Abstract
Anesthetics are commonly used in fish for surgery and to facilitate capture, handling and transport in aquaculture and experimental procedures. In research, the selection of the anesthetic depends on its properties and on the recovery time. Eugenol has been pointed out as an effective anesthetic for fish, alternative to traditional drugs. Although Cnesterodon decemmaculatus is widely used as a model in ecological bioassays, no anesthetic protocol has been reported for this species. The aim of the present study was to evaluate the induction time (i.e. time to reach anesthetic stage VI) and recovery time in individuals of C. decemmaculatus subjected to eugenol at a fixed concentration, according to sex and pregnancy status. Forty-one fish were divided into three groups: males, pregnant females and non-pregnant females. They were measured for total length, standard length and weight, and the condition factor (K) was calculated. No significant differences in induction and recovery times were found for sex, pregnancy status and K between groups. Results are a contribution toward the development protocol of a standard anesthetic protocol for C. decemmaculatus.
Introduction
Anesthetics are commonly used to facilitate capture, handling and transport in aquaculture and research 1 and are essential for surgery and other invasive procedures. 2
Recently, several protocols have been investigated to provide effective anesthesia in fish. The choice of the anesthetic is determined by different factors such as economic feasibility, utility, effectiveness and ethical considerations. 3 Also, potentially painful events result in behavioral and physiological changes in fish, 4 and anesthetics are useful to ensure care and animal welfare.5,6
On the other hand, the optimal dose must not be aversive to the fish and must be sufficient to induce unconsciousness rapidly, thereby reducing stress. 7
Regardless of the objective of the research, the latter response is desirable in most cases, while the criteria for selecting the recovery time differ according to the situation. Thus, a long recovery time is desirable for fish collected in the wild or handled for some time in the laboratory, while a rapid recovery is required in other circumstances to immobilize fish, such as handling, blood collection, sorting and tagging.8,9
Other anesthetics (e.g. benzocaine, lidocaine, MS-222 and 2-phenoxyethanol) are applied via the immersion technique to induce anesthesia. 7 Clove oil has emerged as an alternative anesthetic used for handling and transport. The active ingredient of clove oil is eugenol (4-allyl-2-methoxyphenol), which represents 70–95% of the total weight of the base clove oil. 1 This is an inexpensive and safe natural oil that does not leave toxic residues in fish flesh or in the environment. 10 However, eugenol has the potential to cause skin sensitivity in human operators, making it important to use appropriate personal protective equipment as a precautionary measure. 2
Eugenol showed to be an effective anesthetic for different species, such as the rainbow trout Oncorhynchus mykiss, channel catfish Ictalurus punctatus, the marine fishes rabbitfish Siganus lineatus and ambon damselfish Pomacentrus amboinensis, 11 common carp Cyprinus carpio, medaka Oryzias latipus, crucian carp Carassius auratus and zebrafish Danio rerio. 1
Based on the idea that a good anesthetic induces sedation in less than 180 s and allows recovery in less than 300 s,12 –14 some authors reported that eugenol resulted in short induction and recovery times1,8,10,15 in comparison with other anesthetics.8,16,17
Despite the fact that fish is the most diverse group of vertebrates comprising a large number of species potentially useful for research, the majority of laboratory work is concentrated on a handful of species. 5 On the other hand, literature regarding the use of anesthetics in fish is scarce. The type of anesthetic or sedative agent, as well as its dosage and exposure time, must be carefully selected according to the characteristics of each species, such as size, natural habitat, behavior and physiology, among others. In particular for fish used in research, species-specific knowledge of the anesthetic effect will contribute to the development of appropriate protocols and procedures. 2
Cnesterodon decemmaculatus has been proposed as a local sentinel for monitoring environmental contamination and as an excellent laboratory model for ecological studies.18 –21 Despite this, no anesthetic protocols have been yet conducted in this species.
Small-sized fish like C. decemmaculatus may have complications related to anesthesia. For example, they are at high risk of overdose of anesthesia when the method of drug administration is through immersion. 22
In addition to variations related to sex, physiological factors like size, weight and pregnancy can impact on the induction and recovery time in both pregnant and non-pregnant females.
Thus, the aim of this study was to evaluate the anesthesia induction time and recovery time produced by a fixed concentration of eugenol in males and pregnant and non-pregnant females of C. decemmaculatus.
