Abstract
An essential aspect of genetically-engineered mice (GEM) is the ability to produce live animals after the appropriate injection procedure. Animals are produced by implantation of manipulated embryos into pseudopregnant females for gestation, parturition, and growth to the weaning stage. This study was carried out to test whether the anesthesia used during surgery could affect the number of pups produced. Anesthetics commonly used for implant surgery include tribromoethanol (Avertin) delivered by intraperitoneal (IP) injection, IP-injected ketamine:xylazine or ketamine:medetomidine mix, and inhaled isoflurane. To determine if the anesthesia used might affect the number of animals produced, each anesthetic agent was tested in implant surgeries and the numbers of pups produced using both wild-type and GEM embryos were assessed. Parallel studies were conducted in institutions in the EU and in the USA. Based on a direct comparison of pregnancy status, number of pups born, and number of pups weaned for each agent, we found no statistical differences among the three anesthetics. We conclude that all three anesthetic agents tested are equally useful for implantation surgery.
The surgical implantation of mouse embryos into recipient dams is an essential procedure for any genetically-engineered mouse (GEM) program. The embryo transfer procedure is used to generate animals containing modified genes, 1 expand colonies by implantation of embryos fertilized in vitro, 2 and eliminate endemic murine pathogens by rederivation of contaminated mouse colonies.3,4 While institutional facilities have generated many GEM, it is the advent of large, worldwide initiatives such as the International Knockout Mouse Consortium/International Mouse Phenotyping Consortium (IKMC/IMPC; www.mousephenotype.org/) and Infrafrontier–European Mouse Mutant Archive (EMMA) (www.infrafrontier.eu) that has exponentially increased the demand for GEM models. Additionally, the recent development of the CRISPR-Cas9 endonuclease approach in animal transgenesis is defining a new era for biology.5–7 Dedicated GEM programs must optimize time and resources to meet increased demand. Procedures must be done efficiently and in a manner that reduces the risk of complication to either the recipient dam or the embryos. Metrics such as numbers and quality of sperm and oocytes produced, in vivo and in vitro fertilization (IVF) rates, embryo yields, and pup viability are essential to determine areas where refinement may enhance production.
Manipulators disagree on the effect that anesthetic agents may have on recipient dams or embryo viability. Historically, embryo transfer techniques often used 2,2,2-tribromoethanol (TBE; previously known as Avertin) to anesthetize dams for implantation.8–11 TBE rapidly induces a depth of anesthesia for durations suitable to complete the procedure, approximately 15–20 min at 250 mg/kg.12,13 Some have reported success in administering a second dose if necessary,8,14 while others have reported an increased incidence of mortality. 15
The length of time to recover ambulation after anesthesia with TBE is prolonged (30–90 min). Also, degradation can occur in the presence of heat or light, producing toxic by-products that can cause peritoneal adhesions, 16 peritoneal and visceral necrosis near the site of injection, and increased risk of mortality. 17 Currently, use of TBE has become problematic, as the eighth edition of The Guide for the Care and Use of Laboratory Animals 18 discourages the use of non-pharmaceutical grade compounds in laboratory species. Despite this, many transgenic facilities in the USA have successfully lobbied their local animal care and use committees to approve the use of TBE, as they fear alternative anesthetics will affect the numbers of pups produced. Additionally, similar restrictions for TBE use have been established by EU country legislatures.
One popular alternative is the combination of ketamine with xylazine (K/X), or with medetomidine (K/M), components which are available in pharmaceutical grade. At a dose of 120:16 mg/kg (K/X) or 75:1 mg/kg (K/M) respectively, intraperitoneal injections of the K/X or K/M mix will induce anesthesia for approximately 20–30 min, with an additional half-hour being required to recover ambulation. 16 Unlike TBE, ketamine is a controlled substance, requiring a Drug Enforcement Administration (DEA) license and drug log for use in the USA. In Italy, ketamine is classified as a psychotropic drug; as such, the user must maintain authorized acquisition and use records, and must store the agent in a locked cabinet. There is evidence to suggest that use of K/X or K/M allows for comparable embryo yields as does TBE, but without inducing the same adverse effects. 19 However, we have observed that the margin of safety appears to be narrower than for TBE. The use of medetomidine allows for partial reversal of anesthesia by the administration of atipamezole (1 mg/kg).
A third alternative involves the use of inhalant isoflurane. Unlike injectable forms of anesthesia, the depth of anesthesia is easily adjusted. Induction is rapid, and suitable anesthetic depth is easy to achieve and maintain. 20 The time to recover ambulation after cessation of this anesthesia is much shorter, approximately 3–5 min. A precision vaporizer is recommended to obtain a calibrated dose of anesthetic. Oxygen tanks, hosing, vaporizers, and scavenger systems are all recommended to facilitate this administration. Regular maintenance is necessary to ensure consistent dosing and to reduce personnel exposure. Thus, inhalant anesthetic delivery systems can require substantially more set-up and maintenance than injectable anesthetics. Positioning of the animal can also be problematic, as the manipulator must ensure that the dam’s nose is adequately exposed to the source of the gas at all times; alternatively, endotracheal intubation may help to maintain a proper anesthetic depth.
