Abstract
It has been shown that embryonated chicken eggs can be used as animal models for experimental infections. The aim of the present study was to investigate pigeon embryonated eggs as animal models for experimental neosporosis. An infection with Neospora caninum Nc1 isolate was conducted in chicken and pigeon embryonated eggs to evaluate LD50. After calculation of LD50, 2LD50 of tachyzoites were injected into the eggs. Macroscopic changes of each embryo were observed, and immunohistochemistry (IHC) and molecular methods were used to investigate the parasitic distribution in the tissues. In the present study, histopathological changes were considered, and sections of those used for histopathological examination including the heart, liver, brain and chorioallantoic (CA) membrane were also subjected to IHC. Pigeon embryos showed more macroscopic changes than chicken embryos. A hemorrhage of the CA membrane was the main gross lesion. Microscopic examination of tissues revealed acute neosporosis due to hemorrhage, necrosis and infiltration of mononuclear inflammatory cells. Based on IHC and molecular results, the parasite DNA was detected in the liver, heart and CA membrane. As with chicken embryonated eggs, these results reinforce the susceptibility of pigeon embryonated eggs to N. caninum, and provide new insights into using an inexpensive and available animal model for N. caninum research. The results of the present study suggest that pigeon embryos may be a good choice for studying the biology of N. caninum in living organisms.
Neospora caninum, the causative agent of neosporosis, is an obligate intracellular apicomplexan parasite causing abortion and neonatal mortality in cattle and severe neuromuscular disease in dogs. Since it was first identified in 1988,1,2 this protozoan has emerged as one of the most important etiological agents of abortion in cattle throughout the world.3–5 The combined presence of dogs and poultry in farms was reported to be among several risk factors associated with N. caninum infection in cattle.6,7 Recently, natural infection has been recognized in birds, particularly the domestic chicken (Gallus domesticus), 8 the house sparrow (Passer domesticus) 9 and the common raven (Corvus corax). 10 Some studies have shown that pigeons are a suitable model for N. caninum infection. The results of these studies demonstrated that N. caninum disseminated through various tissues of pigeons experimentally infected with N. caninum induced parasite-specific immunoglobulin (IgG) seroconversion. 11
Having proved the sensitivity of birds to N. caninum, previous studies have investigated experimental N. caninum infection using embryonated eggs of domestic birds, and have proposed embryonated eggs as a completely suitable animal model for neosporosis. 12 Based on the above-mentioned results, the aim of the present study was to compare pigeon with chicken embryonated eggs as models for acute neosporosis.
Animals
All animal work was performed in accordance with the National Institutes of Health (NIH) guidelines for the use of experimental animals, and the study protocol was approved by the Animal Ethics Committee at Shahid Chamran University. This study consisted of two separate steps. The first step was to calculate the LD50. For this calculation, a total of 30 chicken and pigeon eggs were used. In the present study chicken eggs were prepared by the Jahad commercial company (Ahvaz, Khuzestan province, Iran), and all the pigeon embryonated eggs were obtained from the same captive source with proper management. In the second step after calculation of LD50, seven embryonated chicken and pigeon eggs were inoculated with 2LD50 of N. caninum tachyzoites. For each type of egg, five embryonated samples were considered as controls. To evaluate the Neospora effects on different tissues of the pigeon embryo, molecular and macroscopic and microscopic studies were performed in comparison with the chicken embryonated egg. Embryos were euthanized in a glass desiccator jar using open-drop anesthesia with chloroform according to the standard animal ethics guidelines of Iran.
Materials and methods
Parasites
In the present study, tachyzoites of the N. caninum NC1 isolate were used for experimental infections. The tachyzoites were cultured in the Vero cell line in RPMI medium (Sigma Co, St Louis, MO, USA). RPMI medium was supplemented with 2% fetal calf serum, an antibiotic/antimycotic solution containing penicillin (10,000 U), streptomycin (100 µg), and amphotericin B (25 µg) (Invitrogen, Carlsbad, CA, USA), then harvested by scraping off the cell monolayer 4–5 days after infection. For preparation of the necessary inoculum doses, the tachyzoites were pelleted by centrifugation, the concentration set with a Neubauer chamber (Superior, Germany), and diluted in RPMI medium. 13
Samples
Experimental infections were conducted using embryonated eggs from broiler chickens (as the control group) and pigeons. All eggs were maintained in an incubator at a controlled temperature, humidity and rotation until day 8 of incubation. In order to ensure that all changes (mortality and histopathological changes) were related to N. caninum inoculation, the samples were screened for Newcastle disease viruses (NDV) and avian influenza viruses (AIV).
Molecular study for NDV and AIV screening
RNA was extracted from the allantoic fluid of each embryonated egg. Briefly, 1 mL of RNX solution (using a commercial RNA extraction kit; CinnaGen, Tehran, Iran) was added to 250 µL of each allantoic fluid sample, and 200 µL of chloroform was then added to the mixture. After centrifugation at 12,000 rpm for 15 min, each aqueous phase was transferred to another tube. RNA was precipitated with the addition of an equal volume of isopropanol. After washing the sediment with 75% ethanol, it was eluted in 50 µL of distilled water and stored at –70℃ until used.
To identify NDV and AIV by reverse transcription–polymerase chain reaction (RT–PCR), specific primers based on the H9 gene of AIV 14 and the F gene of NDV were used. 15 The cDNA was synthesized using the AccuPower RT PreMix kit (BioNeer Corporation, Daejeon, Republic of Korea) according to the manufacturer’s instructions. Five microliters of total RNA and one microliter of each 20 pmol primer were used for cDNA preparation. The same primers were used in the RT– PCR. Each 20 µL RT–PCR reaction for NDV and AIV screening contained 2 µL PCR buffer 10 ×, 0.6 µL MgCl2 (50 mM), 0.2 µL dNTPs (10 mM), 1 µL of each 20 pmol primer, 5 µL cDNA, 10 µL distilled water, and 0.2 µL Taq DNA polymerase (5 IU/µL) and 2 µL PCR buffer 10 ×, 0.3 µL MgCl2 (50 mM), 0.2 µL dNTPs (10 mM), 1 µL of each 20 pmol primer, 5 µL cDNA, 10.3 µL distilled water, and at the end, 0.2 µL Taq DNA polymerase (5 IU/µL), respectively. For NDV, RT– PCR cycling included an initial denaturation step at 95℃ for 3 min, followed by 40 cycles of denaturation at 95℃ for 30 s, annealing at 55℃ for 60 s, and extension at 72℃ for 60 s. This was followed by a final extension step at 72℃ for 10 min. For AIV, thermal cycling was done as follows: 94℃ for 5 min (as initial denaturing), 30 cycles of 94℃ for 30 s, 50℃ for 30 s, 72℃ for 35 s, and 4 min heating at 72℃ as the final extension. These programs were performed in a Quanta biotech cycler (Quanta Biotech, Surrey, UK). The products were analyzed in 1% agarose gel containing Safe stain, using an ultraviolet transilluminator. RT–PCR was performed to amplify a 488 bp fragment of the AIV H9 gene and a 330 bp fragment of the NDV F gene.
LD50 calculation and experimental infections with 2LD50
Calculation of Neospora LD50 in chicken embryonated eggs using the Reed and Muench mathematical technique.