Material and methods
All research procedures were approved by the Institutional Animal Care and Use Committee (CICUAL//IACUC) of the Faculty of Veterinary Sciences, National University of La Plata (NULP), Argentina, under the registration number 108-4-20T, in compliance with the Guide for the Care and Use of Laboratory Animals of The National Research Council (2011). 23
Experimental organism
C. decemmaculatus (Cyprinodontiformes, Poeciliidae) is an endemic Neotropical fish widely distributed in water bodies of South America. It is a small, ovoviviparous, micro-omnivorous, benthic-pelagic and non-migratory fish with internal fertilization and sexual dimorphism. Females are larger than males, and the male anal fin is modified into a gonopodium. 24 This species is easy to handle and acclimatize to laboratory conditions.
C. decemmaculatus specimens were collected from the Bosque Lake, a non-polluted water body in La Plata, Buenos Aires, Argentina (34°54′ S; 57°56′ W), transported to the laboratory and kept in a 20 l tank with a housing density of two individuals per liter. The offspring, which were used as experimental animals, were transferred to a 20 l tank with continuously aerated, dechlorinated tap water (pH = 7.6, dissolved oxygen 7.5 mg l−1, conductivity 1135 µS cm−1, hardness 245 ppm CaCO3/l) under constant environmental conditions (21 ± 1°C; 14:10 light:dark photoperiod). Fish were fed daily with commercial fish flakes (Shulet carassius®, Argentina). Feces were siphoned followed by a 50% water exchange once a week. The environment was enriched with artificial plants for fish to express their natural behavior. Females and males were housed together; some females became pregnant.
Experimental design
The experiment was conducted with 41 healthy specimens of C. decemmaculatus, females and males 1, 5: 1, one year old. The fish were divided into three groups according to sex and pregnancy status (pregnant and non-pregnant). Sexing was performed by visual inspection of the male copulatory organ (gonopodium) and pregnant females identified by enlarged abdomen and confirmation after euthanasia. Group 1 included 16 males (M); group 2 included 15 pregnant females (F) and group 3 included 10 non-pregnant females (A). Before anesthesia, all individuals were measured for total length (LT), standard length (SL) (mm) and weight (W) (mg) and the body condition or condition factor (K) was calculated. Animals were measured for their SL and TL with a professional Halminton Brand, China caliper, weighed with a Marsboy I-2000, China scale. Condition factor index (K) was calculated as (W (mg)/SL3 (mm) × 100) according to Young et al. (2017). 24 Before the beginning of the trial, all animals underwent physical examination to look for signs of illness (i.e. erratic swimming, presence of lesions and malformations or neo-formations in the tegument) and measured (LT, SL, weight). Sample size was estimated to detect a mean difference in the time to recovery from anesthetic stage VI, of 22.5 s between males and pregnant females assuming a standard deviation of 20 s in the population.
Animals were anesthetized following the methodology of Munday and Wilson (1997). 8 The anesthetic used was eugenol (99% purity; Eugenol Dickinson®, Argentina). Eugenol was diluted in 96% ethanol, resulting in a 1:10 stock solution. 10 Just before the experiment, fish were individually selected at random, sexed and the parameters of interest were recorded. Then, fish were individually placed in an anesthetic water bath at a concentration of eugenol of 50 mg l−1 and the time to reach anesthetic stage VI (hereafter referred to as induction time) was recorded. The eugenol concentration of 50 mg l−1 was selected according to the criteria established by Grush et al. (2004) 1 and Javahery et al. (2012). 9 The onset of this stage was determined based on criteria of Martins et al. (2016) 25 to reach anesthesia stage VI. Next, the fish recovered individually in a 1 l aquarium. Recovery was recognized based on criteria of Ross and Ross (2008) 26 adapted by Small 2003 27 (Table 1).
The induction and recovery times were recorded using a digital stopwatch. After recovery, all animals were transferred to a new 20 l tank, not subjected to any other treatment, and monitored for 48 h. Finally, survivors were euthanized using an overdose of eugenol (150 mg l−1). Mortality was recorded during the recovery and post-recovery times. All procedures were conducted by the same trained operator (MCV).
Statistical analysis
Descriptive analysis was performed with summary measures and by plotting the frequency distribution of SL, weight and K in males and non-pregnant and pregnant females. The induction and recovery times were estimated by fitting a Kaplan–Meier survival probability curve for each group and the log-Rank test was used to test for significant differences between groups.
To estimate the differences in the hazard (i.e. instantaneous risk) to reach stage VI of anesthesia and time to recovery depending on K values, a Cox proportional hazards model was fitted including K and the three sex categories as fixed predictors. Results were presented as hazard ratios with 95% confidence intervals (CIs). To assess the proportional hazard assumption, the Schoenfeld residuals were correlated with time to recovery. Statistical analyses were performed using SAS software 9.4 (SAS Institute, Cary, NC, USA).