Relatively little in vivo research on the effects of different anesthetics on embryo transfer and recovery of pups has been performed. Multiple studies have documented the detrimental effects of isoflurane on two-stage embryos in vitro,21–23 and another study examined the effects on pups conceived by dams administered different anesthetics during gestation. 24
Ultimately, the goal of embryo implantation is to obtain live offspring. This study examines these common anesthetic regimens for their effects on pregnancy rate and pup yield. Direct comparison of these methods revealed no statistical differences among the three anesthetic agents in their effects on pregnancy rate or pup production.
Materials and methods
Animals
MD Anderson Cancer Center (MDA)
Female (36–45 g, 2–5 months of age) and male (2–6 months of age) CD-1® mice were obtained from Charles River Laboratories (CRL; Wilmington, MA, USA). Males were housed singly and females housed in groups of five in ventilated microisolator cages (Tecniplast USA, Exton, PA, USA) at a temperature of 20 ± 2℃ and a humidity of 55 ± 15% with 12–15 air exchanges per hour, on corncob bedding (Bed O’Cob; Andersons Lab Bedding, Maumee, OH, USA). All mice were provided with filtered and chlorinated water ad libitum. Males and unmanipulated females were given irradiated Purina PicoLab Rodent Diet #5053 (Purina LabDiet, St Louis, MO, USA). Surgically-treated females were singly housed and given irradiated Harlan–Teklad Breeder Diet #7926 (Harlan Laboratories Inc, Madison, WI, USA). Animals were maintained on a 12:12 h light/dark cycle. Based on sentinel health surveillance, the housing area was free of mouse parvovirus, minute virus of mice, mouse hepatitis virus, mouse norovirus, GDVII, enzootic diarrhea of infant mice, Sendai virus, pneumonia virus of mice, Reo virus, Mycoplasma pulmonis, lymphocytic choriomeningitis virus, mouse adenovirus, ectromelia virus, K virus, polyoma virus and Helicobacter spp. Sentinels are screened annually by bacterial culture or polymerase chain reaction (PCR) for Bordetella bronchiseptica, Corynebacterium kutscheri, Salmonella sp., Pseudomonas sp., and Citrobacter rodentium. Non-contact sentinels are screened quarterly for Syphacia obvelata, Aspiculuris tetraptera, Myocoptes musculinus, Myobia musculi, and Radfordia affini. All animal experiments were approved by the Institutional Animal Care and Use Committee at the University of Texas MD Anderson Cancer Center (an Association for Assessment and Accreditation of Laboratory Animal Care [AAALAC] International accredited institution).
CNR–Institute of Cell Biology and Neurobiology (CNR)
For recipients, female (30–35 g, 2–4 months of age) CD-1 mice were supplied by the CNR–EMMA–Infrafrontier unit (Monterotondo Scalo, Rome, Italy). As embryo donors, GEM of four different backgrounds (C57BL6/NCnrm, C57BL6/JCnrm, BALB/cByJCnrm and FVB/NJCnrm) were used by the EMMA/Infrafrontier Network. Mice were kept in individually-ventilated cage (IVC) racks and containment high-efficiency particulate arrestance (HEPA) biosafety cabinets (Tecniplast, Gazzada, Italy), at a temperature of 20 ± 2℃ and a humidity of 55 ± 15% with 12–15 air exchanges per hour using a 12:12 h light/dark cycle. Certified wooden dust-free spruce particles (scobis one; Mucedola, Settimo Milanese, Milan, Italy) were provided as bedding. Mice were fed a standardized diet, or a diet enriched with fat and protein (4RFN and Emma 23; Mucedola) depending on the strain. All mice were provided with filtered and chlorinated water ad libitum. Experimental and sentinel mice were maintained free of excluded pathogens according to the Federation of European Laboratory Animal Science Associations (FELASA) recommendations. 25
Superovulation, collection of germplasm, in vitro fertilization (CNR only) and cryopreservation
MDA: Superovulation and cryopreservation
CD-1® females (>35 g, 3–6 months of age) that had been scheduled for removal from the colony were used as embryo donors. Females were superovulated with 5 IU of pregnant mare’s serum gonadotropin (PMSG; (Harbor–UCLA Medical Center, Torrance, CA, USA) followed 47–49 h later by 5 IU of human chorionic gonadotropin (HCG; Sigma–Aldrich, St Louis, MO, USA). 26 Females were mated to proven stud males immediately following HCG injection. Females were humanely euthanized by cervical dislocation the following morning for embryo isolation. MDA personnel have ≥25 years of experience performing cervical dislocation, and have demonstrated their proficiency to an institutional veterinarian. Embryos were isolated and cultured for 24 h in KSOM embryo culture media (Millipore, Billerica, MA, USA) under embryo-tested mineral oil (Sigma–Aldrich) at 37℃ in 5% CO2. Upon reaching the two-cell stage, embryos were cryopreserved in propylene glycol. Cryopreserved embryos were thawed the morning of surgery and cultured in KSOM as noted above.