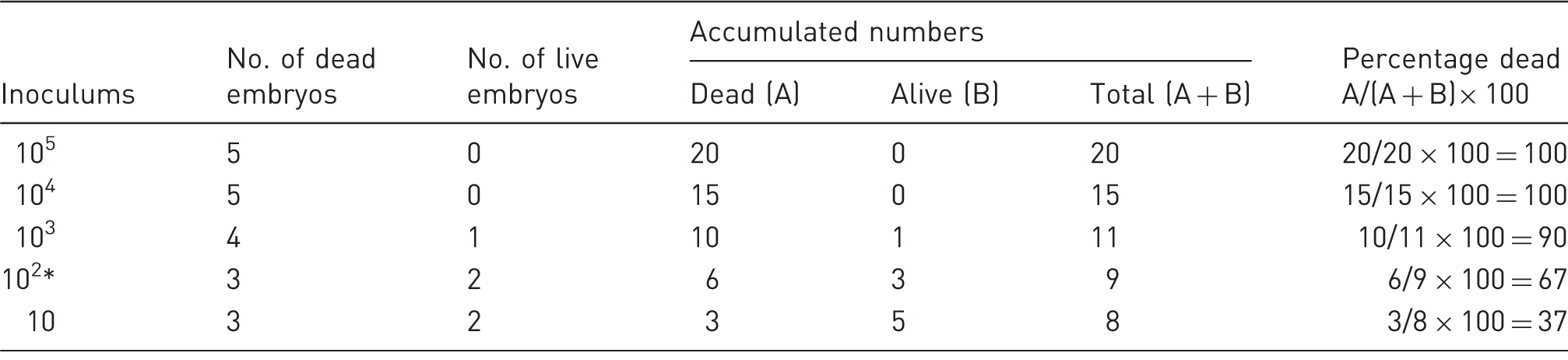
Index: (% dead at inoculums immediately above 50%) – 50% / (% dead at inoculums immediately above 50%) – (% dead at inoculums immediately below 50%): 67 – 50/67 – 37 = 0.6. Apply the index calculated using this formula to the inoculum that produced the infection rate immediately above 50% = 102.6 (mark inoculum with *).
Calculation of Neospora LD50 in pigeon embryonated eggs using the Reed and Muench mathematical technique.
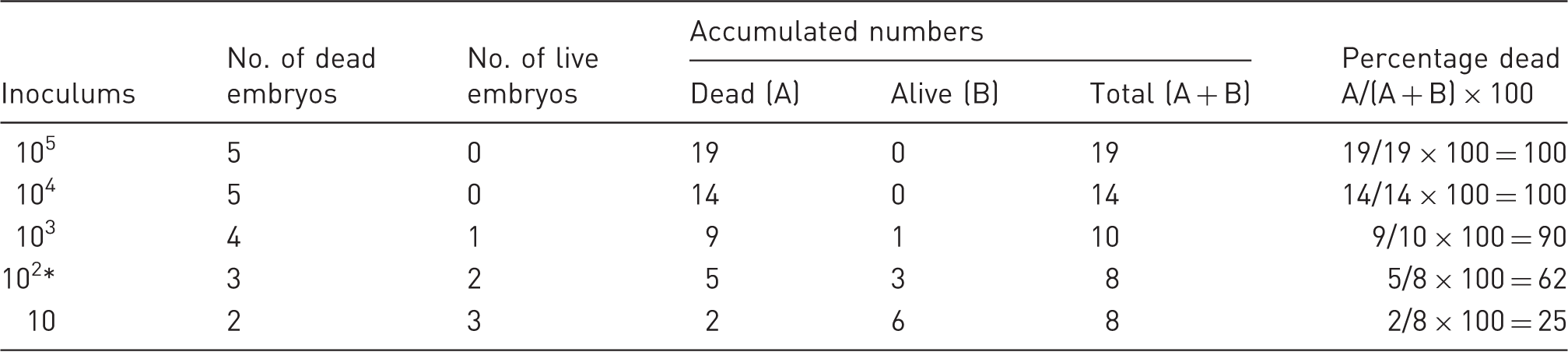
Index: (% dead at inoculums immediately above 50%) – 50% / (% dead at inoculums immediately above 50%) – (% dead at inoculums immediately below 50%): 62 – 50/62 – 25 = 0.3. Apply the index calculated using this formula to the inoculum that produced the infection rate immediately above 50% = 102.3 (mark inoculum with *).
After LD50 calculation, seven embryonated chicken and seven embryonated pigeon eggs were inoculated with 2LD50 of N. caninum tachyzoites. For each type of egg, five embryonated samples were inoculated with sterile RPMI medium as control groups. In consideration of the 3 R principles (replacement, reduction and refinement), specifically reduction, fewer embryonated eggs were included in the control groups. Embryos were euthanized in a glass desiccator jar using open-drop anesthesia with chloroform according to the standard animal ethics guidelines of Iran. Dead or euthanized embryos were necropsied and gross lesions in different tissues were recorded.
Molecular study for N. caninum screening
For the PCR procedure, samples of the liver, heart, brain and CA membrane were collected and DNA was extracted from 50 mg of the different tissues using a genomic DNA purification kit (SinaClon Bioscience, Karaj, Iran). For detection of N. caninum, primers targeting the Nc5 gene were selected from the literature. 17 Primers (Bioneer) used in the reaction were the forward primer Np 21 with the sequence 5'-CCCAGTGCGTCCAATCCTGTAAC-3’ and the reverse primer Np 6 with the sequence 5'-CTCGCCAGTCAACCTACGTCTTCT-3’, yielding a 338 bp product. PCR reactions included a negative control, consisting of the reaction mix and 2 µL of DNase/RNase-free water instead of DNA and a positive control consisting of DNA sample from the tachyzoites of N. caninum NC1 isolate. All PCR were performed in a reaction of 25 µL consisting of 12.5 µL Taq DNA Polymerase Master Mix Red (Ampliqon, Odense, Denmark), 1 μM primer and 50 ng DNA templates. PCR cycling included an initial denaturation at 94℃ for 4 min, followed by 30 cycles of denaturation at 94℃ for 50 s, annealing at 56℃ for 50 s, and extension at 72℃ for 60 s. This was followed by a final extension at 72℃ for 5 min. PCR products were electrophoresed in 1.5% agarose (SinaClon Bioscience) in Tris–acetate–EDTA (TAE) buffer, stained with Green Safe stain (SinaClon Bioscience) and visualized under ultraviolet light.
Histopathological and immunohistochemical studies
For histopathological and immunohistochemical analyses, samples were taken from the liver, heart, brain and CA membrane. Tissue sections were fixed in 10% neutral buffered formalin, dehydrated in graded ethanol, embedded in paraffin wax, sectioned at 5 µm, stained with hematoxylin and eosin, and studied using a routine light microscope. Severity of microscopic lesions was scored by the evaluating pathologist on an ascending scale of 0–4. Grade 1 pathological changes were considered to be minimal, Grade 2 lesions mild, Grade 3 lesions moderate, and Grade 4 changes severe. In a given group of tissue specimens, an overall average severity score was calculated by summing the severity grades for each affected tissue and dividing by the total number of tissues evaluated in that group. For IHC staining, the anti N. caninum antibody (primary antibody) was produced by preparing killed tachyzoite N. caninum antigen (immunogen) and immunizing two rabbits subcutaneously with a mixture of antigen and complete and incomplete Freund’s adjuvant and without adjuvant in three injections with a two-week interval; and finally, detection of the polyclonal antibody titer by the modified agglutination test (MAT). Consecutive sections to those used for histopathological examination were subjected to IHC. After dewaxing and hydrating the sections, antigen retrieval was carried out using citrate buffer. Slides were incubated with primary antibody for 120 min, and visualized with an EnVision + Dual Link System HRP (Dako, Glostrup, Denmark) for 45 min at room temperature. The color was developed with a freshly prepared 3,3′- diaminobenzidine (DAB) solution and the sections were lightly counterstained with Mayer’s hematoxylin, dehydrated and then mounted. Positive samples in PCR were considered as positive controls of IHC, and in negative controls primary antibodies were substituted with phosphate-buffered saline (PBS).
