Abstract
Coagulation parameters are usually included in clinical and preclinical safety studies to evaluate the effect of xenobiotics on the extrinsic or intrinsic pathways of coagulation. The analysis is generally performed at the time of terminal sacrifice where many activities are scheduled. Chances of delay in analysis are likely particularly when blood is collected for coagulation via the abdominal vena cava. This experiment was planned to assess the variations in coagulation parameters caused by delay in analysis as well as by storage conditions. Blood was collected from the posterior vena cava under isoflurane anesthesia, and the plasma was separated immediately. Coagulation parameters were evaluated at 0, 6, 24 and 48 h from the plasma stored at room temperature, as well as plasma stored under refrigerated and freezing conditions. Stability of the analytes in blood was also evaluated under refrigerated conditions for 6 h. All parameters were analyzed using a semi-automated coagulometer. Prothrombin time (PT) was stable under all three storage conditions for up to 6 h. Although statistically significant differences were observed for activated partial thromboplastin time (APTT) at room and refrigeration temperatures for up to 6 h, the difference was clinically non-relevant. Fibrinogen was found to be the most stable parameter that showed consistency in results even up to 48 h under all three storage conditions. Plasma for PT can be stored and analyzed without any significant changes for up to 6 h from the actual blood collection, while fibrinogen level testing can be extended for up to 48 h after collection under any storage condition. For reliable APTT results, plasma samples should be run immediately after collection.
Prothrombin time (PT), activated partial thromboplastin time (APTT) and fibrinogen are common analytes used to evaluate the effect of any xenobiotics on hemostasis and usually included in preclinical safety studies as recommended by the Regulatory Affairs Committee (RAC) of the American Society for Veterinary Clinical Pathology (ASVCP). 1
PT is a universal screening test that measures the efficiency of the extrinsic coagulation system. It contains factors I, II, V, VII and X. APTT is also a universal test which measures the competence of the intrinsic system and is used to investigate the abnormalities in factors I, II, V, VIII, IX, X, XI, and XII. 2 Fibrinogen is a protein that originates in the liver and is converted to fibrin during the blood clotting process. Evaluation of fibrinogen abnormalities aids in the diagnosis of suspected clotting or bleeding disorders. 3
Pre-analytical variables including specimen collection, anticoagulant type and concentration, hematocrit, filling status of the sampling tube, transportation, centrifugation, as well as storage and assay methods can all affect coagulation test and factor analysis results. According to the National Committee for Clinical Laboratory Standards (NCCLS) guidelines, coagulation tests should be done within 2 h if samples are kept at room temperature, 4 h if they are stored at 2–4℃ and two weeks for those preserved at −20℃. Several studies recommended that PT and APTT determinations may be constant for periods longer than those currently suggested in the NCCLS guidelines. 2 In addition, these guidelines do not stipulate whether samples should be kept as whole blood or whether plasma should be separated prior to storage. 4
The Clinical and Laboratory Standards Institute (CLSI) H21-A5 has recommended that specimens should be analyzed within 24 h for PT, and 4 h for APTT and other assays if stored at room temperature (25℃). Nevertheless, they have not recommended any storage time for refrigerated samples (2–8℃). 5 In addition, there is some concern about the effect of time on coagulation parameters used in toxicological studies, since post-sampling change (one of many pre-analytical conditions) can lead to false high values, which in turn may lead to misinterpretation of the test compound effect in toxicological studies. 6
Analysis of coagulation parameters during preclinical studies is generally done at the time of terminal sacrifice where a number of activities are also scheduled. Thus chances of delay in analysis are likely particularly when blood for these parameters is collected via the abdominal vena cava, causing a long delay between the first and last sample collection. Based on our literature search, we could not find any studies which describe in detail the stability of coagulation parameters viz. PT, APTT and fibrinogen in rats which are the most commonly used laboratory animal species in toxicological studies. This experiment was planned with the intention of investigating the variation in coagulation parameters that may occur due to delay in analysis as well as due to storage conditions.
