Abstract
Breast cancer is the most common type of cancer diagnosed in women, and the second most common cause of cancer-related deaths in women in North America. The use of laboratory mice in research is an essential tool for the study of breast cancer biology and for pre-clinical therapeutic development. While subcutaneous flank injections of cancer cells are widely used for studying breast cancer biology and for exploring novel therapies, orthotopic xenografting of tumors into the mouse mammary gland allow for the study of breast cancers in a biologically relevant microenvironment. In this study we report a modification of the method of orthotopic injections of cancer cells into the mouse mammary gland which greatly reduces the effects of surgery in mice including decreased wound size, procedure time and anesthesia. It also removes the risk of accidentally puncturing the peritoneal cavity. Consequently post-operative animal handling and stress are significantly reduced. All of these advantages are present without compromising procedure success rate. Therefore, this modification makes orthotopic mammary gland injection a more efficient procedure and greatly improves animal welfare.
Keywords
One in eight women in North America will develop breast cancer in her lifetime, and a substantial proportion of these patients will develop resistance to therapy and will eventually succumb to the disease. 1 The use of laboratory mice in cancer research is an essential tool for understanding cancer biology, and to facilitate pre-clinical therapeutic testing. The subcutaneous flank injection of human breast cancer cells into immunocompromised mice is a relatively non-invasive technique widely used for studying tumor biology and therapeutic responses. This technique, however, has limitations in that it does not take into account the impact of the microenvironment of the mammary gland on cancer growth. While there are differences in the composition of the mammary stroma between mice and humans, 2 mammary fat pad injections in mice are thought to, at least in part, recapitulate the original microenvironment of the human breast. Classically, this procedure involves a V-like incision from the midline to the mid-ventral region, continuing laterally and caudally towards the mouse thigh. The skin is then carefully detached from the peritoneal cavity exposing the abdominal and inguinal fat pads. After injection of the desired cells the incision is carefully closed. Staples or sutures are then removed after 10–12 days.3,4 In this study we report a modification of this widely used technique which is more rapid, and importantly reduces incision sizes of 1.5–3 cm to 3 mm, allowing the wound to be closed using tissue adhesive, thus eliminating the need for staples or sutures and significantly reduces animal handling and stress.
Materials and methods
Immunocompromised 3–6-week-old female mice (The Jackson laboratory/ Bar Harbor, ME, USA) are first anesthetized using 3% inhalant isoflurane. Eye lubricant is applied, hair around the abdominal and inguinal fat pads is trimmed and the skin is sterilized (Figure 1a). With the aid of magnifying surgical loupes (RS-6698; Roboz Surgical Instrument Co, Gaithersburg, MA, USA), a small incision of less than 3 mm is made externally and caudally to the fourth nipple with the tip of micro-dissecting scissors (Figure 1b). The nipple is lifted, and with the tip of the micro-dissecting scissor blade, the skin is detached from the fascia below through the small incision in an external orientation (towards the fat pad). Once detached the skin is gently glided to locate the fourth fat pad below. With the aid of micro-dissecting forceps, the ventralmost part of the fat pad is gently pulled out and exposed through the small incision (Figure 1c). The desired cells can then be injected using an insulin syringe with a volume ≤120 µL (Figure 1d). Successful injection is confirmed by the swelling of the tissue. With the aid of a Q-tip dipped in 1 × penicillin–streptomycin–neomycin antibiotic mixture solution (15640055; Thermo Fisher Scientific, Waltham, MA, USA), the exposed fat pad is gently pushed inward allowing it to glide back into place (although most times it glides back on its own; Figure 1e). The small incision is sealed using tissue adhesive (Vetbond; 3 M, St Paul, MN, USA) (Figure 1f). Finally post-operative analgesia (bupivacaine) is applied. All procedures involving animal subjects have been approved by the Institutional Animal Care and Use Committee (IACUC) at the Dana-Farber Cancer Institute.