Statistical analysis
Fisher’s exact test was run to compare the number of positive samples detected by PCR and IHC. Also, Mann–Whitney U-test was used for comparison of microscopic lesion grades in the different tissues. P values of < 0.05 were considered to be statistically significant.
Results
Mortality rate of experimental infection and LD50 calculation
In the 10, 102, 103, 104 and 105 tachyzoite chicken groups, three, three, four, five and five embryos died, respectively. And in the 10, 102, 103, 104 and 105 tachyzoite pigeon groups, two, three, four, five and five embryos died, respectively. According to the mortality rates and calculations done using the Reed–Muench method, 16 LD50 in chickens and pigeons was between 102 and 103 doses, and about 102.6 and 102.3 tachyzoites/100 µL, respectively (Tables 1 and 2).
Molecular findings
Number of different infected tissues detected by polymerase chain reaction (PCR) and immunohistochemistry (IHC) in chicken and pigeon embryonated eggs and grade of microscopic lesions.
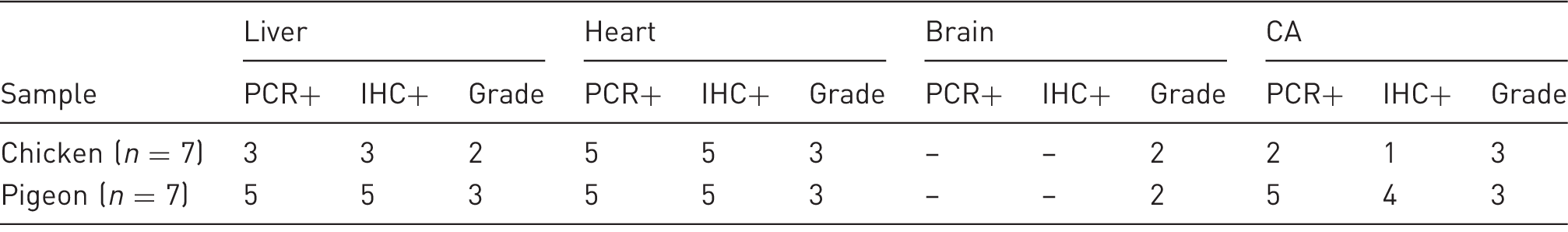
Grade 1: minimal, Grade 2: mild, Grade 3: moderate, Grade 4: severe.
Macroscopic changes
After inoculation with 2LD50 tachyzoites, no macroscopic lesions in the heart, liver and brain of the embryos were found. Cerebral edema was seen in only one chicken embryo whereas three pigeon embryos showed acute generalized edema, and in one pigeon embryo edema throughout the brain and body with severe jaundice was observed (Figure 1). Hemorrhages associated with the thickening of the CA membranes were seen in one chicken embryonated egg and four pigeon embryonated eggs.