Animals, materials and methods
Thirty-six Wistar rats of a conventional type (microbiological status not specified) were procured from RCC India Ltd (Hyderabad, India) and maintained on autoclaved extruded rodent feed (Nutrilab; Provimi Animal Nutrition India Pvt Ltd, Bangalore, India) and potable water (water which has passed through a water purification system) ad libitum. The rats were allowed to acclimatize for seven days to experimental conditions. The rats were 6–8 weeks of age with average body weights of 205 ± 7.5 g for males and 174 ± 4.4 g for females at the time of blood collection. The rats were housed one per cage in sterilized solid bottom polycarbonate cages (Tecniplast, Buguggiate, Italy) of dimension 425 × 266 × 185 mm and a floor area of 800 cm2 with a stainless steel grill top for the feed and water bottle. Autoclaved corn cob (Sparcobb; Sagar Industries, Bangalore, India) was used as bedding material. The cages were suspended on a stainless steel rack. The rats were maintained in well regulated environmental conditions: with a temperature of 21 ± 3℃, relative humidity of between 30 and 70%, air changes of 13–15 per hour and a 12:12 h light–dark cycle. Animal handling and experimental procedure were approved by the Institutional Animal Ethical Committee, and the experiment was performed in accordance with the Guide for the care and use of laboratory animals (National Institutes of Health (NIH), 1985) and the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), India.
Anesthesia and blood collection
Eighteen male and 18 female rats were randomized into three groups: six males and six females for each storage condition. All the rats were fasted (water allowed) overnight. Rats were anesthetized using isoflurane, and blood samples were collected using a 24 gauge needle attached to a 2 mL syringe from the posterior vena cava through a laparotomy incision. Blood samples were collected in tubes containing 3.2% sodium citrate (approximately one part of citrate and nine parts of blood) which was thoroughly mixed. Animals were then humanely euthanized by exsanguinations under isoflurane anesthesia. Blood was centrifuged for 10 min at 4000 rpm immediately after collection. Plasma was separated without delay and aliquots of plasma were prepared and stored at room temperature (22 ± 3℃), refrigerated (4℃) and freezing (−20℃) temperatures for 6, 24 and 48 h. The stability of analytes in blood was also evaluated (n = 6) under the refrigerated condition for 6 h after blood collection.
Coagulation parameters
The coagulation parameters, viz. PT, APTT, and fibrinogen concentrations from all the storage conditions, including fresh plasma, were analyzed using a semi-automated coagulometer (Stago Start 4; Diagnostica Stago, Asnières-Sur-Seine, France) with reagents supplied by Stago (Diagnostica Stago). Standard control samples were analyzed to ensure the quality of analysis before and after analysis of samples for each analyte. All the values obtained from the standard control for the different coagulation parameters were always within the expected ranges. All essential precautions were taken to avoid any pre-analytical, analytical and post-analytical errors.
Prothrombin time test
Fifty microliters of citrated plasma was incubated for one minute at 37℃, 100 µL of the PT reagent (Neoplastin CI Plus; Diagnostica Stago) warmed at 37℃ was added to the incubated plasma. Time from the plasma–reagent mixing to clot formation was measured in seconds.
Activated partial thromboplastin time test
Fifty microliters of citrated plasma was incubated for 3 min at 37℃ with 50 µL of warmed APTT reagent (CK Prest; Diagnostica Stago), 50 µL of calcium chloride warmed at 37℃ was added to the incubated sample. Time from addition of calcium chloride to clot formation was measured in seconds.
Fibrinogen determination
One hundred microliters of diluted plasma (diluted 1:20 with a combination of 50 µL of plasma +900 µL of Owren-Koller buffer; Diagnostica Stago) was incubated for one minute at 37℃, 100 µL of the fibrinogen reagent (Fibri-Prest; Diagnostica Stago) warmed at 37℃ was added to the incubated plasma. Time from the plasma–reagent mixing to clot formation was measured in seconds. The time (in seconds) was automatically converted into mg/dL by the semi-automated coagulometer Stago Start 4.
Statistical analyses
All the results were expressed as mean ± standard deviation (SD). Significant differences were determined from a one-way analysis of variance (ANOVA) and utilized by a Dunnett’s comparison of each time-point with the basal level (0 h). Stability of coagulation parameters in blood kept under the refrigerated condition and gender difference on coagulation parameters were compared by t-tests (for gender effect and the average of zero-hour data from all three storage conditions were considered). Data were analyzed using GraphPad Prism Software (version 4; GraphPad Software, San Diego, CA, USA).
Results
Effect of various storage conditions on prothrombin time (PT).