CNR: Sperm isolation
Males were humanely euthanized by cervical dislocation. CNR personnel have ≥15 years of experience performing cervical dislocation, and have demonstrated their proficiency as per requirements from the Animal Welfare and Ethical Review Body. Each cauda epididymis and vas deferens was placed in the sperm collection dish containing 500 µL of mouse vitro fert (MVF) medium (Cook Medical, Brisbane, Australia) covered with oil. Sperm were allowed to disperse from the tissue for 10 min and subsequently frozen using cryoprotective medium (CPM) of 18% raffinose (w/v), 3% skim milk (w/v) and 477 µM monothioglycerol (MTG). 27 Four aliquots of 12 µL of sperm + CPM were loaded into 0.25 mL French straws (IMV Technologies, L’Aigle, France) and the straws were loaded into cassettes. Cassettes were placed onto a polystyrene raft of liquid nitrogen (LN2) vapors for 10 min before being plunged in LN2.
CNR Oocyte isolation and in vitro fertilization
IVF was performed using the protocol described by Ostermeier et al. 27 Sperm samples were thawed in a 37℃ water bath for ∼30 s. Approximately 12 µL of the CPM + sperm was added to 500 µL of equilibrated MVF medium and incubated for ≥40 min under 5% CO2. Three–four-week-old female Hsd: athymic nude-nu (nu/nu) and C57BL/6N mice were used as oocyte donors. Donors were superovulated by intraperitoneal injections of 5 IU PMSG (Intervet, Milan, Italy), followed 48 h later by 5 IU HCG (Intervet). At 12–14 h post-HCG, females were humanely euthanized by cervical dislocation. Oocytes were isolated and added to the IVF drop of 500 µL of MVF medium + sperm. After 4 h of co-incubation, the presumptive zygotes were washed through drops of 150 µL of MVF medium. Those appearing normal were cultured overnight in 150 µL of MVF. Approximately 18 h later, two-cell embryos were cryopreserved in 1.5 M propylene glycol. 28 Cryopreserved embryos were thawed when needed the morning of surgery and cultured in KSOM.
Embryo implant surgery
CD-1® females (26–33 g, <5 months of age) (CRL [MDA]; EMMA/Infrafrontier, Italy [CNR]) for implant surgery were identified by the presence of a copulation plug after mating to vasectomized CD-1® males (CRL [MDA]; EMMA/Infrafrontier, Italy [CNR]). At the MDA site, implants took place over six separate days; at the CNR site, implants took place on four separate days. Females were anesthetized for surgery by either 2.5% Isothesia® (isoflurane gas) to effect (after induction at 4–5% isoflurane) (Butler Schein Animal Health, Dublin, OH, USA [MDA]; IsoFlo Abbott Laboratories Ltd, Maidenhead, Berkshire, UK [CNR]), K/X or K/M (Fort Dodge Animal Health, Fort Dodge, IA, USA; Lloyd Laboratories, Shenandoah, IA, USA [MDA]; Imalgene1000 Merial Italia and Domitor, Orion Pharma, Finland [CNR]) at 100:10 mg/kg (K/X) and 75:1 mg/kg (K/M), or a TBE:tert–amyl alcohol mix (obtained from MD Anderson Pharmacy Research Laboratory [MDA]; Merck Italia [CNR]) at 250 mg/kg TBE. 29 A 2% working solution of TBE was prepared by adding sterile water to a 50× stock solution at 37℃ and then mixed thoroughly. This working solution (250 mg/kg) was administered and the remainder immediately discarded. The stock solution was stored at 4℃ in the dark and was used within six months after receipt. No discoloration was observed during the experimental period.
After anesthetic induction, the surgical site was prepared by shaving. Females were moved to a 37℃ warming plate during surgery and the site prepared with chlorhexidine:alcohol scrubs. A 1 cm skin incision was made over the spine and the surgical window was moved laterally to visualize the ovary. A 3–5 mm incision was made into the peritoneum and the ovary/oviduct complex was extracted. The fascia covering the oviduct was torn and the infundibulum was located. Fifteen (CNR) or 30 (MDA) embryos in a minimal amount of KSOM were implanted unilaterally (CNR) or bilaterally (MDA) into the ampulla of the oviduct. (Numbers of embryos implanted varied two-fold between CNR and MDA. The embryo numbers implanted were based on results observed over the past five years at each facility.) The oviduct was returned to the peritoneum and the central wound was closed using suture or surgical staples.2,29 Surgical duration was less than 10 min per animal, after which the animal was moved to a warming cage.29,30 After surgery, the animal was given Buprenex®/Temgesic® 31 (Reckitt Benckiser Pharmaceuticals Inc, Richmond, VA, USA [MDA]; Intervet, Italy [CNR]) at 0.1 mg/kg, placed on a 37℃ warming plate, and monitored until ambulatory. At the CNR site, atipamezole (1 mg/kg) (Elanco Animal Health, Florence, Italy) was administered to reverse the clinical effects of the medetomidine. Females were singly housed for gestation either immediately post-surgery (MDA) or after becoming visibly pregnant (CNR). (Single housing to assess pup numbers for each implanted female was required for statistical purposes in this study.) At the MDA site, two bacon-flavored meloxicam tablets (0.0125 mg/tablet; Bio-Serv, Flemington, NJ, USA) were placed in the housing cage for ongoing analgesia. Animals were monitored daily for any complications. Housing cages were given identification numbers that did not indicate the anesthetic used, and the key was maintained in confidence by the surgeon.