Acute generalized edema with severe jaundice in pigeon embryo experimentally infected with Neospora caninum tachyzoites (right embryo); left embryo: control.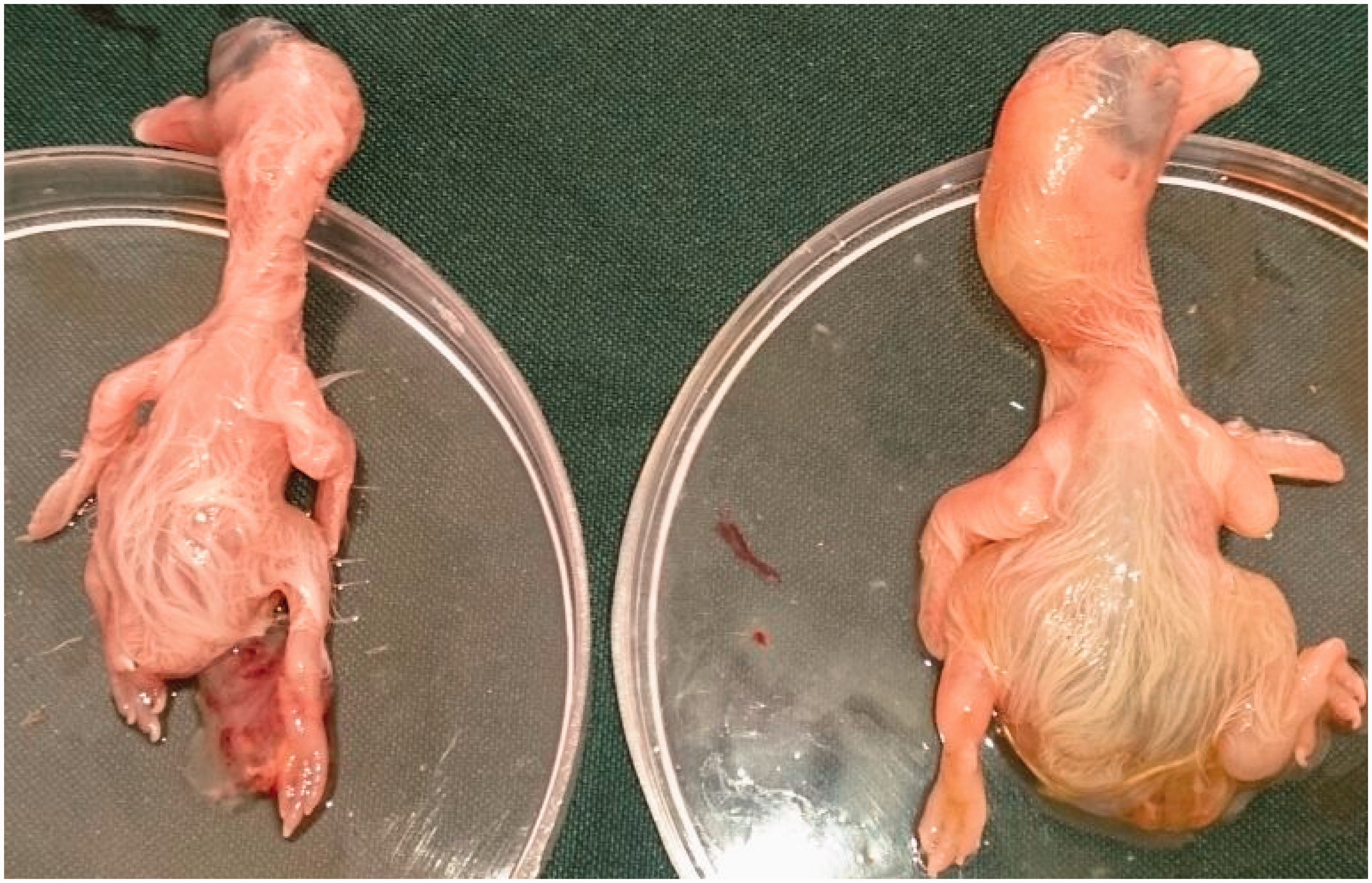
Histopathological and immunohistochemical findings
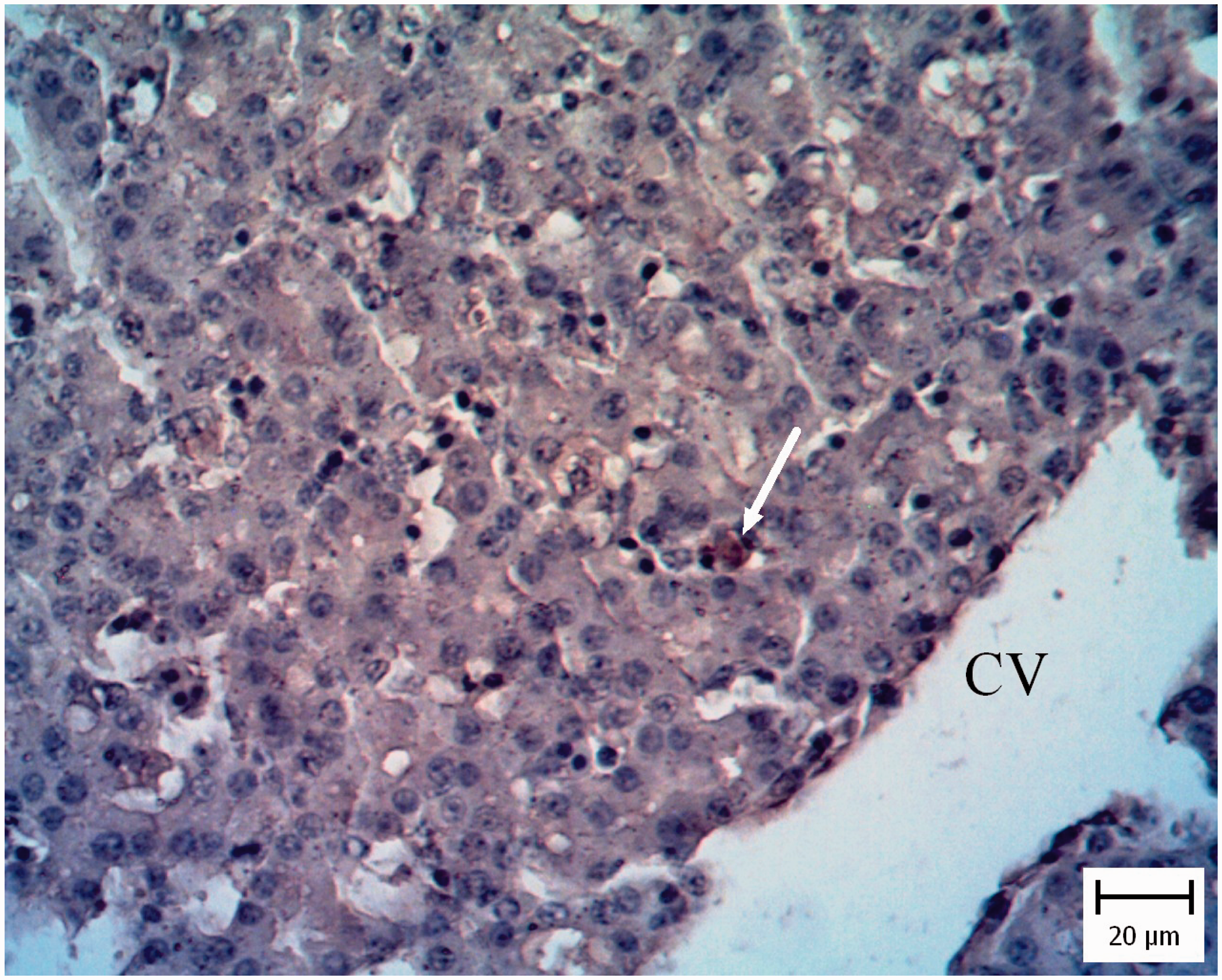
In five pigeon and three chicken embryo livers, the hepatic architecture was moderately disrupted. There were multiple random foci of coagulation necrosis variably associated with light heterophilic infiltration. Also hyperemia and moderate mixed mononuclear inflammatory cell infiltration were present in the portal regions. Moderate tachyzoites were located among the inflammatory cells and lining in the sinusoidal spaces (Figure 2). Presence of tachyzoites was confirmed by IHC (Figure 3).