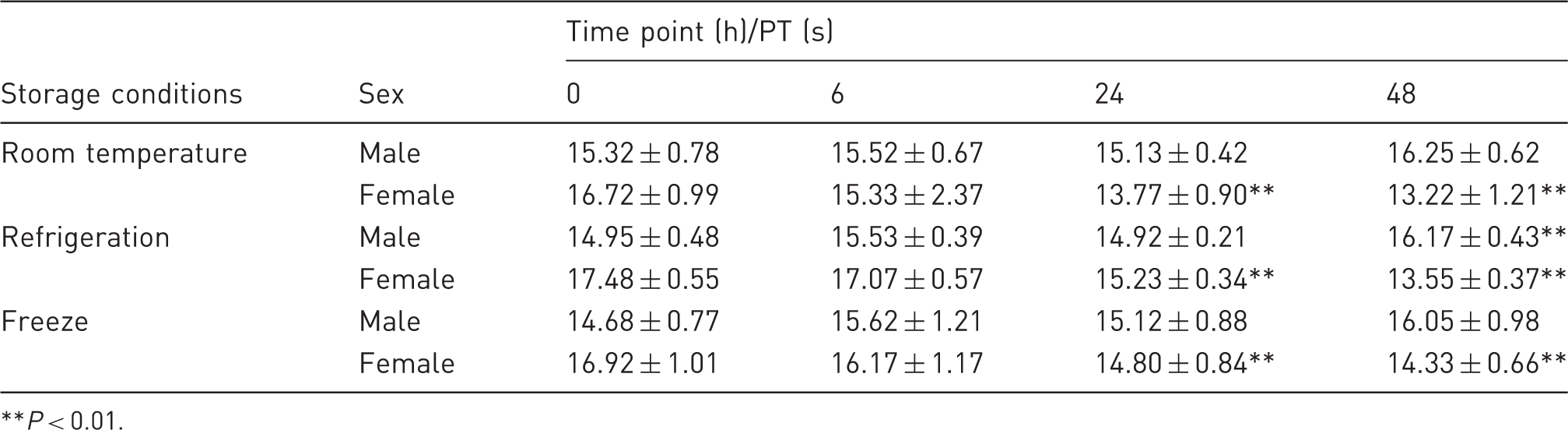
P < 0.01.
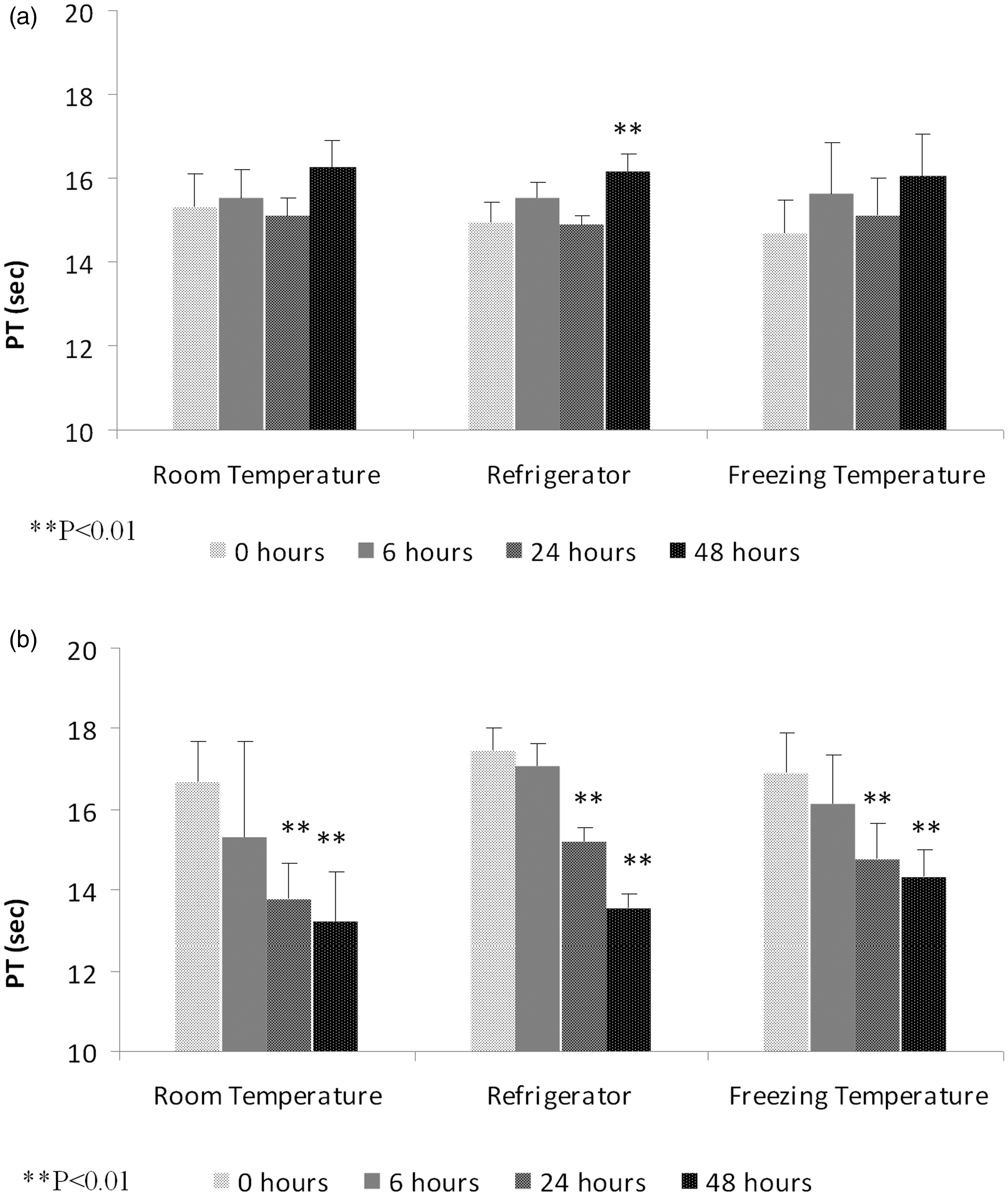
Effect of various storage conditions on prothrombin time. (a) Protrombin time in male Wistar rats. (b) Protrombin