Data collection
Data collection was performed by participants blinded to the anesthetic modality received by the implanted females. Pregnancy status was assessed by trained animal technicians on days 4–5 prior to the expected delivery date, and positive status was denoted by appearance. Births were noted, when possible without disturbing the cage, on the expected delivery date. Pup numbers were obtained on day 5 after birth and again at weaning.
Statistical methods
Group size of recipients was determined by statistical analysis of five-year historical birth numbers. Based on one-way analysis of variance (ANOVA) method, with a sample size of 18 mice per group, the experiment has 80% power to detect an effect size of 0.2 for the number of pups among the anesthetic groups, assuming a type 1 error rate of 0.05 (nQuery Advisor 7.0). The effect size is the variance of the means divided by the square of the common standard deviation.
Mean and standard error (SE) were provided for continuous variables (e.g.: number of pups at day 5 and number of pups at weaning). The one-way ANOVA procedure (F-test) was used to examine overall differences among the means of the multiple groups. Before performing ANOVA analysis, data distributions and the assumptions of the ANOVA analysis were checked. The data of pup numbers at day 5 and pup numbers at day 21 were normally distributed (P values = 0.10 and 0.13 based on normality test). The residuals from the ANOVA models were also normally distributed (P values > 0.10 based on normality test). For pair-wise comparisons, the differences between means were provided. To control the overall type 1 error rate, Tukey’s honestly significant difference (HSD) test was used for pair-wise comparisons to determine differences among groups. Frequencies and