Pigeon embryo liver. Five days after Neospora caninum inoculation. Note presence of tachyzoites in sinusoidal space (arrows) (hematoxylin and eosin). Pigeon embryo liver. Five days after Neospora caninum inoculation. The brown tachyzoites are obvious between hepatocytes (arrow). (Immunohistochemical staining for N. caninum, hematoxylin counterstain.)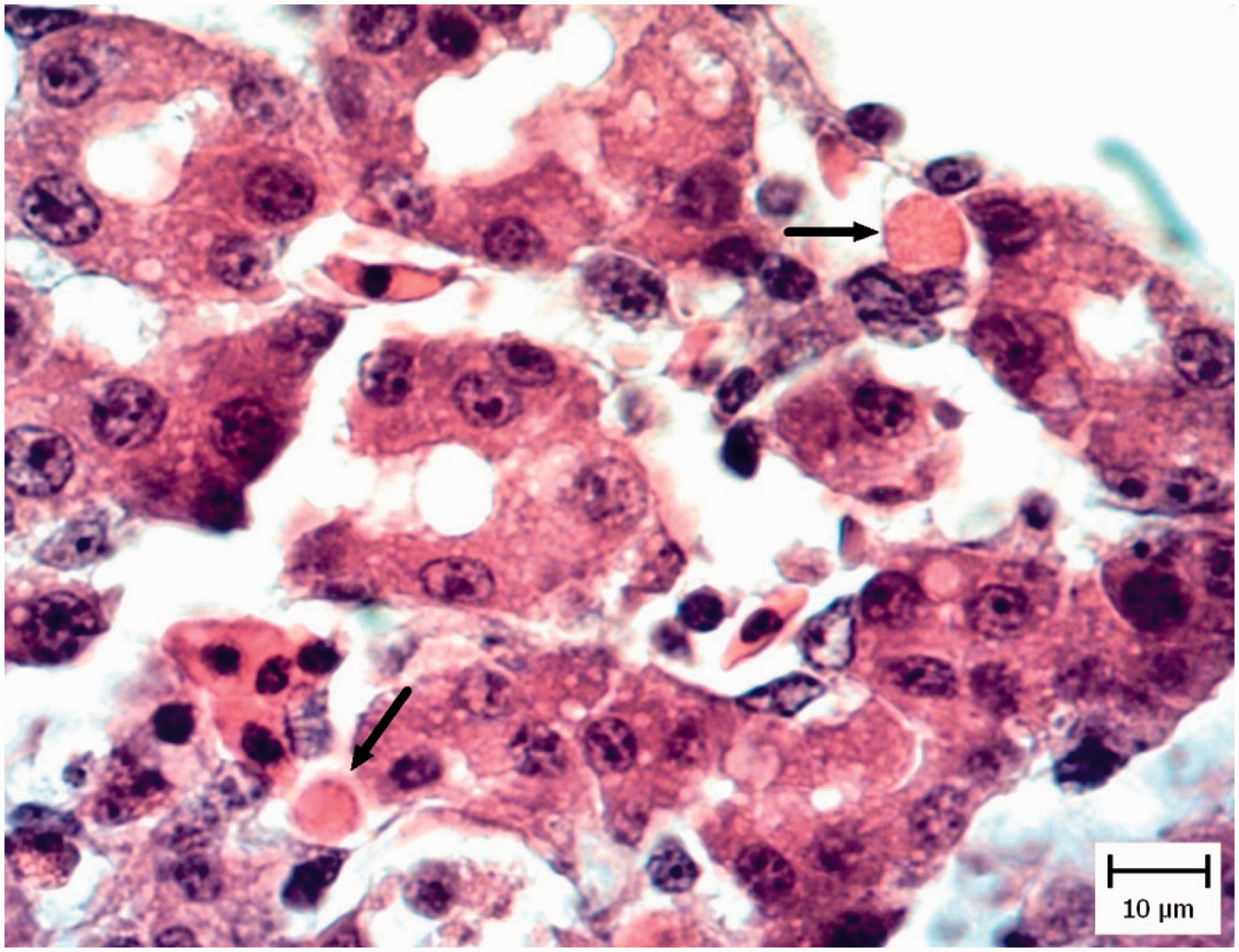
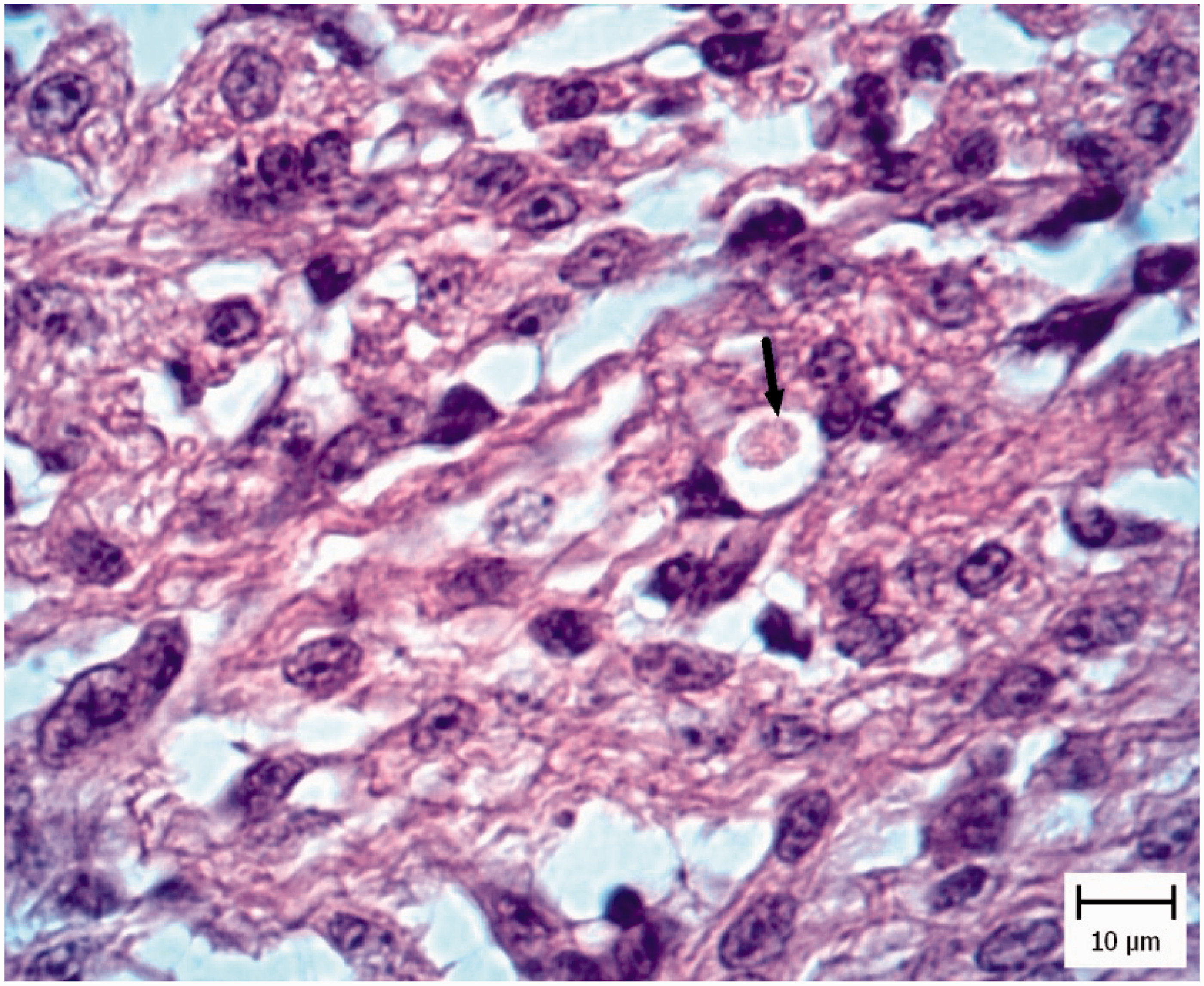
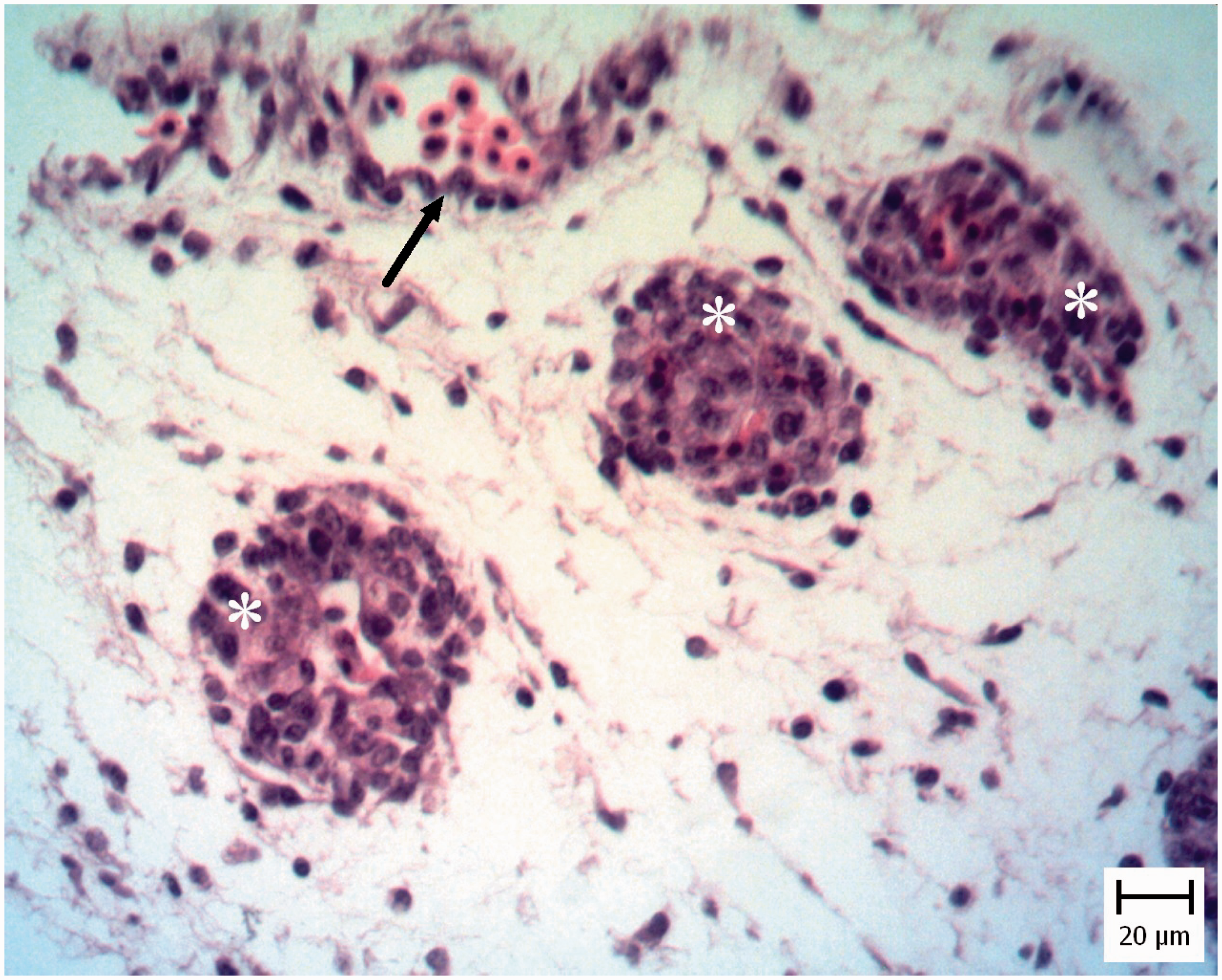
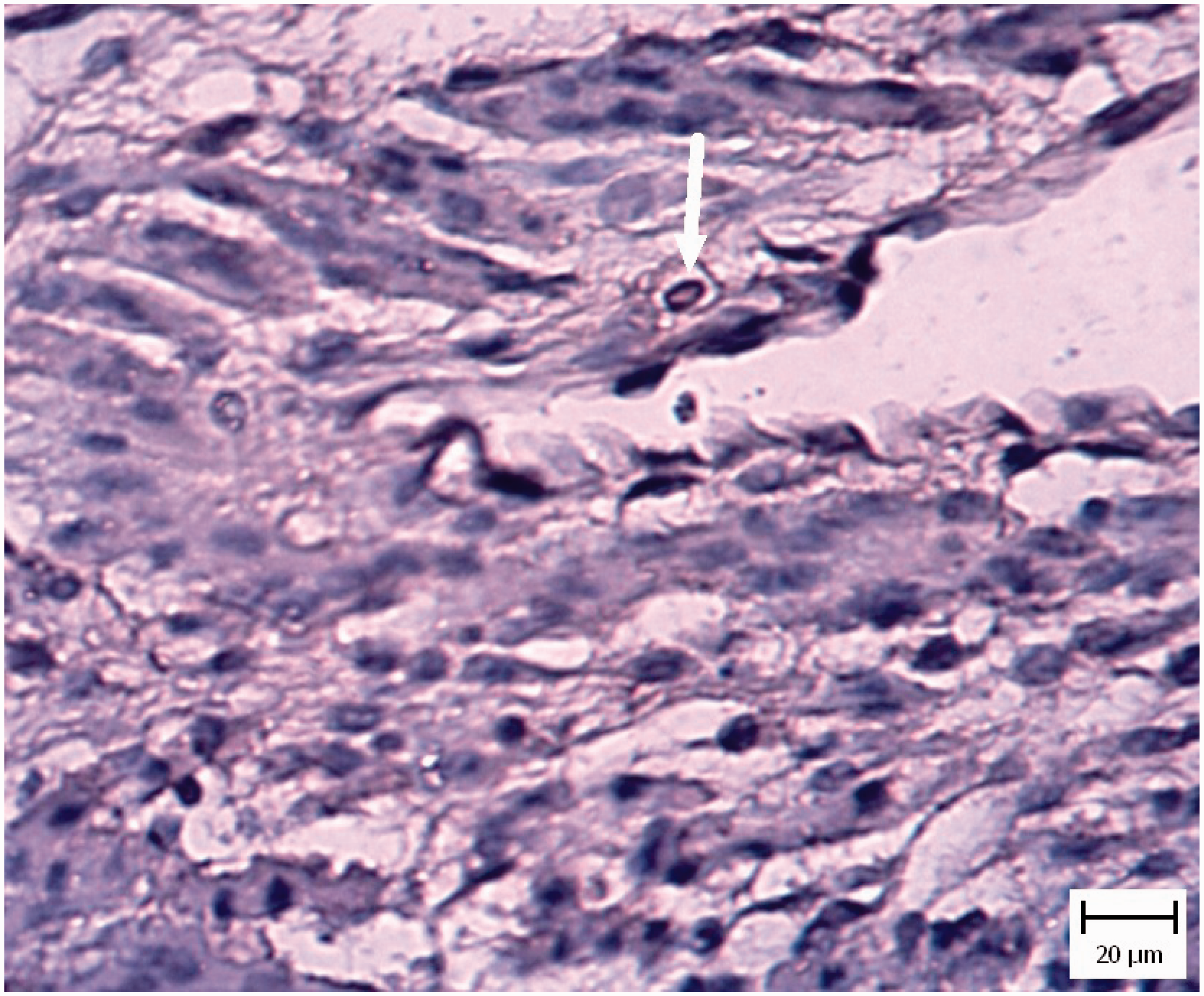
In five chicken and five pigeon embryos, cardiomyocyte necrosis with mononuclear inflammatory cell infiltration was seen (Figure 4). Also tachyzoites were present between myocytes (Figure 5) and they were confirmed by IHC staining (Figure 6).
Pigeon embryo heart. Five days after Neospora caninum inoculation. Note necrosis of myocytes (arrows) (hematoxylin and eosin). Pigeon embryo heart. Five days after Neospora caninum inoculation. Note the