time in female Wistar rats. **P < 0.01.
Effect of various storage conditions on activated partial thromboplastin time (APTT).
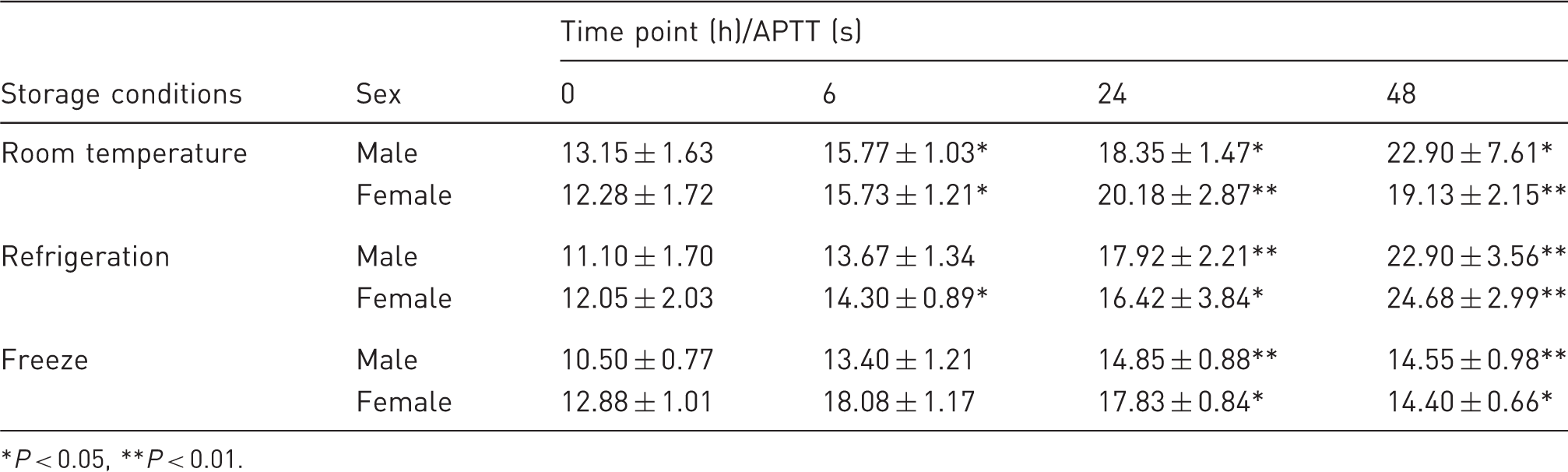
P < 0.05, **P < 0.01.