percentages were reported for categorical variables (e.g.: treatment group and pregnancy status). Fisher’s exact test was used to evaluate the association between treatment and pregnancy status. All tests were two-sided. P values less than 0.05 were considered statistically significant. All analyses were conducted using SAS (version 9.3; SAS Institute Inc, Cary, NC, USA). Due to the different types of embryos used for implantation at the MDA and CNR sites, statistical analyses were conducted separately for each institution.
Results
Implant results for each anesthetic.
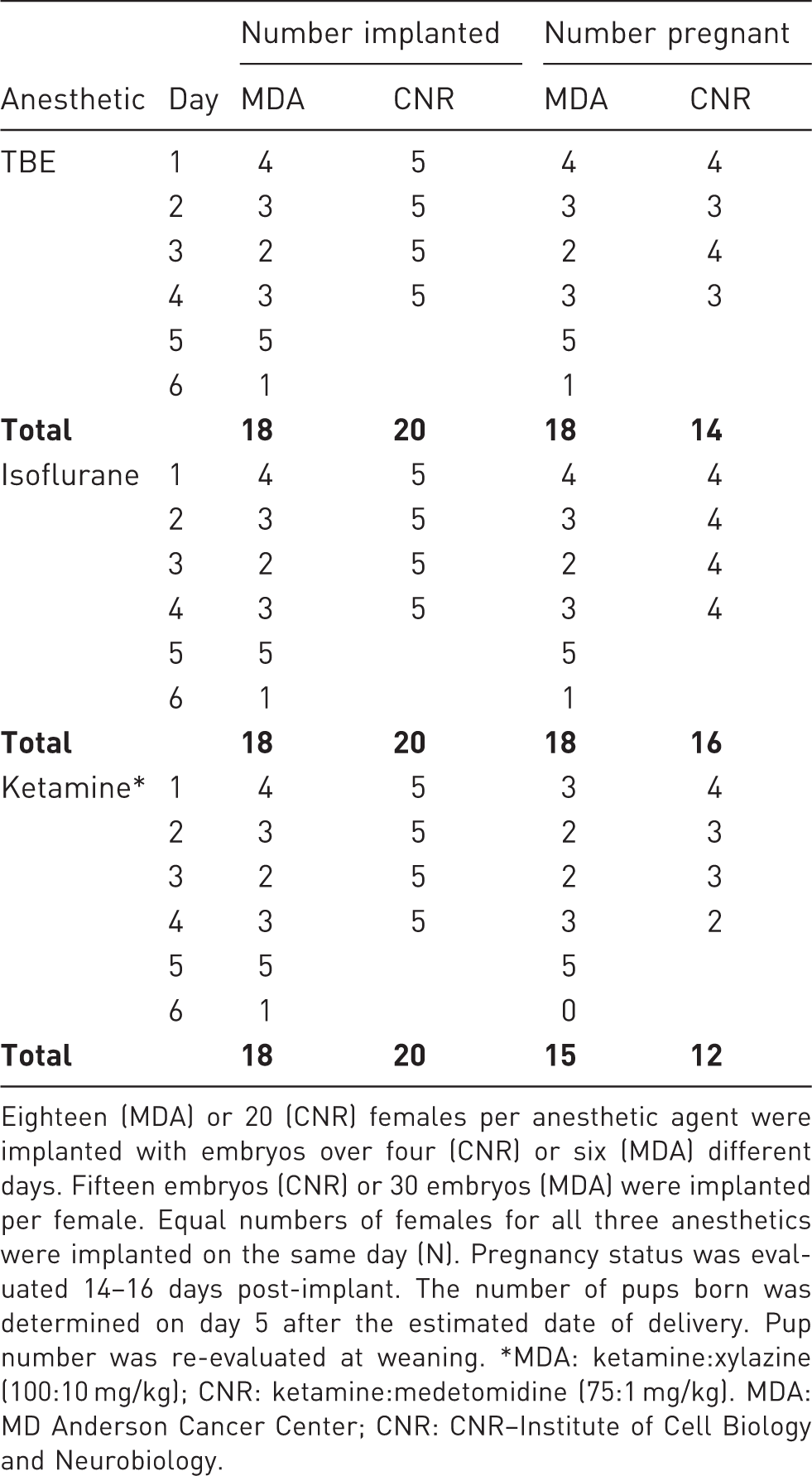
Eighteen (MDA) or 20 (CNR) females per anesthetic agent were implanted with embryos over four (CNR) or six (MDA) different days. Fifteen embryos (CNR) or 30 embryos (MDA) were implanted per female. Equal numbers of females for all three anesthetics were implanted on the same day (N). Pregnancy status was evaluated 14–16 days post-implant. The number of pups born was determined on day 5 after the estimated date of delivery. Pup number was re-evaluated at weaning. *MDA: ketamine:xylazine (100:10 mg/kg); CNR: ketamine:medetomidine (75:1 mg/kg). MDA: MD Anderson Cancer Center; CNR: CNR–Institute of Cell Biology and Neurobiology.
Association between pregnancy status and anesthesia treatment.
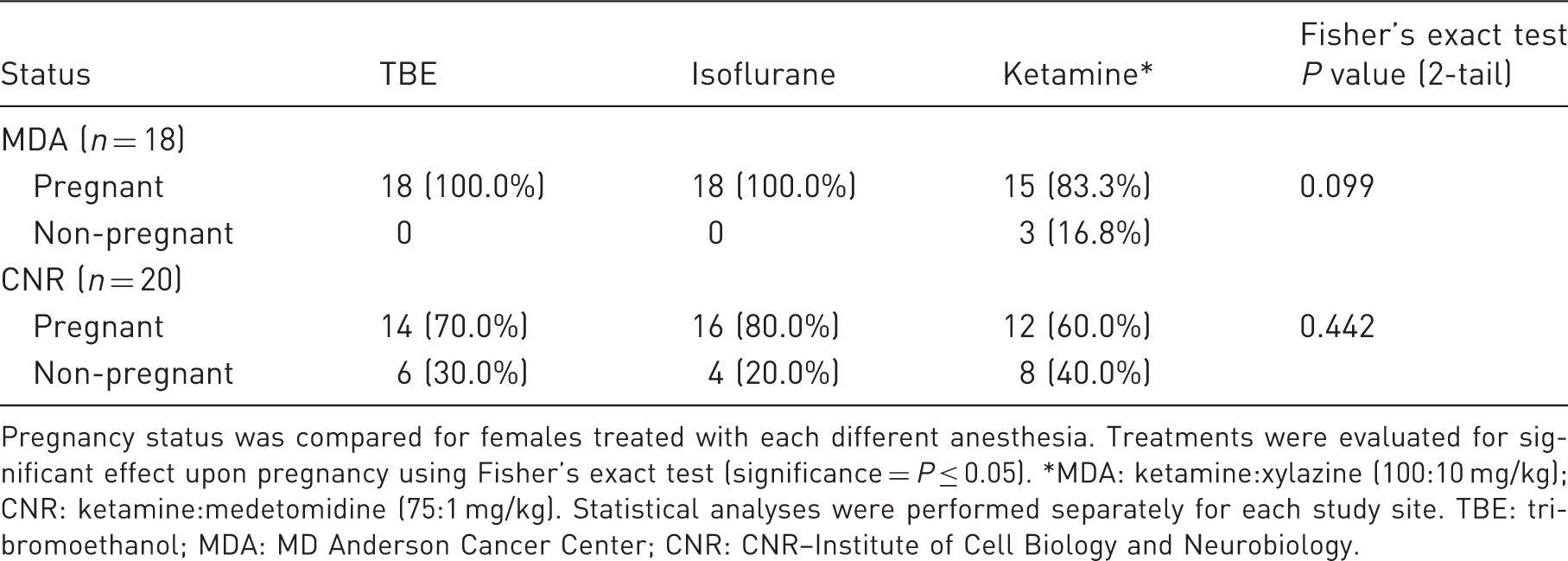
Pregnancy status was compared for females treated with each different anesthesia. Treatments were evaluated for significant effect upon pregnancy using Fisher’s exact test (significance = P ≤ 0.05). *MDA: ketamine:xylazine (100:10 mg/kg); CNR: ketamine:medetomidine (75:1 mg/kg). Statistical analyses were performed separately for each study site. TBE: tribromoethanol; MDA: MD Anderson Cancer Center; CNR: CNR–Institute of Cell Biology and Neurobiology.