obvious tachyzoite in a vacuole (arrow) (hematoxylin and eosin). Pigeon embryo heart. Five days after Neospora caninum inoculation. Note the obvious brown tachyzoites between myocytes (arrow). (Immunohistochemical staining for N. caninum, hematoxylin counterstain).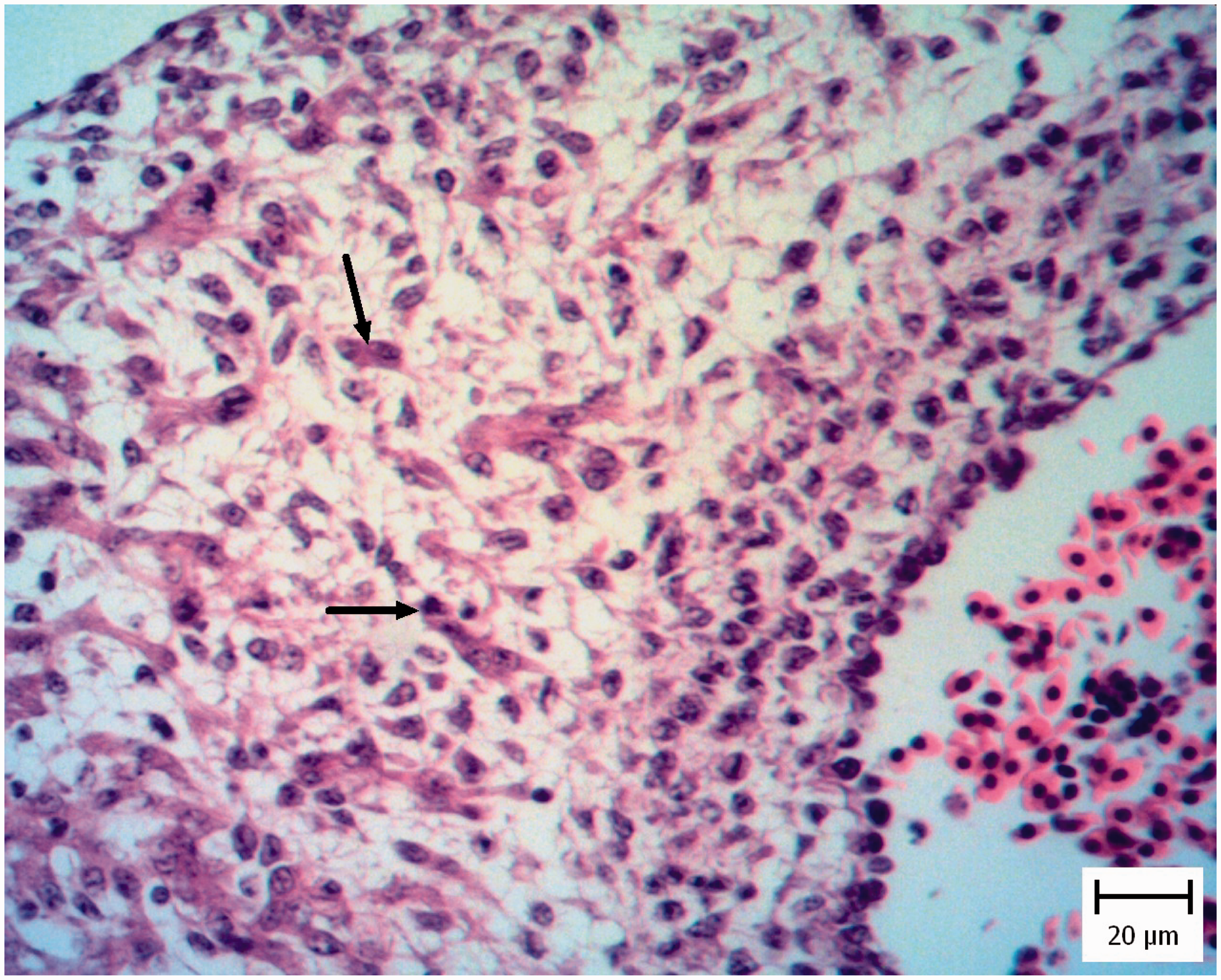
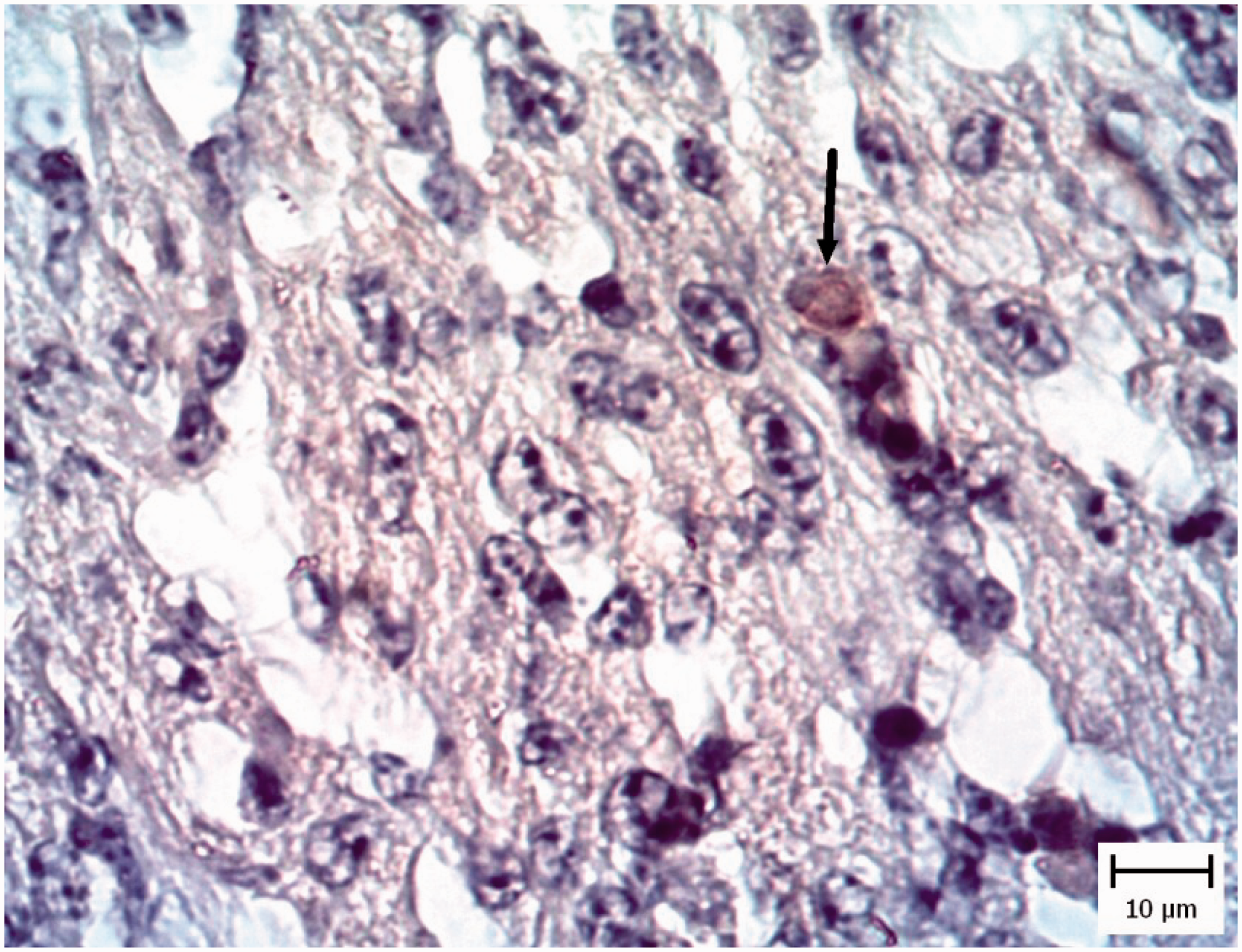
In one chicken and three pigeon embryos, cerebral edema with dilated Virchow–Robin spaces was seen but IHC results showed no evidence of tachyzoites.
Histopathological investigation of CA membranes revealed non-inflammatory edema in four pigeon embryos and one chicken embryo (Figure 7). Also vasculitis was obvious and it was specified with mononuclear inflammatory cells and heterophil infiltration in the vessel walls (Figure 8). Tachyzoites were detected between connective tissues by IHC (Figure 9).