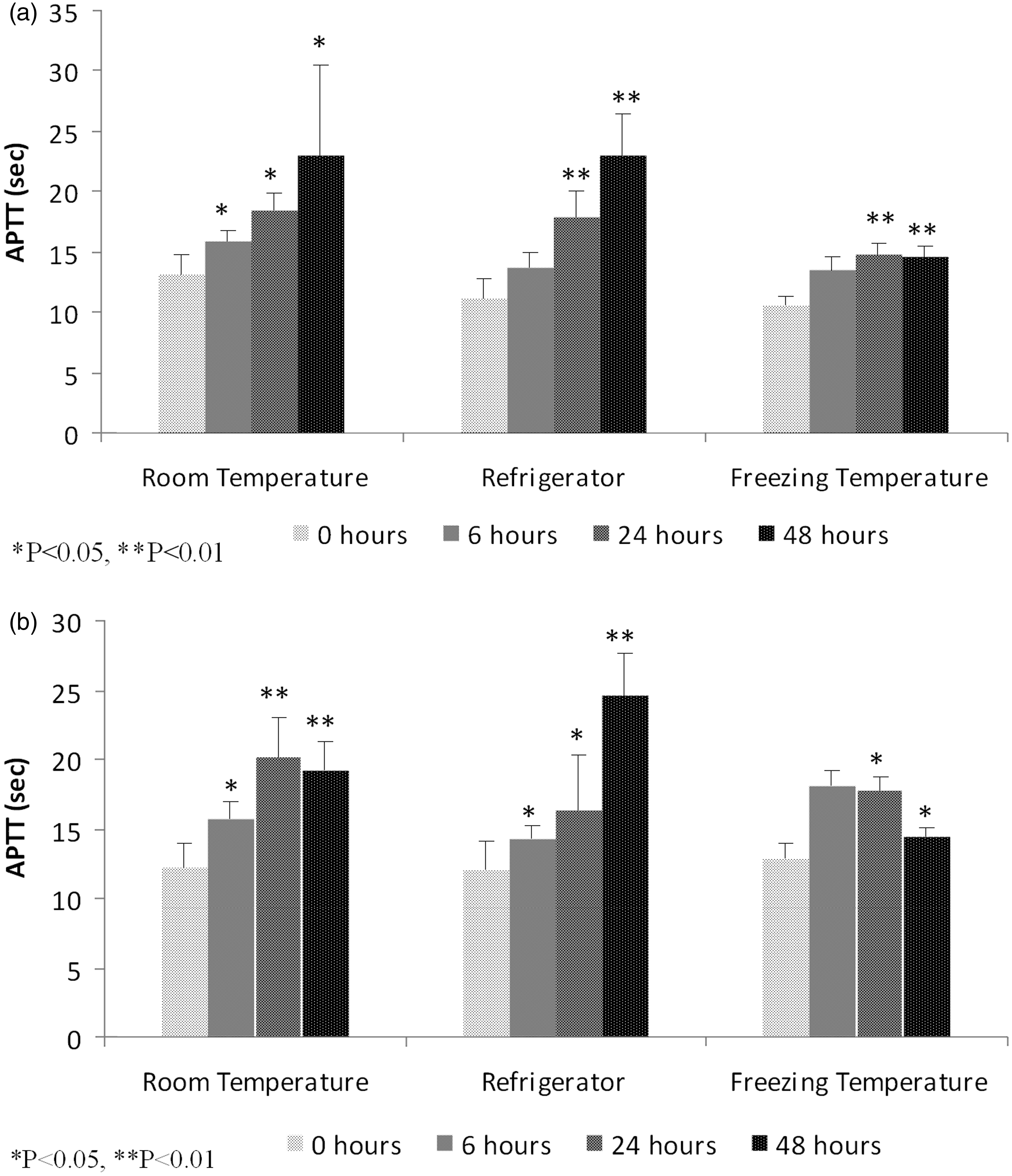
Effect of various storage conditions on activated partial thromboplastin time. (a) APTT in male Wistar rats. (b) APTT in female Wistar rats. *P < 0.05, **P < 0.01.