Comparison of pup numbers at day 5.
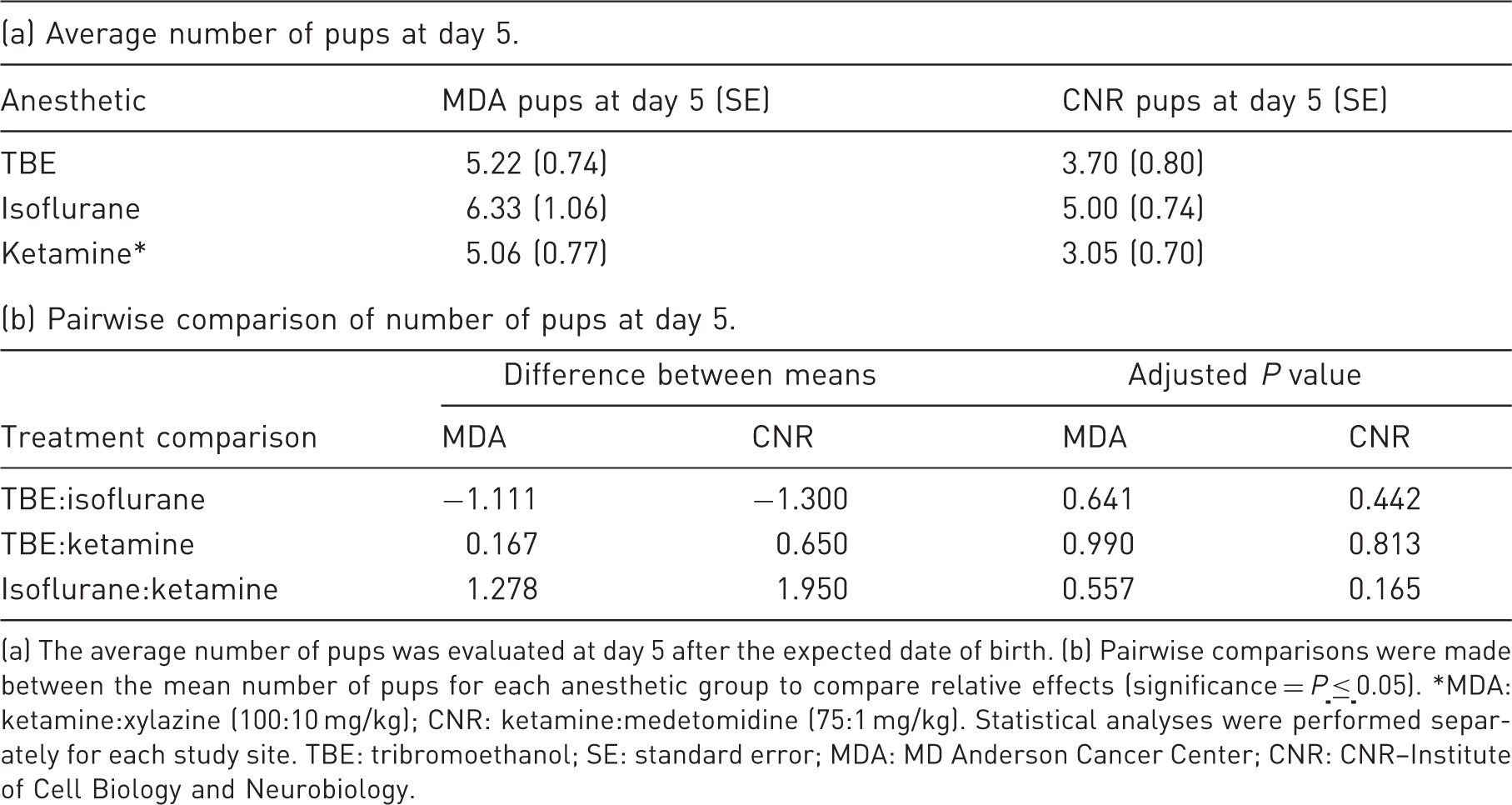
(a) The average number of pups was evaluated at day 5 after the expected date of birth. (b) Pairwise comparisons were made between the mean number of pups for each anesthetic group to compare relative effects (significance = P
Comparison of pup numbers at weaning.
