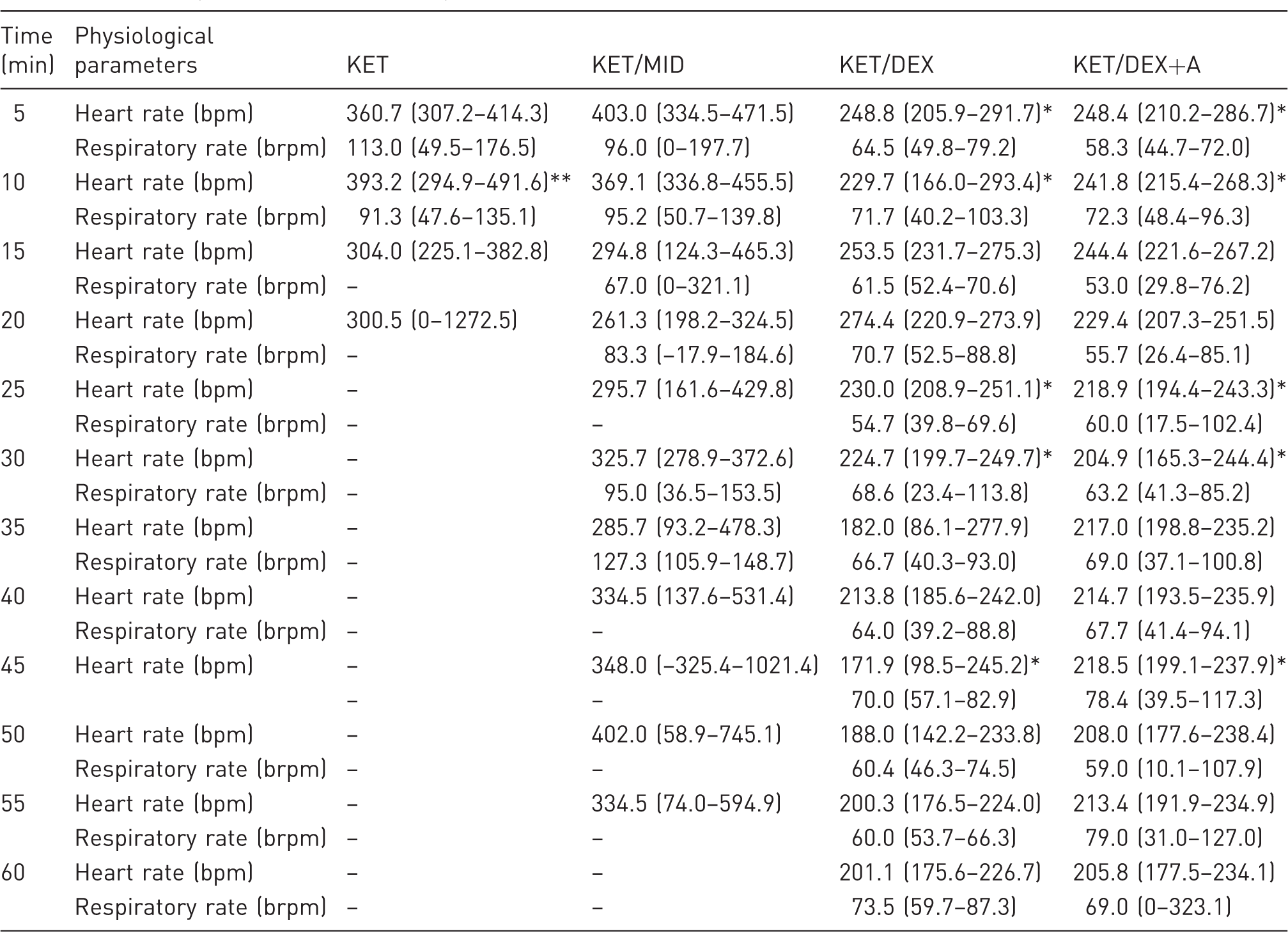
KET: 100 mg/kg of ketamine; KET/MID: 100 mg/kg of ketamine and 5 mg/kg of midazolam in a single injection; KET/DEX: 100 mg/kg of ketamine and 0.25 mg/kg of dexmedetomidine in a single injection; KET/DEX+A: 100 mg/kg of ketamine and 0.25 mg/kg of dexmedetomidine in a single injection, and 5 mg/kg of atipamezole one hour post-anaesthesia; bpm: beats per minute; brpm: breaths per minute. Data are shown as median (95% confidence interval). *P ≤ 0.043 for comparison with KET/MID; **P = 0.039 for comparison with KET/DEX+A. When no value is exhibited this implies that the animals were too superficial, and the equipment could not measure the variable.