Results
Table 2 shows the means and standard deviations of K, weight and SL for the three groups. The frequency distribution of these variables for each fish category was plotted and a smooth line was fitted (Figures 1 –3).
Mean condition factor, weight and standard length values for Cnesterodon decemmaculatus males, pregnant females and non-pregnant females.

K = W (mg)/SL3 (mm)). Young et al. (2017). 24
K: condition factor; SL: standard length; W:weight
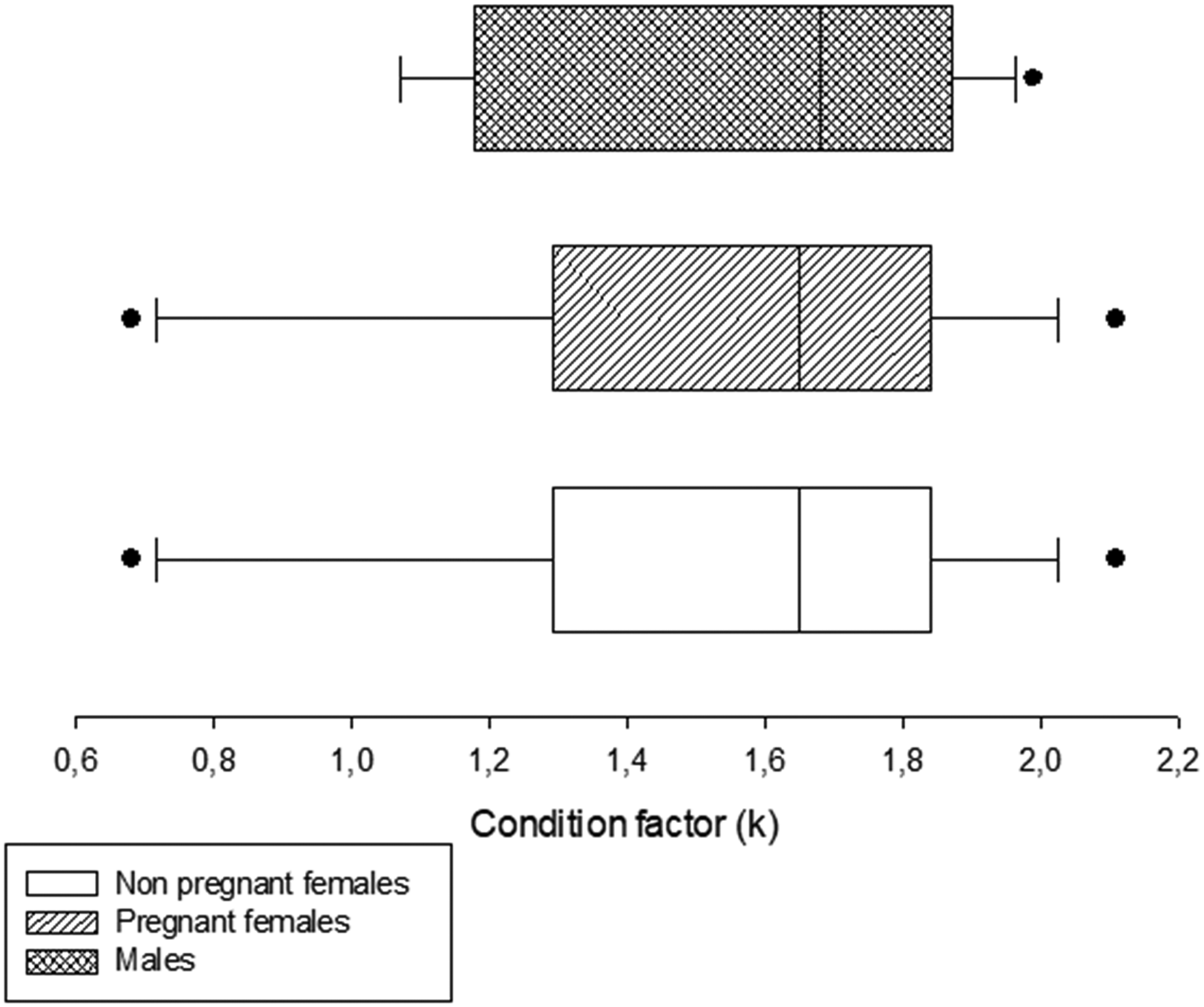
Frequency distribution of the condition factor (K) (W (mg)/SL3 (mm) × 100) in males, non-pregnant and pregnant females of Cnesterodon decemmaculatus.