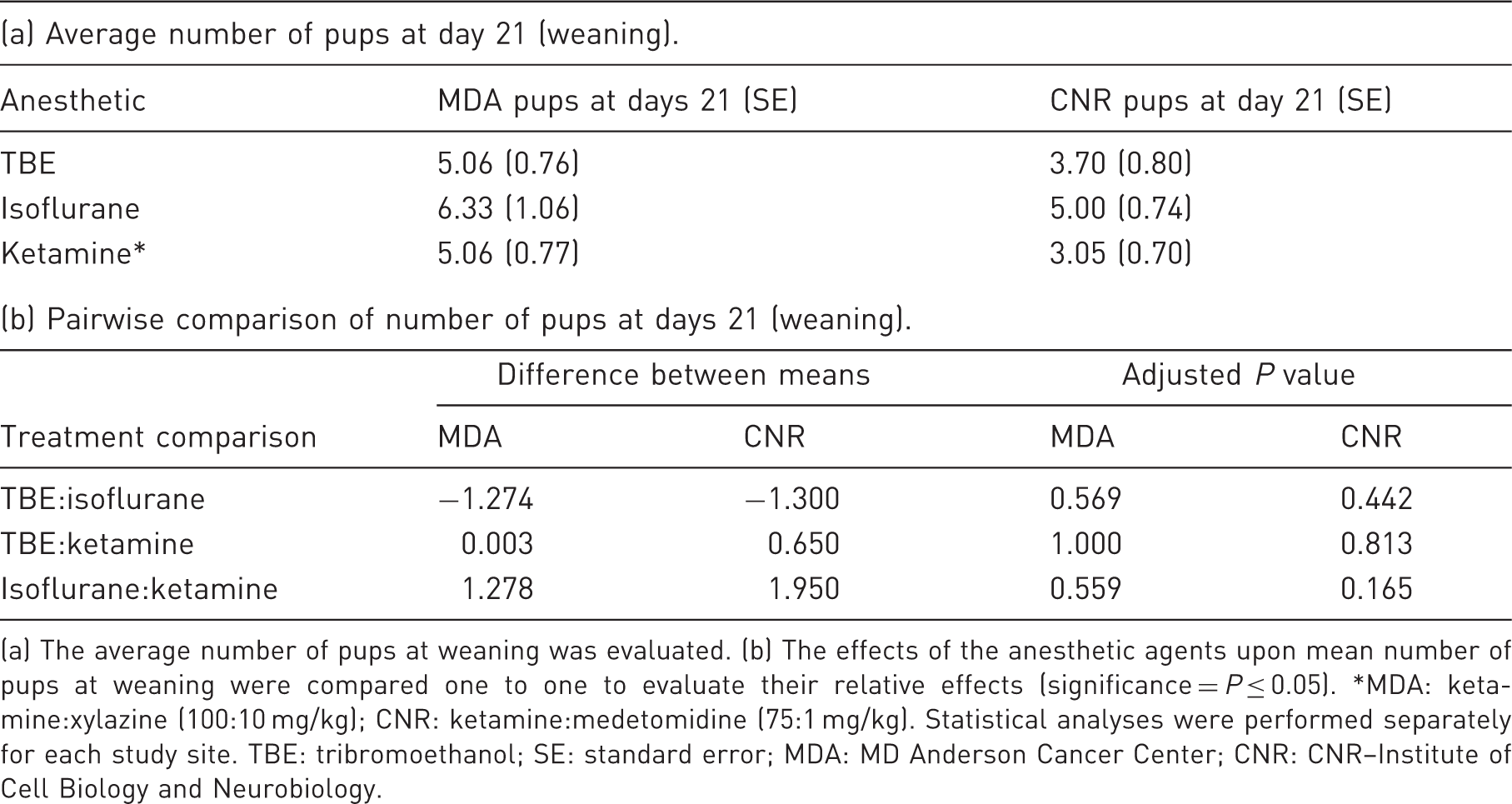
(a) The average number of pups at weaning was evaluated. (b) The effects of the anesthetic agents upon mean number of pups at weaning were compared one to one to evaluate their relative effects (significance = P ≤ 0.05). *MDA: ketamine:xylazine (100:10 mg/kg); CNR: ketamine:medetomidine (75:1 mg/kg). Statistical analyses were performed separately for each study site. TBE: tribromoethanol; SE: standard error; MDA: MD Anderson Cancer Center; CNR: CNR–Institute of Cell Biology and Neurobiology.
Discussion
Highly productive mouse transgenic facilities have detailed anesthetic protocols that reliably produce good pup numbers after surgery. However, new regulations and novel agents must be considered as they arise. Successful procreation is an important criterion for animal well-being; if pup numbers are already high, animal surgeons are often reluctant to change procedures. Additionally, studies to identify and modify mouse genes have exponentially increased the international exchange of mouse mutant resources. Surgical refinements are crucial to saving animal lives, and surgeons should consider flexibility in anesthetic approaches to achieve optimal results.
Our findings on the effects of TBE, ketamine, and isoflurane on embryo transfer suggest that comparable pregnancy rates and pup yields can be obtained regardless of the anesthetic used. This is consistent with previous research in which recipient dams anesthetized with K/X yielded comparable numbers of pups to those anesthetized with TBE. 19 By comparison, in this study we observed a higher percentage of dams in the TBE group with surviving offspring at one site, but not at the other, although this difference was not significant when compared with the other anesthetic cohorts. Additionally, the number of embryos implanted varied between sites in this study. While the numbers of pups obtained at the MDA site were generally two-fold higher than at the CNR site, this was anticipated due to the 2× increase in the number of embryos implanted. The non-significant differences observed among all of the studies, including this one, are likely due to housing, differences in surgical techniques, and the improved chemical isolation techniques developed since 1998.