Behavioural tests
Experiment 1
Animals ranging from 205 to 315 g were used in all behavioural tests; no body weight loss higher than 10% was observed.
The analysis of the radial arm maze data revealed a main effect of day for response latency [F(1,22) = 30.995, P < 0.001], for working memory [F(1,22) = 12.01, P < 0.001] and reference memory [F(1,22) = 27.551, P < 0.001] errors. However, there was no main effect of treatment (P > 0.05) or treatment/day interactions (P > 0.05). In all groups the number of reference and working memory errors decreased across learning sessions (Figure 1a and b).
Reference (a) and working (b) memory errors tested in radial maze in rats exposed to ketamine alone, or combined with midazolam or dexmedetomidine. Results are presented as means + SEM (n = 6) for groups: saline solution (CONTROL); 100 mg/kg ketamine (KET); 100 mg/kg ketamine and 5 mg/kg midazolam (KET/MID); and 100 mg/kg ketamine and 0.25 mg kg dexmedetomidine (KET/DEX); block session was calculated as average measured on five trials. No differences between groups were observed.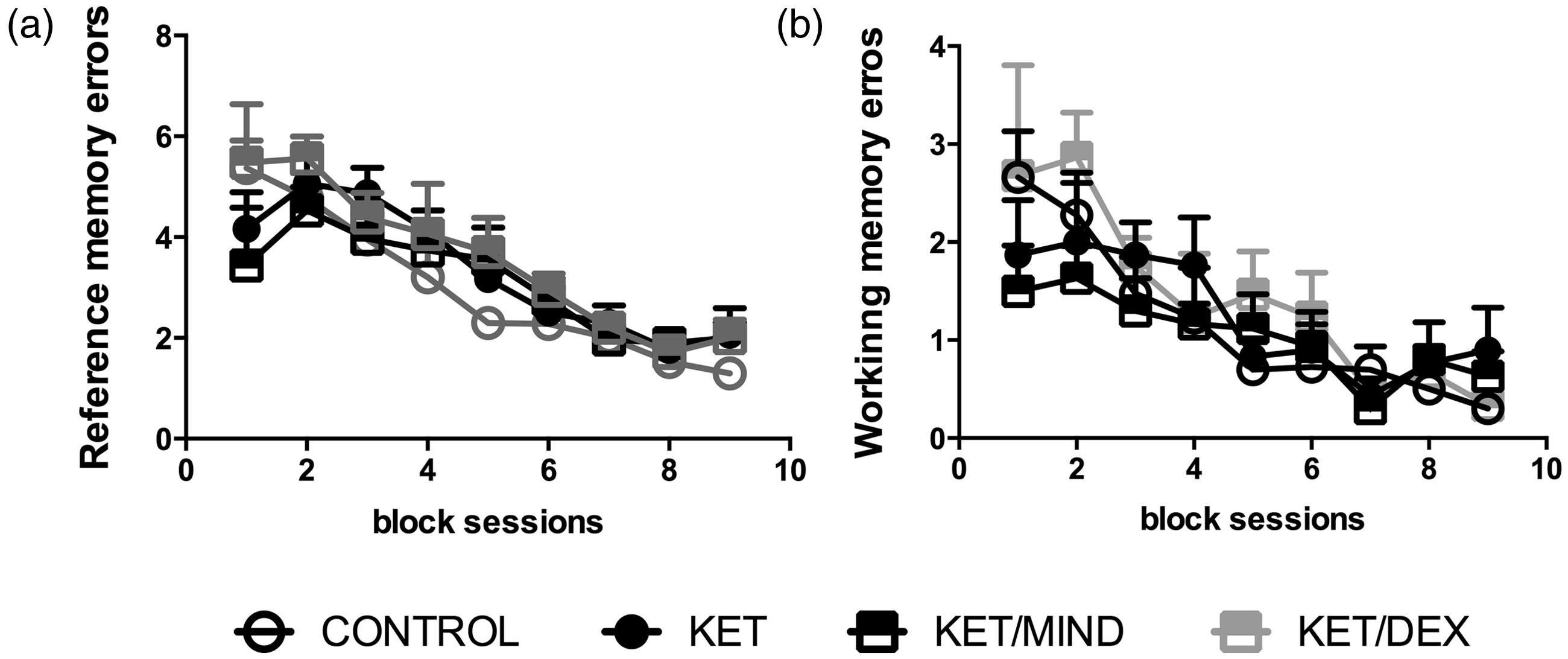