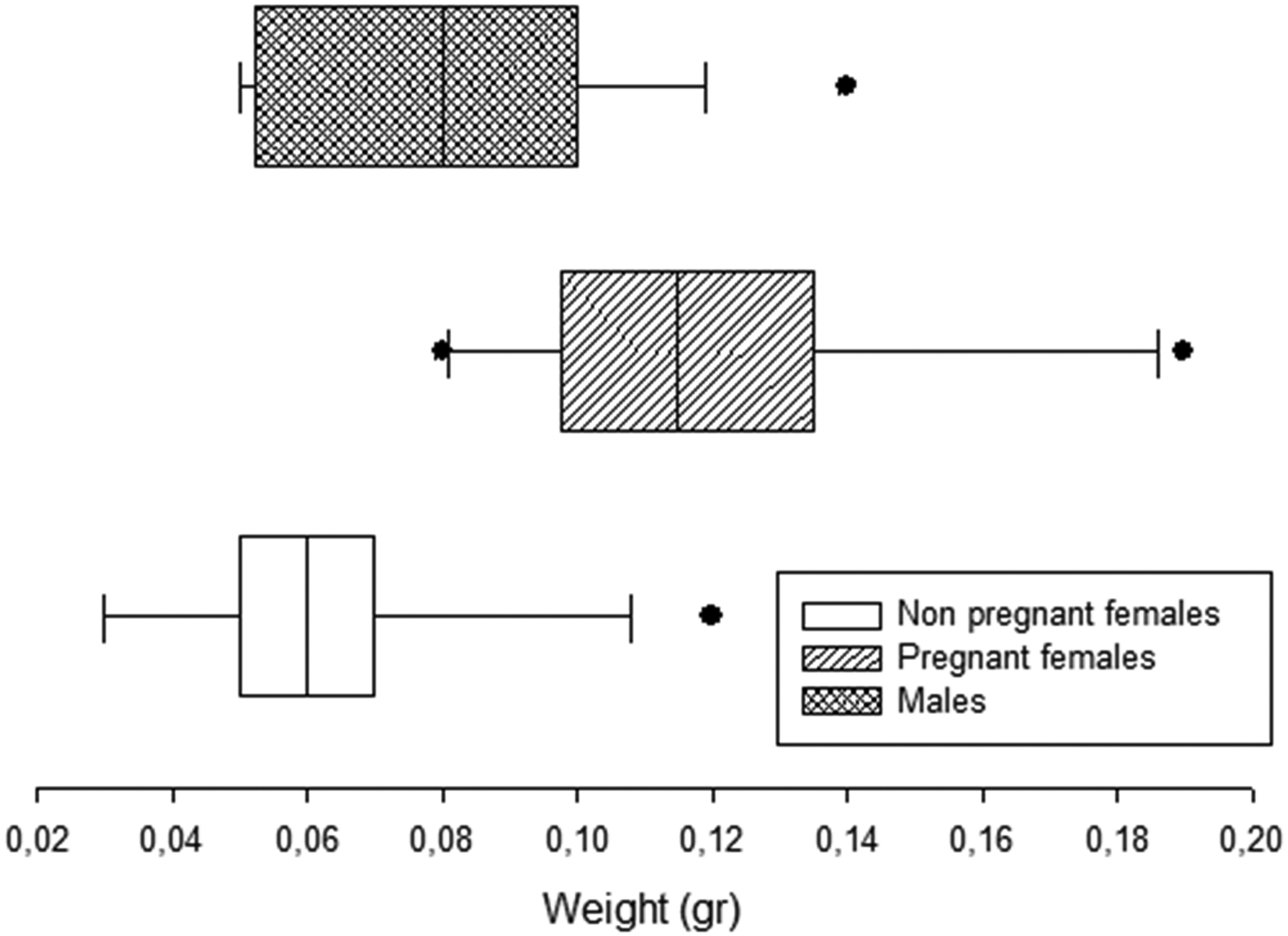
Frequency distribution of weight in males, non-pregnant and pregnant females of Cnesterodon decemmaculatus.