Notably, the ketamine cohort showed reduced pregnancy rates both at the MDA and CNR sites, despite differences in housing conditions and in stock/strains of embryos (C57BL6/NCnrm, C57BL6/JCnrm, BALB/cByJCnrm, and FVB/NJCnrm embryos at the CNR site versus CD-1 embryos at the MDA site). The regimen at CNR also used medetomidine, which was reversed with atipamezole. As both institutions noted similar trends, these differences were unlikely to be the cause. However, while we noted an overall trend for ketamine use, the effect was not statistically significant. It would be worthwhile investigating this further, as ketamine is widely used for implant surgery.
We sought to determine whether isoflurane administration would result in the same number of pups as other, more widely used anesthetics. Isoflurane, combined with morphine, has been shown to be effective for the production of transgenic rats. 32 Although previous research on murine fetal development has demonstrated comparable rates of pregnancy using isoflurane versus TBE or K/X, 24 our study marks the first time that the impact of isoflurane on live mouse pup yield has been compared head-to-head with either of these more commonly used anesthetics. Notably, previous studies have identified adverse impacts of isoflurane on two-cell embryos in vitro.22,23 In those studies, gas was applied directly to the embryos, likely changing the pH of the solution and hampering embryo development. In this study, isoflurane was administered through a well-fitted nose-piece with a controlled rate vaporizer and utilizing the required scavenging. Anesthetic durations were kept consistent with what would be expected for each anesthetic (e.g. approximately 10 min for isoflurane and 30–90 min for K/X; K/M and TBE) rather than exposing the dams to isoflurane for the extended periods of unconsciousness induced by TBE and K/X or K/M. Our results demonstrate that, when used in vivo and scavenged properly, isoflurane does not adversely affect the viability of embryos or the resulting pups.
The analgesics given at both institutions were uniform, with the exception of meloxicam tablets given post-operatively at the MDA site. This difference should not influence pup yield at birth or weaning, as previous research has indicated that multimodal opioid–non-steroidal anti-inflammatory drug (NSAID) administration has no effect on these values relative to the administration of opioids alone. 33 And, although meloxicam has been shown to affect fetal growth when administered to a dam at fertilization, it does not affect litter size. 34
Whereas previous studies have reported pup numbers at a single time point, we recorded numbers at day 5 post-delivery and at weaning. We had previously noted that disturbed new dams have a greater propensity to cannibalize their offspring. Thus, we chose the five-day post birth assessment as one that could be safely used to determine pup survival post-partum. Even so, we noted instances of positive pregnancy status followed by a lack of pups at day 5. Interestingly, three of five instances (MDA) were noted for dams treated with isoflurane, with the other two regimens having one instance each. We chose not to assess pup numbers by delivery via Caesarean-section (C-section), as we wished our study to be applicable to transgenic facilities, and this is not standard procedure for such facilities. Future studies could utilize C-section delivery or detection tools to record the number of live births, as well as to determine the specific cause of death of deceased or cannibalized pups.
Previous research has identified stock/strain variations in response to different anesthetics.35,36 For this reason, at the MDA and CNR sites, we compared the impact of the anesthetic agents using just one recipient stock. CD-1® is an outbred stock commonly used for embryo transfer in mouse transgenic facilities. At the MDA site, frozen/thawed CD-1® embryos were implanted, thus, restricting the strain variation for both the embryos and the recipients. At the CNR site the embryos came from multiple strains (C57BL6/NCnrm, C57BL6/JCnrm, BALB/cByJCnrm and FVB/NJCnrm) obtained from fresh and frozen batches, allowing a comparison of different strains of embryos despite using only one stock of recipients. The statistical variation among anesthetic groups, examined separately for each institution, showed no differences, thus confirming that the anesthetic used had no significant effect on implantation success whether the embryos were matched to the recipient or came from unmatched strains/stock. Additional studies could incorporate additional recipient strains commonly used for implant surgery (e.g. B6D2F1, CBAD2F1, etc.)
Our results indicate that comparable numbers of pregnancies and pups should result regardless of whether TBE, ketamine, or isoflurane is chosen as the anesthetic for implant surgery. With this information, transgenic facilities can make more informed decisions regarding their choice of anesthetic for embryo transfer. For labs struggling to acquire or accommodate certain agents, alternatives can be sought, given that the impact to their production program will be minimal to non-existent.
Footnotes
Acknowledgements
At MDA, we thank Earnessa Edison for animal care and data collection, Dr Suzanne Craig for assistance with study design, Drs Katherine Naff and Mary Robinson for their editing, and Dr Peggy Tinkey for support for animal purchase and care. At CNR, we thank Dr Renata Paoletti, the Cryo-Lab staff and Fabrizio Bonaventura who did most of the experiments at EMMA–Infrafrontier, Italy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by a Comprehensive Cancer Grant to MD Anderson Cancer Center, NCI #CA016672 (GEMF) and the INFRAFRONTIER-I3 project under the EU contract Grant Agreement Number 312325 of the EC FP7 Capacities Specific Programme at CNR Institute of Cell Biology and Neurobiology.