After the habituation phase (Figure 2a), data from OR/L showed no differences between groups. All subjects recognized the novelty of the object, as indicated by the significant increase in exploration of the new object (i.e. robust recognition memory) during the short delay of 15 min (OR2; CONTROL t = −6.529, P < 0.001; KET t = −7.260, P < 0.001; KET/MID t = −6.422, P < 0.001; KET/DEX t = −5.053, P < 0.01), and during the long-term delay of 24 h (OR3; CONTROL t = −10.736, P < 0.001; KET t = −5.33, P < 0.01; KET/MID t = −6.024, P < 0.01; KET/DEX t = −3.532, P < 0.05). Furthermore, no differences between groups were found in the discrimination index (proportion of exploration time) in OR2 and OR3 (Figure 2b and c). Rats from all experimental groups also recognized the object displacement (new location), as indicated by the preferential exploration compared with the object in the familiar location (OL2; CONTROL t = −10.253, P < 0.001; KET t = −9.067, P < 0.001; KET/MID t = −7.832, P < 0.001; KET/DEX t = −8.543, P < 0.001). No differences between groups were found in the discrimination index in OL2 (Figure 2d).
Object recognition (OR) and object location (OL) memory tests in rats exposed to ketamine alone, or combined with midazolam or dexmedetomidine. Schematic representation of the experimental design: 2A: habituation (5 days); 2B: OR1 – sample phase of OR; OR2 – choice phase, delay 15 min, graphs represent duration of object exploration and discrimination index for object exploration in OR2; 2C: OR3 – choice phase, delay 24 h, graphs represent duration of object exploration and discrimination index for object exploration in OR3; 2D: OL1 – sample phase of OL; OL2 – choice phase, delay 15 min, graphs represent duration of object exploration and discrimination index for object exploration in OL2. Individual values are shown for each experimental group, n = 6. The large horizontal bars represent mean values for each group, and the vertical bars represent the SEM. Groups: saline solution (CONTROL); 100 mg/kg ketamine (KET); 100 mg/kg ketamine and 5mg/kg midazolam (KET/MID); and 100 mg/kg ketamine and 0.25mg/kg dexmedetomidine (KET/DEX). No differences between groups were observed. *P < 0.05, **P < 0.01 and ***P < 0.001 indicate significant differences within groups.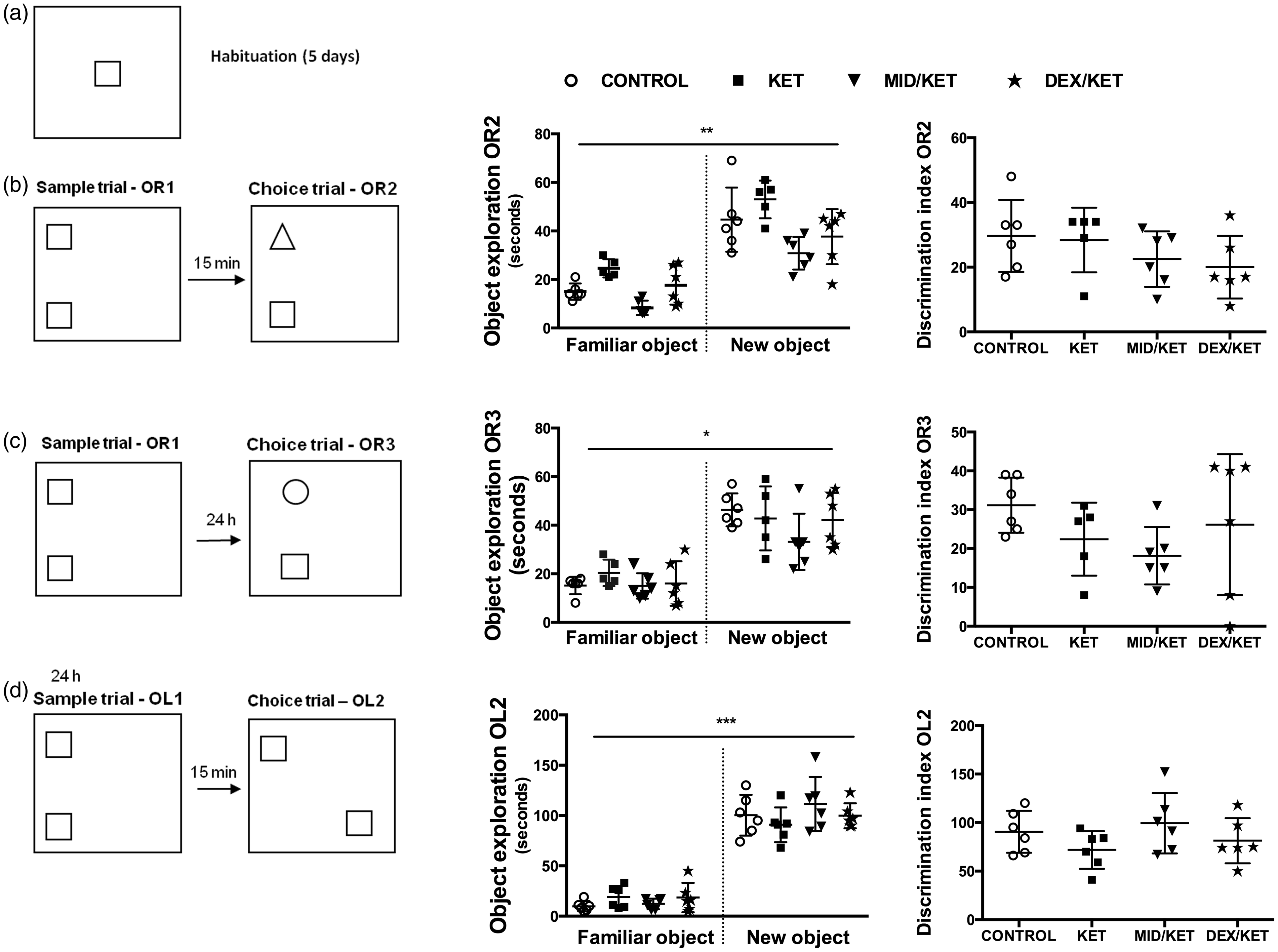
Experiment 2