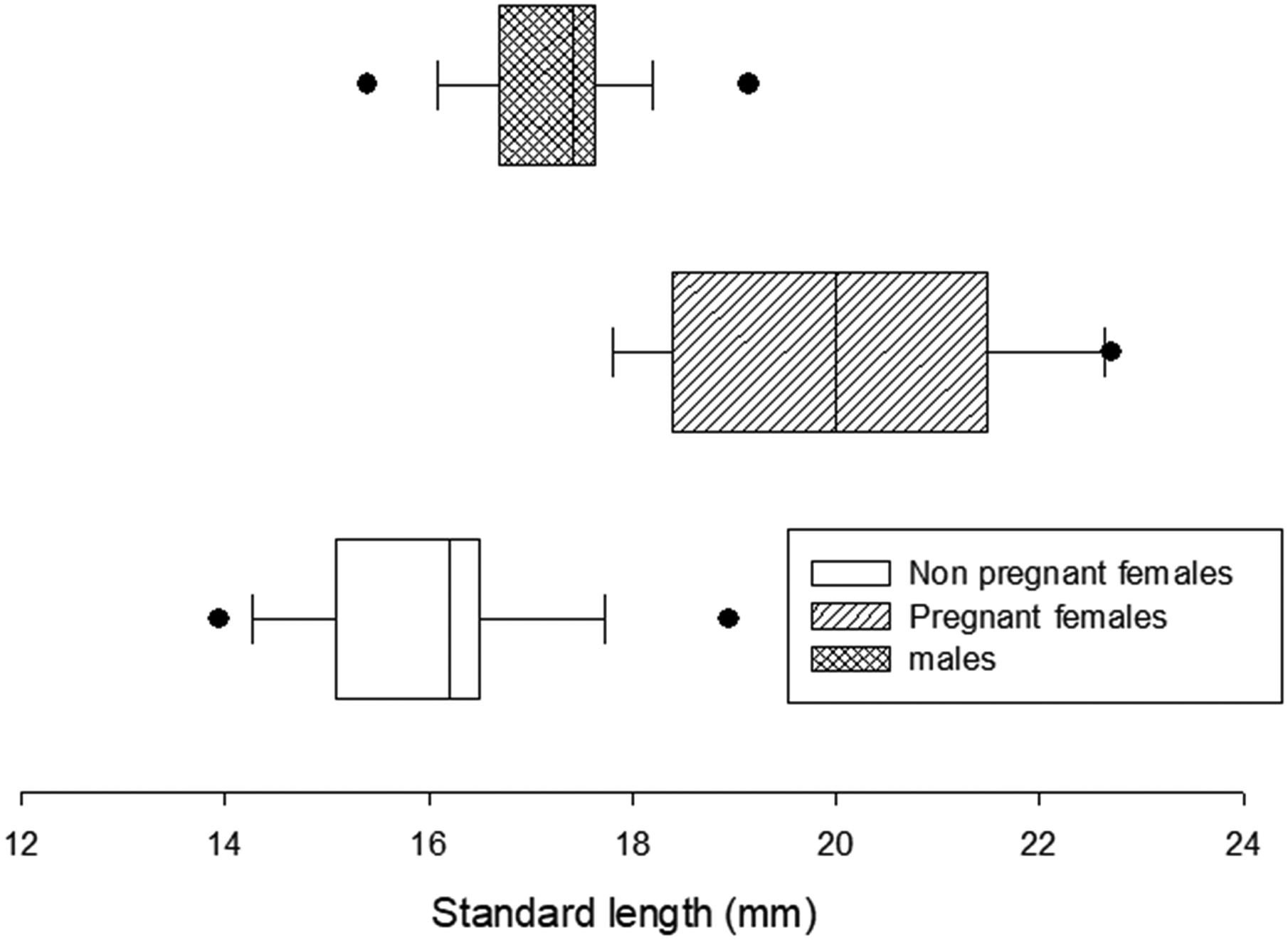
Frequency distribution of total standard length in males, non-pregnant and pregnant females of Cnesterodon decemmaculatus.
The fitted survival functions showed that the median induction time was 37.31 s (95% CI 20–42) for males, 34.40 s (95% CI 10–45) for pregnant females and 38.53 s (95% CI 26–40) for non-pregnant females. No significant differences in induction time were found between groups (log-rank test, p = 0.8401). The Cox proportional hazards model, which estimates the instant risk to reach stage VI of anesthesia, showed no significant differences between K values (hazard ratio = 1.316; p = 0.557) and between categories (hazard ratio A vs. hazard ratio F = 0.811 (95% CI 0.356–1.848); hazard ratio A vs. hazard ratio M = 0.985 (95% CI 0.478–2.029); hazard ratio F vs. hazard ratio M = 1.214 (95% CI 0.540–2.731)) (Figure 4).