Effect of various storage conditions on fibrinogen concentration.
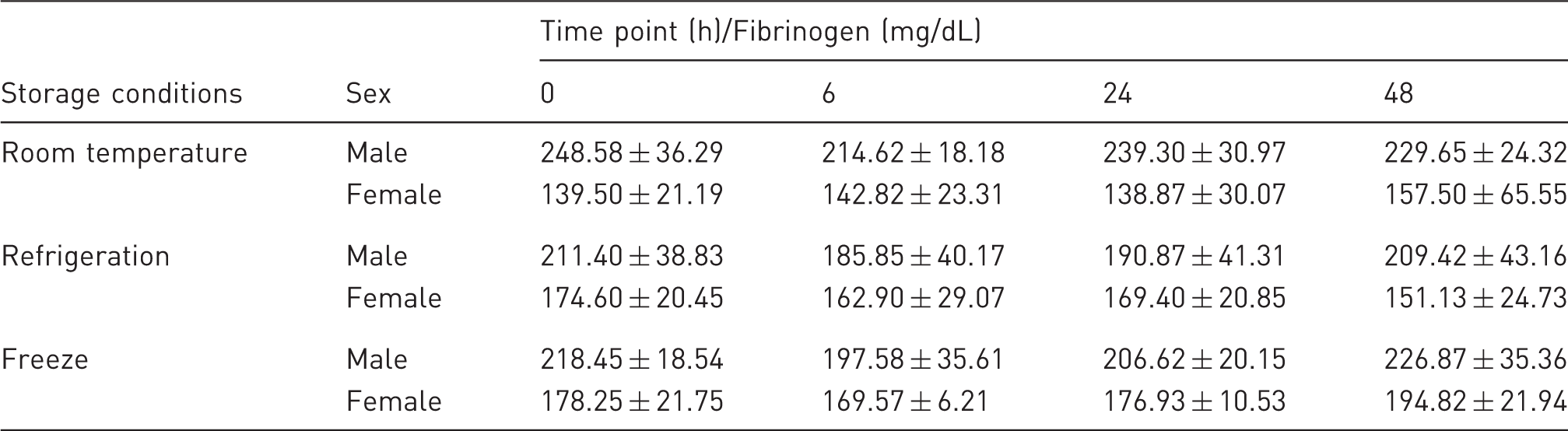
Effect of blood refrigeration on coagulation parameters.

PT: prothrombin time, APTT: activated partial thromboplastin time.
Sex difference on coagulation parameters.

P < 0.01. PT: prothrombin time, APTT: activated partial thromboplastin time.
Discussion
Coagulation parameters viz. PT, APTT and fibrinogen are usually included in clinical and preclinical safety studies to evaluate the effect of xenobiotics on the extrinsic or intrinsic pathways of coagulation. Carrying out these tests as soon as possible after blood collection is an ideal situation; however, in practice a delay in analysis is sometimes inevitable. Therefore, it is deemed necessary to evaluate the effects of storage temperature and time on the outcome of results. Most of the published reports are based on either humans or domesticated animals, and very little is reported involving laboratory animals. Hence, the present study was undertaken to demonstrate the effects of various storage conditions on coagulation parameters. The results of the present study provide new insight regarding the milieu of plasma storage and its effects on coagulation parameters.