Descriptive analysis, median, confidence interval (CI) and P value for the parameters evaluated in the new object exploration (NOE) test.
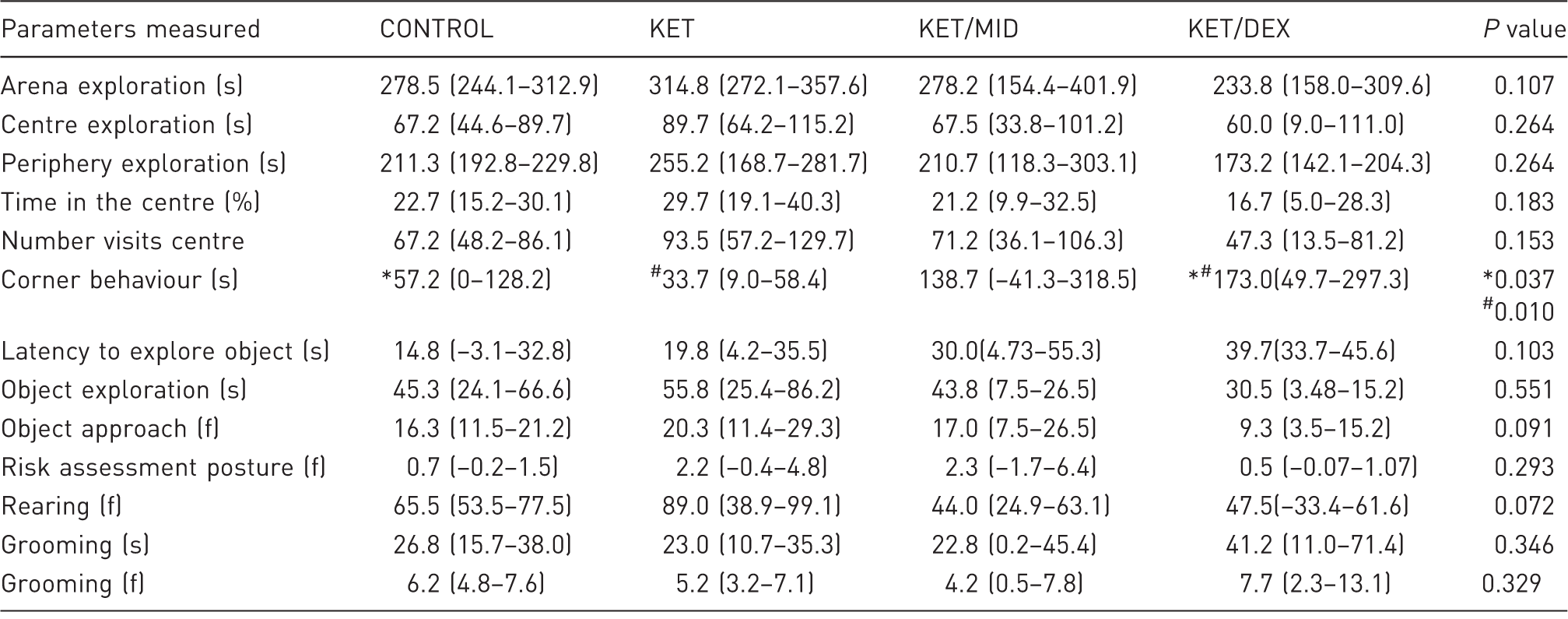
KET: 100 mg/kg of ketamine; KET/MID: 100 mg/kg of ketamine and 5 mg/kg of midazolam; KET/DEX: 100 mg/kg of ketamine and 0.25 mg/kg of dexmedetomidine; CONTROL: saline injection; n = 6; s: duration of the behaviour; f: frequency of the behaviour. Data were analysed with Kruskal–Wallis test followed by the Mann–Whitney U-test and expressed by median and 95% CI. *P < 0.05 indicates significant differences between control and KET/DEX; #p < 0.05 indicates significant difference between KET and KET/DEX.
Parameters measured in the elevated plus maze test.
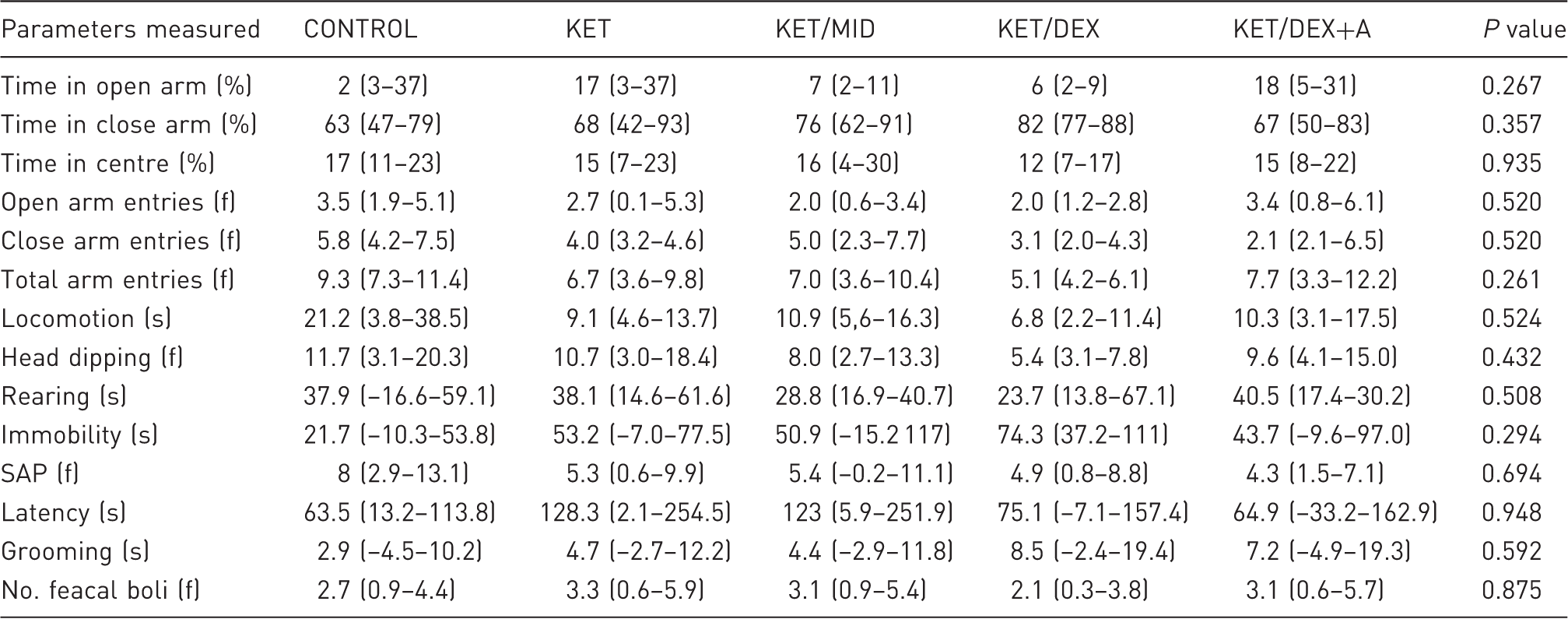
KET: 100 mg/kg of ketamine; KET/MID: 100 mg/kg of ketamine and 5 mg/kg of midazolam; KET/DEX: 100 mg/kg of ketamine and 0.25 mg/kg of dexmedetomidine; KET/DEX+A: 100 mg/kg of ketamine and 0.25 mg/kg of dexmedetomidine reverted with 5mg/kg of atipamezole one hour post-anaesthesia; CONTROL: saline injection; SAP: Stretched Attend Posture; n = 7, except for control with 6; f: frequency of the behaviour. Data are reported as median and 95% confidence interval.