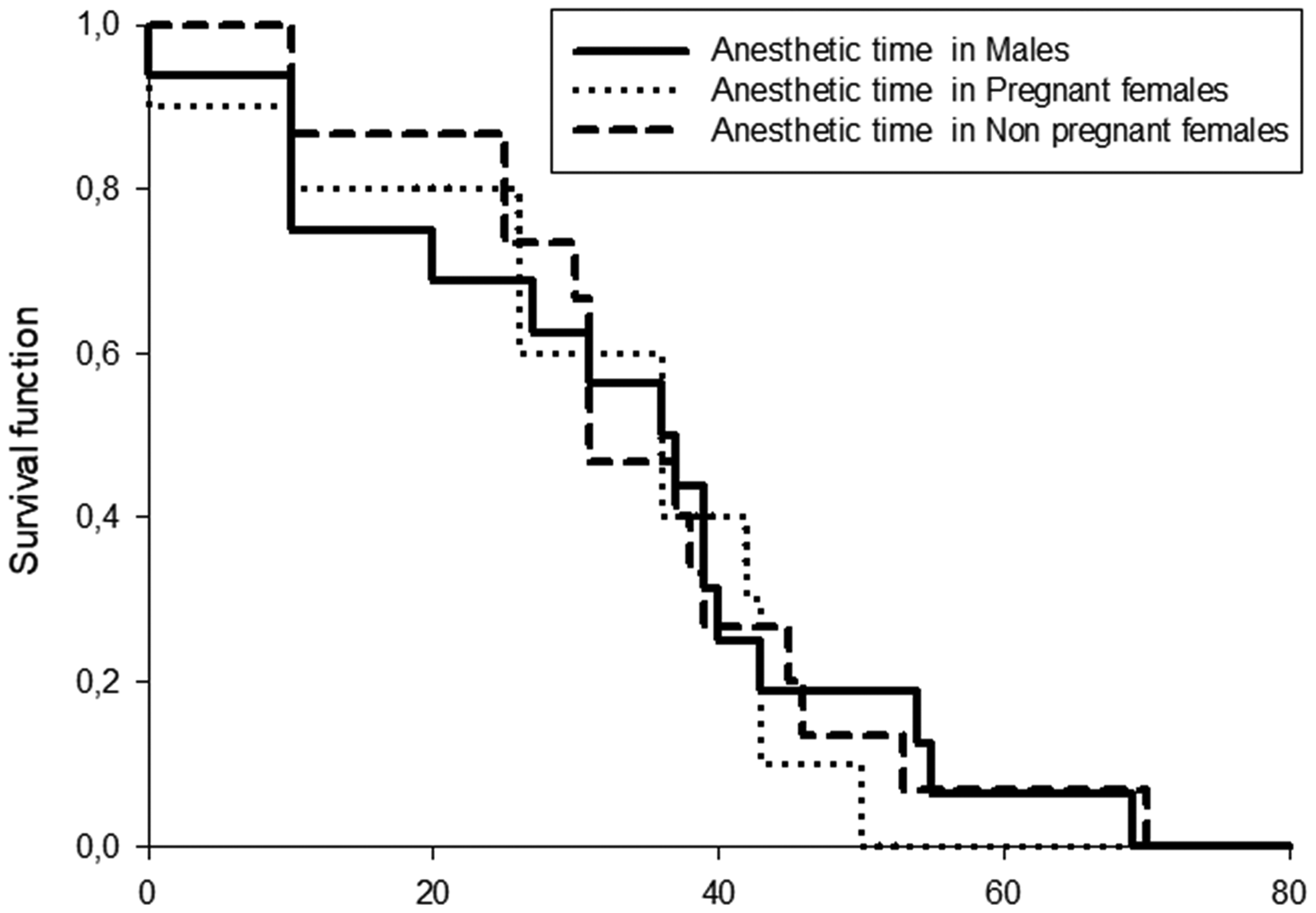
Kaplan–Meier survival analysis that estimated the induction time according sex categories (males, non-pregnant females and pregnant females) of Cnesterodon decemmaculatus.