Pigeon embryo chorioallantoic membrane. Five days after Neospora caninum inoculation. Note the obvious free spaces between the connective tissue (asterisks) and the thickness of membrane has been increased (hematoxylin and eosin). Pigeon embryo chorioallantoic membrane. Five days after Neospora caninum inoculation. Vasculitis which is characterized by infiltration of inflammatory cells in the wall of chorioallantoic membrane vessels is evident (asterisks). Arrow shows normal vessel (hematoxylin and eosin). Pigeon embryo chorioallantoic membrane. Five days after Neospora caninum inoculation. Note the obvious brown tachyzoites between connective tissue (arrow). (Immunohistochemical staining for N. caninum, hematoxylin counterstain).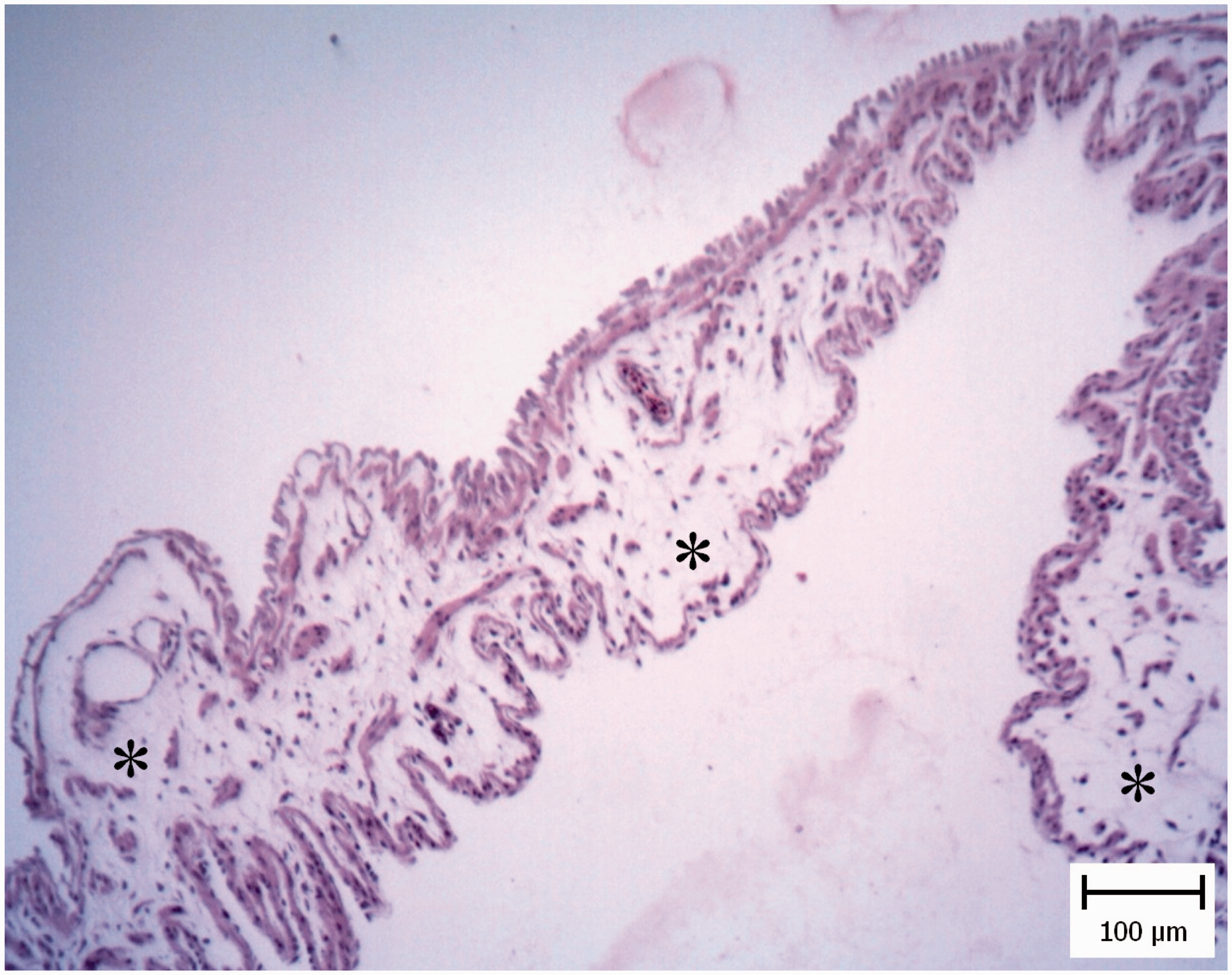
Considering the pathological changes in the different tissues, the liver of pigeon embryos showed more significant changes than in chicken embryos (P < 0.05).
Number of positive samples detected by IHC in different tissues is presented in Table 3. Based on IHC results, the CA membranes of pigeon embryos were more infected than in chickens (P < 0.05), however in other tissues the differences were not significant.
Discussion
The aim of the present study was to compare pigeon with chicken embryonated eggs as models for Neospora infection. In the present study the LD50 of broiler chickens and pigeons were 102.6 and 102.3, respectively. Based on the results, pigeon embryonated eggs were found to be susceptible to Neospora infection. In seeking to develop a laboratory animal model for neosporosis, scientists have found that inbred mouse strains vary in their susceptibility. Most mouse strains are resistant, but BALB/c and C57BL/6 mice develop clinical neosporosis and meningoencephalitis after inoculation with the parasite. 18 Death of animals during the study and congenital infections have been problematic. Embryonated eggs have been used over decades for studying protozoan biology and also as models for protozoan isolation and propagation.19–21 The use of chicken embryos to produce Toxoplasma gondii antigens from the late 1940s until early 1970s was a feasible method due to their low cost and sterile nature. This experimental model also has the advantage of avoiding euthanasia of birds that have hatched. 19 Moreover, different parasitic stages can be studied in the embryonated eggs. For example vertebrate and invertebrate specific stages of Trypanosoma cruzi are propagated inside egg yolks and CA membranes, respectively. 22 Recent studies have demonstrated that chickens may be intermediate hosts of N. caninum. Based on the results from previous studies in laying hens, no signs of vertical transmission were observed. Furthermore, embryonated eggs inoculated by the allantoic cavity route demonstrated susceptibility to infection, and dogs became infected after ingestion of a certain amount of inoculated eggs, producing either oocysts or specific IgG antibodies. Their results demonstrated that embryonated eggs could be a useful model for studying the parasitic biology. 13 In another study, LD50 of N. caninum was determined to be between 102 and 103 doses, and about 102.3 tachyzoites which were administered in the CA liquid of broiler chicken embryonated eggs. 23
After inoculation of 2LD50 tachyzoites in the present study, cardiomyocyte and hepatocyte necrosis was observed in a proportion of embryos infected with Neospora tachyzoites. Whereas three pigeon embryos showed acute generalized edema (especially cerebral edema), brain edema was seen in only one chicken embryo. It is notable that in the molecular and IHC results, evidence of tachyzoites presence was not observed in the brain. Edema can result from heart and liver dysfunction and it can be concluded that liver and heart impairment due to Neospora infection may cause generalized edema. After inoculation of 2LD50 tachyzoites, hemorrhages associated with the thickening of the CA membranes were seen in one chicken and four pigeon embryonated eggs. It should also be considered that pigeon embryos showed more severe macroscopic changes than in chicken embryos. In the current study, based on the molecular and IHC results, Neospora tachyzoites were detected in livers, hearts and CA membranes. N.caninum is more frequently reported in the brain,8,24 but our results are in accordance with the study that demonstrated parasite aggregation in the brain was not predominant in birds. 25 Trafficking of leukocytes to disseminate intracellular parasites via a Trojan horse-type mechanism has been postulated for apicomplexan parasites. It is likely that immune cells, such as mononuclear phagocytic cells, are responsible for Neospora transport. 26
The microscopic findings of our study in broiler chickens and pigeons showed hemorrhage, necrosis and infiltration of the mononuclear inflammatory cells and tachyzoite aggregations in the heart, liver and CA membrane. In other experimental study, in embryos that received 105 and 106 N. caninum tachyzoites, intense parasite multiplication produced extensive inflammation throughout the CA membranes, followed by widespread necrosis and the marked presence of parasitophorous vacuoles in tissues, especially in the brain, heart and liver. 13 But based on the IHC and molecular results in the present study, tachyzoites were not detected in the brain. Based on the LD50 values, macroscopic changes, PCR and IHC results and microscopic lesions, pigeon embryonated eggs were more sensitive, and injury intensities were statistically more than in chicken embryonated eggs.
Conclusions
Previous studies have shown that columbiform birds are sensitive to Neospora and may be good candidates for studying N. caninum infection. 27 In the present study, pigeon embryonated eggs were found to be suitable models for neosporosis and can be used as replacements for birds as models of disease to avoid adult bird euthanasia. Further investigations are required to validate this claim.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the research grant provided by Shahid Chamran University of Ahvaz.