Discussion
This study shows that ketamine alone or in combination with midazolam or dexmedetomidine does not affect spatial and recognition memory or emotional reactivity in rats. We chose to perform the tests 48 h after anaesthesia, since we intended to study not the direct effect of the substances, but the possible long-term and side-effects when the anaesthetics were no longer in the animals. This time frame was chosen since the half-life of ketamine administered intramuscularly is 33 min, 25 and the half-life of midazolam administered intravenously is 29.4 min 26 in rats. Dexmedetomidine has been described with a half-life of approximately 2 h. 27
Results from anaesthesia variables revealed that animals anaesthetized with ketamine / dexmedetomidine had a lower heart rate and a greater depth of anaesthesia when compared with ketamine / midazolam treated rats. 28 Indeed, the pedal withdrawal reflex was only lost in rats from the KET/DEX and KET/DEX+A groups, implying that surgical anaesthesia was only achieved with these combinations. Nevertheless, the other protocols have been proven to be useful in other situations. For example, ketamine alone causes a loss of righting reflex and induces a shallow plane of anaesthesia for 30 min, which is suitable for imaging procedures when only immobility is required. In addition to imaging, ketamine/midazolam anaesthesia may also be adequate for minor procedures such as blood collection, venepuncture/catheter placement, and subcutaneous implantation of devices.
The heart rate of animals treated with KET was higher than that of the KET/DEX and KET/DEX+A rats. In general, these results are in agreement with those found in the literature. Ketamine is expected to oppose the depressant actions of α2-agonists and benzodiazepines on the circulatory system.16,29,30 In humans, the combination of ketamine with dexmedetomidine provides a stable anaesthesia, with a reduction of adverse effects caused by ketamine, such as post-anaesthetic delirium. 16 Furthermore, the use of α2-agonists reduces anaesthetic requirements and improves perioperative haemodynamic stability.31,32 In addition, ketamine/dexmedetomidine anaesthesia has the advantage of allowing rapid recovery with the administration of atipamezole, a specific α2-antagonist available for animal use, which immediately reverses dexmedetomidine effects. 33 Both animal and human studies have shown that ketamine combined with midazolam and diazepam results in fewer side-effects and better recovery than the use of ketamine alone.34,35 However, there is still a lack of information about potential secondary effects on cognitive functions of these combined drugs.
Although the scope of this study was to evaluate the effects of ketamine combinations, the use of 100 mg/kg of ketamine alone was important as a control dose for the anaesthetic combinations used. As has already been mentioned, several studies have reported ketamine-induced neurotoxicity and psychotic symptoms. Therefore, if anaesthetic combinations result in memory impairment or changes in anxiety levels, it is imperative to rule out the possibility of a direct influence of ketamine alone. In this sense, the results of animals treated with ketamine alone may provide insight for other researchers.
The behavioural analysis performed in this study demonstrated that spatial and recognition memory were unimpaired in adult rats 48 h post-anaesthesia with ketamine alone or in combination with midazolam or dexmedetomidine. All groups learned the radial maze task, as shown by the decreasing number of working and reference memory errors over time. Furthermore, all animals showed a decrease in response latency, a measure that seems to reflect appetitive motivational demands. 36 Thus, it is suggested that the anaesthetic protocols used did not induce alterations in the motivation to locate the rewards, which is essential for a proper performance of the complex learning tasks used in neuroscience research.