The fitted survival functions showed that the median time to recovery from stage VI of anesthesia was 288.43 s (95% CI 200–300) for males, 280.40 s (95% CI 135–345) for pregnant females and 297.13 s for non-pregnant females. Differences in recovery time were not significant (log-rank test, p = 0.9713). The Cox proportional hazards model showed no significant differences in instant risk to recovery between K values (hazard ratio = 0.563; p = 0.212) and between the studied categories (hazard ratio A vs. hazard ratio F = 0.827 (95% CI 0.360–1.901); hazard ratio A vs. hazard ratio M = 0.869 (95% CI 0.409–1.844); hazard ratio H vs. hazard ratio M = 1.050 (95% CI 0.464–2.379)] (Figure 5).
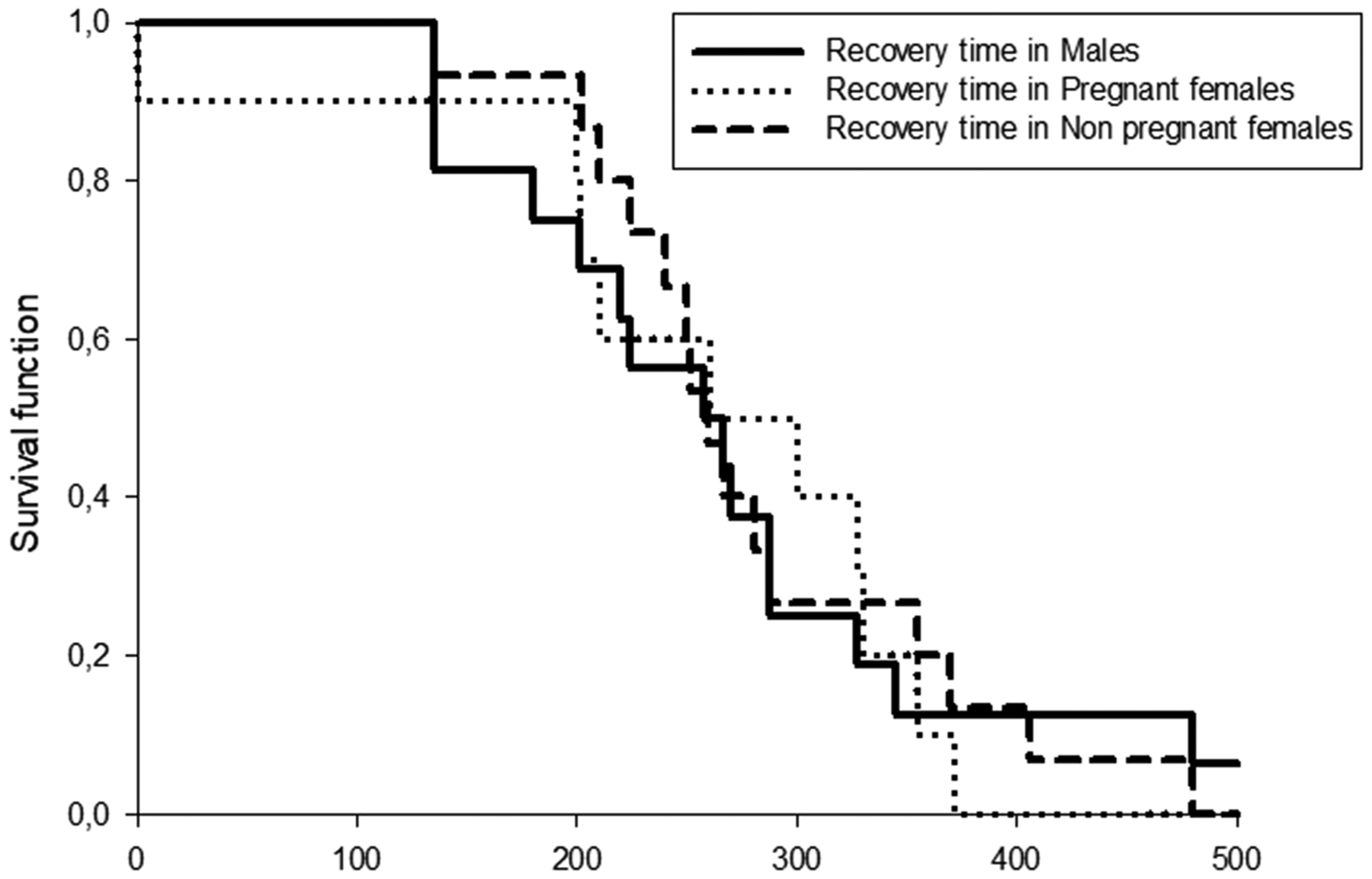
Kaplan–Meier survival analysis that estimated the time to recovery according sex categories (males, non-pregnant females and pregnant females) of Cnesterodon decemmaculatus.