Less invasive procedure for orthotopic injection of breast cancer cells into the mouse mammary gland. (a) Abdominal mammary gland and nipple. (b) Small incision ≤3 mm (external and caudal) below the fourth nipple. (c) Exposed fat pad. (d) Exposed fat pad can be injected with a volume ≤120 µL. (e) Fat pad is allowed to go back to its position. (f) Incision is sealed using tissue adhesive.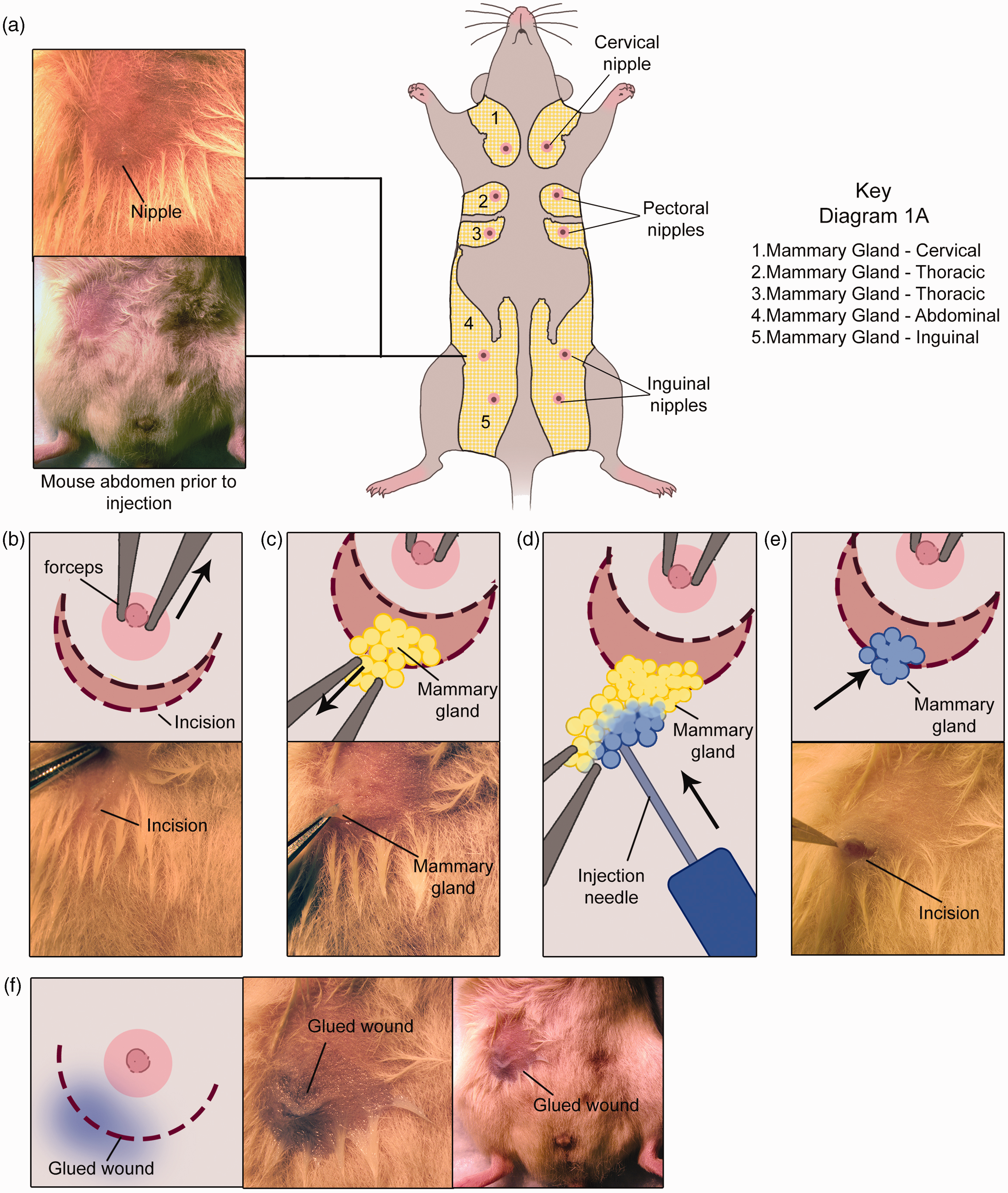
Discussion
Orthotopic injections of human breast cancer cells into the mouse mammary gland provide an invaluable tool in breast cancer research and for the development of new breast cancer therapeutics. This is an involved surgical procedure, and causes substantial stress in prepubertal 3–6-week-old mice including staple or suture removal 10–12 days post-surgery. Other modifications of orthotopic mammary gland injections have been developed previously.2,3,5 Unlike these other methods used for less invasive injections (such as intraductal injections), our modification method allows for full visualization of the fat pad and therefore reduces the risk of injection outside the fat pad. Brill and colleagues have developed an excellent sparing procedure for cleared fat pad transplantation analysis that can also be used for orthotopic injections. 5 While our method cannot be used for fat pad clearing, the sparing procedure used in this study is less stressful for the mice as it does not require further animal manipulation needed for staple removal. Although successful xenograft take rate depends on the intrinsic properties of the cells injected and the hormonal environment of the mice (e.g. adequate administration of estrogen in the study of estrogen receptor positive xenografts) we observed no apparent differences in experimental success (tumor take) when comparing the classical, more invasive method with the present modification (which is ∼98%) using MCF-7 cells. We present here a modification of the orthotopic injection method to the mouse mammary gland procedure which resulted in a considerable improvement to animal welfare as it requires a significantly smaller incision (reduced from ∼3 cm to ∼3 mm), thus lessening the effects of surgery. In our hands, the classical procedure can take >20 min depending on the experimentalist skill level while this new modification method usually takes less than 10 min. Since this is more time efficient, it reduces the amount of anesthesia needed. In the classical procedure, the skin is carefully detached from the peritoneal cavity to expose the mammary gland. However, it is not uncommon (∼15% of the time) to accidentally tear the peritoneal cavity as the skin is being lifted, thus requiring sutures. As the peritoneal cavity is not exposed using the modification method, it eliminates the risk of any accidental peritoneal cavity perforation and potentially lethal complications. Finally, because the small incision is sealed with tissue adhesive it removes animal handling during staple or suture removal and therefore reduces stress to the animal. Since this modification method does not decrease tumor take rate and does not require extra training to perform the procedure, we consider this to be a significant improvement to the existing mouse mammary gland orthotopic injection procedure.
Footnotes
Acknowledgements
We are extremely grateful to Ana Tavera Mendoza for the design and drawing of the figures. We would also like to acknowledge the staff of the Dana-Farber Animal Facility, namely Michael Terrasi, Antonia Garcia, Daisy Moreno, Carmen Da Silva, Elsy Moreno and Catherine A Sypher for their overall excellence in animal care.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