Data concerning the effects of the combined administration of ketamine with midazolam or dexmedetomidine on spatial memory in rodents are scarce. Nevertheless, a previous study using the T-maze test showed that ketamine alone or in combination with medetomidine had no effects on spatial learning in mice, 20 and also that ketamine alone did not interfere with radial maze learning. 24 Similar results were obtained in mice treated with ketamine/midazolam when tested in the radial maze. 18 Nevertheless, sub-anaesthetic 37 and anaesthetic 38 doses of ketamine, administered minutes before the spatial tasks caused impairment in the acquisition of the referred tasks. With the same anaesthetic dose (100 mg/kg), our results shows that ketamine alone has no effect on spatial memory, suggesting that an increased time period between administration and training could be enough to avoid these ketamine-induced effects on memory acquisition.
In the present study, animals from the different experimental groups exhibited a robust recognition memory, as defined by the preferential exploration of the novel object/location over the familiar object/location, and a similar discrimination index. In the test phase of the OR trial, animals have to detect and to encode a novel object, while making the update and reconsolidation of the familiar object. 39 Even in the more demanding trials, with a long delay (24 h) between the sample and the choice phase, this cognitive process was not impaired using our anaesthesia protocols. Corroborating our results, Pitsikas and colleagues 40 have reported that acute administration of ketamine (100 mg/kg) did not induce impairments in recognition memory, 48 h or 72 h after treatment and with a 24 h delay for object recognition or object location, respectively. Hou et al. 41 have also reported that the same ketamine dose did not interfere with the retention of the novel object recognition memory in mice. Furthermore, this anaesthetic procedure had no effect, up to seven days post-treatment, 41 on neurons of the prefrontal cortex and hippocampus, which are the brain regions responsible for recognition, spatial memory, 42 and working memory. 43 It has been previously demonstrated by our group that chronic lower doses of ketamine (5 mg/kg) impaired performance in the OR/L test. 13 To our knowledge, the present work is the first study to assess how a single dose of both combinations of ketamine/midazolam and ketamine/dexmedetomidine affects object recognition memory.
In the NOE test, animal responses may reflect anxiety and/or fear, which is reflected in the avoidance activity or exploring the new object. 44 Here, all groups exhibited a similar response to the new object; in general, all treatment groups had emotional reactivity similar to control levels 48 h post-anaesthesia. However, rats in the KET/DEX group spent significantly more time in corner behaviour than the CONTROL and KET groups. This behaviour represents a thigmotactic response, a natural defensive response in which rats remain close to vertical surfaces. Therefore, this result suggests that ketamine/dexmedetomidine treated animals show an increased anxiety and/or fear-like behaviour.
In order to corroborate the NOE results, a more robust and classical test for anxiety, EPM, was performed. The space occupation in this task reflects a conflict between the rodent's preference for protected areas (e.g. closed arms) and their innate motivation to explore novel environments, such as the open arms. 45 An increased time spent and a higher number of entries in the open arms are generally interpreted as a decrease in anxiety-like behaviour. 46 Since the differences detected in the NOE test involved the group (KET/DEX) that had anaesthesia duration more than three times longer than the other groups, we also tested the KET/DEX+A group. These animals were treated with ketamine/dexmedetomidine and, after one hour, anaesthesia was reverted with atipamezole (an α2-adrenergic antagonist). This procedure was included in order to control anaesthesia duration, which could be a variable inducing the results previously referred to. Our data showed that the different anaesthetic protocols used did not affect space occupation in the EPM apparatus. Other ethological measures registered, such as the number of rears, head dips, faecal boli, freezing or stretched-attend postures, were also not affected by the anaesthetic protocols.