Conflicting results have been reported in the literature on the effect of storage conditions on different coagulation parameters. In equines, PT, APTT and fibrinogen concentrations have been reported to be stable for 24 h at −20℃; while in canines, it has been reported that storage of plasma for up to 24 h at 8℃ has a significant effect on fibrinogen concentrations, while having an insignificant biological effect on PT and APTT.3,7 Similarly, some of the published work on humans has suggested that plasma for PT and fibrinogen can be safely stored for 24 h at both 4℃ and 25℃, and for APTT for 12 h at 4℃ and 8 h at 25℃. 5 Others also have similarly concluded that PT and APTT in humans can be analyzed without any significant changes for up to 24 h and 12 h, respectively, both at room temperature and under refrigerated conditions. 4 Another study has reported that prolonged storage of human plasma for 1, 2 or 3 weeks at −20℃ results in significant differences on PT and APTT. 8 Some of the researchers have opined that the magnitude of PT changes in rats, mice, rabbits, dogs, monkeys and humans up to 96 h of storage at 4℃ and 25℃ is usually so small as to be of no major clinical importance. Prolongation of APTT in rats is rapid and marked, with times doubling within 24 h post-sampling storage at 4℃ and 25℃. Similar findings were noted in our experiment where APPT was increased significantly (∼1.5×) after 24 h under all the storage conditions. Plasma APTT in humans and monkeys were also affected, but to a lesser extent; however no effects were observed in mice, rabbits and dogs. 6 Increased concentration of fibrinogen has been associated with shortened PT and APTT in humans and dogs, but not in rats. 9 It is believed that for reliable results, samples should be processed and run immediately after collection. 10 Results derived from our experiment also suggest that PT can be analyzed without significant effects for up to 6 h post collection if the plasma is stored under any of the three storage conditions. Similarly, the value of APTT is most reliable if the plasma is analyzed instantly or is stored for not more than 6 h, while fibrinogen level testing can be extended up to 48 h after collection under any of the three storage conditions. The variations noted in stability of coagulation parameters in different studies may be attributed to different study populations, study design, reagents, instruments, and collection containers. However, all findings in stability of coagulation parameters in humans are more or less similar to our findings.
The question arises of what makes the PT and APTT relatively less stable. The answer rests on the fact that PT and APTT reflect the activities of multiple clotting factors, and it has been shown that a significant decrease in any one factor must occur before the PT or APTT is significantly prolonged. 3 Delay in transport may also affect a particular labile factor (factors V and VIII), leading to prolonged clotting time. 11 A decrease in activity of factor X is associated with prolonged PT and APTT in dogs when samples were stored for 48 h. As factor X activity affects both PT and APTT results, a marked loss of factor X activity may explain the statistically significant changes in both the hemostatic tests. 3 Stability in PT and APTT is circuitously dependent on stability of blood clotting factors. The effect on stability of APTT parameters in the present study could be due to the effect of stability of blood clotting factors. In parity with our findings, false prolonged values for APTT in rats, humans and monkeys were obtained due to a loss of activity of coagulation factors on prolonged standing. 6 In the present study, a minimal decrease in PT noted at 24 h after storage under all three conditions is in accordance with studies in humans12,13 and dogs. 14 This instability may be the result of clot-induced activation of proteolytic enzymes that are responsible for the slow degradation of factors VIII, IX and XI. 15
Small statistically significant differences between the mean values for control and treated animals for PT and APTT assays (e.g. <2 s) are observed occasionally in toxicology studies. Although usually not toxicologically meaningful, interpretation of these small differences requires the consideration of many factors. 16 The magnitude of a slight variation in results of approximately 2 s, which is observed in APTT at room and refrigerated temperatures of up to 6 h in the present study may have statistically significance but it is biologically acceptable based on the scientific justification such as its fall in the range of historical data or reference range for the species and in scrutiny undertaken in the experiment. In toxicity studies, if animals are not randomized before sample collection, the processing and analysis of samples of one dose may be performed earlier than the control or other doses, which may impact the study results. To circumvent this kind of pre-analytical variable, randomization of animals is advisable to counteract storage time-related bias in results.
Consistent with our findings, gender differences in haemostatic parameters (such as increased PT and decreased fibrinogen values in female Wistar rats) have also been reported, with the influence of steroid sex hormones. 17 In disparity with our findings, sex differences in PT and APTT values were not observed in F344 rats, mice, rabbits, dogs, monkeys and humans under the non-fasting condition, but under the fasted condition PT and APTT values in female F344 rats were found to be less than in male rats, and the difference resulted from the vitamin K-deficient syndrome in male rats. 6
In conclusion, the results of this study suggest that PT and APTT can be analyzed without significant effect for up to 6 h post collection if the plasma is stored under any of the three storage conditions. Fibrinogen level testing can be extended up to 48 h after collection under any of the three storage conditions. Blood can be stored for up to 6 h at the refrigerated temperature for analysis of PT, APTT and fibrinogen. In summary, it is better to analyze samples collected on the same day to distinguish any treatment-related changes in hemostatic parameters during toxicity studies.
Footnotes
Acknowledgement
The authors are thankful to Mr Venkat Jasti, CEO and Chairman of Suven Life Sciences Limited for providing the necessary support and facilities for this experiment.
Declaration of conflicting interests
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of this paper.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