Mortality post-anesthesia occurred only in males (two individuals during the monitory period 48 h after recovery).
Discussion
Our results indicate that 50 mg l−1 of eugenol administered to adults of C. decemmaculatus by immersion in an anesthetic bath resulted to be an effective concentration to be used in a laboratory setting. This dose produced short induction and recovery times and fish resumed normal swimming in the three groups. However this dose showed mortality only in males (12.8%) during the post-exposure period. In agreement with Martins et al. (2019) 2 and Sladky et al. (2001) 17 mortality may occur in fish with the use of eugenol if the concentration exceeds the security range caused by ventilatory failure. Moreover, the mortality in fish increases with increasing exposure time.8,14 In our study, according to the raw data, both dead males had been exposed to eugenol (55 s and 70 s, respectively) for a longer time than the median induction time (37.31 s). In addition, Martins et al. (2019) 2 reported other side effects of eugenol such as prolongation of cardiovascular recovery, medullary collapse caused by neurotoxic and hepatotoxic properties. Further studies are necessary to perform tests with different concentrations as has been done in other species in order to reduce side effects.1,2,10,13,14,22,32
According to Hoseini et al. (2019), 12 Millán-Ocampo et al. (2012), 13 Okamoto et al. (2009) 14 a good anesthetic induces sedation in less than 180 s and allows recovery in less than 300 s, which is in line with the results obtained with 50 mg l−1 of eugenol in our study.
The induction and recovery times following eugenol anesthesia have been assessed for different fish species,1,8,10,13 –15,28 –31 but the present study is the first report to include adult individuals of C. decemmaculatus of both sexes, including pregnant and non-pregnant females.
Different factors such as water quality, species, body size and weight, sex, season, condition, health status and stress affect physiological responses to anesthetics.9,33,34 In addition, the condition factor and health status influence the metabolic rate, therefore modifying the effectiveness of eugenol. 13 In turn, fish physiology is known to modulate induction time.10,35 On the other hand, different developmental stages (embryos and larvae) of zebrafish exposed to eugenol may affect the optimal concentration and likelihood of negative effects. 36 Further studies are needed to investigate the effect of these factors on the response of C. decemmaculatus to eugenol.
In the present study involving C. decemmaculatus anesthetized with eugenol, no significant differences were found in either induction time or recovery time between sexes and between pregnant and non-pregnant females. However, non-pregnant females showed a tendency toward lower hazards of recovery (hazard ratio A vs. hazard ratio F and M = 0.827 and 0.869) as compared with the other two categories (pregnant females and males), and further study with a larger sample size of individuals is required to confirm this finding.
Since the condition factor K is used to compare the ‘condition’ or ‘well-being’ of fish37,38 it appears to be more accurate than size, weight and the physiological status to assess the effect of eugenol (Figure 1), in agreement with Millán-Ocampo et al. (2012). 13 K seems to be the most suitable parameter for adjusting eugenol doses because it is more homogeneously distributed than weight and length population of C. decemmaculatus. This parameter is particularly useful for this sexually dimorphic species, with females being larger and more robust than males. 19 It is possible that variations in K can be used to indicate the need to adjust anesthesia and recovery times. Future studies are necessary to address the possible relationship between K variations and induction and recovery times in C. decemmaculatus anesthetized with eugenol. However, in our study, no significant differences were observed in size between males and females. This observation could potentially be attributed to the differences between studies conducted in a natural environment,39 –41 in contrast to controlled environmental conditions under which the fish were reared.
Huntingford (2004) 42 mentioned that strains selected for rapid growth exhibit higher rates of food consumption compared with the offspring of wild fishes from the population of origin that were reared in the hatchery. In addition, other studies suggested that ambient temperature and circannual rhythms significantly influence the feeding behavior and sizes of fish.43 –46 Differential growth or developmental rate could explain to influence sex differentiation.
Given that there are no previous studies of eugenol on C. decemmaculatus and we are aware of the need for further testing before recommending a full protocol, we are expecting this study be a contribution toward the development of a standard anesthetic protocol for this laboratory species to ensure care and animal welfare.
Footnotes
Acknowledgement
We thank the Centro Parasitologico y de Vectores (CEPAVE-CONICET, Argentina) for providing facilities and equipment to carry out this work.
Data availability statement
Authors agree to make data and materials supporting the results or analyses presented in this paper available upon reasonable request by email to the corresponding author. It was uploaded to the Zenodo database doi: 10.5281/zenodo.7968590.
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.