Overall, our results clarified the NOE test outcomes, supporting the absence of effects of the anaesthetic protocols used on emotional behaviour. Low ketamine doses, such as 20 mg/kg, may be anxiogenic, 47 while doses which are higher, but lower than our dose, result in anxiolytic-related behaviour. 48 Nevertheless, these results were obtained when ketamine was administered a few minutes to 2 h before the EPM test. In humans, studies of anxiety induced by ketamine are few; and in these studies ketamine does not seem to interfere with anxiety issues, even three days after administration. 49 These studies are in agreement and support, to some extent, our data obtained 48 h post-anaesthesia. Concerning ketamine combinations, little is known about their effects on anxiety, except that dexmedetomidine and midazolam are considered to be anxiolytics.16,30 In humans, both drugs may be given to ketamine-anaesthetized patients in order to induce sedation and anxiolysis; however, these effects are not usually evaluated in the long term, such as after discharge from the post-anaesthesia care unit. 16
Therefore, besides the described ketamine side-effects, 6 it is important to highlight the absence of ketamine memory impairment and alterations on emotional reactivity in our results. These outcomes contradict several reports in rodents which can be explained by methodological differences such as different dose effects (sub-anaesthetic versus anaesthestic doses 37 ), age (young and elderly may be more susceptible to anaesthetic toxicity50,51), frequency of administration (acute versus chronic 13 ), time between anaesthesia and behavioural evaluation, 38 study of different memory processes, 52 and temperature. 40 In accordance, human studies have consistently reported that acute ketamine administration induces short-term psychotic events and transient memory impairment without causing sequelae. 53 With these reports, the variables inherent in clinical settings, painful and stressful procedures, and the large diversity of individuals must all be taken into consideration. The use of animal models enables us to overcome these limitations by using a controlled environment, and no surgery or other invasive procedures in order to study the sole effects of anaesthetics.
As previously stated, the potential effects of anaesthesia in neurobehavioural experiments depend on several factors; thus, there is not simply one appropriate time interval from anaesthesia to behavioural assessments to avoid anaesthesia-related effects. Nevertheless, neurobehavioural experiments may be conducted without expecting long-term anaesthesia effects 48 h after a single injection of 100 mg/kg ketamine and its combinations (midazolam, medetomidine, dexmedetomidine).18,19,20,24 Other anaesthetic protocols may be used for this purpose, such as high concentrations of isoflurane,54,55 sevoflurane and desflurane. 56 However, injectable anaesthesia has the advantage of not requiring any special equipment as volatile anaesthesia does. In addition, injectable anaesthetics are needed to promote balanced anaesthesia. Other anaesthetic protocols such as propofol/medetomidine/fentanyl,57,58 and ketamine combined with other benzodiazepines (diazepam) and α-2 agonists (xylazine) are suitable for use in laboratory rodents; 59 however, further studies are needed regarding their effects at the neurobehavioural level.
In summary, our results show that despite the unconsciousness time varying across the anaesthetic protocols used (ketamine alone, or in combination with midazolam or dexmedetomidine), spatial, recognition memory and anxiety-like behaviours are not affected in adult rats. These results indicate that, in adult rats, the tested anaesthetic combinations and doses do not induce long-term effects regarding memory. Although midazolam and dexmedetomidine are considered to have anxiolytic properties, our results show that after 48 h, these drugs do not affect emotional behaviour. Thus, our results suggest that these combinations are useful for rat anaesthesia and/or sedation procedures in clinical and neurobehavioural research without compromising the quality of the outcomes. Therefore, the use of anaesthesia in laboratory rodents, namely these protocols, should always be considered in order to refine procedures and decrease rodent pain and distress.
Footnotes
Acknowledgements
A Magalhães was responsible for study design, data collection, data analysis and drafting of the article; A. Valentim was responsible for study design, data collection, data analysis and article revision; M Pereira was responsible for data analysis; P Melo was responsible for data collection and article revision; C Venâncio was responsible for study design, data collection and article revision; T Summavielle was responsible for study design and article revision; L Antunes was responsible for study design, article revision and approval of the final version to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research project was financed by Fundação para a Ciência e Tecnologia (FCT) through the research grant PTDC/CVT/099022/2008 and financed by COMPETE: -01-0124- FEDER -009497 (Lisbon, Portugal). Teresa Summavielle was supported by Programa Ciência – Programa Operacional Potencial Humano (POPH)-Promotion of Scientific Employment, ESF and MCTES and program Investigador FCT, POPH and Fundo Social Europeu. Ana Magalhães was supported by PTDC/MHC-PAP/5304/2012. Luis Antunes and Carlos Venâncio were supported by the European Investment Funds by FEDER/COMPETE/POCI- Operacional Competitiveness and Internacionalization Programme, under Project POCI-01-0145-FEDER-006958 and National Funds by FCT under the project UID/AGR/04033/2013.
